Abstract
Post-COVID conditions, commonly referred to as long COVID, has become of increasing interest to biomedical and public health communities. Patient advocacy is critical to not only advancing quality care outcomes for patients affected by prolonged effects of the disease, but also to contribute to our understanding of lived perspectives to ensure individual voices inform future care approaches. While there has not been a “one-size fits all” approach to standardized care, disease heterogeneity has challenged previous views that the virus is limited to single organ system infections. Stakeholders at all levels have the most impact when they collaborate and organize strategies to offer assessment and treatment in multidisciplinary settings. I propose a framework in which population- and individual-level models are integrated to enable quality outcomes while helping frontline practitioners improve their decision-making in all environments.
Since the COVID-19 pandemic began in December 2019, the health care environment for treating patients has been evolving and dynamic. A majority of patient care was initially provided in emergency rooms and intensive care units (ICUs) in the symptomatic management of acute illness. As COVID prevalence increased, a growing number of patients required follow-up care in outpatient settings to manage chronic symptoms. As of July 2022, more than 88 million cases and 1 million deaths have been recorded in the United States (Johns Hopkins Coronavirus Resource Center [JHU], 2022).
The disproportionate focus of health recommendations for those dealing with active COVID infection has silently excluded individuals affected by its long-term effects. Many patients continue to experience chronic health problems despite having met the Centers for Disease Control and Prevention (CDC) definition for recovery after completing isolation or convalescence periods (Centers for Disease Control and Prevention [CDC], 2022a). Early studies noted close to 30% of recovered patients suffered from at least one symptom up to 3 months after infection, but new research indicates this value may be close to 54% (Groff et al., 2021; Nehme et al., 2021). Pathological symptoms can include neurological, pulmonary, cardiovascular, psychological, constitutional, or other system deficits (Groff et al., 2021). The growing number of patients experiencing chronic illness along with varying clinical manifestations from native and variant viral types will pose growing challenges for years to come.
While robust investigations have been attempted to learn more about COVID pathophysiology, prevention, and treatment, less attention in the literature has been directed toward improving social and community-based outcomes. In this article, I examine some of the underlying elements affecting care models and how reframing practitioner–patient relationships can improve experiences. I also offer a framework in which collaboration in practice can address qualitative features of the community experience while stimulating greater dialogue for future scientific advancement. For the purposes of this article, I use
Early Care Environment
The rapid expansion of COVID in early 2020 categorized a period in which health centers across the United States experienced intense resource and staffing demands. Metropolitan areas such as New York City experienced the highest levels of transmission as care quickly transitioned away from elective procedures to acute COVID treatment for patients experiencing terminal stage disease (Ramaswamy et al., 2020). As the available supply of resources such as trained medical personnel, diagnostic testing supplies, and infection control consumables lagged behind demand, health systems struggled with triage capacity in high-risk patient care. Even as limited resources were prioritized for health staff, many remained vulnerable due to overwork, occupational exposure, and psychological stress.
Acute care centers at the time offered two strategies for severely ill patients: invasive mechanical ventilation and extracorporeal membrane oxygenation (Selvaraj et al., 2022). This was done to address respiratory complications often provoked by inflammatory responses to viral infection. Many health staff gained valuable ICU experience through these means, but this may also have influenced early views of the course of disease. Patients without acute symptoms were informed to self-isolate at home to preserve health care capacity and prevent further disease transmission. However, the exclusion to the range of COVID clinical manifestations early in the pandemic resulted in a filtered, partial understanding in the medical community to the diversity and complexity of disease.
Current Care Environment And The Void Created
New vaccines and treatment options such as antivirals and monoclonal antibodies offered promising ways to level the playing field in prevention and care management. These tools provided a method to systematically treat infection while reducing hospitalizations via vaccine-induced immunity. The resulting reduction in COVID-related health care demand, in combination with declining transmission risk across the United States, allowed recovering patients to access care services more broadly. These included telemedicine and outpatient in-person visits. Post-COVID care centers (PCCCs), or locations where patients could be seen for a variety of COVID-related complaints, also emerged in the summer of 2020. As of June 2022, 257 PCCCs are registered in the United States (Survivor Corps, 2022).
Despite the creation of these special resources, health systems have poorly adapted to provide continuity for long COVID patient needs. Growing waitlists, lack of care accessibility or availability, strict referral requirements, and other administrative barriers have hindered timely care through the PCCC model alone. These problems are further compounded by general inexperience of many health care workers outside PCCCs in diagnosing multisystem COVID-related symptoms. Patients may feel dismissed when relaying their complaints. Taylor et al. (2021) reported this phenomenon was prevalent among health care workers who sought care for long COVID themselves, indicating the significance of overcoming structural barriers to novel disease treatment.
Overcoming these care barriers requires active participation from clinicians, public health experts, and patients to reframe interactions and experiences. How resources are organized and how patient feedback is received is just as critical as to what resources and knowledge are available. Long COVID patients seek confidence in health care staff being transparent with what they do not know about novel disease, often as a prerequisite in building trust. Nimmon and Stenfors-Hayes (2016) previously recommended practitioners become familiar with how to use reflective and contextual approaches to tailor their communication to patients, underscoring the importance of transparency to mitigate the impacts of a virus defined by many unknowns. Even as scientific data proliferates at a rapid pace, frontline clinicians should be clearly aware not all the information accessible is scientifically and clinically sound and therefore be cautious of bias in their decisions based on current published research alone. There is no unified diagnostic standard for long COVID and its disease heterogeneity requires a very tailored approach to each encounter.
There are opportunities to explore diverse strategies to close the gap in long COVID specialty care inequities at a systems level. Health networks which prioritize models to accommodate these populations will seek ways to normalize long COVID care in their institutions. This has the added benefit of reducing overreliance on PCCCs in communities while also acclimating patients to care with their primary care managers. By exchanging and identifying novel information or symptoms, all stakeholders can explore various avenues to aid in the diagnosis and treatment of heterogeneous disease.
Bringing the Framework Together: Clinical Practice Meets Population Approaches
The following multidisciplinary framework (Figure 1) offers several recommendations to improve upon the general long COVID patient care environment.
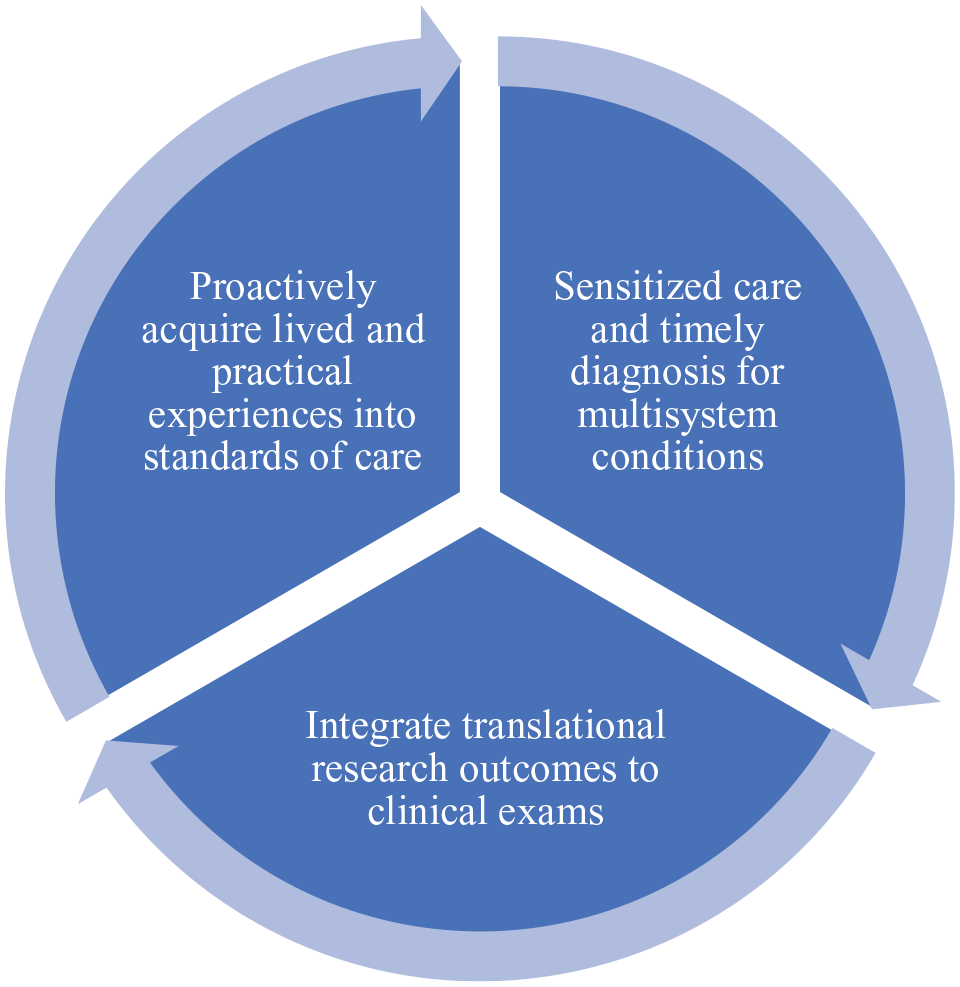
Multidisciplinary Framework for Improved Long COVID Care
Improve Patient Experiences With Timely Diagnosis and Sensitized Care to Multisystem Conditions
Multiple definitions with varying temporal requirements have been created to categorize post-infection symptoms. This has led to confusion as to when patients experience enduring disease effects. Clinicians who suspect a nexus to long COVID symptoms in patients after initial convalescence should apply diagnostic guidelines with the shortest timeline possible to support timely triage of care. In 2021, the CDC endorsed the approval of a new International Classification of Diseases, Tenth Revision (ICD-10) code, U09.9, post-COVID-19 condition, unspecified (American Academy of Physical Medicine and Rehabilitation [AAPM&R], 2021). Appropriate use of this new code promotes legitimacy among patient populations while reducing administrative barriers when seeking specialty treatment.
Varying Definitions and Criteria of Long-Term Effects Related to COVID-19 Infection

It is more important to sensitize those in clinical and public health communities to the increasingly common nature of long COVID than to provide precise diagnostic instructions for all cases. Staff should be sensitive to patient experiences and incorporate attitudes of inclusion in communication. Over time this will allow for ubiquitous exposure to the breadth of clinical manifestations and associated benefit of improved care capacity to offer specialty assessment and treatment. Failing to expand this knowledge to broader audiences outside long COVID specialists presents quality outcome risks. Patients’ health complaints may go unheard and they may not receive referrals for specialty care.
Restore Use of Detailed Clinical Examinations and Tailored Research to Support Quality Outcomes
The absence of standardized testing for specific biomarkers presents opportunities to return to the basics of quality clinical history taking. Clinicians should devote greater time to these principles and use a low to high guidance scale approach. Patients presenting with detailed histories including infection and symptom timelines range on the low scale whereas those with less recall fall on the high scale and may require guided interviewing to obtain a clinical history. The CDC’s
Public health practitioners should also recognize the direct role they have on influencing dialogue on patient care. While their work in various areas may support population approaches, their work in research equally can also be a conduit to individual clinical decisions. Because much of the clinical literature is case related, expounding upon study limitation publication narratives, to include clinical implications for practice, when possible and in scope should be done. For long COVID, this will filter the increasing saturation of literature and reduce the need for clinicians to extrapolate relevant information, thereby improving research translation and its use in patient cases (Caulfield et al., 2021).
Turn Lived and Practical Experiences Into Future Education for All Stakeholders
The long-term outlook for care experiences will be dependent on clinical and scientific advances from lessons learned. Experienced long COVID specialists are best positioned to cross this frontier in coming years. As primary and specialty care providers get more experience in managing care, long COVID specialists may shift their attention to education and training. The first PCCCs began operations in mid-2020, with many long COVID specialists contributing great insight to present knowledge.
Accreditation bodies such as the Liaison Committee on Medical Education and the Council on Education for Public Health should consider the contributions from field experts not only include direct scientific knowledge as it is discovered but also recommendations on reducing inequities through the provision of highly sensitive care. As this framework has emphasized improving community-based outcomes through care equity and advocacy, future educational programs, both individual level and population level, should preserve these principles. Long COVID specialists may be poised to lead initial dialogue; however, it is expected all stakeholders be given a voice due to the inherent value of individual experiences.
Conclusion
The growing prevalence of individuals suffering from chronic illness after COVID-19 infection will require innovative approaches for years to come. Adaptive care models which provide quality care outcomes work best when incorporating inclusive long COVID surveillance and early treatment through a variety of professions. Allowing patients to perceive commonality in their experiences as opposed to an isolated condition will help build their confidence in health systems and reduce the risks of loss to care. As these experiences become more normalized in communities, stakeholders can expect to build greater knowledge through collaboration on the course of disease.
