Abstract
Objective:
The purpose of this feasibility pilot study was to evaluate safety and adherence of a wearable brain sensing wellness device designed to reduce stress among healthcare professionals (HCP).
Methods:
A total of 40 HCP were invited to participate in an open-label pilot study. Participants were asked to use a brain sensing wearable device (MUSE-S™) on a daily basis to reduce their stress, for a total of 90 days. Total study participation duration was 180 days. Study enrollment began in August 2021 and ended December 2021. The exploratory outcomes included stress, depression, sleep, burn-out, resilience, quality of life, and cognition.
Results:
Among the 40 HCP in study, the majority were female (85%), white (87.5%) and with an average age of 41.3 ± 11.0 years (SD). Participants used the wearable device an average of 23.8 times over a 30-day period with a mean duration of 5.8 min with each use. Study results demonstrate the positive impact of guided mindfulness using the wearable device MUSE-S™ and its accompanying application (APP). A statistically significant improvement was found for a reduction in stress (P < .001) and improvement in resilience (P = .02), quality of life (P = .003), and cognition (P < .001). The majority (91.9%) of the participants indicated they felt more relaxed after using the device, and 73% indicated they would continue to use this device at end-of-study. No adverse effects were reported.
Conclusion:
Study results show that 3 to 10 min of guided meditation during work hours through the use of a brain sensing wearable device is safe and acceptable, with associated health benefits for HCP.
Introduction
Healthcare professionals (HCP) encounter patients who are experiencing a variety of ailments at the time of the face-to-face clinical visit. Providing medical care has been associated with inherent stressors, often related to ensuring that patient’s needs and expectations are met, and exacerbated by a multitude of socio-economic, environmental, and other factors. Among these stressors are lack of sleep, increased stress, burn-out, reduction in cognition and depression, all of which may be reflective of a decrease in resilience and reduced quality of life. The purpose of this study was to introduce a new approach to HCP for addressing these health issues.
Even prior to the COVID-19 pandemic, physicians were reported to be experiencing symptoms of burnout, with a negative impact on physician health.1-3 A multitude of causes can result in physician burnout, such as adopting/learning new electronic medical records, 3 administrative responsibilities taking time away from clinical practice, 4 lack of autonomy, changes in reimbursement model 5 amongst other factors.
COVID-19 added yet another dimension to the stress experienced by physicians. Whereas stress is defined as the reaction to a particular situation in question, the accumulation of stress over time can impact the emotional state of a person by eroding the sense of wellbeing. This latter impact is often referred to as distress. A recent study 3 showed that front line healthcare workers in China were experiencing a high degree of distress in the form of depression (634 [50.4%]), anxiety (560 [44.6%]), insomnia (427 [34.0%]), and distress (899 [71.5%]). 3 The stress of caring for others while worrying about the potential ramifications of their care management decisions and the possibility of personally contracting COVID-19 and inadvertently transmitting the disease to others (friends and family included) created significant physical and mental health consequences for HCP. 6 Additional stressors that resulted from COVID-19 included the rapid evolution in the practice of medicine. 7 The use of barrier-protection, adoption of telemedicine, and decrease of income also affected the way physicians practice medicine.
Over the years, the Integrative Medicine (IM) specialty has introduced many mind-body therapies for addressing mental stress. The top recommended stress relievers include getting active, eating a healthy diet, avoiding unhealthy habits, meditation, laughing more, connecting with others, asserting yourself, practicing yoga, getting enough sleep, keeping a journal, exploring music, being creative, and seeking counseling. 8 These IM strategies can be utilized to reduce stress and potentially positively impact burnout during the COVID-19 pandemic.
Whereas it has been touted that mindfulness can be a benefit to physicians in the front line, 9 little guidance exists as to how this can be learned and practiced by HCP during such a busy time with little respite (such as during the COVID-19 pandemic). Mobile and wearable technology now offers new avenues for technology-supported meditation practice and learning. 10 Some of the current smart-applications are used as a feedback mechanism for wearables and others are designed solely as self-help applications without feedback. Biofeedback has been used as a technique to reduce stress in multiple studies, however, these were typically in laboratory settings, so the efficacy of at home biofeedback is not clear. 11 The multitude of applications marketed to deliver mindfulness meditation makes it challenging to determine the most efficacious application. 12
Muse-S™ (InteraXon Inc, Ontario, Canada) is a novel biofeedback-assisted meditation device which can increase calm state brain activity in physicians expressing high stress during the pandemic when used multiple times throughout the workday. 13 This has been tested in a prior study by some of the current study team with patients undergoing breast cancer surgery. 14 In that study, patients preparing to undergo surgery for breast cancer were randomized to 1 of 2 groups—guided meditation with the MUSE device or CD-based stress reduction education. The study found that patients in both groups had significant reduction in fatigue and stress when compared to baseline, but the sample size was too small for meaningful comparisons to be made between groups. This current pilot study was designed to test the safety, acceptability of and adherence to mindfulness self-therapy through the use of a brain sensing device among HCP. An exploratory aim was to assess changes over time in psychological outcomes among participants using this device.
Methods
Study Overview
The aim of this open label feasibility pilot study was to evaluate the safety, acceptability, adherence, and impact on health benefits of guided meditation using a brain sensing wellness wearable device by HCP during a time of high stress in a pandemic. This was a pilot study whereby participants were asked to use a wearable device (MUSE-S™) on a regular basis to reduce their stress.
In accordance with the Declaration of Helsinki, this study was reviewed and approved (ID 20-007207) by the Institutional Review Board (IRB). IRB approved written informed consent to be obtained for all study participants prior to study participation.
Setting
Potential participants were HCP in the frontline of the COVID-19 pandemic. They were recruited from a large healthcare facility in the Midwestern United States. Enrollment took place from August 16, 2021 to December 22, 2021. Study participation concluded on August 15, 2022.
Participants
Eligibility criteria for this study included those who were currently employed at our healthcare facility as HCP (physicians or nurse practitioners or physician assistants), not pregnant at time of consent, and no contraindicated comorbid health conditions as deemed by the clinical investigator. They were excluded from the study if they were currently practicing mindfulness training on a weekly or regular basis; undergoing additional interventions to improve quality of life (QoL); enrolled in another clinical or research program which intervenes on the patient’s QoL, stress or anxiety; or having an unstable medical or mental health condition such as existing eye strain, seizures, dizziness, or nausea. In addition, to be included in this study, participants needed to be able to tolerate the wearable device and conduct the assigned mindfulness meditation on a regular basis (ie, daily).
All interested HCP underwent a 10-min phone pre-screen whereby the study details were discussed, and an interview took place for study eligibility. Those who passed the pre-screen interview were invited to attend an in-person consent visit. After signing an informed consent, participants would then complete a mini-mindfulness session with study staff to ensure they understood the Muse-S™ device. Total study participation was approximately 26 weeks (screen phase was 1-2 weeks, intervention phase was 3 months, and post intervention follow up was 3 additional months). Following completion of the trial participants were allowed to keep their MUSE-S™ devices. No other compensation was provided.
Of the 43 HCP who expressed an interest in the study, 3 failed the study pre-screen (2 were enrolled in a concurrent mindfulness program and 1 self-reported pregnancy at time of pre-screen). Of the 40 who went on to be consented and enrolled in the study, 4 terminated the study during the treatment phase and 1 additional participant terminated the study early, after treatment phase was completed. All 4 who terminated early cited the reason for early termination was lack of time for using the wearable device. Two of the 3 also cited resulting headaches from the MUSE-S™ headband system—citing it did not fit properly (N = 1) and lack of sleep (N = 1).
Intervention
Muse-S™ is a clinical-grade, headband-style wireless electroencephalography (EEG) system designed to interact with a mobile device (a smartphone or tablet) ( https://choosemuse.com ). In combination with the Muse-S™ Application (App) running on iOS or Android smartphones, Muse-S™ converts EEG signals measured over the frontal and temporal cerebral cortices into measures of brain state. At the same time, Muse-S™ can also guide the user through the meditation steps. Calibrated to an individual user, Muse-S™ distinguishes between active and calm brain states to provide real-time performance feedback, and helps users realize the benefits of mindfulness practice in an engaging and accessible manner. Muse-S™ also has a photoplethysmography (PPG) sensor that tracks heartrate during meditation sessions as well.
The Muse-S™ Headband system uses a series of focus attention training methods to train the brain. Each meditation session can vary in length (as chosen by the participant), and has been shown to reduce stress, anxiety, and improve focus and productivity. 14 The Muse-S™ Headband system has 7 sensors with 4 channels of EEG data for an accurate, high-quality experience.
In this study, all participants were educated on the use of the Muse- S™ Headband systems and the accompanying Muse-S™ App. They were instructed to use the Muse-S™ Headband systems and the accompanying App about 5 to 10 min on a daily basis. During the study, participants were able to access and choose the meditation type they desired while using the Muse-S™. Under Biofeedback section the options included Body, Breath, Heart, and Mind Meditation. There was also an additional section on Guided Meditation which included over 35 meditations—including burn-out and Go-to-Sleep Meditation. The biofeedback used soundscapes to give real time feedback between active and calm brain states. Body and Breath Meditation included breathing exercises where different breathing techniques, (such as box breathing, 6-4 breathing, etc.), were taught to help the individual regulate their breath. Heart Meditation utilized an immersive soundscape wherein the participant’s heartbeat was played back in real-time in fusion with a rhythmic drum sound by synchronizing the heartbeat with the rhythmic drum beat the participants finds stillness and calm. Guided Meditation involved classic focused attention on the breath, coupled with realtime feedback to signal to participants when they were experiencing focused attention or when their mind was wandering. If mind wandering was detected, the Muse-S™ cued the individual to return to a state of focused attention (meditation state). Go-to-sleep Meditations provided different experiences that helped participants fall asleep and fall back asleep if they wake in the night.
Data Collection
Data collection included frequency of device use as well as duration of each use and choice of program accessed. In addition, the study collected data on demographics and lifestyle history; burnout as assessed by using 2 single-item measures adapted from the validated Maslach et al Burnout Inventory (MBI).15,16 QoL was measured with the validated Linear Analog Self-Assessment (LASA).17-20 Stress was measured by in 2 ways, both of which have shown good validation and reliability. The first is the 1 item Perceived Stress Scale (PSS1) 21 used at baseline as part of their lifestyle history and the 10 item Perceived Stress Scale (PSS-10)22,23 was used during the study. Resilience as evaluated through the validated Connor-Davison Resilience Scale 10 (CD-RS10).24,25 Sleep quality was evaluated by the validated Pittsburgh Sleep Quality Index (PSQI).26-29 Depression was measured by the Primary Care Evaluation of Mental Disorders (PRIME-MD) Questionnaire,30-32 which was derived from the Patient Health Questionnaire (PHQ).33,34 Cognitive testing used 3 of the 12 Creyos Health (formerly known as CambridgeBrain Science) web-based tests ( https://creyos.com ) which have been validated. The patient satisfaction survey used was adapted, and individualized, to this specific pilot study from Was it Worth it Questionnaire (WIWI). 35
The MBI was used under license with Mindgarden, Inc., the 2 single-time measures were scored on a scale from 0 (Never) to 6 (Every day). A score of 4 (Once a week) or greater was considered a “yes,” while anything less is classified as “no.” For the overall MBI score if either question was a “yes” then the overall score was classified as “yes.”), LASA is made up of 6 Likert scale questions from 0 (as bad as it can be) to 10 (as good as it can be). The PSS-1 is a validated scale of 1 question with an 11-point VAS (0 = no stress and 10 = very high stress). The PSS-10 is a validated 10 question survey with a 4-point Likert scale from 0 (Never) to 4 (Very often). CD-RS10 has 10 Likert scale questions from 0 (Not true at all) to 4 (True nearly all the time). The overall LASA, PSS, and CD-RS10 scores are calculated by adding up all survey questions. The 3 cognitive tests used in this study were: Double Trouble, Feature Match and Grammatical Reasoning. Participants were asked to undertake these 3 cognition tests at 3 time points: baseline, 3 months (end of treatment) and 6 months (end of study). An introduction was provided at the beginning of each test, which included a tutorial video. All participants were asked to complete the introduction prior to initializing the tests.
Data Analysis
The sample size of N = 40 participants for this pilot study was chosen based on the primary aim of assessing feasibility and adherence. Descriptive statistics were reported using frequencies for categorical variables and mean, standard deviation, median, and range for continuous variables. For the questions that made up the PRIME MD survey and MBI survey, McNemar’s tests were conducted between the baseline value, day 90 values, and day 180 values. Total scores for the PSS, CD-RS10, and LASA surveys were compared over time using a linear mixed model. The model used an auto-regressive 1 covariance matrix, and each participant had their own intercept. Time was the only fixed effect in the model. Linear mixed models were also used to compare the scores over time for the 3 different tests: feature match, double trouble, and grammatical reasoning. Results from a satisfaction survey are reported. All P-values <.05 were considered statistically significant. Analyses were performed using SAS (SAS 9.4).
Results
Demographics and lifestyle characteristics of the respondents are summarized in Table 1. The majority were female (85%), white (87.5%) and with an average age of 41.3 ± 11.0 years (SD). Among the 40 participating HCP, 17 (42.5%) were physicians, 17 (42.5%) were nurse practitioners or physician assistants, and 6 (15%) were therapists (N = 5) or researchers on staff (N = 1). Using the 0 to 10 Visual Analog Scale (VAS) of the PSS-1 the majority of the participants had stress levels of 5 and above (85%) at baseline.
Demographics.

The average usage was generally consistent across the different MUSE-S™ programs except for the sleep programs which were longer in duration when participants wore the headbands through the entire sleep cycle. The Guided Meditation program was the one used most often with an average usage of 43 times during the 90-day study period, followed by the Heart Meditation program (average of 12 times in the 90-day period). When evaluating use frequency, we found that the average number of times used over the first 30-day period was 23.2 (±12.4) and 57.8 (±30.6) over a 90-day period. The duration of each individual use (in minutes) was 5.8 (±2.2) for the first 30-day period and 5.9 (±1.9) for the 90 days-period (respectively). There was no correlation between average time spent using MUSE-S™ and the change in scores for resilience, stress, or QoL (data not shown).
The satisfaction survey at the end of the study showed an overall positive experience for participants. A total of 83.8% found study participation to be worthwhile, while 13.5% were unsure. An overall 94.5% thought the experience was “the same as expected” (45.9%) or “better than they expected” (48.6%) and 89.2% would recommend it to others. A total of 91.9% of participants reported feeling more relaxed after using the MUSE-S™ as well as agreeing that the intervention was useful to them. Finally, 73% of the participants wanted to use the device for a longer period of time, and the same number planned to use the device past their study participation (Supplemental Table A). When asked about improvements in the program, the comments ranged from “no changes” to more reminders to use it (Supplemental Table B).
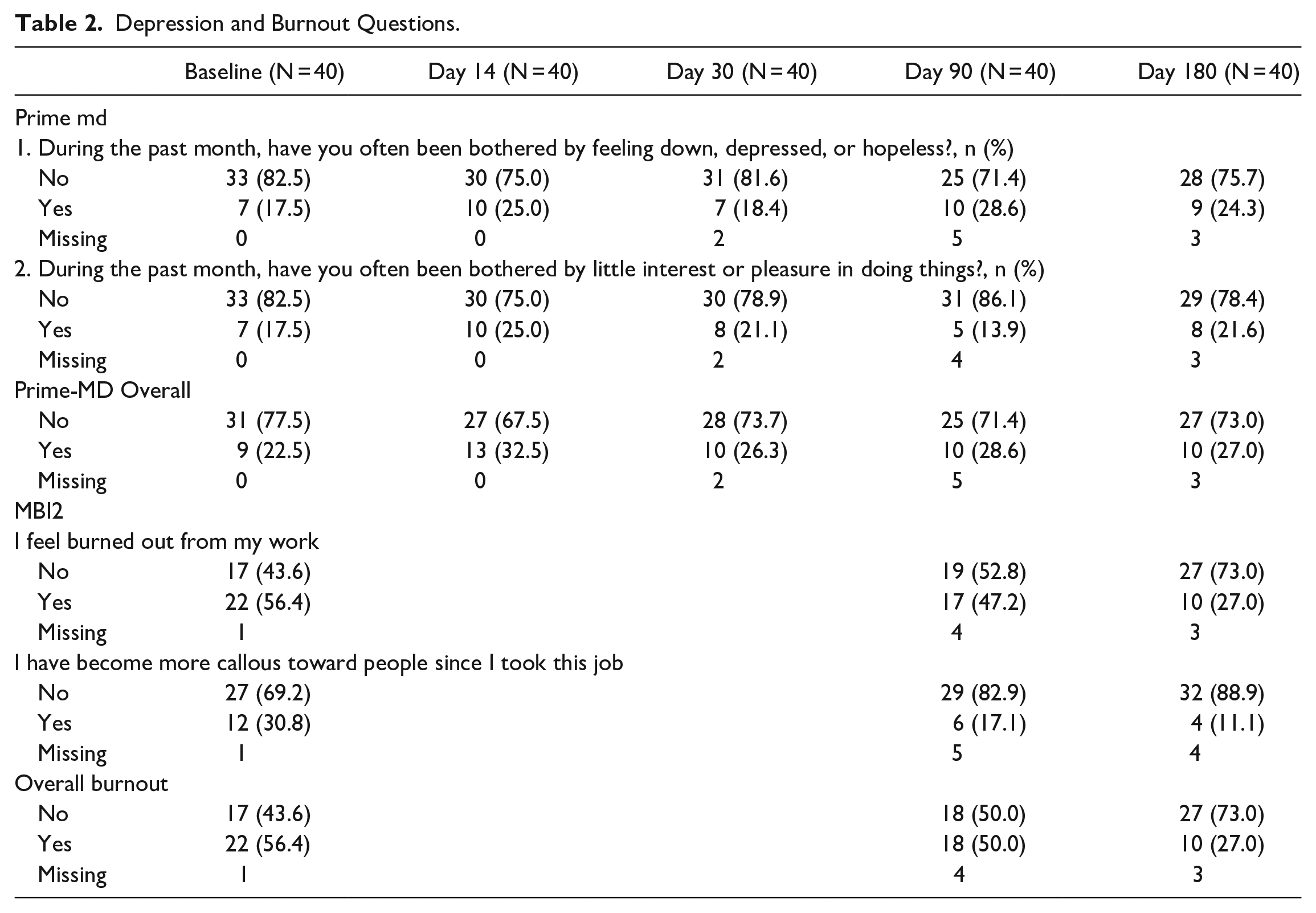
Among the exploratory variable’s evaluation, overall depression measured by the PRIME MD was compared from baseline to end-of-treatment and then to end-of-study. For end-of-treatment, there were 10 participants who changed from baseline. Seven participants experienced depression measured by the PRIME MD at the end-of-treatment but not at baseline, while 3 participants no longer experienced depression at the end-of-treatment (P = .206). For end-of-study 5 participants changed to experience depression while 2 participants changed to no longer experience depression (P = .257). Likewise, the overall burnout as measured by the MBI2 from baseline to end-of-treatment had 3 participants change to experience burnout and 5 participants changed to no longer experience burnout (P = .480). At end-of-study 1 participant changed to experience burnout and 12 participants no longer experienced burnout (P = .002) (Table 2).
Depression and Burnout Questions.
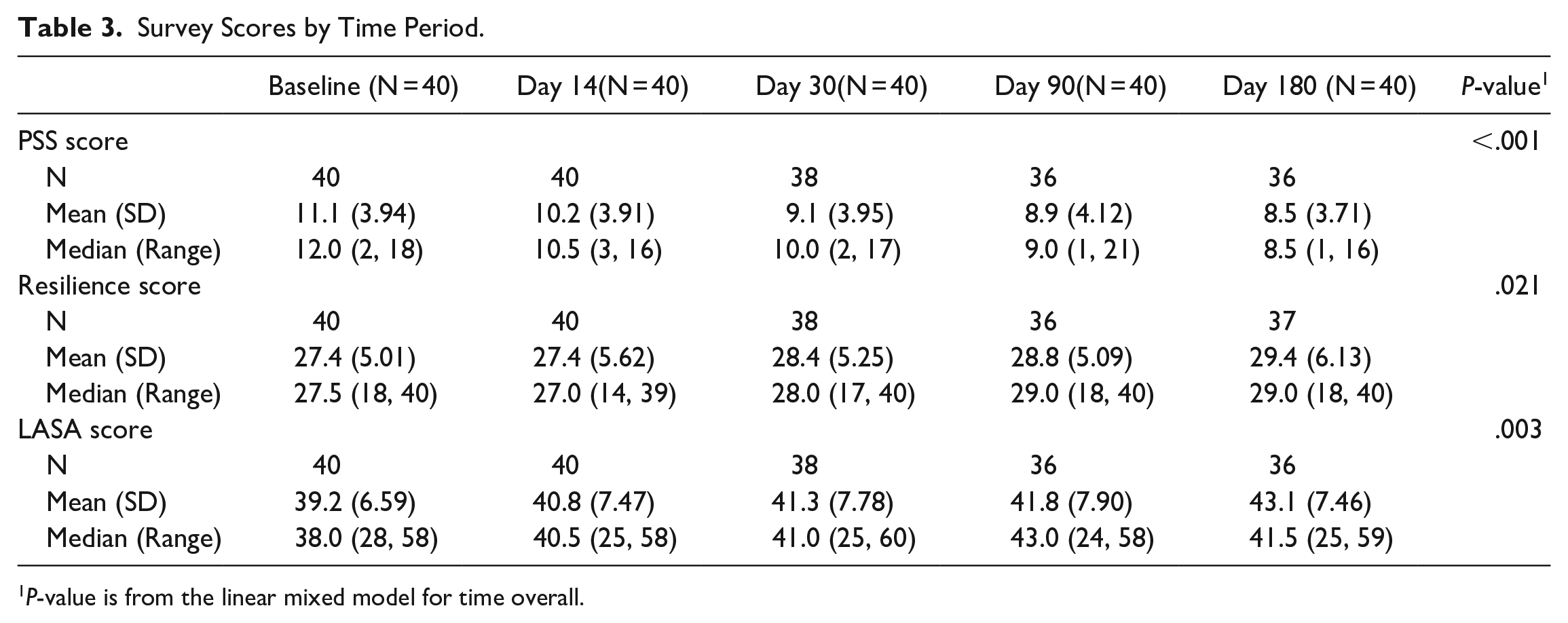
Between baseline and both end-of-treatment and end-of-study, a statistically significant improvement was observed in stress, resilience and QoL (Table 3). Stress declined from a mean of 11.1 at baseline to 8.9 at end-of-treatment (P < .001) (Table 3 and Figure 1); Resilience improved from a mean of 27.4 at baseline to 28.8 at end-of-treatment (P = .02); and QoL improved from a mean of 39.2 at baseline to 41.8 at end-of-treatment (P = .003). These improvements for all 3 outcomes persisted when testing with linear mixed model to adjust for repeated measures (Table 3).
Survey Scores by Time Period.
P-value is from the linear mixed model for time overall.
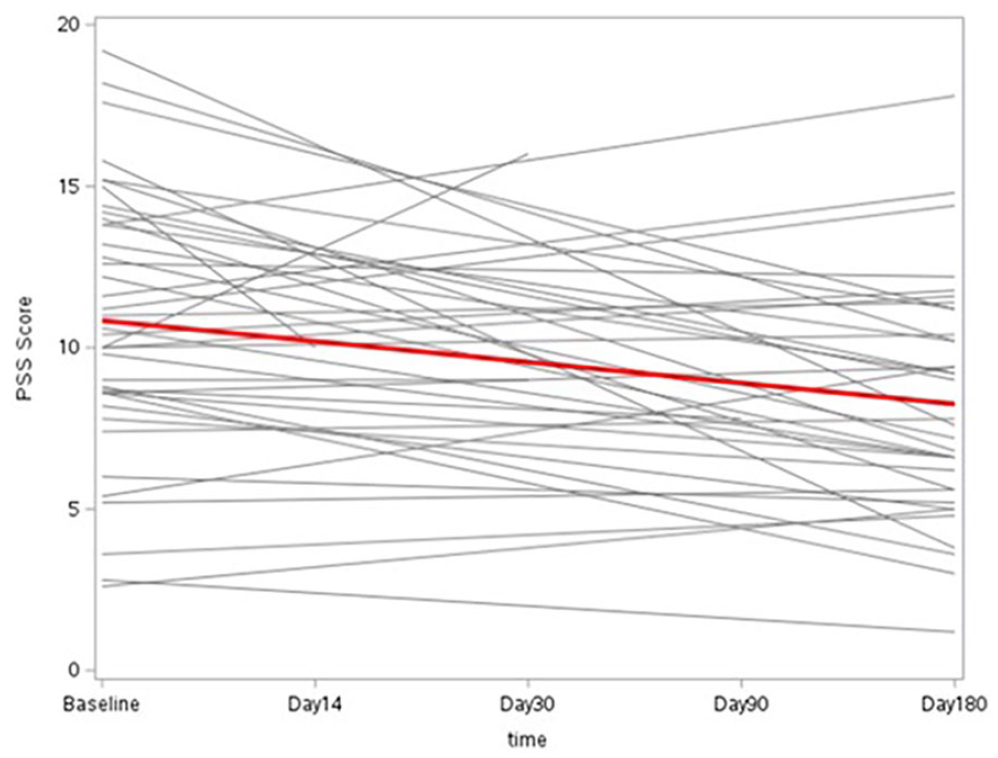
PSS-10 scores over time for each participant (red line is the trend line).
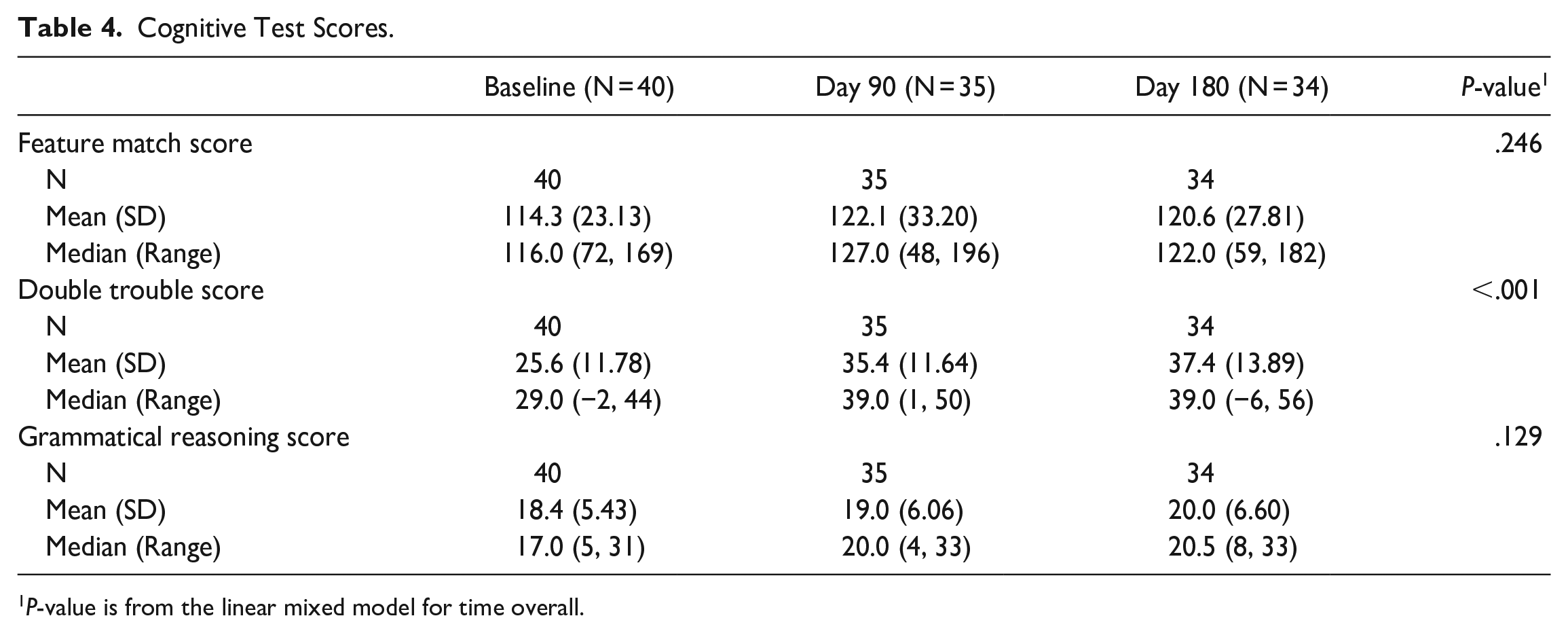
Cognitive testing did show an improvement in all 3 tests, but only 1 test showed the improvement to be statistically significant (Table 4). Cognition as tested by the Creyos test Double-Trouble, showed a statistically significant improvement from a mean of 25.6 at baseline to 35.4 at end of treatment (P < .001). These trends persisted when analyzing the data using linear mixed modeling to adjust for the repeated measures (Table 4).
Cognitive Test Scores.
P-value is from the linear mixed model for time overall.
Discussion
This study provides evidence that integrating an individualized mindfulness program through a brain sensing wearable device is safe, acceptable, and beneficial for HCP. Participant satisfaction suggests that integration of this MUSE program was well received. Adherence was high with 37 out of 40 (92.5%) HCP completing all aspects of the training indicating that this wearable device was perceived as valuable. Benefits include reduction in perceived stress, improvement in resilience and QoL, and improvement in cognition.
The improvements in stress and resilience as well as QoL observed in our current study using the Muse-S™ correlates with the extensive research on the benefits of mindfulness training which has demonstrated beneficial health impact on depression, anxiety, stress, and burnout.36-38 The reduction in stress and improvement in QoL and resilience as well as the improvement in cognition was sustained at 3 months (end-of-treatment) and 6 months (end-of-study). A recent systematic review and meta-analysis of 25 studies evaluating the effectiveness of mindfulness in reducing burnout and stress among physicians has similarly shown that mindfulness training is effective and sustainable over a minimum period of 5 months. 39
Similar to our study, there have been a number of other studies testing the delivery of mindfulness through meditation applications. A recent study randomized 68 adults to meditate with and without auditory feedback from an EEG headband and, similar to our study, found that the auditory feedback was associated with greater mindfulness by addressing the mind wandering in real-time. 40 The portability and simplicity of the system was found to be ideal for our HCP to use as needed when they had time in-between their practice responsibilities, and as needed during times of high stress. This is corroborated by a few other studies using the MUSE-S™ headband system. In one study, a total of 10 orthopedic clinicians tested the MUSE-S™ and concluded that the device’s feedback system was able to track data in real-time while providing feedback regarding the exercise and adherence but noted challenges concerning technical accuracy and tailoring the MUSE sessions/feedback to fit the rehabilitation schedule. 41 In another feasibility study, 30 breast cancer patients were randomized to MUSE-S™ or a CD-based stress reduction education from 4 days prior to surgery to 3 months post-surgery. 14 This study found that both groups had a reduction in fatigue and stress as well as improved QoL. Unfortunately, due to the small sample size the study was not powered to allow for comparison between groups. The study did find the MUSE-S™ to be feasible, effective, relatively low-cost, low risk, and practical since it required very little or no training and integrated with the person’s lifestyle. The improvement in QoL and reduction in stress were also noted to have lasting effects at 3 months post-surgery. All of these findings corroborate data from our study of MUSE-S™ among our HCP.
The current study demonstrated some significant health benefits for the HCP who used the MUSE-S™ and associated App. But it should be noted that despite attempts to recruit HCP from all race, marital statuses and sex, we did not control for any of these characteristics and due to the nature of the study sample, most of the participants were female, white, and married, therefore the generalizability of study findings are limited. In addition, the research study design inherently has its own limitations. The study team chose a single group pre-post design so that all HCP who participated would act as their own controls and everyone would receive the benefits of MUSE -S™ intervention, but—with this single group pre-post design, no data from a concurrent control data was available for comparison. At the same time, the small sample size and the decision to stay flexible with how the HCP used this intervention created some inherent limitations. This study was designed as a feasibility pilot study with exploratory aim of evaluating the device’s impact on selected health measures among HCP. With feasibility as the primary aim, the study lacked power to detect small changes in the psychological outcomes. Therefore, a larger study is needed to detect full impact of the device on HCP health. In this study, the HCP were given access to several types of guided meditations (eg, resilience, focus, sleep, burnout, etc.). Although data was collected on the specific type of meditation accessed by each individual HCP and the frequency of its use was assessed, this data could not be analyzed further due to the small sample size and variety of options given to the participants. In addition, we could not correlate if the intervention was being used during particularly stressful times since this was not tracked on a per use basis. Finally, as indicated above, though most of the HCP found the MUSE-S™ easy to use and adapt, a few participants had issues with the equipment (the headband was too tight, too loose, the sensors wore out, etc.). Additional barriers included not remembering to bring it with them during time away from work or problems finding the time to use it. Despite some of these limitations, the HCP found the intervention to be easy to use, adaptable to their work environment and helpful in improving their mental health during a stressful clinical practice during the pandemic.
Conclusion
The COVID-19 pandemic has highlighted the need for interventions to improve HCP well-being through reduction of stress. Wearable devices have a unique opportunity to deliver integrative strategies to improve QoL and resilience. Brain sensing technology is a unique type of wearable that is showing promise to positively impact the lives of HCP by delivering mindfulness techniques in an efficient and innovative way. This report of the use of a wearable brain sensing wellness device to HCP showed that the device was safe and acceptable to participants with a high level of adherence to the device. Participants noted a reduction in perceived stress and improvement in resilience and QoL. The implications of these findings include the inclusion of adding MUSE to the HCP tool kit for addressing episodic perceived stress. Further research in larger and diverse populations is warranted as a next step to better understand the benefits and expand the applications of this device.
Supplemental Material
sj-docx-1-jpc-10.1177_21501319231162308 – Supplemental material for Mindfulness Using a Wearable Brain Sensing Device for Health Care Professionals During a Pandemic: A Pilot Program
Supplemental material, sj-docx-1-jpc-10.1177_21501319231162308 for Mindfulness Using a Wearable Brain Sensing Device for Health Care Professionals During a Pandemic: A Pilot Program by Karthik Ghosh, Sanjeev Nanda, Ryan T. Hurt, Darrell R. Schroeder, Colin P. West, Karen M. Fischer, Brent A. Bauer, Shawn C. Fokken, Ravindra Ganesh, Jennifer L. Hanson, Stephanie A. Lindeen, Sandhya Pruthi and Ivana T. Croghan in Journal of Primary Care & Community Health
Footnotes
Acknowledgements
The study team would like to thank, Ariel Garten, Founder of InteraXon ( choosemuse.com ) and Mike Batista from Creyos Health ( creyos.com ) for their support and patience during this study We would also like to thank Ms. Leslie Hassett, MLS, Outreach Librarian, Education Administration, for her support during the literature search for this study and the General Internal Medicine Research team for their help in organizing and coordinating the data collection and study visits. A special thanks to all study participants; without their participation, this study would not have been possible.
Author Contributions
All the authors participated in the study concept and design, collection, analysis and interpretation of data, drafting and revising the paper for important intellectual content, and have seen and approved the final version of the manuscript. Each author will take public responsibility for the entire work.
Availability of Data and Materials
All data supporting the study findings are contained within this manuscript and within Clinicaltrials.gov (NCT04743973).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by Mayo Clinic Department of Medicine, General Internal Medicine and Integrative Medicine & Health. InteraXon provided all the MUSE -S™ devices at no cost and Creyos Health provided free access to cognition online testing for all participants. The data entry system used was REDCap, which is supported in part by the Center for Clinical and Translational Science award (UL1 TR000135) from the National Center for Advancing Translational Sciences (NCATS). Dr. Bauer’s participation was supported in part by a general grant from The Head Foundation, Singapore.
Ethics and Consent to Participate
In accordance with the Declaration of Helsinki, this study was reviewed and approved by the Mayo Clinic Institutional Review Board (IRB). Mayo Clinic IRB approved written informed consent was obtained for all study participants prior to study participation.
Consent to Publish
Not applicable.
Trial Registry Information
Trial registration: NCT04743973. Registered February 8, 2021.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
