Abstract
Osmotic demyelination syndrome is a rare condition reported mainly in the case of rapid correction of hyponatremia, but it can occur even in the case of complicated diabetes mellitus either during rapid correction of hyperglycemia or anytime during the complicated diabetes mellitus. We report a case of complicated diabetes mellitus developing osmotic demyelination syndrome. The patient had presented with altered sensorium and seizure, which was initially diagnosed as hyperglycemia, but during his treatment, the magnetic resonance imaging of brain revealed central pontine myelinolysis. Our search on the causes of osmotic demyelination syndrome other than rapid correction of hyponatremia has revealed several other causes like autoimmune liver disease, Sjogren’s syndrome and non-Hodgkin’s lymphoma in addition to diabetes mellitus.
Keywords
Introduction
Osmotic demyelination syndrome (ODS) is non-inflammatory demyelination of neurons due to apoptosis of oligodendrocytes and infiltration of myelin degrading macrophages. Based on location of demyelination, ODS is classified into central pontine myelinolysis (CPM) and extrapontine myelinolysis (EPM).1,2 ODS is a rarely reported disease, and studies have shown its prevalence of about 0.06% among all hospitalized patients. Despite being rare, it is a serious issue which can even lead to severe disability or death. ODS is usually reported in cases of chronic alcohol use, alcohol withdrawal and rapid correction of hyponatremia. 1 ODS in case of diabetes mellitus (DM) has not got as much concern in literature as in case of hyponatremia although DM seems to be a significant cause of ODS. We report a rare case of ODS with CPM which has complicated DM as underlying cause.
Case description
A 24-year-old male, who is a known case of type II DM for 5 years, presented with loss of consciousness, abnormal behaviour (shouting and aggressiveness), four episodes of seizure and altered sensorium for 5 days. The first seizure episode was of sudden onset of jerking of upper and lower limbs with loss of consciousness that occurred 5 days back while he was lying on bed and talking with his family members. There was up rolling of eyes with frothing of saliva; however, no tongue bite or incontinence was found. Patient’s consciousness was regained after 10 min, but drowsiness persisted for about 30 min. A similar second seizure episode repeated next day with patient remaining unconscious for about 40 h for which he was admitted to nearby hospital. When patient woke up on the fourth day of symptoms onset, he still had altered sensorium. He had two further attacks of seizures on the fourth and fifth days. Patient had three episodes of vomiting which was non-projectile, associated with nausea. At local hospital, the patient was treated for 4 days with intravenous insulin, potassium supplement, intravenous anticonvulsant (Sodium Valproate 500 mg and Levetiracetam 500 mg twice a day) and intravenous antibiotics (Ceftriaxone 2 g twice a day). Then, patient was referred to our centre for further management. There was no history of bowel or bladder incontinence. He was under insulin for 2 years with poor compliance and had missed his recent insulin dose for 1½ months. Family history of diabetes was only positive in his grandmother. There was no prior history of electrolyte imbalance, correction of hyponatremia, liver transplantation, alcohol use or malnutrition. On general examination, he was ill-looking and fatigued. Body built was average with body mass index of 23.5 (weight 70 kg and height 1.73 m). His vitals were normal with Glasgow coma scale of 15. 3 Cranial nerve functions were intact, except with mild slurring of speech and action tremor in both hands. However, patient had no difficulty holding things in hands or feeding by himself. Bulk, tone and power of all limb muscles were 4+/5 according to Medical Research Council (MRC) 4 power grading. Sensory examination showed no abnormal findings, and there was no sign of cerebellar abnormalities. Deep tendon reflexes were brisk in all the joints with bilateral plantar extensor response. Patient could walk without support but was unable to walk at a fast pace.
His previous reports showed serum sodium concentration had increased from 140 to 152 mEq/L in the first 3 days in the local hospital. Serum glucose and electrolytes returned to normal range after getting care from our centre, and no significant fluctuation in these parameters were seen during the patient’s stay in the hospital. Serum biochemical parameters of the patient at the time of admission in our centre are given in Table 1, which shows increased sodium level up to 156 mEq/L.
Serum biochemical parameters at the time of admission.
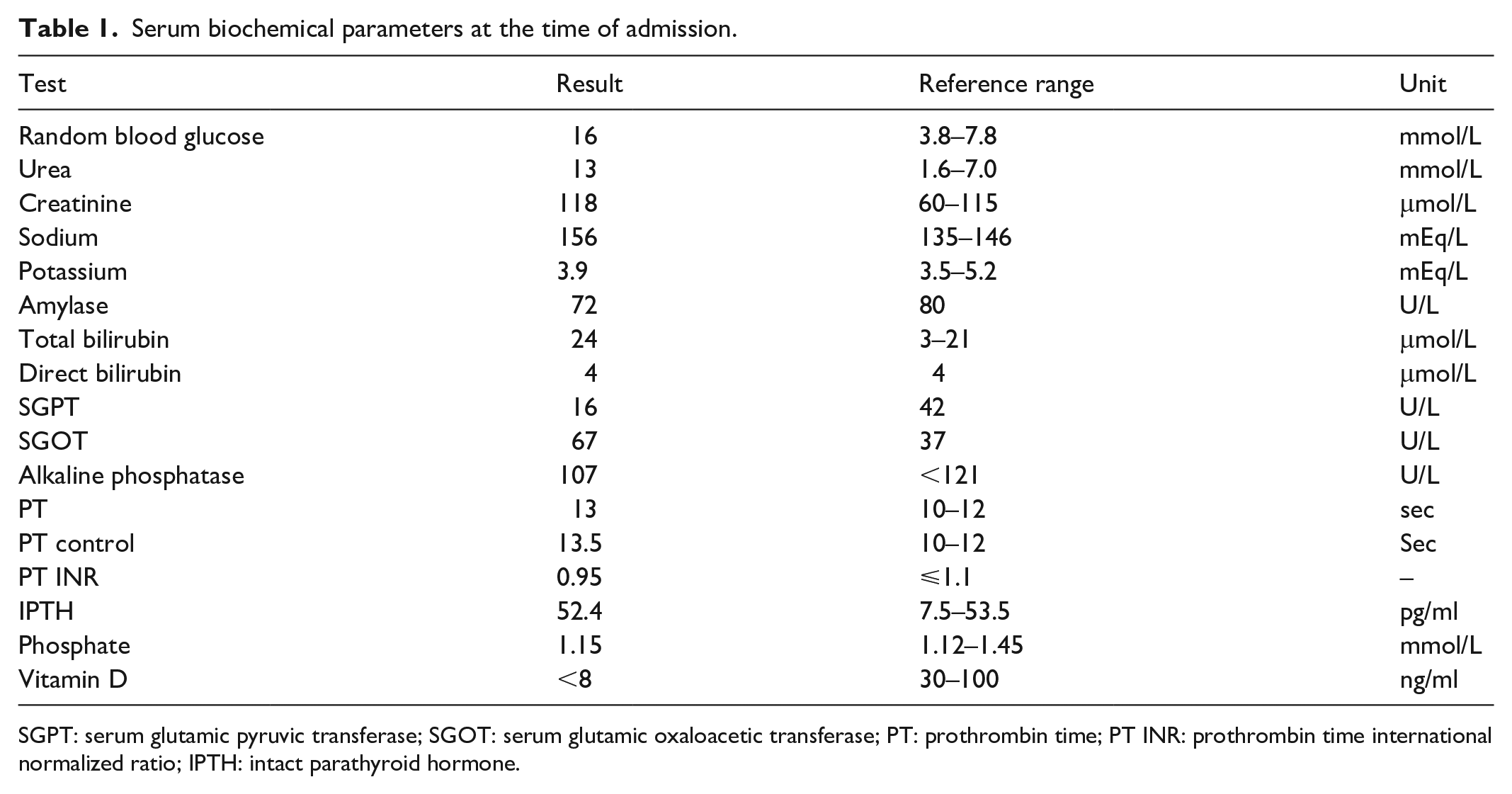
SGPT: serum glutamic pyruvic transferase; SGOT: serum glutamic oxaloacetic transferase; PT: prothrombin time; PT INR: prothrombin time international normalized ratio; IPTH: intact parathyroid hormone.
After admission, patient was managed symptomatically for high blood sugar level, that is, 16 mmol/L and seizure. Intravenous 1 g levetiracetam was administered, normal saline was infused at a rate of 100 mL/h and subcutaneous insulin under basal bolus regimen insulin was administered. Furthermore, patient was supplemented with 500 mg intravenous thiamine per day along with weekly 60,000 units of oral vitamin D. Diabetic retinopathy screening and ocular coherence tomography revealed bilateral severe proliferative diabetic retinopathy with neovascularization of the iris. Pan-retinal photocoagulation was planned for the treatment of proliferative diabetic retinopathy. Ultrasonography of abdomen and pelvis revealed no significant abnormalities.
No abnormalities were noted in plain computed tomography (CT) of brain, which was done 4 days prior to visiting our centre. Brain magnetic resonance imaging (MRI) was done on third day of admission at our centre which showed hyperintensity area in central pons (Figure 1) and right crus of midbrain (Figure 2) in T2 and Flair sequences suggestive of demyelination. Correlating clinical scenario and neuroimaging findings, diagnosis of ODS with CPM was made. With supportive management, patient gradually improved to the normal state with no limb weakness or fatigue or slurring of speech, and he was discharged after 21 days of hospital stay. In 1-month follow-up, patient was fine with no deficits, and his both fasting and postprandial sugar levels were normal. However, deep tendon reflexes were brisk in all the joints and bilateral plantar had extensor response.

Hyperintensity centrally and right anterolateral aspect of the pons in axial FLAIR sequence (right) and T2 weighted MRI (left).

Hyperintensity in right-side midbrain tectum in axial flair sequence (right) and T2 weighted MRI (left).
Discussion
In previous studies, chronic alcohol use and alcohol withdrawal were reported being the main cause of ODS. Recent studies have revealed hyponatremia followed by its rapid correction to be the most common cause of ODS although any kind of electrolyte imbalance can be the cause of ODS. 1 Liver transplant recipients are at the highest risk of developing ODS. 5 In our patient, there was no history of chronic alcohol consumption, correction of hyponatremia and liver transplantation. In our case, a rise in serum sodium was noted from 140 to 152 while the patient was admitted in previous centre. Despite hyperglycemia, the increment in sodium could be due to less intake or supplement of free water to the patient then. Occurrence of ODS having complicated DM as its underlying cause is rare, and we could identify only 16 such cases reported in English literature till date.6–21 In a recent case report by Kusumoto et al., 6 malnutrition and severe illness with fluctuations in osmolarity were also considered risk factors for onset of ODS which was ruled out in our case.
Demyelination involves region-specific oligodendrocytes, that is, oligodendrocytes of pons, basal ganglia, mesencephalon and deep cortical layers. The glial cells of ODS-resistant brain regions are not affected by change in osmolarity. 2 Spectrum of clinical features varies among patients of ODS and are associated with the anatomy of the lesion. 22 Dysphagia, dysarthria, hemiplegia or paraparesis or quadriparesis, decreased deep tendon reflexes, ataxia, altered mental status, confusional state, and behavioural changes like agitation and disorientation were noticed in the patients developing CPM in case of complicated DM.6,8–10,13,15,17–20 Paroxysmal painful spasms have also been reported in a case of ODS related to non-ketotic hyperglycemia. 23
Diagnosis of ODS is confirmed by T2 and T2 FLAIR sequences of brain MRI in which areas of demyelination are demonstrated. 1 The symptoms and signs, that is, loss of consciousness, altered sensorium, abnormal body movement, mild limb weakness and increased plantar reflex were present in our patient. There was history of missed insulin therapy, high blood glucose level, hypernatremia during treatment, and MRI findings in T2 and FLAIR sequences showing hyperintensity in pons and tectum suggestive of complicated DM with CPM. This diagnosis was considered after ruling out differentials such as stroke, encephalitis, meningitis, Wernicke encephalopathy, hepatic encephalopathy, primary brain tumours, metastases, radiotherapy, chemotherapy and multiple sclerosis. 24
Ten out of 16 case reports have shown the hyperosmolar hyperglycemic state (HHS) as the cause of ODS.6,7,14–21 Six of these case reports6,7,14–17 have described that the ODS was caused by rapid correction of serum glucose from hyperglycemic state to normal or hypoglycemic state. Three of these case reports18,19,21 have stated that the ODS was accompanied by HHS prior to when the patients visited hospitals. Ramineni et al. 25 reported ODS in de novo type 2 DM with HHS on presentation suggesting its association even without sodium imbalance and its correction. Jalalzadeh et al. 21 has reported prior history of several hypoglycemic and hyperglycemic states and indicated the fluctuations in serum osmolarity due to corresponding fluctuation in serum glucose concentration as the cause of ODS. Hegazi and Mashankar 20 have indicated severe prolonged hypernatremia secondary to HHS as the cause of ODS. In our case, hypernatremia developing only after the onset of symptoms and later during treatment indicates hyperglycaemia rather than hypernatremia as the cause of ODS.
In the remaining 68–13 out of 16 case reports, complicated DM without HHS and any other risk factor of ODS was the underlying cause of ODS. The case of Sharma et al. 8 had already presented with clinical features of ODS while visiting hospital. Gourine et al. 10 hypothesized either rapid increase of glucose after stopping insulin therapy or rapid correction of glucose as the cause of ODS. Rest four cases developed clinical features of ODS during the rapid correction of serum glucose. Our case had history of noncompliance to insulin like the case of Talluri et al. 26 In the case of Talluri et al., the noncompliance to insulin is supposed to be the cause of alteration in glycaemic status leading to ODS.
Age of patients in the 16 case reports varied from 18 months to 93 years. Although the ODS is rarely reported in children, its occurrence in 4-year-old 11 and 18-months-old 12 children indicates that any age group can suffer from ODS.
Proposed mechanism for development of ODS during rapid correction of sodium is that osmolytes can easily diffuse from cytoplasm to extracellular fluid during the development of hyponatremia thus compensating the decreasing osmolarity of extracellular fluid, but the same osmolytes cannot diffuse back fast enough into the cytoplasm from extracellular fluid as well as cannot replicate within the cells fast enough to balance the increasing osmolarity of extracellular fluid during the correction of hyponatremia. As the hypertonicity created in extracellular fluid during correction of hyponatremia goes uncompensated, the oligodendrocytes are degenerated.1,2 This difference in diffusion of osmolytes in the two directions clarifies why ODS develops while correcting hyponatremia but contrasts with development of ODS during the correction of hyperglycemia because in the latter, it is decreasing osmolarity, rather than increasing osmolarity, as the cause of ODS. We suggest both increasing or decreasing osmolarity can be the cause of ODS.
Furthermore, ODS has been found in autoimmune liver disease, 27 Sjogren’s syndrome, 28 and non-Hodgkin’s lymphoma. 29 The pathophysiology for ODS occurring in case of autoimmune diseases, that is, autoimmune hepatitis and Sjogren’s syndrome, has not been cleared but in case of non-Hodgkin lymphoma, it is supposed to be due to systemic and metabolic stress induced by non-Hodgkin lymphoma.
There is no specific therapy for ODS, and only some patients of ODS can fully recover and remyelinate within months. 2 Treatment under recent understanding of ODS has shown 33%–50% have better prognosis with good outcome while 33%–55% of patients require some or fully dependent care or die. 1 All the 16 patients in our review, including this case, were treated supportively under controlled serum glucose since there is no specific treatment for ODS; however, correction of osmolarity under appropriate correction rate can prevent ODS in case of hyponatremia correction. 1 Two patients14,21 died during the treatment, and others had significant prognosis. Our case presented in healthy state without any neurological deficit in a follow-up after a month of discharge.
Conclusion
Not only rapid correction of hyponatremia, but also sudden or rapid change in extracellular fluid osmolarity due to complicated DM can cause ODS. The change in osmolarity causing ODS can be in both increasing or decreasing order as well as any age group can be affected. However, the exact mechanism for the development of ODS and appropriate glycaemic correction rate needs further research to be revealed. ODS is rare, and it can be missed in clinical setting, and as it can be fatal or debilitating, it must be considered in any kind of disorder associated with change in osmolarity. The specific treatment for ODS needs to be established, till then early supportive therapy may have good outcome in ODS patients.
Footnotes
Acknowledgements
The authors are thankful to the patient and his family for cooperation.
Author contributions
SKY, SK and SP collaborated in collecting the data, interviewing the patient and patient party, and preparing the manuscript. RO, RK, BPG, RR and NP managed the case and guided throughout the process of writing this article. NG and AS were involved in management of the case, and preparing and review of the manuscript. AJ helped in interpretation of radiological findings, and preparing and review of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
