Abstract
Objective:
This study evaluated the feasibility and satisfaction of using a wearable brain activity sensing device for stress reduction among patients experiencing Long COVID (LC).
Patients and Methods:
Patients with LC (N = 45) were invited to participate in an open-label pilot study. Participants were asked to use a brain-sensing electroencephalogram (S-EEG) wearable device (Muse-S™) daily for 90 days and followed for an additional 90 days (180 days total participation). Study enrollment began June 28, 2022, and ended July 28, 2023.
Results:
Patients were predominantly female (82%), white (96%), and with an average age of 47.6 (±12.5) years. At the end of treatment (90 days), cognition, sleep, self-efficacy, and quality-of-life quality of life improved (P < .001 to P = .04). In addition, stress and anxiety were significantly reduced (P < .001). These significant changes persisted by end-of-study (180 days). For stress (P = .023) and anxiety (P = .033), a change from the baseline during follow-up was significantly associated with the amount of S-EEG usage. The end-of-study satisfaction survey indicated that patients were very satisfied with the S-EEG device, and 97% planned to continue using it.
Conclusion:
Results suggest that the S-EEG device improved cognition in LC, which is one of the main severe symptoms we see in our LC clinic. In addition, it helped reduce stress and anxiety. These findings have important clinical implications for LC patients using S-EEG as an adjunct to concurrent LC clinical care which includes pharmacotherapies.
Introduction
Post-acute sequelae of SARS-CoV-2 infection (PASC), also known as Post-COVID Syndrome, or Long-COVID (LC), is a significant cause of morbidity and disability in patients who have had COVID-19 with an estimated 10% to 30% of patients developing LC with symptoms lingering for months and even years. 1 While LC is a heterogenous condition, symptoms often include fatigue, dyspnea, cognitive dysfunction, headache, palpitations, orthostatic intolerance, and gastrointestinal symptoms, thereby significantly impairing quality of life (QoL).
Mind-body techniques, including yoga, mindfulness/meditation, biofeedback, and progressive muscle relaxation, have been proposed for use in patients with complex chronic conditions such as cancer, PTSD, depression, anxiety, and LC targeting symptoms.2 -7 Given the limited exercise tolerance experienced by most patients with LC, mindfulness/meditation, and biofeedback have the lowest barrier to treatment.8,9 Muse-S™ is a clinical grade, headband-style wireless electroencephalogram sensing (S-EEG) system designed to interact with a mobile device using an accompanying smartphone application (App). In combination with the S-EEG device App, the headband converts EEG signals measured over the frontal and temporal cerebral cortices into measures of brain state, allowing the S-EEG device to guide the user through the meditation steps. Calibrated to an individual user, the S-EEG device distinguishes between active and calm brain states to provide real-time performance feedback. It helps users realize that it allows them to benefit from biofeedback practice in an engaging and accessible manner. The S-EEG device also has a photoplethysmography (PPG) sensor, which is worn on the device and tracks heart rate during meditation sessions. This has been tested in a prior study by some of the current study team, with patients undergoing breast cancer surgery 10 and healthcare clinicians during the height of the COVID-19 pandemic10,11 and is currently being tested in patients with fibromyalgia who concurrently experience pain (ITC and RG). The S-EEG can be used as an adjunct therapy in established standard LC treatments, including pharmacotherapies, physical therapies, pulmonary rehabilitation, counseling, and other off-label therapies such as low-dose naltrexone (LDN), guanfacine, and devices such as vagal nerve stimulation (VNS).
We hypothesize that daily use of this novel biofeedback-assisted S-EEG wearable device can provide mindfulness training to increase calm-state brain activity in LC patients, activating the parasympathetic nervous system.12,13 The longer the patient uses this tool, the greater the effect on LC-associated autonomic dysfunction, which is hypothesized to decrease symptom burden.
The purpose of the present study was to assess the impact of the S-EEG device as an additional treatment on LC symptoms such as cognition and focus (aka—“brain fog”), sleep, stress, anxiety, QoL, and resilience in patients who were seen and in an LC clinic and were given standard LC treatment.
Methods
This study was designed as an open-label clinical trial with no control group (aka—all study participants received the intervention). The purpose was to collect preliminary data on the adherence and satisfaction of a wearable brain S-EEG device on LC. All participants accrued to the study received the wearable S-EEG device headband system and an additional wrist wearable device at the study entry. At entry, the participants also download the device accompanying the smartphone app for study purposes. They were then instructed to always wear the wrist wearable device unless they were charging it and to use the S-EEG headband device at least 4 times per week, up to 10 min each time over a period of 90 days. They were followed for 180 days (90 days of treatment and 90 days post-treatment). Study outcome data was collected before initiating treatment (baseline), during treatment (days 30 and 60), end of treatment (day 90), and post-treatment (day 180).
Following the Declaration of Helsinki, the Institutional Review Board (IRB) reviewed and approved this clinical trial study. The IRB-approved written informed consent was obtained for all study participants before study participation. During the study, all significant changes to study design and procedures were appropriately filed, reviewed, and approved by the IRB.
Setting
Invited participants were patients presenting at 1 of 2 post-COVID clinics at Mayo Clinic—Rochester: Post-COVID Care Clinic (PCoCC) and the COVID-19 Activity Rehabilitation Program (CARP). Enrollment took place from June 28, 2022, to July 28, 2023. Study participation concluded on October 31, 2023.
Study Population
A total of 55 patients with LC who were scheduled for a visit within 1 of 2 post-COVID clinics in 1 Medical Center in the Midwest of the United States of America were invited to participate in this open-label study. Of the 55 patients, 45 participated in this study. All patients were diagnosed with LC, 18 years of age or older, identified with LC, not pregnant at the time of consent, had the ability to provide informed consent and complete all aspects of the trial, had access to an iPhone/iPad or Android/mobile tablet device and did not have any contraindicating co-morbid health condition which would interfere with the proper use of the headband system, as determined by the clinician. Potential participants were excluded if they were on another investigational drug within 30 days of enrolling in this study, if they were on an unstable dose of medication for anxiety, depression, or sleep (if they were on any of those meds), if they were concurrently practicing/using another form of mindfulness training on a weekly/regular basis or enrolled in another program focused on patients QoL or stress. Finally, patients were also excluded if they were diagnosed with an unstable medical or mental health condition as determined by the clinician investigator.
Patients were categorized as fatigue, orthostatic intolerant, or pain-predominant phenotypes based on the assessment of their treating LC physician. A total of 10 patients did not meet the study criteria (4 were practicing mindfulness, 5 were enrolled in a formal complementary alternative medicine (CAM) program, and 1 did not have access to a smart device (iPad/iPhone or Android/mobile tablet) and were excluded from the study. The current analysis is based on data from the 45 patients who met inclusion criteria (Figure 1).
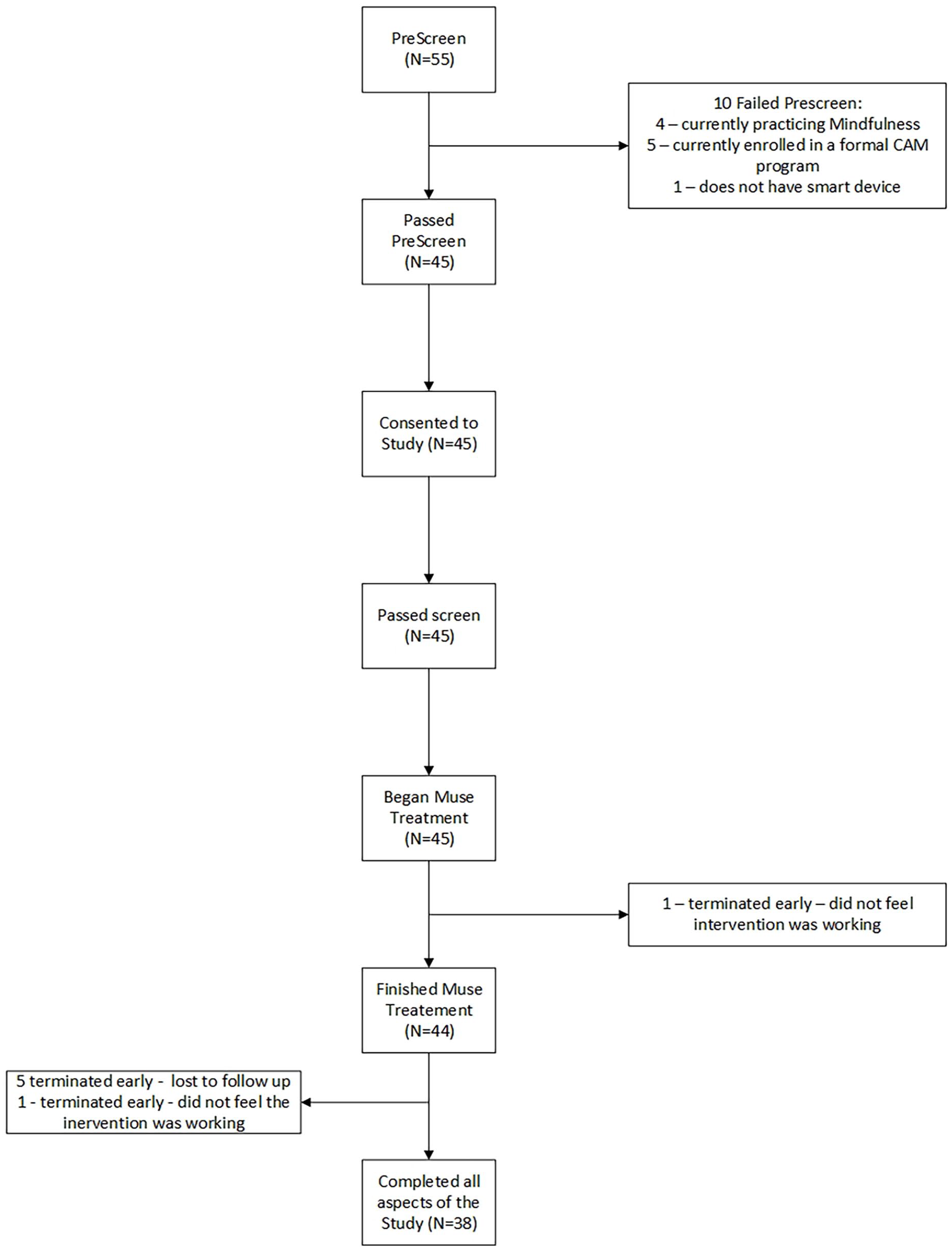
Study consort diagram.
Intervention
Muse-S™ is a wearable brain S-EEG device which is a clinical-grade, headband-style wireless EEG system with 7 sensor and 4 channels of EEG data. It uses Bluetooth to interact with an associated app on a smart device. In combination with the smartphone app, this device converts EEG signals measured over the frontal and temporal cerebral cortices into measures of brain state. The app walks the users through the meditation steps, by using a series of focus attention training methods to train the brain of the user. This device and app are described in detail elsewhere.10,11
Once the S-EEG device calibrates to an individual user, it can distinguish between active and calm brain states to provide real-time performance feedback. The S-EEG device also has a PPG sensor that tracks heart rate during meditation sessions. For a visual look at the Muse-S™ S-EEG device, please refer to Supplemental Figures 1 to 3.
In this study, all participants were educated on using the S-EEG Headband and the accompanying app. They were instructed to use the S-EEG Headband and the accompanying app at least 4 times per week up to 10 min each time. Within the app there are several options to choose from: Body, Breath, Heart, and Mind Meditation Guided Meditation, Body and Breath Meditation; Heart Meditation Guided Meditation and Go-to-sleep Meditations. These options, as well as the app itself, are described in a prior publication. 11
Data Collection
In addition to demographics and lifestyle history, medical history (per their LC diagnosis) was abstracted from the electronic medical records. Data on adherence, including frequency of device use, duration of each use, and choice of program accessed, were collected from the app dashboard download. Survey data was collected from the returned surveys which utilized the REDCap14,15 electronic system to automatically send the surveys to the participants’ email at the appointed time (baseline, day 30, day 60, day 90, and day 180) during the study participation. The Cognitive Testing Scores were downloaded from the Creyos Dashboard and the wrist wearable data (Fitbit) were downloaded from the Fitbit app dashboard.
QoL was measured with the validated Linear Analog Self-Assessment (LASA).16 -18 The LASA-6 is a 6-item measure of QoL, in which QoL is conceptualized as a multidimensional construct with 5 domains (physical, functional, emotional, spiritual, and social). The LASA uses an 11-point visual analog scale (VAS). LASA-6 is made up of 6 Likert scale questions from 0 (as bad as it can be) to 10 (as good as it can be). Overall scores can range from 0 to 60, with higher numbers being good quality of life and lower scores being bad QoL.
Anxiety was measured using the State-Trait Anxiety Inventory (STAI-Y1)19 -21 This 20-item self-report measure indicates the intensity of feelings of anxiety; it distinguishes between state anxiety (a temporary condition experienced in specific situations) and trait anxiety (a general tendency to perceive situations as threatening). This uses a 4-point visual analog scale, and the participant indicates how they feel in the moment (1 = not at all to 4 = very much so). Overall scores can range from 20 to 80, with higher numbers being high anxiety and lower scores indicating low anxiety.
Stress was measured using 2 approaches, which have shown good validation and reliability. The first is the 1-item Perceived Stress Scale (PSS1) 22 used at baseline as part of their lifestyle history. The PSS1 is comprised of 1 question concerning stress using an 11-point VAS (0 = no stress and 10 = very high stress). Using this scale at baseline, the stress score could range from 10 (highest imaginable stress) to 0 (no stress at all).
During the study visit, stress was tracked using the validated 10-item Perceived Stress Scale (PSS-10).23,24 The PSS-10 is a validated 10-question survey with a 5-point Likert scale from 0 to 4. With responses being “Never” (“0”) to “Very Often” (“4”), the scoring is reversed for specific items. The score could range from 0 to 40, with the higher numbers indicating high stress and the lower numbers indicating low or no stress.
Resilience was evaluated through the validated Connor-Davison Resilience Scale 10 (CD-RS10). 25 This validated scale uses a 5-item VAS. The scoring of each of the 5 questions is a 5-point VAS from 0 (Not true at all) to 4 (True nearly all the time). Overall, the total score could range from 0 to 40, with the higher numbers indicating high resilience and the lower numbers indicating low or no resilience.
The PROMIS t-score reflected sleep quality. We adapted the ASCQ-Me® v2.0 Sleep Impact—Short Form.26,27 to assess the sleep quality in this study. It should be noted that the ASCQ-Me short form was validated in patients with sickle-cell and not in other patient populations. The questions did not need to be altered, and it was easy to administer and adapt for our study. This questionnaire focuses on sleep impact on overall health in adults in the prior 7 days. It consists of 5 questions scored on a 5-point VAS (1-5). The responses are “never” to “always,” but the scoring is reversed depending on the item. Overall, the total score could range from 5 to 20, with the higher number indicating problems with sleep and lower numbers indicating no problems with sleep.
Self-efficacy was measured using the validated PROMIS Self-Efficacy Short Form 4a. 28 It is designed to assess one’s confidence in performing certain tasks. It consists of 4 items measured on a 5-point VAS from 1 (not at all confident) to 5 (very confident). Overall, the total score could range from 5 to 20, with the higher number indicating high self-efficacy and lower numbers indicating little or no self-efficacy.
Emotional Distress—Anxiety was measured using an adapted version of the validated Emotional Distress Short Form 7a. 28 . Each item on the measure is rated on a 5-point scale (1 = never; 2 = rarely; 3 = sometimes; 4 = often; and 5 = always) with a range in total score from 7 to 35, with higher scores indicating greater severity of anxiety. The original form instructed participants to think about the past 7 days, but for purposes of this study, we reworded the question to focus on the current time (“now”).
The Patient Health Questionnaire (PHQ)-2 measured depression.29,30 The PHQ-2 is a validated and frequently used self-report measure of depressive symptoms comprising 2 questions (each with a yes/no score) derived from the PHQ-9.31,32
Self-perceived Cognitive Function was measured using 2 validated PROMIS forms: the Cognitive Function—Short Form 4a and the Cognitive Function—Abilities—Short Form 4a. 28 Each validated form asked 4 questions scored with a 5-point VAS. The 5-point VAS for the Cognitive-Function Short Form ranged from 1 (“Very Often”) to 5 (“Never”), and the VAS for the Cognitive-Abilities Form ranged from 1 (“Cannot Do”) to 5 (“None”). The overall score for each component ranges from 0 to 20, with the higher number being better cognition.
Objective cognitive testing was completed using 3 of 12 online-based tasks provided by the Creyos platform, all of which have been validated. Double Trouble, Feature Match, and Monkey Ladder are 3 tasks among a battery of 12 neuropsychological tasks, which measure core elements of cognition. Demos of the tasks are found at https://creyos.com/online-cognitive-tasks. Double Trouble measures response inhibition and asks the player to match color rather than words; Feature Match measures attention and is a version of “Spot the Difference”; and Monkey Ladder measures memory. In this task, boxes with numbers appear on a screen for a short period of time; the player memorizes which number is in which box, and the numbers disappear. The player then has to click the boxes in numerical order. In essence, it tests temporary memory. Participants were asked to undertake these cognition tests at 3 time points: baseline, 90 days (end of treatment), and 180 days (end of study). An introduction, including a tutorial video, was provided at each test’s beginning. All participants were asked to complete the introduction before initializing the tests.
Participants were asked to wear their Fitbit® 24/7 except during recharging and to download the Fitbit® App. Some of the data collected by this app included heart rate, steps, and blood oxygen saturation level (SpO2). 33
The patient satisfaction survey used was adapted and individualized to this specific pilot study from The Was It Worth It Questionnaire (WIWI). 34
Data Analysis
Demographic characteristics are described using mean and standard deviations or frequencies and percentages based on the type of variable. For the primary aim of assessing feasibility, the primary outcome was the frequency of S-EEG usage. Descriptive statistics are presented for data obtained on frequency and duration of use over the 90 days of treatment. Outcomes obtained at 30, 60, 90, and 180 days are summarized using mean and standard deviation and compared to baseline using the paired t-test. Linear mixed models were used to assess whether changes in outcomes were associated with the S-EEG device usage in the week prior to the assessment. For each outcome, data collected at 30, 60, and 90 days were analyzed in a mixed model with the subject included as a random effect, the number of the S-EEG device sessions in the prior week included as the explanatory variable, and the baseline value the given outcome included as a covariate. For the additional cognitive tests that were obtained at 90 and 180 days, linear regression was used to assess whether scores at 90 days were associated with the S-EEG device usage in the week prior to the 90 days assessment. For these models, the test score was the dependent variable, the number of the S-EEG device sessions in the prior week was the explanatory variable of interest, and the baseline value of the given test was included as a covariate. All modeling results are summarized by presenting the point estimate with a 95% confidence interval for the mean change in the outcome for 1 additional S-EEG device session in the prior week. A sample-size of N = 45 was chosen for this pilot study in order to provide enough information to assess feasibility and also provide preliminary data for changes in outcomes. In all cases, P < .05 was considered statistically significant. SAS statistical software was used for all analyses (SAS version 9.4, SAS Institute Inc.).
Results
Among the 45 patients who consented and were accrued to study, the majority were female (37/45, 82%), non-Hispanic white (43/45, 96%), married or living as married (27/45, 60%), had some college education or more (44/45, 97%), and had a mean ± SD age of 47.6 ± 12.5 years (Table 1).
Subject Characteristics.

One subject was African American, and 1 subject was Hispanic.
Among the 45 post-COVID patients who participated in this study, the majority of the patients had a fatigue-predominant phenotype (34/45, 76%) versus the orthostasis-predominant phenotype (5/45, 11%) and a pain-predominant phenotype (6/45, 13%; see Table 1). The average number of days ± standard deviation (SD) from COVID infection to the first visit to our post-COVID clinic was 347 ± 229 days, and the total number of days from the first visit to the post-COVID clinic to enrollment was 93 ± 143 days (data not shown).
The percent of people who used the S-EEG device throughout the study ranged from 100% (week 1) to 73% (week 13). The number of weekly sessions changed from a median of 9 in week 1 to 3 in week 13. This was consistent with the number of minutes spent in S-EEG device meditation, which changed from 542 (among all 45 patients) in week 1 to 29 min in week 13 (Table 2).
Device Adherence According to Study Week (N = 45) a .

Any use is summarized by presenting the number (%) of subjects who used the S-EEG device for at least 1 session during the given week. Number of sessions and total minutes are summarized by presenting the median (25th, 75th) values with all subjects included (ie, those who did not use the S-EEG device in the given week are included with 0 sessions and 0 min).
Improvement at end-of-treatment, compared to baseline, was noted for perceived stress, using the validated PSS-10 scale (mean ± SD change from baseline −5.0 ± 5.3, P < .001), sleep using the adapted ASCQ-Me® v2.0 Sleep Impact (3.0 ± 5.9, P = .003), anxiety using the PROMIS Emotional Distress Scale (−4.5 ± 5.5, P < .001), cognitive function using the PROMIS cognition scale (3.5 ± 5.1, P < .001), self-efficacy (2.8 ± 8.4, P = .040), and quality of life using the validated LASA-6 (1.0 ± 1.6, P < .001). These continued to persist at the end of the study (180 days from baseline or 90 days post end-of-treatment) whereby, all continued to be significantly improved when compared to baseline: PSS-10 (−5.2 ± 6.2, P < .001), ASCQ-Me (4.1 ± 5.7, P ≤ .001), PROMIS Emotional Distress (−5.2 ± 5.2, P < .001), PROMIS Cognitive Function (4.1 ± 5.7, P < .001), PROMIS Self-Efficacy (3.4 ± 9.1, P = .028), and LASA-6 (1.4 ± 1.6, P < .001). No significant differences were found for anxiety using the State-Trait Anxiety Inventory (STAI-Y1) assessment for the resilience Connor-Davison Resilience Scale 10 (CD-RS10; Table 3).
Outcomes.

For perceived stress and emotional distress, the change from baseline during follow-up was significantly associated with the S-EEG device usage. For perceived stress, the estimate (95% C.I) for the mean change for 1 additional S-EEG device session in the prior week was −0.26 (−0.48, −0.04), P = .023; and for emotional distress, the estimate (95% C.I) was −0.25 (−0.48, −0.02), P = .038. (Table 4). Of the 3 additional cognitive tests, only “Double Trouble” was found to be improved from baseline to end of treatment, and none were associated with the S-EEG device usage (Table 5).
Association of the S-EEG Device Usage (Number of Sessions in Prior Week) With Outcomes.

For each dependent variable, data collected at 30, 60, and 90 days were analyzed in a linear mixed model with the subject included as a random effect. The number of the S-EEG device sessions in the week prior to the assessment was the explanatory variable of interest and the baseline value was included as a covariate. Data are summarized by presenting the estimate (95% Confidence Interval) for the mean change in the dependent variable for 1 additional S-EEG device session in the prior week.
Additional Cognitive Tests.

Linear regression was used to assess whether scores at 3 months were associated with the S-EEG device use in the week prior to the 3-month (90-day) assessment. For these models, the test score was the dependent variable, the number of the S-EEG device sessions in the week prior to the assessment was the explanatory variable of interest, and the baseline value of the given test was included as a covariate. Data are summarized by presenting the estimate (95% Confidence Interval) for the mean change in the given test score for 1 additional S-EEG device session in the prior week.
The end-of-study satisfaction survey indicated that most patients felt it worthwhile to participate in the study (92%) and would recommend it to others (95%). The majority of participants thought they were more relaxed after using it (87%) and would like to use it daily for more extended periods (82%). Overall, about 97% had plans to continue to use the S-EEG device post-study (Tables 5 and 6).
Study Satisfaction a .

39 of the 45 subjects completed the end-of-treatment satisfaction survey.
Discussion
The findings from this small feasibility study demonstrate statistically significant decreases in symptom burden across multiple domains, including cognitive function, stress, sleep, anxiety, self-efficacy, and QoL in patients using the S-EEG device. Further, the improvement was greater for stress and anxiety when the S-EEG device was used more frequently. The device was well-tolerated with no patients reporting side-effects, a high percentage of patients (87%) indicating that the device was useful, and nearly all (97%) indicating they planned to continue using the device after the study was completed, While device use diminished with time, raising concerns about long-term therapy adherence, the changes from baseline for many outcomes remained statistically significant at 90 days. Taken together, these findings suggest that the S-EEG device for biofeedback was effective and well tolerated in patients with LC. This is highly important as our armamentarium currently has limited tools against LC, rendering treatment difficult and often ineffective. The ability to deliver meaningful therapy with an app-enabled wearable device in an asynchronous manner also has great potential impacts on health access and equity, particularly in patients with LC who live in under-resourced areas, those with fatigue, and those who struggle with reliable transportation.
Mindfulness techniques such as those delivered by the S-EEG device have been associated with brain changes, including increased functional connectivity. 35 These changes also include numerous brain structures, specifically increased gray matter, improved connectivity between the salience and default mode networks, 36 and increased hippocampal connections. 12 Mindfulness techniques are also associated with mood, attention, memory, and emotional regulation improvements. 37 Further, mindfulness techniques have improved several common symptoms in LC, including fatigue,38,39 cognitive dysfunction, 40 autonomic dysfunction,41,42 and pain 43 —including headache.44,45 The S-EEG device also incorporates biofeedback from an EEG channel and a PPG sensor. Biofeedback has also been shown to help alleviate many of the symptoms common to LC, with efficacy noted against pain, 46 dysautonomia, 47 and cognitive impairment (in cancer patients). 48 While biofeedback has been studied for fatigue, it seems to improve fatigue perception without altering muscle performance activities such as climbing stairs. 49
While there remains a paucity of clinical trials on patients with LC, there have been a few trials of mindfulness and biofeedback in this population. Indeed, our data is similar to that seen in a small study of 13 patients with LC dysautonomia who underwent heart rate variability (HRV) biofeedback training for 4 weeks. 47 In this group, there were significant reductions in the modified Yorkshire rehabilitation scale, Composite Autonomic Symptom Score (COMPASS-31), WHO Disability Assessment Schedule (WHODAS), and EuroQoL 5 dimensions. A randomized controlled trial on 34 patients with LC showed improved speed of cognitive processing in patients treated with a 4-week neuro-meditative program, 40 which is again similar to our data. Combining biofeedback and mindfulness training elements, the S-EEG device intervention provides significant benefits to symptom burden in LC patients across multiple domains.
This intervention may directly exert some of its impact on symptoms through regulation of the dysautonomia typically associated with LC, wherein patients will demonstrate increased sympathetic and decreased parasympathetic tones. 50 In fact, in a previous study, improvement in these autonomic parameters, as evidenced by changes in heart rate variability, has been associated with decreased orthostatic hypotension. 51 In the clinical trial of HRV biofeedback above, the root mean square of successive differences between heartbeats (RMSSD) increased significantly, which indicates improvement in the LC dysautonomia. 47 The effects of biofeedback and mindfulness on brain organization and central neuroinflammation may modulate the effect on dysautonomia. While brain connectivity and central neuroinflammatory markers were not directly measured in our study, previous data on mindfulness interventions suggest the possibility of enhancing neuroplasticity, thereby leading to improved connectivity and gray matter remodeling.12,35,36,52 This is putatively important in patients with LC who have been demonstrated to have significant reductions in cortical gray matter, particularly in those experiencing sleep difficulties,53,54 as well as neuroinflammation.55 -57
Our study has several limitations. First, the majority of the patients in our study were white and well-educated, which reflects the referral population to our mid-west medical center. Therefore, the generalizability of our findings to other racial and ethnic groups is limited. Given the limited sample-size used for this pilot study, the results should not be considered definitive. Additionally, one of the inherent limitations of this study design is the lack of a control group. We opted for a pre-post design which allowed all the LC patients to benefit from the S-EEG device intervention while allowing each patient to act as their own control. However, given the unblinded nature of the intervention some of the change in outcomes could have been influenced by a placebo effect. Since so little is known about the use of the meditation type, we chose to stay flexible with how the patients used the S-EEG device app intervention. In this study, the participants were given access to several guided meditations (eg, resilience, focus, sleep, and burnout). Although data was collected on the specific type and frequency of meditation accessed by each individual, this data could not be analyzed further due to the small sample size and variety of options given to the participants. Finally, although most of the patients enjoyed using the device and stated they would continue to use it, a few participants had issues with the equipment—the cause of the issues were not investigated in detail, so whether or not they were due to mechanics of the devices, user error, or issues with local internet is unknown. What is known is that the patients needed the study team’s support long-distance and replacement devices were sent through the postal mail on a few occasions when the original devices stopped working. Despite these limitations, the patients found the intervention to be easy to use, adaptable, and helpful. This study was designed as a feasibility pilot study with the exploratory aim of evaluating the device’s impact on selected health measures. Therefore, a larger study is needed for these findings to be considered representative of the general population with LC.
Conclusion
The results of our study suggest that a combination biofeedback approach may be delivered remotely and asynchronously alongside standard LC treatment in an effort to effectively reduce symptom burden in patients with LC. Our study adds to the literature supporting LC treatment with both biofeedback and mindfulness, which has been limited by small sample sizes. These findings have important clinical and research implications for the management of patients with LC as there is no evidence for at-home adjunct treatment using an app-enabled wearable, which will assist these patients not only with symptom burden but also with energy management and reducing transportation burden.
Supplemental Material
sj-jpg-1-jpc-10.1177_21501319251325639 – Supplemental material for Using a Wearable Brain Activity Sensing Device in the Treatment of Long COVID Symptoms in an Open-Label Clinical Trial
Supplemental material, sj-jpg-1-jpc-10.1177_21501319251325639 for Using a Wearable Brain Activity Sensing Device in the Treatment of Long COVID Symptoms in an Open-Label Clinical Trial by Ryan T. Hurt, Ravindra Ganesh, Darrell R. Schroeder, Jennifer L. Hanson, Shawn C. Fokken, Joshua D. Overgaard, Brent A. Bauer, Bright Thilagar, Christopher A. Aakre, Sandhya Pruthi and Ivana T. Croghan in Journal of Primary Care & Community Health
Supplemental Material
sj-jpg-2-jpc-10.1177_21501319251325639 – Supplemental material for Using a Wearable Brain Activity Sensing Device in the Treatment of Long COVID Symptoms in an Open-Label Clinical Trial
Supplemental material, sj-jpg-2-jpc-10.1177_21501319251325639 for Using a Wearable Brain Activity Sensing Device in the Treatment of Long COVID Symptoms in an Open-Label Clinical Trial by Ryan T. Hurt, Ravindra Ganesh, Darrell R. Schroeder, Jennifer L. Hanson, Shawn C. Fokken, Joshua D. Overgaard, Brent A. Bauer, Bright Thilagar, Christopher A. Aakre, Sandhya Pruthi and Ivana T. Croghan in Journal of Primary Care & Community Health
Supplemental Material
sj-jpg-3-jpc-10.1177_21501319251325639 – Supplemental material for Using a Wearable Brain Activity Sensing Device in the Treatment of Long COVID Symptoms in an Open-Label Clinical Trial
Supplemental material, sj-jpg-3-jpc-10.1177_21501319251325639 for Using a Wearable Brain Activity Sensing Device in the Treatment of Long COVID Symptoms in an Open-Label Clinical Trial by Ryan T. Hurt, Ravindra Ganesh, Darrell R. Schroeder, Jennifer L. Hanson, Shawn C. Fokken, Joshua D. Overgaard, Brent A. Bauer, Bright Thilagar, Christopher A. Aakre, Sandhya Pruthi and Ivana T. Croghan in Journal of Primary Care & Community Health
Footnotes
Acknowledgements
The study team would like to thank Ariel Garten, Founder of InteraXon (choosemuse.com) and Mike Batista from Creyos Health (creyos.com) for their support and patience during this study. We would also like to extend a special thanks to all study participants, without their participation, this study would not have been possible.
Author Contributions
All the authors participated in the study concept and design, participant enrollment, data collection, analysis and interpretation of data, drafting and revising the paper for important intellectual content, and reviewed and approved the final version of the manuscript. Each author will take public responsibility for the entire work.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RTH is a consultant with Nestle Nutrition. All other authors declare no support from any organization for the submitted work, no financial relationships with any organizations that might have an interest in the submitted work in the previous 3 years, and no other relationships or activities that could appear to have influenced the submitted work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by the Mayo Clinic Department of Medicine, General Internal Medicine and the Research Hub. General Internal Medicine provided support and oversight for clinical trial conduct and data collection and the Research Hub provided support for data analysis. In addition, InteraXon provided the Muse-S™ devices used in this study at no cost, and Creyos Health provided free access to cognition online testing for study participants. Data were collected and managed using REDCap (Research Electronic Data Capture) hosted at Mayo Clinic and supported in part by the Center for Clinical and Translational Science Award (UL1 TR000135) from the National Center for Advancing Translational Sciences (NCATS).
Ethical Approval and Consent to Participate
In accordance with the Declaration of Helsinki, this study was reviewed and approved (21-009820) by the Mayo Clinic Institutional Review Board (IRB). Mayo Clinic IRB approved written informed consent was obtained for all study participants prior to study participation.
Availability of Data and Materials
All data supporting the study findings are contained within this manuscript and within Clinicaltrials.gov (NCT05199233).
Trial Registry Information
Trial registration: NCT5199233. Registered 18 Jan 2022
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
