Abstract
Objective:
Calculate rates of nirmatrelvir-ritonavir prescription and assess area-level social deprivation as an effect modifier of patient characteristics on nirmatrelvir-ritonavir for the treatment of COVID-19.
Methods:
Retrospective cohort study December 22, 2021 to June 30, 2023 using electronic health record (EHR) data of primary care patients from the American Family Cohort (AFC). Eligible patients had documented COVID-19 between December 15, 2021 and February 28, 2023; ≥1 healthcare encounter between January 1, 2020 and December 14, 2021; and ≥1 healthcare encounter between December 15, 2021 and June 30, 2023. Of the 3 678 474 AFC patients observed during the study period, 86 043 met inclusion criteria. Social deprivation was measured by census tract of patient residence using 2020 Social Deprivation Index (SDI) values; quintiles were calculated using national census tract distribution with lower values indicating less deprivation. The primary statistical analysis estimated the probability of nirmatrelvir-ritonavir prescription using logistic regression. The secondary analysis assessed area-level social deprivation as an effect modifier of patient characteristics on nirmatrelvir-ritonavir prescribing.
Results:
Of the cohort, 26 797 (31.1%) patients were prescribed nirmatrelvir-ritonavir. Asian patients were more likely than White patients to receive a nirmatrelvir-ritonavir prescription in the lowest 2 quintiles of social deprivation (Q1: OR = 1.33 (95% CI = 1.10, 1.59); Q2: OR = 1.29 (95% CI = 1.02, 1.61)). Non-Hispanic/Latino patients in the lowest quintile had 1.32 times the odds of nirmatrelvir-ritonavir prescribing (95% CI = 1.12, 1.56) compared to Hispanic/Latino patients; however, in the third, fourth, and fifth quintiles, non-Hispanic/Latino patients had a reduced odds of a nirmatrelvir-ritonavir prescription compared to Hispanic/Latino patients (Q3: OR = 0.83 (95% CI = 0.73, 0.95); Q4: OR = 0.67 (95% CI = 0.60, 0.75); Q5: OR = 0.67 (95% CI = 0.56, 0.80)).
Conclusions:
Our research suggests intersectionality between patient race/ethnicity with economic status – with meaningful implications for understanding access to new medications within primary care, as they relate to treatment for communicable diseases.
Background
The US Food and Drug Administration (FDA) declared an Emergency Use Authorization (EUA) for nirmatrelvir and ritonavir tablets co-packaged for oral use on December 22, 2021; issuing authorization for the drug as a treatment of mild to moderate COVID-19 among individuals at risk of severe disease. 1 Distribution of the drug ramped up quickly, with shipment volumes almost quadrupling between March and July of 2022.2,3 However, there were disparities in dispensing rates by level of social vulnerability in the early part of 2022. From April 24 to May 21, 2022, 122 courses were dispensed per 100 000 patients in areas of high social vulnerability compared to 274 zip codes with low social vulnerability. 4 While dispensing disparities by zip-code were largely eliminated by August 2022, it is unclear whether prescribing disparities persisted. Despite this general evidence for underutilization, there have been limited studies to investigate individual and subgroup access to nirmatrelvir-ritonavir.5,6
In the context of a pandemic when there is an impetus to respond quickly and equitably, it is important to consider demographic risk factors and their interactions that may influence health outcomes.
The current study had 3 principle aims: (1) calculate prescription rates of nirmatrelvir-ritonavir in primary care from approval of the EUA in December 22, 2021 to June 30, 2023; (2) calculate prescription rates among subgroups defined by patients’ clinical and demographic characteristics; and (3) evaluate whether area-level social deprivation modifies the relationship between patient demographic characteristics and nirmatrelvir-ritonavir prescription. Data for the study come from the American Family Cohort (AFC), a research dataset of electronic health records (HER) derived from the PRIME Registry, a Qualified Clinical Data Registry for primary care (AFC RIF OMOP, v2.0).10,11
Methods
We conducted a retrospective cohort study. Eligible clinical practices contributed data to the PRIME Registry for the entire study period, January 1, 2020 to June 30, 2023; have evidence of nirmatrelvir-ritonavir prescribing; and 10 or more patients with a documented COVID-19 diagnosis as evidence of sufficient patient volume. Patient eligibility criteria included an episode of COVID-19 between December 15, 2021 and February 28, 2023; 1 or more primary care visits between January 1, 2020 and December 14, 2021 in order to assess underlying medical conditions and 1 or more visits between December 15, 2021 and June 30, 2023 to ensure active seeking healthcare at the clinic during the study period. There were 855 active practices within AFC during the study period and 3 678 474 patients (Supplemental Figure 1). Of those, 86 043 patients at 326 practices met inclusion criteria.
We used diagnosis codes to identify documented COVID-19 (Supplemental Table 1). All COVID-19 diagnoses between January 1, 2020 and December 14, 2021 were considered prior to the study period and are noted as a history of COVID-19. We used drug codes to identify nirmatrelvir-ritonavir prescriptions for all patients between December 22, 2021 and June 30, 2023 inclusive (Supplemental Tables 2 and 3–6). Patient-level characteristics including gender, race, date of birth, Hispanic/Latino ethnicity, and census tract of residence were extracted from the EHR. Social deprivation was calculated using the Social Deprivation Index (SDI), an area-level measure which summarizes 7 socioeconomic indicators into a composite value using U.S. Census, American Community Survey data. 12 The SDI values for the current analysis were calculated for each US census tract using 2020 American Community Survey data. 13 Quintiles of SDI were calculated using the national distribution as a reference and then each patient was assigned a quintile based on their census tract SDI value.
The observation period for underlying medical conditions was 2020-01-01 to 2021-12-14. We assessed conditions thought to increase risk of severe COVID-19 and/or post-COVID conditions informed by previously published literature. 14 Risk categories combined patient age and number of underlying medical conditions: age ≥50 years and ≥2 conditions was considered “severe risk,” age <50 years with 0 conditions was “low risk,” and remaining combinations were considered “moderate risk.” We fitted a multivariable logistic regression model using robust standard errors clustered by clinical practice and adjusted for patient-level and practice-level characteristics.
Results
Of 86 043 patients with COVID-19, 31.1% received a prescription for nirmatrelvir-ritonavir; (Table 1). Patients prescribed nirmatrelvir-ritonavir were 9.7 years older on average, were more likely to be Asian or White, and less likely to be American Indian or Native Alaskan, Black, or Native Hawaiian and other Pacific Islander compared to patients not prescribed the drug. Underlying medical conditions were observed in 62.1% of patients. The patients were distributed across 326 practices located in 41 US states and spread across the US Census regions: 50 practices in the Midwest, 34 in the Northeast, 190 in the South, and 52 in the West. The states with the largest number of practices were Texas (n = 48), California (n = 22), Florida (n = 21), Oklahoma (n = 20), and Virginia (n = 20).
Demographic Characteristics of Primary Care Patients With COVID-19 by Nirmatrelvir-ritonavir Prescription Status.
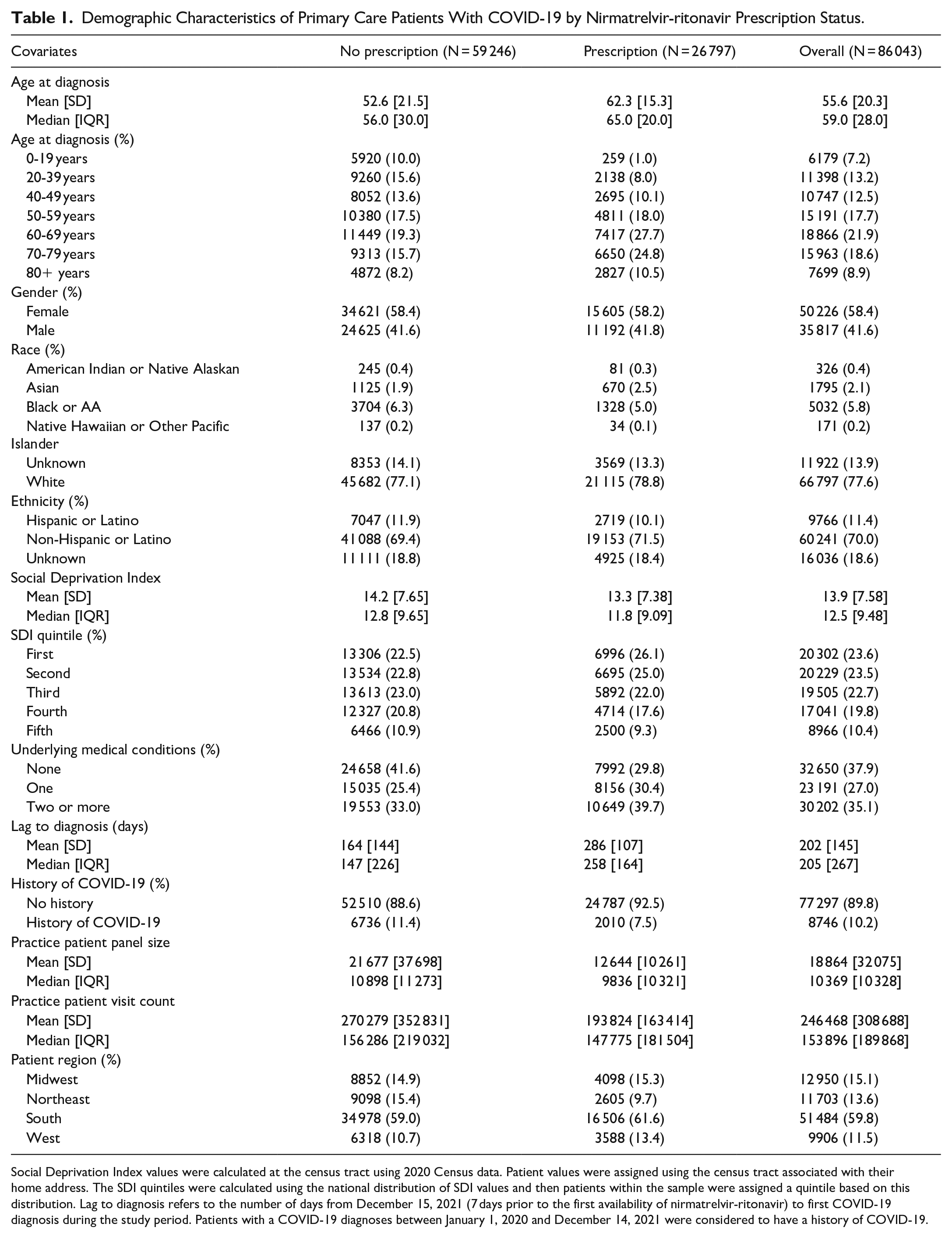
Social Deprivation Index values were calculated at the census tract using 2020 Census data. Patient values were assigned using the census tract associated with their home address. The SDI quintiles were calculated using the national distribution of SDI values and then patients within the sample were assigned a quintile based on this distribution. Lag to diagnosis refers to the number of days from December 15, 2021 (7 days prior to the first availability of nirmatrelvir-ritonavir) to first COVID-19 diagnosis during the study period. Patients with a COVID-19 diagnoses between January 1, 2020 and December 14, 2021 were considered to have a history of COVID-19.
Rates Stratified by Social Deprivation
Rates of nirmatrelvir-ritonavir prescribing decreased within race, ethnicity, and comorbidity subgroups as social deprivation increased (Figure 1). That is, prescription rates were higher among quintiles with greater affluence. However, there were notable exceptions in the pattern. Among American Indian or Native Alaskan patients, there were 188.2 more prescriptions per 1000 patients in the fourth quintile compared to the second. Among Black/AA patients, rates were clustered around 265 prescriptions per 1000 patients with overlapping confidence intervals across all quintiles. Among Native Hawaiian and other Pacific Islander patients, rates ranged from 142.9 in the third quintile to 282.1 in the second; however, confidence intervals across all quintiles overlapped. For Hispanic/Latino patients, the relationship was reversed, such that the lowest rate 242.6 (95% CI = 216.3, 269.0) was observed among the first quintile while the highest rate 296.2 (95% CI = 279.4, 313.0) was observed among the fifth quintile.

Nirmatrelvir-ritonavir prescriptions per 1000 primary care patients with COVID-19 stratified by Social Deprivation Index (SDI) quintile, December 2021 to June 2023.
Rates by Time and Social Deprivation
Rates of nirmatrelvir-ritonavir prescriptions across the study period followed common trends across all SDI levels (Figure 2). Both raw counts and rates were generally less than 100 prescriptions per 1000 patients until mid-March 2022. We observed differences by SDI quintile in the time to initial ramp up to 500 or more prescriptions per 1000 patients. Rates among all quintiles initially surpassed 400 in May 2022. However, rates surpassed 500 in the first SDI quintile the week of June 12, 2022 followed by the third quintile the week of June 19, 2022. Compared to the first quintile, reaching a rate of 500 prescriptions was 4 weeks later for the second quintile, 28 weeks later for the fourth quintile, and 15 weeks later for the fifth quintile.

Weekly prescription frequencies (left
Odds Ratios of Nirmatrelvir-Ritonavir Prescription
The estimated odds of nirmatrelvir-ritonavir after adjustment for patient and practice criteria were largely consistent with the bivariate comparisons described above (Table 2). Patients 20 to 49 years had 0.65 (95% CI = 0.62-0.68) times the odds of nirmatrelvir-ritonavir while patients ≥65 years had 1.11 (95% CI = 1.06-1.15) times the odds compared to patients 50 to 64 years of age. Asian patients had 1.22 times the odds (95% CI = 1.09-1.36) and Native Hawaiian and other Pacific Islanderpatients had 0.57 times the odds (95% CI = 0.36-0.91) compared to White patients. Non-Hispanic/Latino patients were less likely to be prescribed nirmatrelvir-ritonavir than Hispanic/Latino patients (0.84, 95% CI = 0.80-0.89). Patients with 1 comorbidity had 17% increased odds of prescription compared to patients without comorbidities; however, the odds between patients with 2 or more comorbidities and no comorbidities did not differ statistically. All social deprivation quintiles compared to the first quintile had reduced odds of nirmatrelvir-ritonavir prescription ranging from 0.90 (95% CI = 0.86-0.94) in the second quintile to 0.70 (95% CI = 0.67-0.74) in the fourth quintile. Finally, for each 1 standard deviation increase in days from the drug EUA to the COVID-19 diagnosis, the odds of nirmatrelvir-ritonavir increased (2.41, 95% CI = 2.37-2.45); this finding is consistent with the descriptive rates by time (Figure 2).
Multivariable Analysis for Receiving a Nirmatrelvir-ritonavir Prescription Amongst Primary Care Patients With COVID-19 (n = 86 043), December 2021 to June 2023.

Social Deprivation Index values were calculated at the census tract using 2020 Census data. Patient values were assigned using the census tract associated with their home address. The SDI quintiles were calculated using the national distribution of SDI values and then patients within the sample were assigned a quintile based on this distribution. Lag to diagnosis refers to the number of days from December 15, 2021 (7 days prior to the first availability of nirmatrelvir-ritonavir) to first COVID-19 diagnosis during the study period. The lag variable was centered and scaled upon inclusion in the statistical model. Patients with a COVID-19 diagnoses between January 1, 2020 and December 14, 2021 were considered to have a history of COVID-19.
Effect Modification by Social Deprivation
We observed evidence that area-level social deprivation modified the effect of patient and practice characteristics on nirmatrelvir-ritonavir prescription (Table 3). Within social deprivation quintiles, there were no statistically significant differences in the odds of prescription by race group except among the first and second quintiles, Asian patients were more likely than White patients to be prescribed nirmatrelvir-ritonavir (Q1, OR = 1.33, 95% CI = 1.10, 1.59; Q2, OR = 1.29, 95% CI = 1.02, 1.61). For ethnicity, the association was reversed across quintiles of SDI such that non-Hispanic/Latino patients in the first quintile had 1.32 times the odds of a nirmatrelvir-ritonavir prescription (95% CI = 1.12, 1.56) compared to Hispanic/Latino patients; however, in quintiles 3 to 5, non-Hispanic/Latino patients had a reduced odds of nirmatrelvir-ritonavir compared to Hispanic/Latino patients (Q3, OR = 0.83, 95% CI = 0.73, 0.95; Q4, OR = 0.67, 95% CI = 0.60, 0.75; Q5, OR = 0.67, 95% CI = 0.58, 0.77). Time from EUA to diagnosis was statistically significant across all SDI quintiles; however, the strength of the relationship was greater at higher levels of deprivation. For each standard deviation increase in the lag, the odds of prescription was 2.28 (95% CI = 2.21, 2.36) within the first quintile compared to 2.76 (95% CI = 2.62, 2.91) the fifth quintile.
Odds of Receiving a Nirmatrelvir-ritonavir Prescription Among Primary Care Patients With COVID-19 (n = 86 043) Stratified by Social Deprivation Index quintile, December 2021 to June 2023.
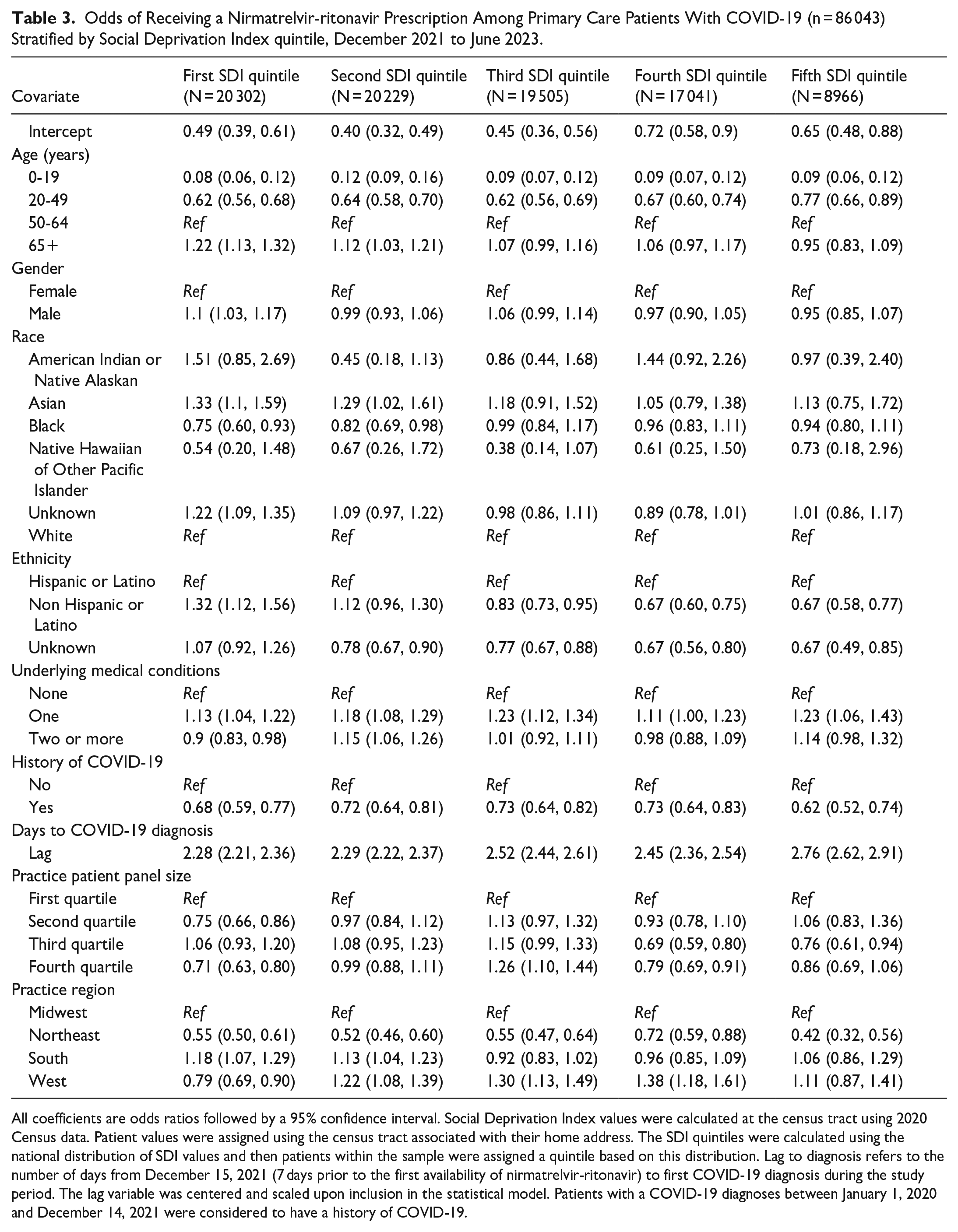
All coefficients are odds ratios followed by a 95% confidence interval. Social Deprivation Index values were calculated at the census tract using 2020 Census data. Patient values were assigned using the census tract associated with their home address. The SDI quintiles were calculated using the national distribution of SDI values and then patients within the sample were assigned a quintile based on this distribution. Lag to diagnosis refers to the number of days from December 15, 2021 (7 days prior to the first availability of nirmatrelvir-ritonavir) to first COVID-19 diagnosis during the study period. The lag variable was centered and scaled upon inclusion in the statistical model. Patients with a COVID-19 diagnoses between January 1, 2020 and December 14, 2021 were considered to have a history of COVID-19.
Discussion
The current study extends previous research by observing nirmatrelvir-ritonavir prescription rates over 18 months and examining the impact of social deprivation alongside patient and practice characteristics. Earlier studies focused on the first year of the drug’s availability, with a 34.3% prescription rate at 30 National Patient-Centered Clinical Research Network sites from January to July 2022, while our study found a lower rate of 19.9% during the same period, mainly due to less than 50 prescriptions per 1000 patients in the first 3 months of 2022. A Veterans Affairs study reported a 12.2% prescription rate for January to December 2022, which is less than half of our estimate of 32.9%. Despite these differences, our findings align with previous studies in showing that prescription rates increase with age and comorbidities, consistent with nirmatrelvir-ritonavir guidelines (Supplemental Figures 2 and 3). 15 Our study revealed some nuances with comorbidities; specifically, the modeling showed no statistically significant difference between having 2 or more comorbidities compared to having none. However, there was a difference between having 1 comorbidity and having none, indicating that further research is needed to understand the drivers of this difference.
Current literature shows disparities in nirmatrelvir-ritonavir prescriptions by area-level social deprivation, race, and ethnicity. White patients were more likely to be prescribed nirmatrelvir-ritonavir compared to non-White patients while Hispanic/Latino patients were less likely to be prescribed nirmatrelvir-ritonavir compared to non-Hispanic/Latino patients. Additionally, those in areas of high social deprivation were less likely to be prescribed nirmatrelvir-ritonavir compared to individuals in low deprivation areas.4 -6 In the current study, we further show that social deprivation modifies the relationship of race and ethnicity on the probability of nirmatrelvir-ritonavir prescription. Asian patients have an increased probability of nirmatrelvir-ritonavir prescription compared to White patients in areas of low deprivation, but no differences were observed in areas of higher deprivation. Hispanic/Latino patients in contrast had a reduced probability of nirmatrelvir-ritonavir compared to White patients in areas of low deprivation but an increased probability in areas of high social deprivation. The diversity within the Asian and Hispanic/Latino populations in the United States including within group differences in income, educational attainment, occupation, immigration status, language use, and regional residence may influence health care utilization and health outcomes.16,17
A key limitation of this work is the unknown reasons for the lack of treatment which may include clinical contraindications, referrals to higher level-care, or delays between symptom onset and healthcare visits. Provider factors, such as familiarity with guidelines, may also influence prescribing. Editorial reports indicate provider hesitancy, particularly for patients with more complex medical needs. 18 Finally, unmeasured patient factors, like concerns of rebound effects or perceived risk of severe COVID-19 may have swayed patient decision-making.19, 20 Other limitations include the source data and selection criteria, as only primary care visits are captured, and inclusion depends on patients’ healthcare-seeking behavior. Nonetheless, the estimates reflect the real-world patterns in this frontline care setting.
This work highlights the need for real-time data of drug distribution, dispensing, cases during a pandemic to swiftly identify underutilization and inform strategies to reduce disparities across patient, provider, and practice characteristics. Simultaneously, it raises warning flags for monitoring the equitable distribution of new drugs outside of the pandemic context when dispersion may be slower and the gaps in treatment adoption between communities may be greater.
Supplemental Material
sj-docx-1-jpc-10.1177_21501319251350935 – Supplemental material for An Intersectional Analysis of Social Deprivation and Patient Characteristics on Nirmatrelvir-Ritonavir for Treatment of COVID-19 in U.S. Primary Care Practices, 2021 to 2023
Supplemental material, sj-docx-1-jpc-10.1177_21501319251350935 for An Intersectional Analysis of Social Deprivation and Patient Characteristics on Nirmatrelvir-Ritonavir for Treatment of COVID-19 in U.S. Primary Care Practices, 2021 to 2023 by Esther E. Velásquez, Neil S. Kamdar, Nathaniel Hendrix, Xiaoyu Wang, Robert L. Phillips and David H. Rehkopf in Journal of Primary Care & Community Health
Footnotes
Acknowledgements
None.
Ethical Considerations
This research was reviewed by the Stanford IRB (Protocol IRB-70828, “Assessing the racial, ethnic, and geographic equitability of COVID-19 treatments in a primary care population: 1U01FD007879-01 Chronic disease in the American Family Cohort.”)
Author Contributions
EV drafted the manuscript and completed the statistical analysis. NK contributed to the study design and analysis. NH contributed to the study design and analysis. XW provided statistical code review. RB contributed to the conception of the work. DR contributed to the conception of the work. All authors contributed to interpretation of the data, edited the manuscript, and approve of the version to be published.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by funding from the Food and Drug Administration (Project Number U01-FD007879).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
