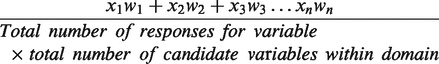Abstract
Background
New-onset atrial fibrillation (NOAF) is common during critical illness and is associated with poor outcomes. Many risk factors for NOAF during critical illness have been identified, overlapping with risk factors for atrial fibrillation in patients in community settings. To develop interventions to prevent NOAF during critical illness, modifiable risk factors must be identified. These have not been studied in detail and it is not clear which variables warrant further study.
Methods
We undertook an international three-round Delphi process using an expert panel to identify important predictors of NOAF risk during critical illness.
Results
Of 22 experts invited, 12 agreed to participate. Participants were located in Europe, North America and South America and shared 110 publications on the subject of atrial fibrillation. All 12 completed the three Delphi rounds. Potentially modifiable risk factors identified include 15 intervention-related variables.
Conclusions
We present the results of the first Delphi process to identify important predictors of NOAF risk during critical illness. These results support further research into modifiable risk factors including optimal plasma electrolyte concentrations, rates of change of these electrolytes, fluid balance, choice of vasoactive medications and the use of preventative medications in high-risk patients. We also hope our findings will aid the development of predictive models for NOAF.
Background
New-onset atrial fibrillation (NOAF) is a common phenomenon in critically ill patients. 1 It has deleterious haemodynamic effects 2 and is associated with thromboembolic events. 3 It is independently associated with intensive care unit (ICU) and hospital mortality.4–6
Many risk factors for developing NOAF during critical illness have been identified. These include demographic and comorbidity-related factors, along with acute factors such as disease severity.4,7,8 The evidence for modifiable predictors is scarce as potential targets for prevention have not been studied in detail. 9
In prognostic factor research, variables are often studied because they are readily available. Systematic reviews are helpful to identify risk factors evident in the available literature. However, they can only identify those factors that have already been studied. They therefore have limited value in identifying direction for future study. This can result in established variables being repeatedly identified, but with limited extension of knowledge.
An expert consensus can help provide informed direction for future study. The Delphi process is an established method to develop expert consensus. 10 The key principles behind the Delphi method are that group consensus is more valid than individual opinion, and that structured communication is an effective way to address complex problems. 11 Five to fifteen experts are considered adequate to achieve content validity.12,13 Delphi processes have previously been used to inform critical care practice including management of COVID-19, 14 preventing pressure injury 15 and identifying core outcome measures after respiratory failure. 16
The aim of this study was to gather expert consensus to identify important risk factors for NOAF during critical illness. By identifying variables that may not have been studied in this population we hoped to guide future research into NOAF prevention.
Methods
We used the Conducting and REporting DElphi Studies (CREDES) guidance to plan and report our Delphi process. 17 The study protocol is available online. 18 We used an online survey tool (Survey Monkey®) for data collection and analysis.
Panel selection
We identified potential participants from the author lists of articles included in a recent systematic review of risk factors for NOAF in patients in an ICU. 9 We also identified experts through national research networks. We selected participants predominantly representing professional groups that are directly involved with patient care.11,19 The final panel included general intensive care physicians, cardiac intensive care physicians and cardiologists. We used the Scopus database 20 to present H-indices demonstrating author-level publication metrics whilst maintaining anonymity. To confirm appropriate panel expertise we performed a search of the Medline database for publications by the panel members focussing on atrial fibrillation. We conducted this Delphi study entirely through electronic contact and data capture.
Procedure
A flow chart illustrating the rounds of the Delphi process is displayed in Figure 1.
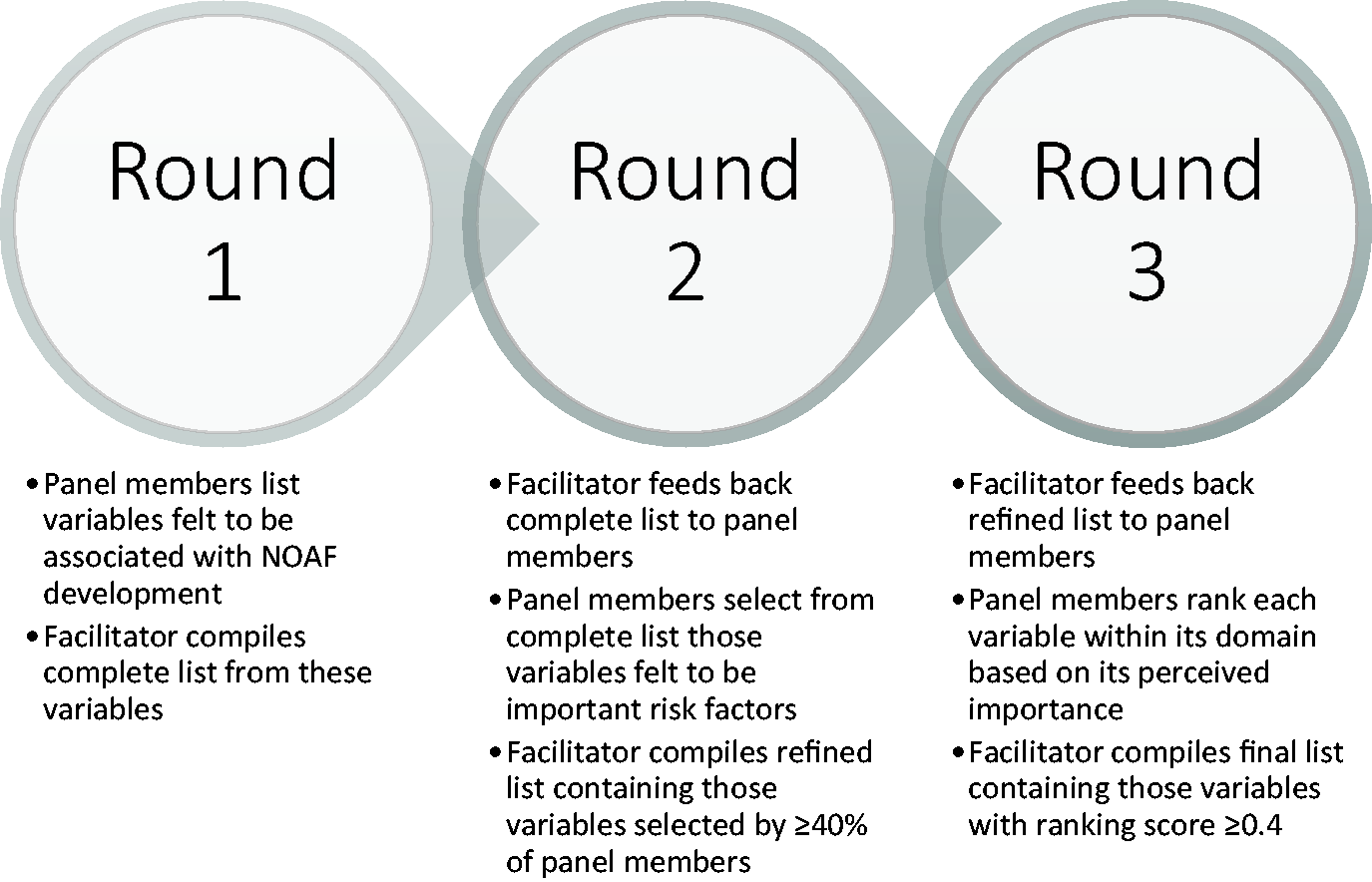
Flow chart of Delphi process.
Round 1: Panel members were asked to spontaneously list factors that affect NOAF risk in critically ill patients. We permitted potential protective factors as suggestions. We offered panel members six categories (demographics; comorbidities; disease, and disease-severity-related variables; investigation results; interventions; and vital signs) to provide structure to their answers. Similar variables were individually retained within domains where they could be studied independently. Similar variables across domains were retained should interest lie in one particular domain.
Round 2: We provided each panel member with the complete list of variables generated by the panel during round 1. We asked each panel member to select variables they felt affect NOAF risk in critically ill patients. We refined the list by only retaining variables selected by at least 40% of panel members.
Round 3: Variables that met retention criteria in round 2 were fed back to the Delphi panel. We asked panel members to rank each variable by perceived strength of association with NOAF development during critical illness. We ranked variables within each variable’s domain (for example we ranked demographic factors and comorbidity factors separately).
Average ranking
We calculated a normalised average ranking score to determine which answers in each domain were most preferred by the panel. 21 Possible scores range from 0-1. The answer choice with the largest average ranking score per domain represented the most preferred choice.
We calculated the ranking score for each variable as follows:
1,2,3…n = rank choice
x = Number of votes for rank choice
w = weight of ranked position. Weights are the inverse of respondents’ chosen ranks. Position 1 receives the highest weight which equals the number of rank choices. E.g. in a question with 3 candidate variables,
To produce a final list of variables felt to be important predictors of NOAF we retained variables with an average ranking score of 0.4 or higher. The average ranking score ensured variable exclusion was not inevitable i.e. all variables within a domain could be carried through to the final list given sufficient spread of votes.
Results
Participants
We approached 22 experts located in Europe, North and South America, and Asia. Of these, 12 agreed to contribute to the Delphi process. All 12 respondents were active clinical researchers based in Europe, North America and South America. We identified 110 publications relating to atrial fibrillation written by members of the panel in a search of the Medline electronic database. H-indices of our panel members ranged from 2–29 (mean = 14.4, median = 14).
We first contacted panel members in November 2019, sent information for round one in December 2019, and the final list of variables was confirmed in July 2020.
Round 1
The panel responded with 126 distinct candidate variables across the 6 suggested domains. The largest domain contained variables relating to investigation results.
Round 2
All 12 of the initial participants responded in round 2. We generated a refined list of 102 candidate variables after excluding variables not selected by at least 40% of panel members.
Round 3
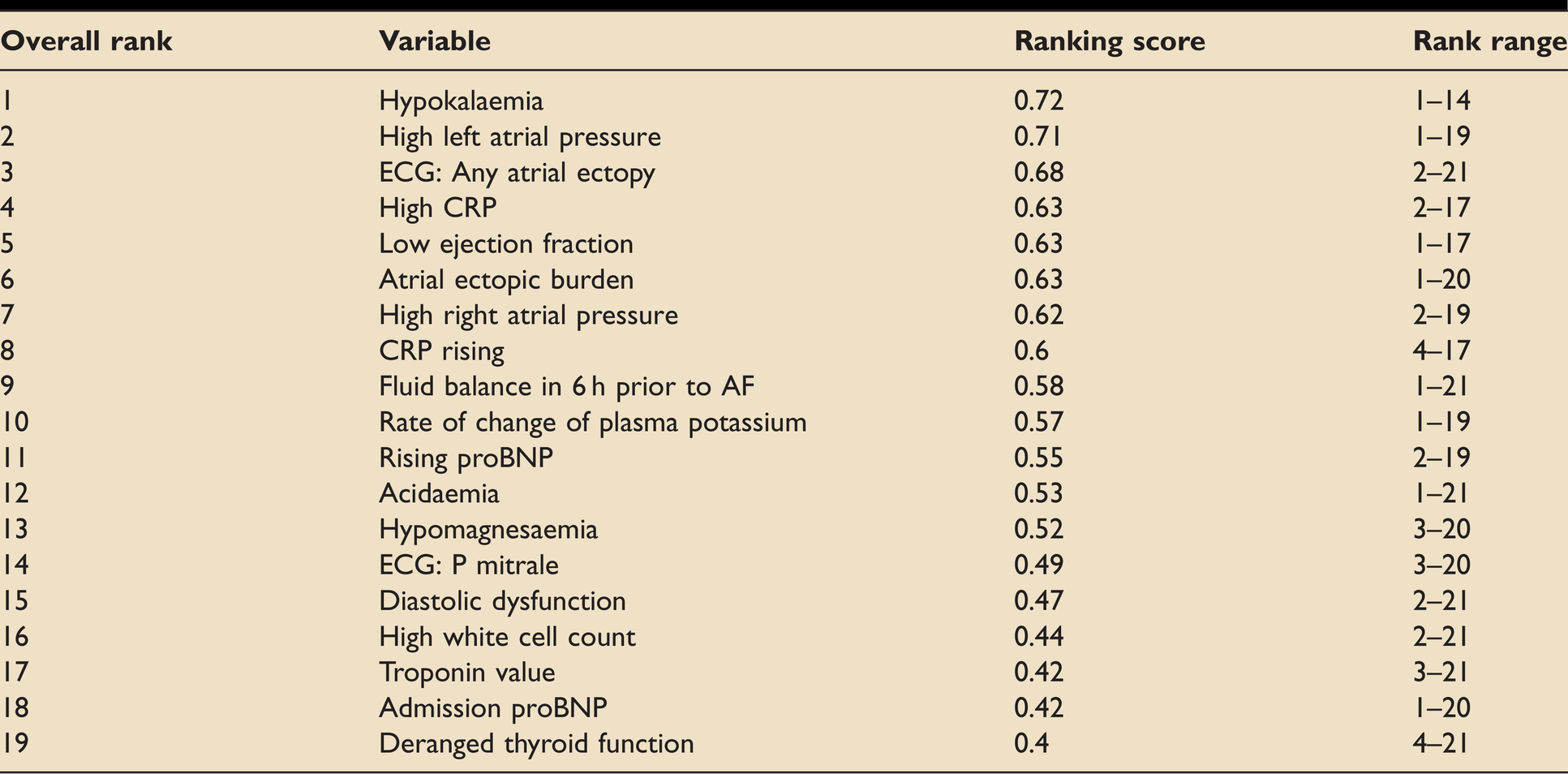
All 12 of the initial participants responded in round 3. We produced a final list of 75 candidate variables after excluding those variables with an average ranking score of less than 0.4. These factors represent the most important variables that the panel of experts felt affect NOAF risk during critical illness. Ranked results for demographic, comorbidity, disease, investigation, intervention and physiological domains are displayed in Tables 1 to 6, respectively.
Ranked demographic variables considered to affect NOAF risk.

Ranked comorbidity variables considered to affect NOAF risk.
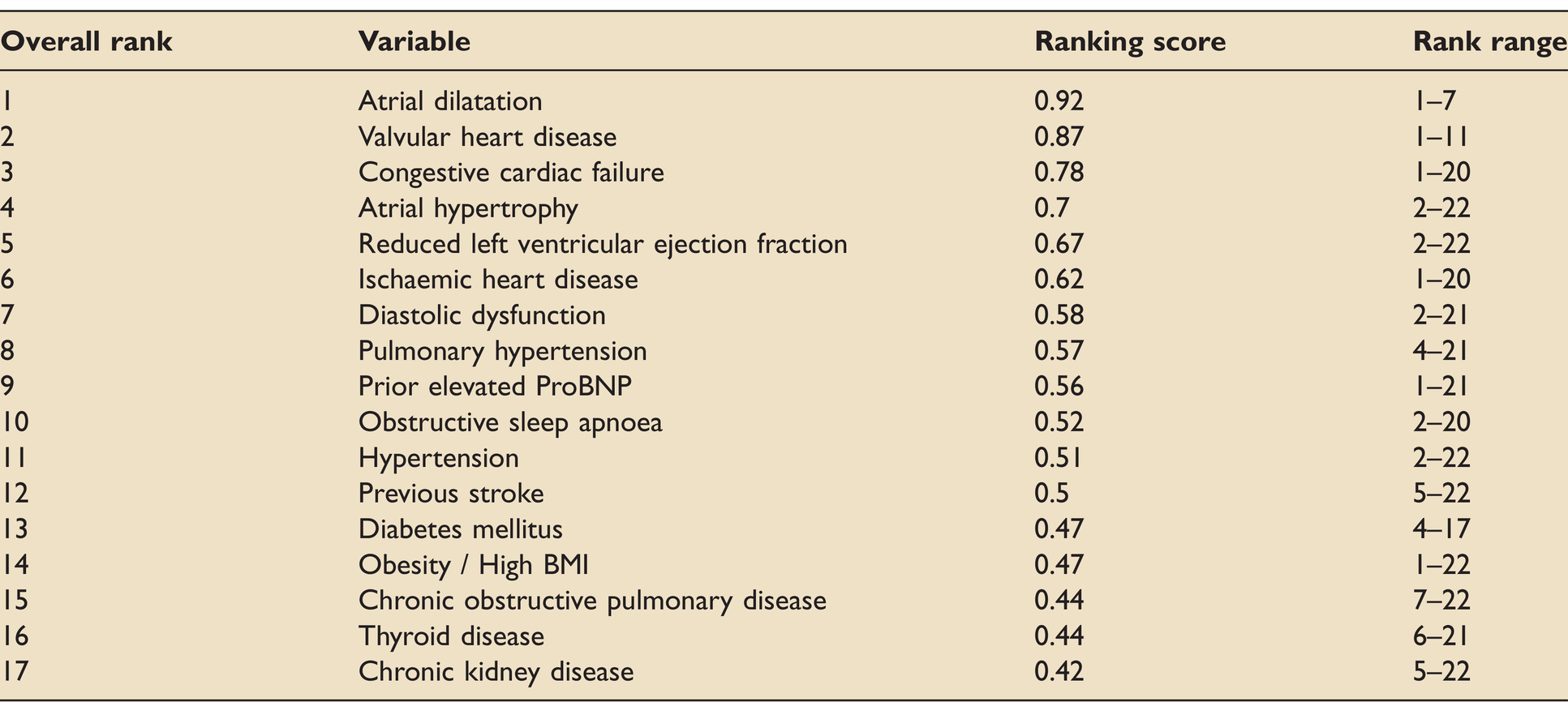
Ranked disease/disease severity variables considered to affect NOAF risk.
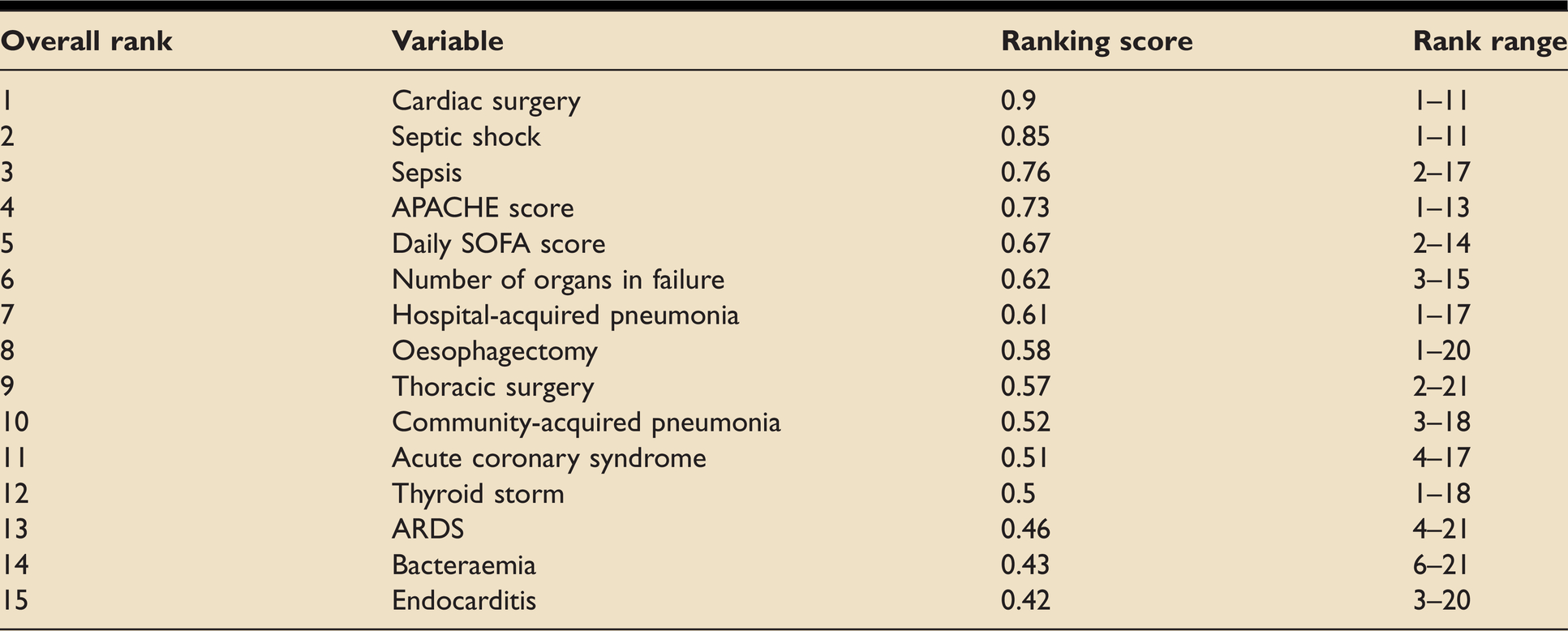
Ranked investigation-related variables considered to affect NOAF risk.
Ranked intervention variables considered to affect NOAF risk.
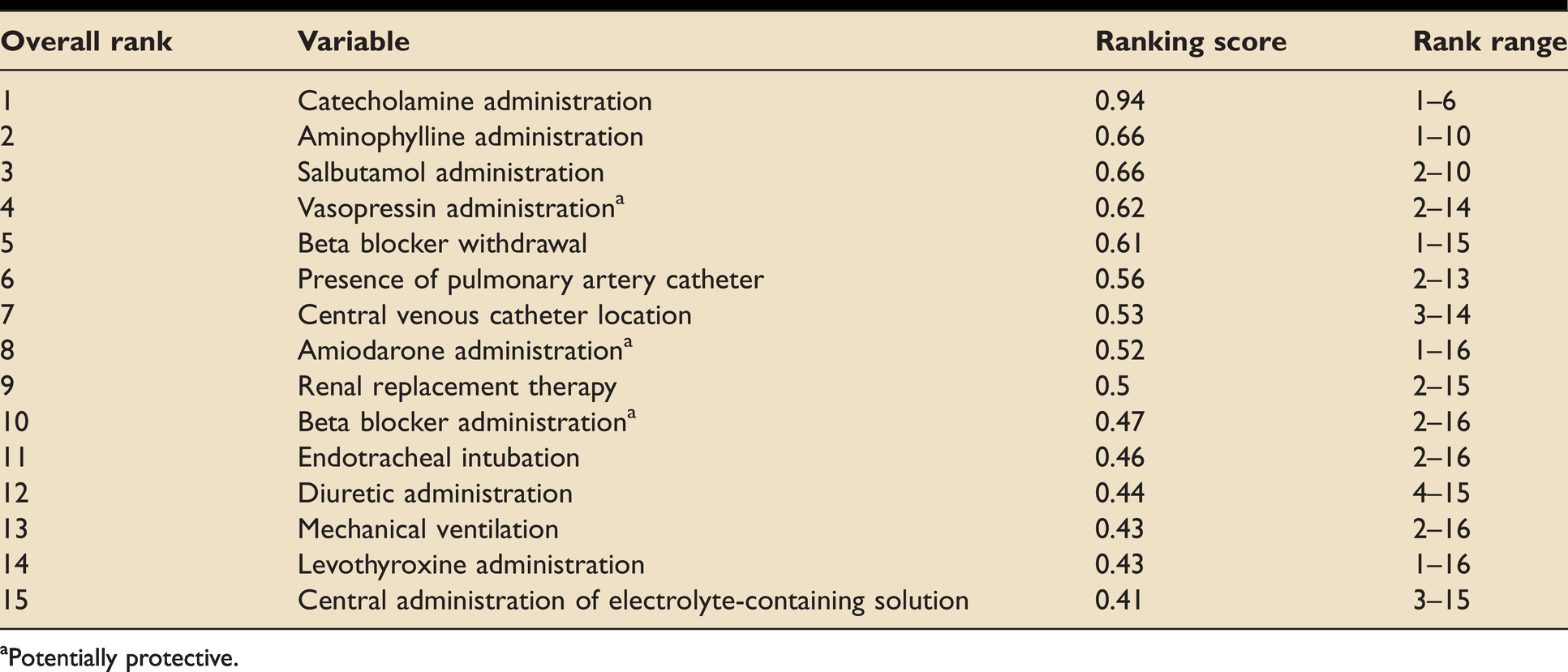
aPotentially protective.
Ranked physiological variables considered to affect NOAF risk.

Discussion
AF in critical illness is an independent risk factor for mortality and hospital length of stay, as well as an increased risk of thromboembolic events. Reducing its incidence may significantly reduce the burden of disease. In this study, we aimed to identify important variables affecting NOAF risk during critical illness. Identifying predictive factors may be used in the development of predictive models, to guide treatment, to inform future randomised trials, or as covariates in prognostic studies.
The feasibility of remote participation facilitated international involvement. The wide range of expertise across the panel provided a broad range of perspectives, maximising the benefits of the group consensus model.
The expert panel identified several factors across a range of categories. Whilst some factors have been extensively studied in the ICU population, some, for example the use of preventative medications, await investigation in this setting.
Non-modifiable risk factors
Demographic factors
The demographic factors highlighted in this study are consistent with those associated with increased risk of AF in patients in the community, such as increasing age. 22 The underlying pathophysiological changes associated with this increased risk are generally well understood. It is associated with atrial fibrosis and changes in ionic currents independent of fibrosis.23,24 These changes affect normal conduction pathways, promoting non-uniform conduction, electrical dissociation and re-entry circuits, leading to arrhythmia.24–26 Increasing age is consistently found to increase NOAF risk in patients in ICU. 9
Male sex is a recognised risk factor for AF in patients in the community. 22 This is also true of NOAF associated with critical illness. 1 Sex appears to modify how other risk factors affect AF risk, with obesity increasing risk more in men than in women. 27 Increasing height also increases AF risk and may relate to increasing atrial size. 28 Sex-dependent ion channel expression 29 and atrial refractory period duration 30 may also contribute. Future studies should consider including sex-disaggregated data when investigating modifiable risk factors for AF.
Comorbidity
The comorbidity-related factors felt to be most strongly associated with NOAF in the critically ill were related to heart disease, consistent with evidence in the community population. Atrial dilatation is a strong predictor of atrial fibrillation in the community, 31 and is thought to be due to mechanisms associated with atrial stretch. Atrial stretch causes a decrease in atrial effective refractory period and an increase in AF inducibility in animal studies. 32 Stretch-activated ion channels may be responsible for these changes. 33 This association has been demonstrated in patients in ICU with left atrial diameter increasing NOAF risk.9,34–36 Atrial wall thickness (as a marker of atrial hypertrophy) correlates with degree of atrial fibrosis, independent of atrial volume. 37 It also correlates with areas demonstrating complex fractionated atrial electrograms (CFAE), implicated in the development and maintenance of AF. 38 Atrial volume may be estimated by transthoracic echocardiography (TTE); however assessment of atrial wall thickness is more difficult. Whilst TTE has been used for this purpose, 39 the spacial resolution tends to be inadequate. Transoesophageal echocardiography, cardiac CT, and cardiac MRI can all be used to provide measures of atrial hypertrophy, 40 though their use is unlikely to be feasible for routine risk stratification for patients on an ICU. If it could be accurately measured non-invasively, atrial wall thickness could become a useful marker of NOAF risk in critically ill patients.
Conditions leading to right heart pressure overload including pulmonary hypertension, obstructive sleep apnoea (OSA) and COPD ranked highly. OSA may predispose to AF due to atrial stretch 41 and large variations in afterload. Furthermore, vagal activation secondary to negative tracheal pressure shortens the atrial refractory period and promotes AF.42,43 Pulmonary hypertension (PH) increases AF risk through increased sympathetic activity 44 and chronic right atrial pressure overload causing atrial stretch. 45 These findings are supported by studies in critical care demonstrating increased NOAF risk in patients with OSA, COPD or PH.5,46,47
Each of these comorbidity-related factors may be exacerbated in the intensive care setting. For example, excessive fluid resuscitation increasing left atrial stretch and thus increasing NOAF risk.
Disease/disease severity factors
Of the factors relating to disease or disease severity, sepsis and measures of organ failure were ranked highest. Sepsis is a known risk factor for NOAF. Understanding of the pathophysiology of sepsis continues to develop. Systemic inflammation plays a role in the initiation and maintenance of atrial fibrillation. Generation of reactive oxygen species,48,49 alteration in potassium currents by circulating cytokines,50,51 and disruption of normal electrophysiology by bacteria are some of the processes that increase arrhythmia risk. 52
Our results propose that certain infections may carry more NOAF risk than others, with hospital-acquired pneumonia ranking highest. The mechanism of a preferential association of pneumonia with NOAF is unclear but if it exists may relate to the type and site of inflammation, or the role of hypoxia in arrhythmogenesis. 53
The expert consensus in this study is consistent with existing evidence that points to sepsis as a risk factor for NOAF. 6 NOAF in sepsis is an independent risk factor for mortality and hospital length of stay.5,6 Future studies should investigate ways in which to mitigate the risk of NOAF in this population.
Heart rate variability (HRV) is a feature of the healthy cardiovascular system. It is controlled by a balance between both autonomic limbs and is reduced in states of autonomic dysfunction. Reduced HRV predicts NOAF in patients in the community. 54 In hospitalised patients, reduced HRV has been noted to precede ischaemic cardiac events 55 and to be predictive of multi-organ dysfunction. It is not clear whether HRV may be predictive of new-onset arrhythmias in critically ill patients.
Cardiac surgery, particularly that involving the mitral valve is a well-established risk factor for post-operative AF. 56 Despite the body of research, tools to predict AF after cardiac surgery (AFACS) have proven unreliable and the topic remains a research priority. 57
Modifiable factors
The study has also identified potentially modifiable factors, such as physiological variables, electrolyte levels, interventions, and preventative medications. Many of these, such as target plasma potassium and magnesium concentrations, are commonly used to reduce risk and treat atrial fibrillation in the absence of strong evidence for this practice. Other measures, such as preventative medications, have demonstrated utility in non-ICU populations and are worth exploring in future studies.
Physiological status/vital signs
The interaction between physiological variables, disease processes, and underlying comorbidities is complex and worth examining.
Fluid balance and increasing weight were felt to be important predictors of NOAF risk. Increasing fluid balance may be a surrogate for atrial stretch and is associated with increased NOAF risk in critically ill patients. 36 High left atrial pressure is ranked highly as a risk factor in this study. This association may not be causal. It may instead reflect that more unwell patients (who often receive larger volumes of fluid) are at higher NOAF risk.
Hypotension is likely to be associated with the development of NOAF when in the context of vasodilation or cardiomyopathy related to systemic inflammation. Independent from systemic inflammation, hypotension may impair tissue perfusion including that of myocardial conduction tissue. 58 Decreased tissue oxygen delivery also stimulates endogenous catecholamines, further increasing the risk of arrhythmias. 59
Investigation results
Hypokalaemia was considered the most important variable in the domain relating to investigation results. This is consistent with current knowledge of cardiac conduction, as well as existing evidence.
Lower plasma potassium concentration leads to increased risk of atrial arrhythmia due to a lowering of cell resting membrane potential which slows intra-atrial conduction, increasing the risk of re-entrant circuits. 60 Furthermore, hypokalaemia reduces the outward repolarising current, increases intracellular [Ca2+], and shortens the refractory period.61,62
Low plasma potassium concentration is associated with a higher risk of developing atrial fibrillation in patients in the community. 63 Hypokalaemia has also been associated with NOAF in patients post-MI and has led to guidelines suggest targeting a plasma [K+] in the upper range of normal in these patients. 64 This practice is often applied to the general critically ill population, although optimal target potassium levels are yet to be determined.
In addition to hypokalaemia, the rate of change of potassium level was considered to be an important risk factor for NOAF. Increasing potassium variability is associated with poorer outcomes in critically ill patients.65,66 Electrolyte supplementation may induce arrhythmias through rapid changes in extracellular concentration, although this requires further study. 65 These findings support further investigation to determine the optimal target for patients’ potassium concentration, and supplementation using considered dosing.
The panel also identified hypomagnesaemia as a risk factor for NOAF. Magnesium supplementation is commonly used in the treatment of NOAF, even in the context of normal plasma magnesium levels.
Extracellular magnesium has effects on inward calcium channels and membrane stability.67,68 Intracellular magnesium plays a role in maintaining the plateau phase of the cardiac action potential. 69 There is a disparity between intracellular and plasma concentrations of magnesium, and equilibrium between these compartments occurs slowly. Plasma levels may therefore not be reflective of intracellular stores. 70 The presence of cardiac arrhythmia is associated with intracellular magnesium deficiency. 71 This association provides plausibility to magnesium supplementation in the context of normal plasma [Mg2+].
Perioperative magnesium administration reduces the incidence of atrial fibrillation after cardiac surgery (AFACS). 72 It has rate-control properties in new-onset AF in hospitalised patients 73 and may promote cardioversion when high plasma concentrations are targeted. 74 However, it is suggested that routine magnesium supplementation in undifferentiated ICU patients has no effect on arrhythmia rates 75 and hyper- rather than hypomagnesaemia is associated with adverse outcomes. 76 To date, there is no evidence supporting an optimal target plasma [Mg2+].
High left atrial pressure (LAP) was also felt to be an important predictor of NOAF during critical illness. Atrial electrical remodelling can occur within minutes of stressors including elevated LAP and predisposes to AF.77,78 Whilst LAP may be inferred using a pulmonary artery catheter, echocardiographic (including transoesophageal) surrogates correlate only modestly. 79 Whether bedside TTE measures of LAP are predictive of NOAF during critical illness is unknown.
Hyperthyroidism is an established risk factor for NOAF. 80 Furthermore, the AF risk is elevated even in patients with high-normal thyroid function. 81 However, it is not yet known whether abnormal thyroid function in the context of critical illness correlates with NOAF risk, independent of illness severity.
Interventions
Of the interventions, administration of medications such as catecholamines, bronchodilators, aminophylline and vasopressin were ranked highest. Adrenergic stimulation from catecholamines and bronchodilators induces after-depolarisations, enhances automaticity and may promote atrial re-entry circuits. 82 Further to phosphodiesterase inhibition, aminophylline is an antagonist at the adenosine A1. receptor Reducing adenosine’s cardiac sympatholytic function adds to tachyarrhythmia risk. 83
Beta blocker and amiodarone therapy were judged to be important modifiers of NOAF risk, as was vasopressin. Vasopressin induces vasoconstriction through action on V1 receptors on vascular smooth muscle. 84 It is often used as a catecholamine-sparing agent in the management of distributive shock. By reducing the requirement for catecholamines, it decreases the stimulation of arrhythmogenic myocardial β1-receptors and thereby reduces arrhythmia risk. 85 This protective effect has been demonstrated in patients during critical illness in a systematic review of studies comparing catecholamines alongside vasopressin vs catecholamines alone. 86
ICU-specific interventions, such as the placement of PA catheters and central venous catheters were also considered important predictors. Two randomised trials of PA catheters did not compare rates of NOAF between intervention and control groups, however both reported dysrhythmias during insertion in 3% of patients.87,88 Atrial fibrillation may be induced by myocardial irritation from malpositioned central catheters 89 or by guidewires inserted into the atrium during catheter insertion. 90 It remains unclear whether central venous catheters pose an increased NOAF risk when well positioned.
Preventative measures
The expert consensus in this study puts forward a role for preventative pharmacological therapies. A recent systematic review found that the use of perioperative beta blockers was associated with a reduced risk of AF after cardiac surgery. 91 Beta-blocker prophylaxis in the general critical care population has not been studied. Timing of re-initiation of beta-blockers for patients who were previously taking them may be important. Restarting beta-blockers early after non-cardiac surgery may be protective. 92
Perioperative amiodarone prophylaxis prevents NOAF after cardiac surgery. 72 This intervention is safe and generally well tolerated 93 and justified given the high risk of NOAF in this cohort. Further study is required as to whether amiodarone prophylaxis may be of benefit to select high-risk patients in the general critical care cohort.
Limitations
A well-designed and run Delphi process is a valuable addition to quantitative data allowing some degree of qualitative evaluation to better assess results in context. Delphi results will be largely subjective and are not supported by a robust quantitative measure developed from primary data. There is therefore an element of trust associated with the presentation of Delphi results that is linked intrinsically to the unknown identities of an anonymous panel. Commonly described limitations of the method include a lack of common guidelines for either definition of consensus, or for panel selection.
For this study we selected panellists purposively to include individuals with different experiences, all relevant to the identification and treatment of AF in the critically ill population. We retained independence throughout. At no point were individual panellists known to each other. At each round consensus was reported in line with pre-determined criteria. Our response rate was relatively low (55%) and responses were not acquired from all geographical regions invited. This may act as a source of bias and limit the generalisability of our findings. Lastly, there was overlap between some variables within and across domains. Certain similar variables were retained uncollated as per pre-specified methods to avoid facilitator influence on our findings.
Conclusion
We present the results of the first Delphi study into factors affecting NOAF risk in critically ill patients.
The Delphi process facilitated opportunities to include variables during critical illness that have not been formally evaluated in clinical studies. Many factors identified by the panel were those with evidence of correlation with NOAF either in the community or during critical illness. Variables that were proposed which do not have a strong evidence base have physiologically plausible mechanisms of action that could alter the risk of NOAF in critically ill patients. We highlight several potential targets for future research including potential biochemical and echocardiographic predictors.
Our results could be used to guide future research into AF prediction, provide a framework for covariates in prognostic studies of NOAF, and inform trials of preventative interventions in high-risk patients.
Footnotes
Acknowledgements
The authors wish to express huge gratitude to the anonymous experts who gave up their precious time to take part in this study.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Peter Watkinson works part-time for Sensyne Health and has received grant funding from the National Institute for Health Research, Wellcome, and Sensyne Health outside the submitted work. The remaining authors confirm they have no conflicts of interest to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and publication of this article: PW is supported by the NIHR Biomedical Research Centre, Oxford. JB is funded by an NIHR Doctoral research fellowship [NIHR300224].