Abstract
Introduction/Objectives:
Insomnia is a common sleep disorders that affects most individuals in the United States, and worldwide. Insomnia is linked with an increased risk of atrial fibrillation (AF) in adults, although the strengths of association were weak, especially in the elderly population. AF is estimated to affect approximately 3 to 6 million people in the United States. We studied the association of AF with insomnia in the elderly population.
Methods:
We reviewed the electronic medical records of elderly patients who received care in an internal medicine office from July 1, 2020 through June 30, 2021. Patients were grouped into AF group, and a group without AF (NOAF). Association of insomnia and other variables were compared between the 2 groups.
Results:
Among 2428 patients, 341 (14%) had AF. Patients in the AF group were significantly older compared to no-AF group (80.3 ± 7.9 vs 76.1 ± 7.4 years;
Conclusion:
AF was associated with insomnia in the elderly population. Higher frequencies of association of AF were also seen with older age, male sex, White race, CVA, TIA, dementia, CAD, CHF, other cardiac arrhythmias, COPD, OSA, CKD, anemia, and cancer.
Keywords
Introduction
Chronic, or long-term insomnia, is a common sleep disorder in which the individuals experience trouble falling asleep or staying asleep on a regular basis. It has been estimated to affect up to 60% of the adult population.1,2 It is defined as insomnia that occurs 3 or more nights per week and that lasts for more than 3 months, which cannot be explained by a comorbidity, or other health problem. Multiple risk factors are associated with insomnia, including age, sex, pain disorders, and psychiatric conditions. 3 Insomnia has been noted to increase the risk of other comorbid conditions and vice versa. The association of insomnia with atrial fibrillation (AF) is not well understood. Some studies have found that insomnia was associated with an increased risk of AF in adults, although the strength of association is weak, especially in elderly individuals.4 -7
AF affects around 3 to 6 million people in the United States.8,9 It has been classified into 4 categories, paroxysmal, persistent, long-standing persistent, and permanent AF.10,11 Paroxysmal AF is intermittent and resolves within a week of onset, persistent AF is continuous and lasts beyond a week, long-standing persistent AF is continuous which lasts beyond a year, and permanent AF is termed when no further rhythm control attempts are made after patient and clinician’s joint discussion. 11 In 2016, the global burden of AF was estimated to be around 46.3 million. 12 In the United States, it is projected that the approximate prevalence of AF will reach 12.1 million by 2030 and 16 million by 2050.9,12
The pathophysiology of AF is multifactorial involving the formation of rapidly firing of ectopic foci by re-entry circuits in the pulmonary veins 13 and cardiac tissue fibrosis that alters the conduction pathway and changes in Ca2+ channels. 14
AF is associated with multiple risk factors and comorbid medical conditions, such as hypertension, coronary artery disease (CAD), increases in atrial pressure from valvular heart disease, congestive heart failure (CHF), chronic obstructive pulmonary disease (COPD), pulmonary embolism (PE), obstructive sleep apnea (OSA), increasing age, chronic kidney disease (CKD), and thyroid disease. 9 Some modifiable risk factors include heavy alcohol use and smoking.9,12 The association of psychosocial factors, such as post-traumatic stress disorder (PTSD), has also been observed in veterans who had greater incidence of AF under psychological stress.12,15 AF increases the mortality risk, usually from associated complications from comorbidities such as thromboembolic events, CHF, CKD, and cancer.9,12 Hence optimizing modifiable risk factors is strongly recommended, both for primary and secondary prevention of AF. 10
There are only a few studies that have reported the association of AF with insomnia,16,17 and none have focused in the elderly population. In this study, our aim was to study the association of AF with insomnia in the elderly patients.
Materials and Methods
Study Design and Setting
This study was a retrospective cohort study of the entire group of elderly patients and who received medical care in a single internal medicine office located in a suburban setting. Using electronic medical records with patients that had an encounter during the timeframe allowed for convenience sampling of the subjects.
Participants
We included patients who were 65-year-old or older and had an office visit from July 1, 2020 through June 30, 2021. Patients who were younger than 65 years of age were excluded.
Variables
For each patient, we retrieved the data on the demographics, social factors, vitals and laboratory parameters, comorbid medical conditions, and use of certain medications. We used Microsoft Excel (2016, Redmond, Washington, USA) spreadsheet to record each data point.
Data Source and Access
Our Institutional Review Board (IRB) approved the study. The material that was collected was only for the research purposes as permitted by the IRB. Informed consent waivers were granted by the IRB. The study complied with the code of ethical standards of the IRB. The study investigators had access only to the selected data based on the unidentified list of patients provided by the medical informatics team, who followed the study inclusion criteria.
Bias
In order to minimize possible confounders and inaccuracies in identifying the diagnosis of chronic insomnia, we excluded patients who had been diagnosed with insomnia in less than 3 months preceding the documentation of diagnosis of AF. Patients who had a diagnosis of insomnia after the diagnosis of AF were also excluded. Additionally, we excluded patients who were terminally ill, or had metabolic encephalopathy.
Study size
The study population consisted of the entire population of 2428 elderly patients.
Statistical Methods
We computed the statistical analysis in Statistical Package for the Social Sciences (SPSS, version 15.01, IBM, Armonk, New York, USA) software. The patients were placed into 2 groups. The first group comprised of patients with AF, while the second group consisted of patients without AF (NOAF). Testing the skewness of data distribution was done for continuous variables. An independent t-test was used for analysis when data was normally distributed with values between 1 and −1. Mann Whitney
Results
There were 2428 patients in this study. The AF group had 341 (14%) patients and the NOAF group had the remaining 2087 patients (Table 1). The mean age was significantly greater in the AF group (AF = 80.3 ± 7.9 vs NOAF = 76.1 ± 7.4 years;
Baseline Characteristics.

Abbreviations: ALT, ALANINE transaminase; AST, aspartate aminotransferase; BMI, body mass index; BP, blood pressure; eGFR, estimated glomerular filtration rate; Hb, hemoglobin; HDL, high-density lipoprotein; LDL, low-density lipoprotein; n, number of patients; SD, standard deviation; TG, triglycerides; TSH, thyroid stimulating hormone; Vit B12, vitamin B12; Vit D, vitamin D.
Analysis of associations with comorbid medical diagnoses revealed a significantly higher frequency of association of insomnia with the group of patients with AF than NOAF (14.1% vs 9.5%;
Comparative Association of Comorbidities.

Abbreviations: Anxiety Dis, anxiety disorder; Bipolar Ds, bipolar disorder; CAD, coronary artery disease; CHF, congestive heart failure; Chronic pain dis, chronic pain disorder; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; CVA, cerebrovascular accident; DM, diabetes mellitus; GERD, gastroesophageal reflux disorder; n, number of patients; OSA, obstructive sleep apnea; Other Rheum Ds, other rheumatological disorders; Parkinson’s Ds, Parkinson’s disease; TBI, traumatic brain injury.

Frequencies of associated medical diagnoses.
The usage of certain medications in the group with AF was higher than the NOAF group, such as antihypertensives (83.0% vs 66.6%;
Medication Use Between the 2 Groups.

Abbreviations: CNS, central nervous system.; LABA, long acting beta2 agonist; n, number of patients; Non-Benz sleep med, non-benzodiazepine sleeping medication; OHA, oral hypoglycemic agent; PPI, proton pump inhibitor; SABA, short acting beta2 agent.
A logistic regression model demonstrated 1.97 times greater odds of AF in patients with insomnia (OR = 1.972, 95% CI = 1.360-2.851;
Influence of Insomnia and Other Comorbidities on AF.
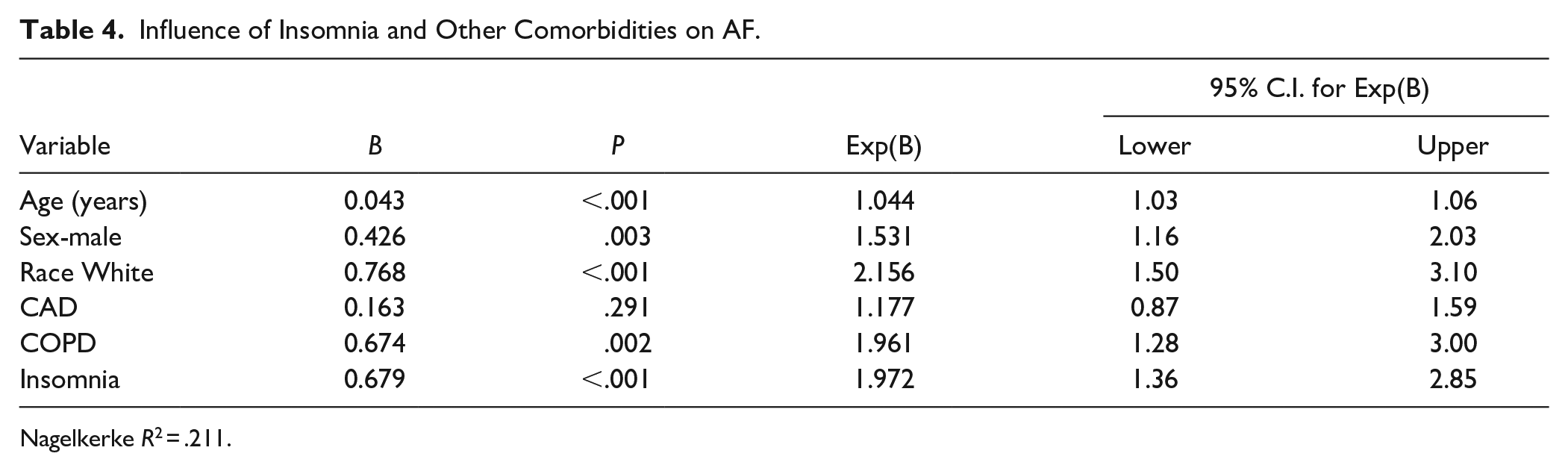
Nagelkerke
Discussion
We found that AF was associated with insomnia among the elderly patients. Additionally, we found that AF was also associated with older age, male sex, White race, CVA, TIA, dementia, CAD, CHF, other cardiac arrhythmias, COPD, OSA, CKD, anemia and cancer in the elderly patients.
Our findings of association of AF with insomnia align with a large cohort study of patients of all ages, majority being younger than 65 years-old, that found a markedly greater rate of occurrence of AF among individuals with insomnia (insomnia group = 2.6% vs. no insomnia group = 2.3%,
Multiple mechanisms have been proposed to explain the pathogenesis of AF in insomnia due to a lack of clear understanding. It has been largely observed that the relationship between AF and insomnia happens to be bidirectional as the cardiovascular changes that might influence sleep. AF can be promoted by autonomic dysfunction from increased sympathetic nervous system (SNS) activity leading to physiologic hyperarousal and sleeplessness in insomnia.6,14,18 SNS upregulation from catecholamines can support re-entry pathways in AF and promote wakefulness in insomnia.6,18 Additionally, it has been shown that insomnia and shorter sleep duration can lead to hypertension. 19 Physiological hyperarousal, marked by the hyperactivity of hypothalamic-pituitary-adrenal (HPA) axis and sympatho-adrenal-medullary axis, is predominantly found in patients with insomnia who have objectively short sleep duration, especially the elderly. 20 This hyperarousal involves increased levels of stress hormones, such as cortisol and adrenaline, contributing to hypertension. In individuals with reduced sleep, negative changes in metabolism, imbalance between the sympathetic and vagal nervous systems, and dysfunction of the endothelium, lead to hypertension, which in turn leads to remodeling, fibrosis and loss of atrial muscles, thereby, predisposing AF. These issues are driven by molecular mechanisms involving inflammatory cytokines and oxidative stress, which interact intricately with the circadian clock genes. 21
Another observation reports that the variations in the HPA axis from the stress of fragmented sleep duration could lead to an increase in cortisol and variability in heart rate, which helps in promoting a more heterogeneous atrial conducting system for AF to persist.6,14 Sleep duration also appears to have some involvement in AF as both decreased and increased sleep durations have been associated with AF, with shorter durations having higher risk. 5 A Doppler study demonstrated that acute sleep deprivation was associated with a decreased left atrial early diastolic strain, which suggests that chronic sleep deprivation might have a greater impact on the left atrial function and development of AF. 22 Our finding of markedly increased odds of AF exclusively among the elderly patients with chronic insomnia is unique and it highlights the need for further research of such association in different settings.
Older age has been reported as a risk factor for AF. About 8.8% of individuals aged 80 years and older in the United States are affected by AF.
23
The lifetime risk of developing AF is approximately 1 in 4 for individuals aged 40 years and older.
24
A retrospective study concluded that that for each unit increase in age, there was 4.5% odds of developing AF (95% CI = 2.2%-6.9%,
Multiple studies have observed that males were at a higher risk for AF than females.9,12,26 Higher level of endogenous circulating testosterone has been reported to be associated with increased risk of AF, 27 while there was a negative correlation in females. 28 While there are known electrophysiological differences between males and females, and limited research in atrial remodeling between sexes, the finding of post-menopausal women having higher incidence of AF suggest that pre-menopausal hormones are protective. 29 Additionally, men typically have a QT interval that is 10 to 20 ms shorter than women. This difference in cardiac muscle repolarization emerges during puberty and is believed to involve the influence of androgenic hormones on heart function. 29 Ethnic differences in left atrial (LA) size could also partially explain differences in the incidence of AF, as other ethnic groups have been seen to have smaller LA size when compared to body mass index. 12 Our finding of higher association of AF with White race is consistent with the current literature.
In our study, there was a significantly increased association of CVA and TIA with AF which has been widely reported.9,10,12,14 In this scenario, AF is the risk factor which promotes embolization of thrombi which forms in the left atrial appendage and results into CVA and TIA.30,31 Additionally, our group of patients with AF had a greater association with dementia. There are reports that risk of dementia increases in AF even in the absence of CVA or TIA.12,14 Compared to those in sinus rhythm, patients with AF without prior CVA have 2.3 time greater risk of dementia and 1.7 to 3.3 time greater risk of cognitive impairment. 14 Patients with AF and a history of CVA are even at a greater risk for dementia. 12 There are reports of an increased risk of dementia in patients with AF diagnosed after CVA compared to those without AF. 32 The major mechanisms includes cerebral hypoperfusion and hypoxia from microembolization.12,14 The AF-SCREEN collaboration proposes that there may be an interplay between cerebral hypoperfusion, microhemorrhage, and inflammation which may lead to cerebral microinfarcts and decreased brain volume. 24 The transient variability in cardiac cycle beat-to-beat due to AF may alter stroke volume, decreasing cerebral perfusion and chronically over time may predispose to dementia. 33 Other mechanisms include repetitive and chronic formation of micro- and macro-clots, or micro- and macro-bleeds due to preventive medical therapies for CVA and TIA, which suggests that cognitive decline and dementia in AF happen to be a part of the disorders that include CVA and TIA. 34
Our findings showed an increased association of AF in patients with CAD. CAD is an established risk factor that increases the risk of AF (population attributable fraction of 3.6%; history of MI: HR = 1.64; genetically predicted CAD: OR = 1.18). 10 In individuals diagnosed with AF, the prevalence of coexisting CAD ranges between 17% and 46.5%. Conversely, among patients with CAD, the prevalence of AF is comparatively low (0.2%-5%). 35 CAD is among the cardiovascular diseases where there is a bidirectional increase risk of development of AF.9,12 AF with rapid ventricular rate can lead to decreased coronary perfusion. 12 Thromboembolism may also predispose to CAD and increase the risk of MI. 12
We found a higher association between AF and the presence of CHF. AF afflicts over half of those suffering from CHF, while CHF is found in over one-third of individuals experiencing AF. 36 CHF is associated with various ion channel current abnormalities including an increased Na+/Ca2+ exchange current, creating a net positive intracellular imbalance. This imbalance predisposes to delayed afterdepolarizations that result into triggered activity and arrhythmias. 37 Additionally, increased pressure in the left atrium and pulmonary veins associated with CHF promotes AF. The stretching of myocytes enhances the heightened electrical activity of the ectopic foci in the pulmonary veins via mechano-electrical feedback. 38 Additionally, myocyte stretch leads to increased fibrosis and structural remodelling, including left atrial enlargement, which predisposes to AF. 39
In our study, there were higher odds of AF with COPD. The prevalence of AF in stable COPD is 4.7% to 15%, but increases notably to approximately 20% to 30% among individuals with severe COPD. 40 A study on COPD and the development of AF revealed that patients with COPD had 28% increased risk of AF, with this risk further exacerbated by frequent exacerbations and an enlarged left atrium. 41 Another study found that COPD was associated with a significantly elevated risk of developing AF (risk ratio = 1.74; 95% confidence interval: 1.70-1.79). 42 Hypoxia plays a pivotal role in COPD, contributing to atrial remodelling and potentially leading to AF. Various molecular pathways activated by COPD-related hypoxia, such as hypoxia-inducible factor 1, transforming growth factor β1 (TGF-β1), vascular endothelial growth factor (VEGF), and matrix metalloproteinases (MMP), are implicated in the fibrotic changes observed in the atrial tissue. 43 Hypercapnia in COPD induces a consistent increase in atrial refractoriness and significantly slows atrial conduction. 44 Both hypercapnia and hypoxemia cause constriction of pulmonary arterioles, resulting in pulmonary arterial and right ventricular hypertension, 45 which may precipitate arrhythmias by promoting dilation of the right atrium and altering transmural pressure on endocardial vessels, leading to disruption of the distribution of blood flow. 46 Additionally, disturbances in electrolyte levels stemming from administration of corticosteroids or beta-blockers might result in hypokalemia in individuals with COPD which contributes to prolonged P-wave duration, which has been reported as a risk factor for AF. 47
We found that AF was associated with OSA. Association of OSA has been described as a comorbidity in 21% to 74% of patients with AF.
48
Other studies have reported 88% increased association of OSA with AF.49,50 A meta-analysis reported a significantly increased risk of AF in patients with OSA (OR = 2.120; 95% CI = 1.845-2.436,
We found an increased association between AF and CKD. This association has been widely reported and established.54 -56 Renal dysfunction promotes initiation and maintenance of AF due to cardiac remodelling, while uncontrolled AF accelerated renal dysfunction.54 -56
Our study demonstrated a significant association between AF and anemia. Anemia has been correlated with an increased likelihood of thromboembolic and hemorrhagic events in individuals with AF who are on anticoagulant treatment. 57 In patients with AF of non-valvular origin, the incidence of anemia has been reported as 13% to 34%. 58 A retrospective study examining critically ill patients found that the incidence of AF was 12.8% in the patients with anemia, compared to 9.9% in those without anemia. 59 Studies have indicated that patients with AF exhibit heightened lympho-mononuclear cell activity, increased myocyte death, elevated inflammatory markers, and higher neutrophil-to-lymphocyte ratios compared to individuals without AF. 60 Inflammatory factors such as interferon-γ, lipopolysaccharide, and tumor necrosis factor-α increase divalent metal transporter 1 expression, enhancing iron uptake by macrophages. 61 This results in excessive iron retention in the reticuloendothelial system, limiting its availability for red blood cell production. Inflammatory cytokines, such as interleukin-1 and tumor necrosis factor-α further exacerbate anemia by inhibiting erythropoietin production and promoting red blood cell destruction. 62 The current body of literature suggest a bidirectional association of anemia with AF.
Finally, we found an increased association of AF with cancer. There are reports that 30% of individuals with cancer develop AF. 63 The occurrence of AF in patients with history of any cancer varies based on the type of cancer, chemotherapy regimen and any surgical intervention. A study reported 12.6% prevalence of cancer in patients with AF compared to 5.6% in patients without AF. 64 Among those, ill-defined cancer (4.3%), prostate cancer (2.7% in males), breast cancer (2.3% in females), colon-rectum cancer (2.1%), and lung cancer (2.0%) were the most prevalent types. Another population-based study reported a 44% higher risk of developing AF a year after the cancer diagnosis, and 8% greater risk of AF 5 years after the cancer diagnosis. 65 Several pathophysiological links explain the association between cancer and AF. Risk factors such as pre-existing cardiovascular disease, aging, obesity, diabetes, alcohol consumption, and smoking can be classified as shared risk factors which may account for a significant part of this association. 66 Additionally, cancer patients experience specific disorders, such as pain, hypoxia, electrolyte imbalances, and malnutrition, which can lead to autonomic and endocrine metabolic abnormalities contributing to AF. 67 A meta-analysis showed that patients with solid tumors had 1.47 times greater odds of developing AF compared to those without. 68 Strong association between cancer and AF emphasizes the need for integrated care approaches that address both oncological and cardiovascular health.
There were a few limitations in our study. Our elderly patients represented a suburban population, hence our findings cannot be generalized. Although the comorbidities were entered chronologically in the problem lists of each patient; nevertheless, some data required further verification by reviewing the progress notes. A large sample of patient cohort from 1 internal medicine office was the major strength of this study, in which all patients were treated by a group of longstanding physicians in the practice who documented all the diagnosis chronologically which helped the study team in identifying the data of the onset of each medical problem.
Conclusions
We conclude that AF was associated with insomnia in elderly population. Higher frequencies of association of AF were also seen with older age, male sex, White race, CVA, TIA, dementia, CAD, CHF, other cardiac arrhythmias, COPD, OSA, CKD, anemia, and cancer. Early diagnosis and treatment of insomnia and additional modifiable risk factors may offer a favorable outcome in the elderly patients.
Footnotes
Acknowledgements
The authors thank Christine Rickette, RN (study coordinator) for her contribution to this study.
Author Contributions
JN and SR made substantial contributions to the study design, drafting, data acquisition, data analysis, and manuscript writing. All authors except PK contributed in data collection and manuscript writing. PK contributed in manuscript writing. KH analyzed the data. SR contributed in revising the manuscript critically for improved intellectual content, and final approval for the version to be published.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable. Being a retrospective chart review study the Institutional Review Board waived the need for informed consent.
Data Availability
The authors declare that data supporting the findings of this study are available within the article.
