Abstract
Introduction:
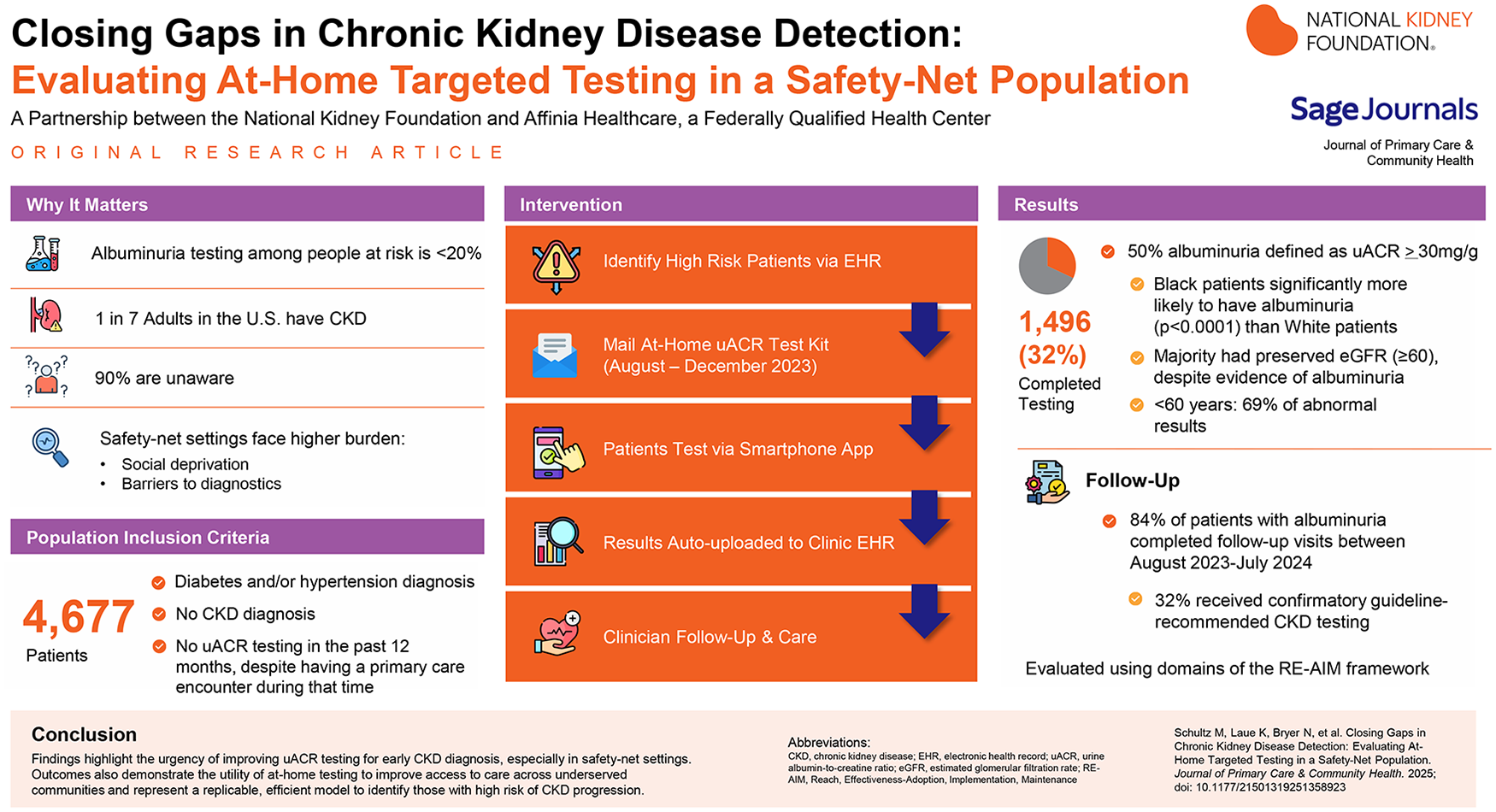
Chronic Kidney Disease (CKD) affects 1 in 7 adults in the United States, yet 90% of those impacted remain unaware of their condition, and fewer than 20% of at-risk individuals are appropriately tested. Safety-net health care settings are disproportionately burdened by CKD, with a patient population enriched for CKD risk factors, social deprivation, and barriers to diagnostic testing which delay access to diagnosis and lifesaving interventions. The National Kidney Foundation partnered with a Federally Qualified Health Center (FQHC) to evaluate an approach to increase guideline-recommended testing among patients at high-risk for developing CKD.
Methods:
Through electronic health record (EHR) data analysis, eligible patients were identified to receive an at-home, semi-quantitative urine albumin-creatine ratio (uACR) testing kit. The kits provided immediate results via a smartphone application, as well as automatically routed to the clinic EHR for the patient’s provider to coordinate necessary follow-up care. This initiative was conducted in Missouri, USA in 2023 and evaluated using domains of the Reach, Effectiveness, Adoption, Implementation, and Maintenance (RE-AIM) framework.
Results:
Results reflect that 1496 of 4677 (32%) eligible patients completed uACR testing with 50% receiving abnormal results indicative of albuminuria. Of those with evidence of albuminuria, 84% had follow-up visits and 32% completed appropriate follow-up testing based on clinical guidelines. Albuminuria was prevalent across all age groups, with 69% of abnormal results appearing in patients under 60 years. Consistent with national data, patients identifying as Black were significantly more likely to have albuminuria in this cohort (P < .0001). Notably, most patients with albuminuria had an eGFR ≥60 mL/min/1.73 m2.
Conclusions:
Findings highlight the urgency of improving uACR testing for early CKD diagnosis, especially in safety-net settings. The findings also demonstrate the utility of at-home testing to improve access to care across underserved communities and represent a replicable, efficient model to identify those with high risk of CKD progression. While the program required significant time and coordination, this can be streamlined for analogous programs. Future opportunities exist to further the impacts including additional quality improvement activities to ensure follow-up testing and close gaps in CKD care.
Keywords
Introduction
Approximately 35.5 million people in the United States (US) have chronic kidney disease (CKD) and up to 90% are undiagnosed and remain unaware, including those established in primary care. 1 Approximately one-third of US adults are at risk for developing CKD, including adults with diabetes and/or hypertension.2,3 These leading risk factors accelerate a decline in kidney function, which in turn significantly heightens the risk for cardiovascular disease (CVD). More than 50% of people with early-stage CKD do not survive to end stage kidney disease (ESKD) due to cardiovascular mortality. 4
CKD is defined by the presence of kidney damage (including presence of albuminuria) or decreased kidney function for at least 3 months, irrespective of the cause. 5 CKD is often asymptomatic and requires annual testing for those with risk factors.6,7 Two laboratory tests are needed to appropriately diagnose and stage CKD—estimated glomerular filtration rate (eGFR) and urine albumin-creatinine ratio (uACR). 8 Both are essential as each independently predicts CKD and CVD risk. 4 While eGFR testing for kidney function is widely ordered as part of routine metabolic panels, annual uACR testing rates remain low nationally, at 11.3%, 32.2%, and 43% among the populations with hypertension, diabetes, and both, respectively. 9 uACR testing is needed for risk stratification and prognosis and is one of the earliest predictors of kidney damage, often before eGFR declines. 10
Gaps in testing inhibit timely access to evidence-based interventions demonstrated to improve health outcomes. As CKD progresses, cost of care increases as a result of increased hospitalizations, readmissions, emergency department utilization, and complications, but timely management can effectively control these costs.11 -14 CKD testing and diagnosis must increase to improve the cost and quality of care for people living with CKD, especially for high-risk patients with diabetes and hypertension.
Inequities in CKD outcomes are common and well-documented, demanding interventions. Black and Hispanic individuals are less likely to receive pre-dialysis kidney care, more likely to advance to ESKD, and less likely to receive a kidney transplant.15 -18 Common social determinants of health (SDoH) barriers (eg, housing instability and environmental risks, limited access to healthy foods or transportation, and physiologic stress caused by racism and discrimination) have been associated with a higher prevalence of CKD and CVD.17 -20 Safety-net health care settings (including Federally Qualified Health Centers [FQHCs]) are particularly challenged by CKD underdiagnosis due to higher proportions of patients with risk factors and higher rates of patient-level barriers to diagnostic testing (eg, time, transportation, and awareness). 21 Patients served by these systems generally have lower socioeconomic status, lower levels of health literacy, and more frequently identify as Black, Indigenous, or a Person of Color (BIPOC). 21 Hence, FQHCs are an ideal setting to implement quality improvement interventions to drive enhancements in equitable CKD care.
At-home testing has demonstrated efficacy in improving completion of CKD testing, especially for uACR, but to the authors’ knowledge, has not previously been evaluated in a safety-net health care setting.22 -24 Therefore, the primary aim of this intervention was to determine the efficacy of at-home testing for increasing rates of guideline-concordant testing for CKD among patients with hypertension and/or diabetes in a safety-net health care setting. Secondarily, the program aimed to evaluate the extent to which patients with evidence of albuminuria (A2 [30-300 mg/g] or A3 [>300 mg/g] 6 ) from at-home tests engage with their primary care provider (PCP) for follow-up care.
Methods
To close gaps in CKD detection, the National Kidney Foundation partnered with Affinia Healthcare, a FQHC in the St. Louis, Missouri metropolitan area, to implement this pilot program. The at-home test utilized was a semi-quantitative uACR test (Healthy.io, Boston, Massachusetts), with results read via an associated smartphone application.
Patient eligibility aligned with clinical practice guidelines;8,25 inclusion criteria consisted of patients who: (1) had a diagnosis of diabetes and/or hypertension (2) did not have a CKD diagnosis, and (3) had not received a uACR test in the past 12 months despite having a primary care encounter during that time. Exclusion criteria were: (1) inability to speak and read English or Spanish, (2) residence in chronic care facility, (3) eGFR <15 mL/min/1.73 m2, and (4) treatment with dialysis or kidney transplant. As albuminuria testing is recommended annually for patients with diabetes and/or hypertension, informed consent was not required. 25
Eligible patients were identified from the electronic health record (EHR) using ICD-10-CM codes and CPT codes as shown in Supplemental Table S1, along with filtering for the last visit date and missing laboratory test results during the time frame. A list of eligible patients was transmitted via a secure data file to the manufacturer who coordinated the mailing of kits. To maximize timely distribution of kits, an ‘opt out’ method was used where all eligible patients received a test and then had the opportunity to decline completion. At-risk patients were notified via a letter signed by their PCP regarding their need for testing. Approximately 15 days later, patients received a test kit via mail with instructions for how to complete the test using their mobile phones. A 60-day window for completion was established to encourage timely testing. At 3 points during that period, a member of the manufacturers’ engagement team completed phone calls, text messages, and/or email outreach to assist patients who had not participated to support them with test completion.
Upon completion, patients received immediate results and guidance on the next steps via a smartphone application that read the test results. Electronic transmission of test results to the FQHC’s EHR was also established to ensure that results were easily viewed and addressed by the PCP. Patients with abnormal results were instructed to schedule a follow-up visit with their provider for confirmatory testing. FQHC staff contacted patients with abnormal results who did not schedule appointments within 7 days.
To accommodate PCP capacity for follow-up, the kits were sent out incrementally over 7 weeks. Patients were divided into cohorts with a relatively equal distribution of patients assigned to each PCP involved in the pilot. Providers received written and verbal communication about the upcoming intervention and a workflow (Supplemental Figure S1), outlining the test process and recommendations for addressing abnormal results. However, no formal training was provided to providers regarding the program or reinforcing best practices for CKD management.
A waiver of informed consent was obtained from the Washington University in St. Louis Institutional Review Board to conduct this retrospective analysis. Data was collected from 2 periods (Baseline Period: February 1, 2022-July 1, 2023, and Intervention Period: August 15, 2023-June 1, 2024). Baseline was defined as the 12-month period prior to the deployment of the intervention. Baseline data included the full cohort of patients receiving test kits. Intervention data was extracted only for those who completed a test and were analyzed comparing sub-populations grouped by test result categories (normal/A1 albuminuria [<30 mg/g] vs abnormal/A2 [30-300 mg/g] and high abnormal/A3 [>300 mg/g]). Changes in individual metrics were assessed for statistical significance by comparing baseline and post-intervention results using the Chi-square test along with percent change comparisons. To address incomplete data (eg, some participants missing blood pressure, A1C, etc.), the analysis for each variable compared overall population trends rather than limiting analysis to individuals with a complete dataset for all variables.
The RE-AIM framework was identified as a useful tool to comprehensively organize and assess the effects of the intervention; however, this paper will only speak to Reach and Effectiveness as described in Tables 1 and 2.
Program Outcomes – Reach Domain.

Laboratory results were unmapped or unable to match the data to the FQHC’s electronic health record.
For age analysis, test kit manufacturer population size for non-completers differs from the FQHC’s electronic health record. Age categorization was not provided by the FQHC out of abundance of caution in the interest of safeguarding protected health information.
Program Outcomes- Effectiveness Domain.

Results
As described in Figure 1, 4997 participants met the inclusion criteria. A total of 4824 test kits were mailed to patients with 147 returned due to an undeliverable address, resulting in 4677 participants receiving kits. Out of these 4677 tests, 1496 (32%) were completed. When comparing completers and dropouts, completion rates by various demographics differed significantly by race (P = .0017), ethnicity (P < .0001), primary language spoken (P < .001), and insurance type (P < .0001) with each demographic analyzed independently (Supplemental Table S2). The majority of test completers identified as Black (73%), had hypertension without diabetes (71%), and utilized Medicaid (37%) as their primary insurer with a median age of 56 years (Table 1).
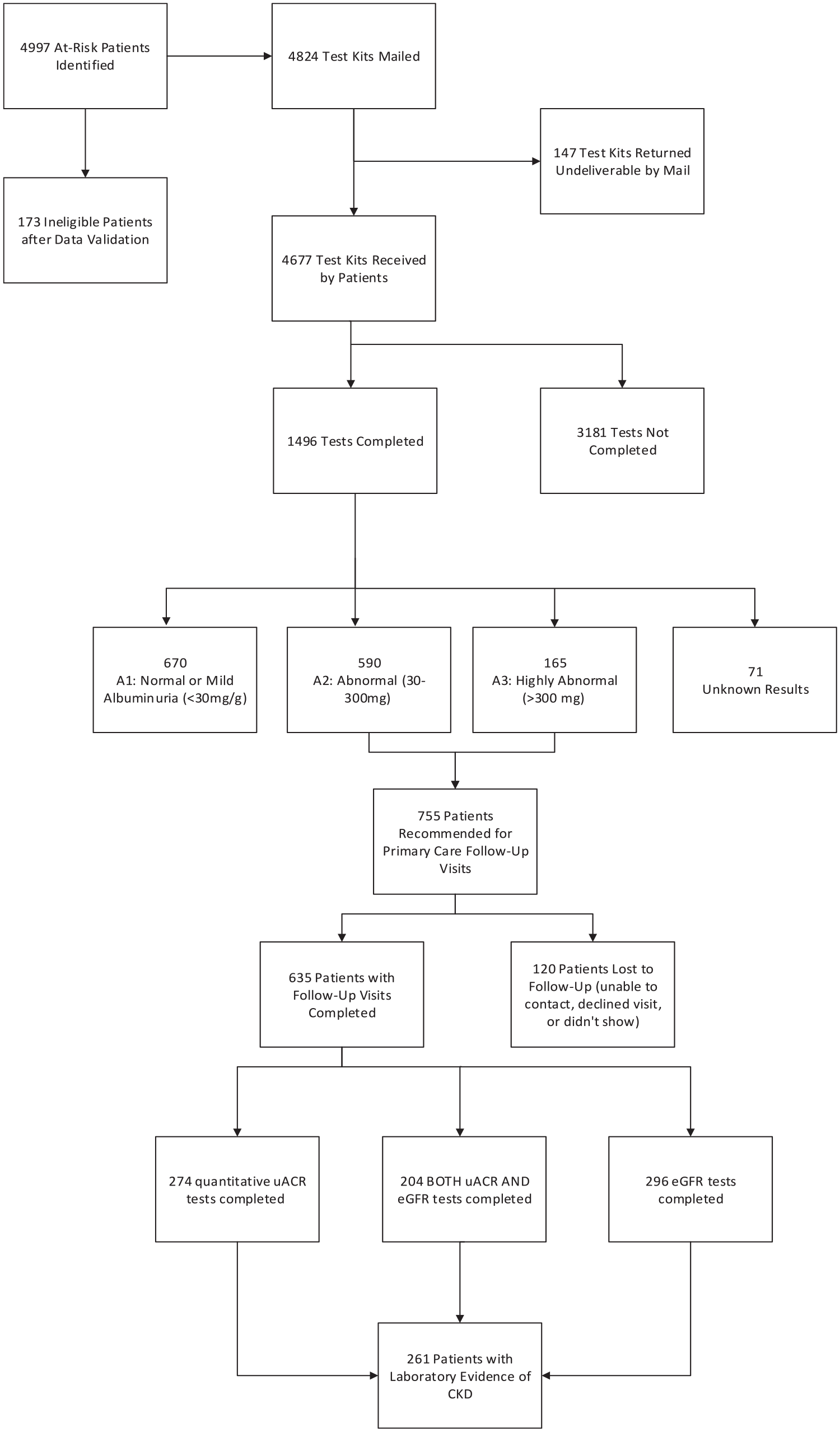
Enrollment and results flow diagram.
Approximately half of the completed tests showed abnormal uACR results (A2 or A3 albuminuria). Positive test results were significantly more common among Black individuals compared to White individuals (P < .0001). The prevalence of albuminuria was relatively consistent across all age groups (Figure 2). Approximately two-thirds of tests completed were in patients under age 60 years, with 521 of these patients found to have albuminuria.

Test results by age groupa.
Of the patients who had abnormal results, 635 (84%) completed a follow-up visit with their PCP, with 366 of those patients (58%) having at least 1 CKD test (eGFR or uACR) completed and 204 (32%) receiving complete CKD testing (both eGFR and uACR; Figure 1). Further details on test completion and results of follow-up testing are provided in Table 2. Ninety-one percent of patients who received a follow-up quantitative uACR test had results that remained abnormal. In total, 261 patients had laboratory evidence suggestive of CKD (uACR ≥30 mg/g and/or eGFR <60 mL/min/1.73 m2) and, pending repeat confirmatory testing, could receive a diagnosis, with 90% falling into a CKD stage G1 or G2 category. However, nearly 20% of patients could not be staged appropriately due to incomplete testing.
Discussion
The goal for this intervention was to rapidly identify patients with CKD risk factors who had evidence of albuminuria. Results demonstrated that at-home testing for albuminuria is feasible and effective in a FQHC population. The EHR data extraction method allowed the health setting to rapidly triage a high-risk cohort for intervention. A majority of the patients tested had evidence of albuminuria, which had previously gone untested. Most patients with evidence of albuminuria received follow-up care. Thus, this program represented a time-effective way to screen a large, high-risk population for CKD and improve access to care across underserved communities.
The program successfully engaged patients who would have remained undetected using traditional screening methods. All participants had been active patients seen in the past year but had not received the tests needed to screen for CKD. Many of the patients in the program exhibited albuminuria with an eGFR ≥60 mL/min/1.73 m2, demonstrating the utility of albuminuria testing to identify CKD early. Given the association between albuminuria and risk of CKD progression and/or CVD complications, this program successfully identified high risk patients. The majority of patients tested were younger than 60 years, despite national data showing a higher prevalence of CKD in adults over 65 years and broad perceptions of CKD being a disease of older adults, 26 demonstrating the utility of this intervention to improve identification in younger patients who traditionally are perceived as lower risk.
This program also illustrates the necessity of improving secondary prevention of CKD in safety-net healthcare settings. In alignment with other literature that shows that the safety-net population is enriched for CKD, 21 the tested population in this program had a high prevalence of proteinuria (50%), compared with national rates among adults with diabetes or hypertension (35%). 27 As described in Table 1, there was a significant association between Black race and positive test results. This aligns with national data indicating that CKD disproportionately impacts Black communities.1,26 Although racial disparities in disease prevalence are primarily attributed to the impacts of SDoH, interventions such as this to improve early identification and diagnosis are essential for addressing inequities in late-stage outcomes.17,28
Completion rates were slightly lower than previously published studies on at-home albuminuria testing.23 -25 Other similar interventions leveraged an opt-in approach which presumably resulted in test recipients being more activated to complete the tests. However, the opt out model in this program eliminated the need for staff to contact patients prior to kit distribution, making it more feasible to replicate and potentially increasing overall reach. The intervention site and cohort demographics also likely contributed to lower completion rates in comparison to similar studies. Although this is, to the authors’ knowledge, the first use of at-home uACR testing in a safety-net setting, at-home testing for colon cancer has been previously deployed in similar settings with comparable return rates (20%-37%).29,30
Another factor for lower completion may be limited awareness of the need for CKD testing or unfamiliarity with home testing among the population. Although patients were informed in advance via letter that they would receive a test kit, some still contacted the clinic with questions regarding its legitimacy or the reason for receiving it. Delays between data extraction and kit mailing also contributed; some of the patients who met inclusion criteria had switched to another primary care facility and a few patients passed away before completing the test. Lastly, the requirement to have access to and comfort using a smartphone, either the patients’ own or that of a care partner or family member, may have impacted test completion for this population. While 91.5% of Missourians have access to a smartphone and 76.6% of those with an income less than $20 000 have broadband internet subscription, 31 there was no verification that all patients in the cohort had access to appropriate technology. Although efforts were made to ensure technology was user friendly, there was no verification that test recipients in this program had the willingness to download the application, or technological competency needed to utilize the tools prior to patients receiving and completing the test.
One of the limitations of the intervention, as described in Table 1, was that some patients (~5%) could not be definitively matched between the FQHC and the test kit manufacturer’s data reports. Because the EHR reflected the most up-to-date patient information, this data was prioritized for analysis over the manufacturer’s data. Another challenge noted was the delay between data extraction and kit deployment resulting in some patients being tested twice in a less than 3-month period.
While the program succeeded in its goal to increase testing, its impacts on CKD diagnosis were limited, in part by FQHC capacity constraints that prevented PCP education on CKD diagnosis and management guidelines prior to the intervention. While a recommended clinical workflow and best practices were provided, there was variability in how providers approached addressing the results. As a result, many patients with evidence of albuminuria from at-home testing did not complete follow-up laboratory testing in the clinic necessary to receive a subsequent CKD diagnosis. This highlights the opportunities to implement additional activities to increase complete testing rates and appropriately render a CKD diagnosis for patients with laboratory evidence. Such activities could include continuing education resources for providers, integration of clinical decision support tools in the EHR, or implementation of a structured quality improvement program such as a CKD Learning Collaborative. 32
Conclusion
This pilot offers a replicable model for addressing critical gaps in access to guideline-concordant CKD care within a FQHC population. There was a significant increase in the clinic’s CKD testing rates, remaining high after the pilot’s conclusion and indicating sustained awareness among both patients and providers. As uACR testing is associated with an increase in guideline concordant care, including utilization of medications that can help mitigate CKD progression and CVD outcomes, this intervention will likely improve quality of care long-term.27,33 Future analysis is needed to verify this hypothesis, as well as impact of the intervention on changes in risk factor control among patients identified with albuminuria.
Supplemental Material
sj-docx-1-jpc-10.1177_21501319251358923 – Supplemental material for Closing Gaps in Chronic Kidney Disease Detection: Evaluating At-Home Targeted Testing in a Safety-Net Population
Supplemental material, sj-docx-1-jpc-10.1177_21501319251358923 for Closing Gaps in Chronic Kidney Disease Detection: Evaluating At-Home Targeted Testing in a Safety-Net Population by Megan Schultz, Katelyn Laue, Nicole Bryer, Andrew Bzowyckyj, Anuja Java, Leslie Lake, Elizabeth Talbot-Montgomery, Brit Sovic, Bri’Anna Watson and Joseph A. Vassalotti in Journal of Primary Care & Community Health
Footnotes
Acknowledgements
The authors recognize and thank the members of the Healthy.io team, especially Adam Russell, who contributed to the program design and provided supplemental data used for evaluation. The authors also recognize and thank Robert Hayden (NKF) for his project management support during the intervention design and deployment phase of the project.
Author Note
Leslie Lake is now with the Veteran Evaluation Services, an affiliate of Maximus LLC., since completing this project.
Ethical Considerations
The Washington University Human Research Protection Office (HRPO) reviewed the project and determined that it does not involve activities that are subject to Institutional Review Board (IRB) oversight on 6/11/2024. Therefore, informed consent to participate was waived as it was not considered human subject’s research.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by grant ID 77351603 through AstraZeneca BioPharmaceuticals, US Medical.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Joseph A. Vassalotti, MD: (1) Novo Nordisk, Inc.—consultant US Nephrology Advisory Board in June 2024 on the FLOW trial for semaglutide (Ozempic) impact on nephrology—no speaking role. (2) Sanofi, Inc. OTC Consultant on the oseltamivir (Tamiflu) approach to over-the-counter use of antiviral medication that requires dose adjustment for CKD with low eGFR—no speaking role. Leslie Lake is now with a new institution since completing the project (see Author Note Section).
Data Availability Statement
De-identified, aggregated data from this intervention will be made available upon reasonable request to the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
