Abstract
Introduction:
Basal and intermediate insulin is available as U-100 (glargine, determir, NPH) or ultra-long-acting (ULA) U-200 (degludec) or U-300 (glargine). Insulins may be substituted with other insulin formulations based on financial factors, formulary preferences, patient preference, and patient response.
Objective:
Evaluate the impact on total daily dose of insulin when switching from an intermediate or basal U-100 to a ULA insulin.
Methods:
A single-center retrospective chart review was performed at a federally qualified health center. Patients switched from a U-100 intermediate or basal insulin to a U-200 degludec or U-300 glargine ULA insulin from 2019 to March 2024 were assessed. Clinical measures assessed were initial intermediate or basal insulin total daily dose, ULA insulin total daily dose at time of switch, at 1, 3, and 6 months, and change in HgbA1c, BMI, and weight.
Results:
When switched from a U-100 to a U-200 or U-300 insulin (n = 53), basal insulin total daily dose decreased by 13.1 units at 6 months (P < .05). At the 6-month mark after ULA switch, HgbA1c decreased (P < .001), but BMI (P = .161) and weight (P = .076) were similar. HgbA1c, BMI, weight, and total daily insulin dose were not significantly different between patients assigned U-200 and U-300 insulins.
Conclusion:
Patients switched from a U-100 basal insulin may utilize a lower total daily dose of a ULA insulin and experience reductions in HgbA1c, BMI, and weight.
Introduction
Diabetes in the United States (U.S.) has been on the rise with a recent prevalence rate in 2021 of 38.4 million or 11.6% of the population having any form of diabetes. 1 The Centers for Disease Control and Prevention estimates 5.7% (1.7 million) adults diagnosed with diabetes in the U.S. have type 1 diabetes (T1D) and use insulin. 2 Of individuals with diabetes in the U.S., around 21% receive care at a Federally Qualified Health Center (FQHC). 3 FQHCs provide thorough health care services to underserved communities including indigent, uninsured, and educationally disadvantaged persons through funding from federal grants from the Bureau of Primary Health Care at the Health Resource and Services Administration (HRSA). 4 Services are provided to those insured (Medicare, Medicaid, Children’s Health Insurance Program (CHIP)) and uninsured where patients can be eligible for a reduced cost utilizing a Sliding Fee Scale. 5 Many FQHCs utilize clinical pharmacists to address HRSA measures and expand access to care. 6
Type 1 diabetes is managed with continuous subcutaneous insulin infusions, or basal insulin with multiple doses of prandial insulin. 7 The initial management of type 2 diabetes (T2D) may involve several glucose lowering medications with the primary outcome to reduce the risk of cardiorenal complications and to maintain glycemic and weight management goals. According to the American Diabetes Association (ADA), for the maintenance of glycemic control in patients with T2D there has been high HgbA1c lowering efficacy associated with glucagon-like peptide-1 receptor agonists (GLP-1 RA; ie, dulaglutide, semaglutide, tirzepatide) and insulin. Second line agents include other GLP-1 RA not listed as first line, metformin, sodium-glucose cotransporter-2 inhibitors (SGLT2i), sulfonylureas, and thiazolidinediones while intermediate efficacy has been shown with dipeptidyl peptidase-4 inhibitors. 7
Approximately 8.4 million Americans with diabetes depend on insulin for survival.
8
Insulin has a unique role due to its high efficacy, general tolerability, and use in severe hyperglycemia cases with or without catabolism (weight loss, hypertriglyceridemia, ketosis).
8
According to the ADA, basal insulin can be added alone or as a combination of basal insulin and GLP-1 RA if the T2D patient is still above the HgbA1c target goal after appropriate dosing and time on a GLP-1 RA and already using 3 to 4 oral therapies. T2D patients with symptomatic hyperglycemia (polyuria or polydipsia), a HgbA1c >10%, marked insulin deficiency, blood glucose levels
Patients started on one type of insulin may switch to another for reasons including delivery systems, dexterity, adverse effects, insurance coverage, and other factors. Concentrated insulin offers the ability to inject larger insulin doses with lower volume, potentially improving absorption. Ultra long-acting (ULA) insulins are structured to release insulin more slowly. This increases the duration of action and lowers the peak of action which increases efficacy and safety compared to long-acting insulin. 10 For example, insulin degludec U-200 has a duration of action of 42 h with a peak maximum action at 9 h, glargine U-300 has a duration of action of >24 h with a peak maximum action at 12 to 16 h, glargine U-100 has a duration of action of 10.8 to 24 h with a peak maximum action at 10 to 12 h, determir U-100 has a duration of action of 6 to 23 h with a peak maximum action at 3 to 9 h, intermediate U-100 insulin has a duration of action of 14 to 24 h with a peak maximum action at 4 to 12 h.11 -16 Therefore, conversion to concentrated U-200 or U-300 ULA insulin may result in reduced daily doses needed. Despite this, the package inserts for insulin glargine (U-300) and insulin degludec (U-100 and U-200) recommend switching from a once daily intermediate or long acting insulin at a one to one unit conversion.11,13
There has been proven benefit of switching from an intermediate or long acting insulin to a ULA insulin from clinical trials. Previous studies have supported that the use of ULA insulins compared to long or intermediate acting insulins have reduced the risk of hypoglycemic events in patients with type 1 and 2 diabetes.17 -19 A meta-analysis comparing ULA, long acting, and intermediate acting insulins in patients with type 1 diabetes, concluded that both ULA and long-acting insulin were superior to intermediate acting in reducing HgbA1c, fasting blood glucose, major or serious hypoglycemia, nocturnal hypoglycemia, and weight gain. 18 Additionally, for fasting blood glucose, ULA insulin degludec administered once daily was superior to basal U-100 detemir or glargine administered twice daily. 18 Real-world outcomes on the switch from long acting to ULA insulin is limited to countries outside of the United States where factors impacting glycemic control and response to therapy differ.20 -25 For example, in the US, variability in insurance status, adjustment of other diabetes-related medications, and assessment in patients who experience social determinants of health in an FQHC setting may impact diabetes-related clinical outcomes.
This study’s primary aim is to determine the impact of switching to ULA insulin from U-100 intermediate or basal insulin on total daily insulin dose for FQHC patients with diabetes. The primary outcome is the mean unit change in total daily insulin dose in units between last U-100 basal or intermediate-acting insulin dose and at 6 months after switching to ULA insulin. This study’s secondary aims are to (1) determine the short-term change in total daily insulin dose based on ULA insulin concentration at 1 and 3 months and (2) estimate the impact of ULA insulin concentration on clinical parameters (BMI, weight, and HgbA1c) at 1, 3, and 6 months in FQHC patients with diabetes, and 3) change in number of antihyperglycemic medications at 6 months between the ULA U-200 and U-300 groups and overall after switching from U-100 insulin.
Methods
Study Design
This study was a single site observational cohort study design via retrospective chart review at an FQHC in Texas. According to the HRSA 2022 data, the percentage of patients at this FQHC with diabetes is 24.68% (1305) and patients who have diabetes with poor hemoglobin A1c (HgbA1c) control are 29.04% (379). 5 HRSA defines poor diabetes HgbA1c control as >9%. 5 The study was submitted to the affiliated university’s institutional review board and was approved as exempt from full review (protocol number: STUDY00004561).
Data Source
The principal investigator utilized EPIC Systems® (Version 052023) electronic health record SlicerDicer® (Version 5.2) data exploration and analytical software. A clinical report was pulled to identify patients with an active prescription for ULA insulin degludec U-200 or insulin glargine U-300 from 2016 to March 2024.
Context
The inclusion criteria consisted of patients from a single-site FQHC in Texas who were at least 18 years of age and switched from an intermediate (NPH) or basal U-100 (glargine or detemir) insulin to an ULA U-200 (degludec) or U-300 (glargine) insulin between 2019 and March 2024. Patients could have any form of diabetes that was insulin-treated. Patients may have been seen by their primary care provider for usual care or an ambulatory care pharmacist for diabetes, insulin management, or disease state education via collaborative drug therapy management. Eighty-nine patients were pulled from the initial report, 40 of which were excluded for reasons such as never being prescribed a basal U-100 insulin, insufficient information in the chart, cost barriers that prevented them from starting a ULA insulin, or no prescription history on file (Figure 1). Patients had to have at least one follow up visit at least 1 month after the basal insulin to ULA switch for data collection.

CONSORT diagram for intermediate or basal U-100 to ULA U-200 or U-300 insulin.
Patient demographic data collected from chart review included age, ethnicity, race, sex assigned at birth, medical insurance status, prescription insurance status, and insurance or program used to cover the ULA basal insulin (Table 1). Clinical data points collected from chart review included height, weight, body mass index (BMI), type of U-100 intermediate or basal insulin prior to switch, total daily dose of intermediate or basal U-100 insulin administered prior to switch and the number of injections of intermediate or basal insulin per day, administration of other prescribed antihyperglycemic medications while on U-100 insulin (including metformin, sodium-glucose cotransporter-2 inhibitors, glucagon-like peptide-1 agonists, dipeptidyl peptidase-4 inhibitors, thiazolidinediones, sulfonylureas, and prandial insulin). Given the integrated care between primary care providers and ambulatory care pharmacists, it was also recorded whether the patient had a visit with a clinical pharmacist at any time point when they were switched from a U-100 to a ULA insulin. Weight, BMI and HgbA1c were all assessed prior to the insulin switch and at 1, 3, and 6 months after switch. The number of antihyperglycemic medications were collected at the last U-100 insulin dose and 6 months after switching to a ULA basal insulin, U-200 (degludec) or U-300 (glargine). Additional data collected included the type of U-200 or U-300 ULA basal insulin at the time of switch, total daily dose of ULA insulin at time of switch and at 1, 3, and 6 months after the switch. Time points were specific and important to allow for adequate time to analyze laboratory and clinical metabolic changes.
Patient Demographics for U-100 to Ultra-Long-Acting (ULA) Insulin Switch.

Statistical Analysis
A 1-sample, 2-sided test for mean power analysis was performed and found a sample of 34 patients was required to achieve 80% power with an effect size of 0.5 (medium effect) with a significance of P = .05. 26 The effect size estimate of 0.471 categorized as “small” (Cohen’s d 0.2 to <0.5), albeit at the upper limit of this category. The primary outcome was analyzed using a 1-sample Kolmogorov-Smirnov test with Lilliefors correction and the data was found to be normally distributed (P = .170). 27 A 2-sided, paired t-test was then used to analyze the primary outcome. Missing values were replaced using linear interpolation and which had no impact on the mean (from 54.902 to 54.902, P = 1.0).28,29 The secondary outcome measures’ distributions for each group were subjected to the same test of distribution and none were found to have both sets be normally distributed for these measures. Secondary outcomes were analyzed using a McNemar test for paired categorical data related to cohort overall and a Mann–Whitney U test when comparing U-200 and U-300 insulin as independent samples. 28 Linear regression was performed to estimate the correlation of demographic predictors on change in the primary outcome. Descriptive statistics were also used. Analysis was performed using IBM SPSS Statistics version 29.0.1.1 (244) and descriptive statistics and charting were performed using Microsoft Excel version 16.86 (24060916).
Results
The chart review yielded 53 eligible patients yielding a power of 94.7% to detect an effect size of 0.5 with a significance of P = .05. An effect size of 0.5 was chosen given it is standard for moderate effect of the outcome.30,31 Of the 53 patients included, 31 (58.5%) were switched to U-200 insulin and 22 (41.5%) were switched to U-300 insulin (P = .216). Patients had a significantly more likely to be white (75.5%, P < .001), female (64.2%, P < .001), had a HgbA1c of 9% or greater at last U-100 visit (62.2%, P < .001), and had met with a clinical pharmacist for insulin management after the switch to ULA insulin (88.7%, P < .001; Table 1). Most patients also identified as “Hispanic or Latino” but not significantly so (69.8%, P = .054). One patient (2%) had T1D, and the remainder, 52/53 (98%), had T2D. Insurance varied for medical, pharmacy, and ULA coverage and included commercial insurance, Medicare, or income-based programs such as patient assistance programs, family assistance programs, and 340B programs. Additionally, few patients were using more than 2 injections of intermediate or basal U-100 insulin before the switch (1.9%, P < .001) and most patients were using no bolus insulin (66%, P < .001).
Primary Outcome
For the primary outcome of change in TDD of insulin at 6 months from last U-100 dose, patients experienced a mean change of −13.1 units (P = .024, d = .471; Table 2). Comparisons in total daily dose overall and between U-200 and U-300 insulin users and mean changes in total daily dose overall between intervals are available in Figure 2. The average TDD for intermediate or basal U-100 insulin in all patients was 68 units per day. When divided out by insulin type, the mean U-100 dose prior to switching to U-300 and U-200 was 72 and 65.16 units, respectively. On switching from U-100 to a ULA insulin, patients’ doses were initially decreased, and then titrated based on response. At 6 months, the average TDD of all patients had decreased from 68 to 54.9 units. The U-300 group’s TDD had decreased from 72 to 57.06 units. The U-200 group’s TDD decreased from 65.15 to 53.53 units.
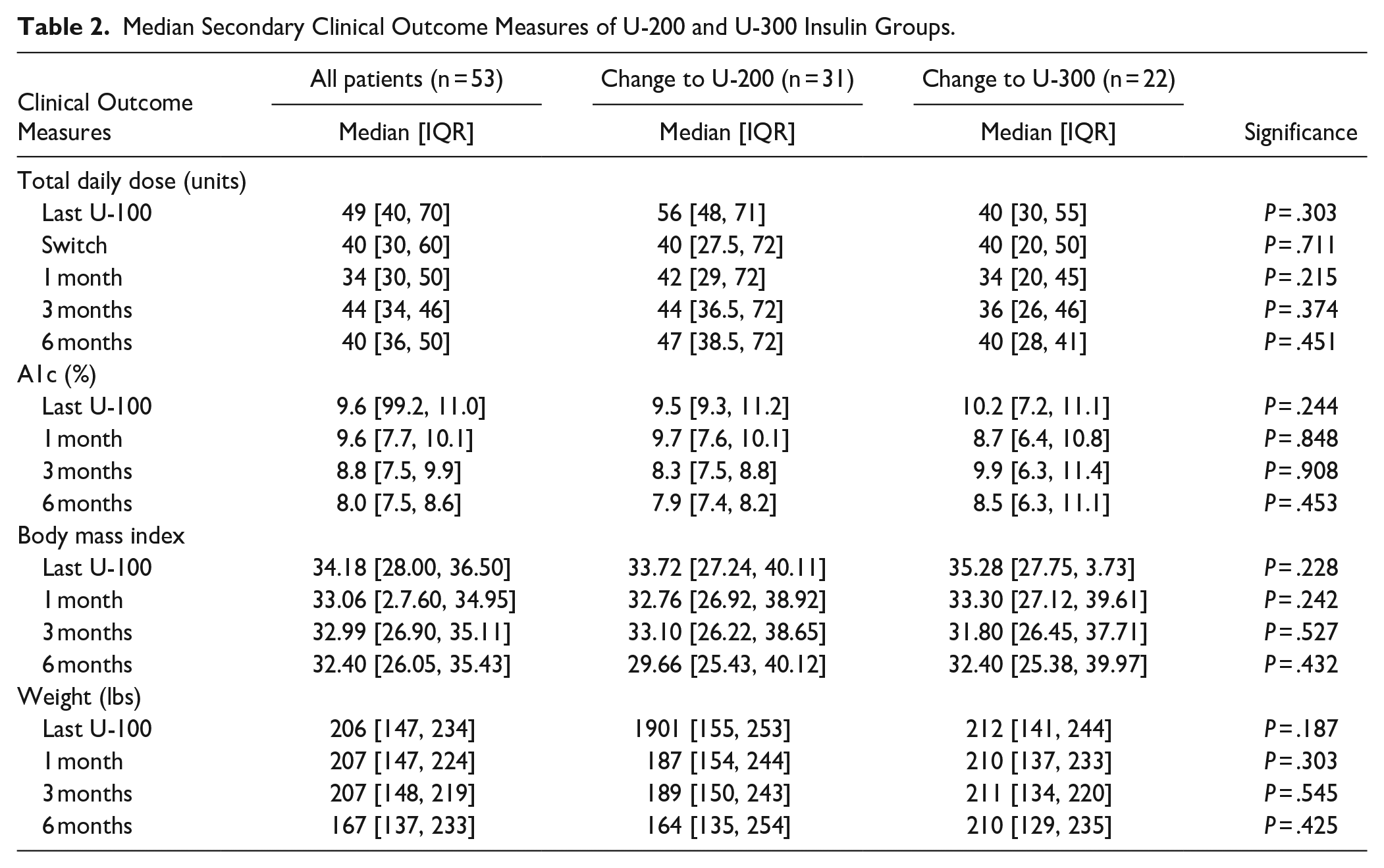
Median Secondary Clinical Outcome Measures of U-200 and U-300 Insulin Groups.

Six-month change in mean total daily dose after switching to ULA insulin.
Secondary Outcomes
Secondary outcomes included change in BMI, HgbA1c, and weight from last U-100 visit to 6 months after the switch to ULA insulin. Overall means changed for HgbA1c by −1.35% (P < .001), BMI by −2.64 (P = .161), and weight by −20.10 lbs (P = .076). Additionally, patients using U-200 and U-300 insulin were compared and no differences were found between patients based on insulin concentration for total daily dose, HgbA1c, BMI, nor weight (Table 2).
Other antihyperglycemic agents were adjusted based on usual care at patient visits. Change in patients using metformin decreased by 28.3% (P = .016) and sulfonylurea use decreased by 1 patient. No other significant changes in other antihyperglycemic agent use were observed (Figure 3). The linear regression model using demographic predictors (ethnicity, race, sex, pharmacist visit, medical insurance, pharmacy insurance, ULA coverage, u100 insulin type, U-100 insulin product, and HgbA1c over 9%) showed poor correlation with change in total daily ULA insulin dose with an adjusted R2 of 0.96 (P = .294).

Change in percentage of patients using additional antihyperglycemic agents.
Discussion
When patients were switched from an intermediate or basal U-100 insulin to a ULA insulin, their TDD of insulin decreased by approximately 13 units per day. The power analysis based on a moderate effect size detects a difference of moderate magnitude should it exist. While the sample size was appropriate, the small effect size suggests that the magnitude of the difference is small but is distinctly present. The decrease in insulin utilization was consistent and observed in both ULA U-200 degludec and U-300 glargine groups which suggests that while switching to a ULA insulin will decrease total daily insulin usage, specific ULA insulin product chosen will not impact this usage. These findings differ from other studies which found a slight increase in overall insulin (basal and bolus) utilization after switching from a basal U-100 to ULA degludec. 21 In Thewjitcharoen’s study, ULA insulin utilization decreased by 1 unit, from 15 to 14 units per day. 21 Other studies have shown only a decrease in basal insulin utilization when switching from a twice daily U-100 to a once daily degludec. 20 This directly impacts patient care as most package inserts recommend a one-to-one conversion when switching from a U-100 basal insulin to a ULA insulin. Findings from our study indicate that utilizing a one-to-one conversion may unintentionally increase the risk of hypoglycemia in patients when switched from an intermediate or basal U-100 to a ULA (U-200 or U-300). This may also be beneficial to patients in FQHC settings who utilize patient assistance programs, 340B medication pricing, commercial insurance, Medicare, or Medicaid to mitigate insulin rationing due to dose changes and cost. 33
Overall, this study showed a statistically significant decrease in HgbA1c when switched from an intermediate or basal U-100 to the ULA insulin. This is clinically significant if substantial HgbA1c reduction is needed in patients with diabetes whose intermediate or basal U-100 insulin regimen is not sufficient to reach HgbA1c goals and prevent diabetes-related complications. Use of ULA insulin also allows an opportunity for decreased injection burden if the patient is injecting the U-100 insulin more than once per day. Reduction of HgbA1c after switching to a ULA is consistent with other studies.18,20 -22,24 Additionally, studies have shown benefit in cardiovascular risk reduction and decrease in severe hypoglycemia events when patients’ intermediate acting insulin was changed to a long acting or ULA insulin.18,33 While not directly assessed in this study, based on previous findings, changing an intermediate acting U-100 insulin to a ULA insulin may benefit patient cardiovascular outcomes and decrease the risk of severe hypoglycemia.
A common side effect of long-term insulin use is weight gain. In this study, when patients were switched from a U-100 to a ULA insulin, BMI and weight decreased overall. Overall, patients experienced a mean weight loss of approximately 20 pounds, which may also lead to improvement in other metabolic parameters; however, there was high variability in weight loss or gain among the group, so the results were not statistically significant due to the high variance in change in weight. Other studies have shown either no significant weight change22,24,25 or modest weight gain or loss for type 1 diabetes and type 2 diabetes, respectively when switching from a basal U-100 to an ULA U-200 or U-300 insulin. 21 The pharmacist-led lifestyle counseling and medication adjustments may have also contributed to the change in weight for patients; however, this was not specifically assessed in this study. Most patients met with a clinical pharmacist (88.7%) at the clinic where diabetes medications could be adjusted based on patient response as a component of usual care via collaborative drug therapy management. Pharmacist-led visits were typically 30 to 60 min, in person or via telehealth, every 2 to 4 weeks based on patient or provider preference or availability, and included patient-specific counseling on diet, physical activity, medication adherence, medication side effects, blood glucose monitoring, and other concerns. For patients on high doses of U-100 intermediate or basal insulin who desire weight loss, switching to a ULA may be a reasonable consideration, though patient specific factors and integration of collaborative care with clinical pharmacy services may impact change in weight.
A statistically significant decrease in metformin and sulfonylurea use was observed at the 6-month mark. Diabetes guidelines previously considered metformin first-line therapy for the treatment of type 2 diabetes in most patients; however, with newer drug classes that have cardiovascular and renal benefit, clinicians are encouraged to initiate pharmacotherapy based on patient-specific factors and comorbidities. 7 This may impact the utilization of metformin in practice as patients may have additional comorbidities that prioritize agents with cardiovascular or renal benefit such as SGLT2i or GLP RAs. 7 This may explain the decrease in metformin and sulfonylurea use in this study. Patients with diabetes may take many medications for diabetes, and associated conditions such as dyslipidemia as primary or secondary prevention, and hypertension. Decreasing pill burden may assist patients with diabetes distress related to taking medications. Lowering the pill burden and injection burden may also minimize undesirable side effects from antihyperglycemic medications, decrease medication copays, and improve medication adherence.
Limitations include a small sample size from the FQHC despite this being the full report of patients on ULA. The effect size of the primary outcome was “small” but may be higher in the population given a decrease in standard deviation. Larger samples with more complex sampling techniques from multiple institutions are needed to determine a more global impact. Additionally, most patients who were able to access the ULA insulins had commercial insurance, Medicare, or Medicaid, which may allow them to have access to additional healthcare related resources compared to patients without access to insurance. While some patients without insurance were able to utilize manufacturer patient assistance programs, not all patients without insurance meet the manufacturer’s eligibility criteria to receive the medications. Hypoglycemia was not formally quantified in this study as patients received usual care from primary care providers or clinical pharmacists who assessed hypoglycemia in the visit and made adjustments based on patient-specific factors and clinical judgment. Adherence was not formally assessed in this study due to challenges including lack of reliable data related validated adherence tools (ie: proportion of days covered (PDC), medication possession ratio (MPR), or Morisky Medication Adherence Scale-8 (MMAS-8)), potential patient self-reporting bias, and lack of documentation due to frequent dose changes or appointment time constraints. The retrospective methodology of this study also introduces selection bias and allows for potential confounders. The investigators attempt to mitigate selection bias were performed through demographic collection and a comparison between agents U-200 and U-300. The demographics were subjected to a linear regression analysis for their impact on the primary outcome to capture the potential impact of confounding demographic variables. Other potential confounders not evaluated include comorbid disease states such as hypertension and cardiovascular disease. Additionally, the observational cohort study design creates challenges such as lack of randomization, selection bias and confounders for self-selection, and the lack of a uniform study period all increase the risk for error.
Future directions include evaluating these clinical outcomes in a larger sample size, other types of health systems in addition to FQHCs, and more equitable access to ULA insulins. A cost analysis may be helpful in future studies to assess the implications of expanding access to ULA insulins in underserved populations given the results of this study. Additional studies with the resources to enroll more FQHC patients at multiple sites may provide insight into the qualities of ULA insulins that are driving the TDD reduction and improvement in clinical outcomes among patients at FQHCs. A multivariate analysis can simultaneously investigate many factors and their relative contribution to these outcomes in the context of one another.
Conclusion
Switching patients’ intermediate or basal U-100 insulin to a ULA insulin decreased total daily dose of insulin utilization at 6 months with overall reduction in HgbA1c, BMI, weight, and pill burden in patients at this FQHC. The concentration of ULA insulin (U-200 degludec or U-300 glargine) did not impact these clinical outcomes. This is meaningful as clinicians adjust insulin doses to minimize the risk of hypoglycemia. Utilization of ULA insulin in underserved populations may improve diabetes-related clinical parameters as shown in this preliminary data. Further studies examining the impact of switching to ULA insulin from U-100 insulin on a larger scale with multivariate analysis or randomized controlled trial in FQHC populations can provide further insight into the factors impacting clinical outcomes.
Footnotes
Author Contributions
Natalie Rosario: Supervision, Project administration, Methodology, Investigation, Data curation, Conceptualization, Writing—Original draft, Writing—Review & editing. Cami Hunger: Investigation, Data curation, Writing—Original draft. Allison Pettijohn: Investigation, Data curation, Writing—Original draft. K’bria Whaley: Investigation, Data curation, Writing—Original draft. Andrea Pabon: Investigation, Data curation, Writing—Original draft. Valeria Hohl: Investigation, Data curation, Writing—Original Draft. Bernadette Asias-Dinh: Conceptualization, Writing—Review & editing. Jodie Gee: Conceptualization, Writing—Review & Editing. Joshua Wollen: Methodology, Data curation, Resources, Formal Analysis, Writing—Original draft, Writing—Review & editing.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Joshua Wollen is a Board of Directors member at Vecino Health Centers where the study took place.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
