Abstract
Introduction:
Antinuclear antibodies (ANA) are the hallmark of many connective tissue disorders (including lupus), which comprise roughly 5% to 10% of chronic debilitating diseases causing morbidity and mortality. In society, fear of these diseases increases illness-related uncertainty because the prognosis of progression is often difficult to determine and at least some symptoms fluctuate unpredictably. In the anti-vaccination movement, the question of the possible connection between vaccination and connective tissue disorders and other autoimmune diseases has grown to rank as an important argument for rejecting vaccination. In 2021, every fifth Polish first-degree nursing student decided to not be vaccinated against COVID-19.
Objective:
This study aimed to explore the prevalence of antinuclear antibodies in students vaccinated and unvaccinated against COVID-19.
Participants and Methods:
A single university cross-sectional study was performed in a small academic centre in Poland, where 210 students were recruited in 2022. All the participants were screened for SARS-CoV-2 IgG and antinuclear antibodies.
Results:
The mean age of the students who rejected vaccination was higher than that of those who were vaccinated. Among nursing students, 30.0% of vaccinated and 58.3% of unvaccinated individuals had COVID-19. The frequency of antinuclear antibodies was 3 times lower in vaccinated students than in unvaccinated students (2/159 vs 2/51; P > .05).
Conclusion:
The results of our study did not confirm the rationality of rejecting vaccinations against COVID-19 for fear of developing autoimmune diseases among healthy students.
Introduction
Connective tissue disorders (CTD), commonly referred to as systemic autoimmune diseases or collagenopathies, comprise roughly 5% to 10% of chronic debilitating diseases that cause morbidity and mortality.1-3 CTD is a wide variety of related diseases characterised by dysregulation of immune homeostasis which enables the activation of immune cells to attack autoantigens, resulting in chronic increased inflammation and multi-tissue damage. Detailed characteristics of more than a hundred CTDs have already been described. The clinical hallmarks of CTD overlap with those of diseases caused by pathogenic microorganisms and other nonimmune-mediated disorders. Patients may experience considerable illness-related uncertainty because the prognosis of disease progression is often difficult to determine and at least some symptoms fluctuate unpredictably.1-3
As these diseases are autoimmune in nature, a plethora of well-described autoantibodies are useful for differential diagnosis. Since their first description, antinuclear antibodies (ANA) have been the hallmarks of many CTD (so-called ANA-associated rheumatic diseases), such as Sjögren’s syndrome, systemic lupus erythematosus (SLE), mixed connective tissue disease, idiopathic inflammatory myopathies, and systemic sclerosis. ANA is also important in the diagnosis of autoimmune hepatitis and primary biliary cholangitis. Trace concentrations of ANA are measurable in approximately 10% to 30% of healthy individuals. 3 Indirect immunofluorescence microscopy (IIF) using human epithelial HEp-2 cell substrates is considered the most accurate method for ANA detection in serum samples. 3 However, many commercial enzymes immunoassay kits with variable performance are available for routine ANA screening.
Vaccines may initiate autoimmunity through mechanisms such as molecular mimicry, T-cell activation without antigen recognition, epitope spreading, and an anti-idiotypic network. However, there are no elaborated criteria for the diagnosis of vaccine-induced CTD, which has to be evaluated on a case-by-case basis. 4 The question of the connection between vaccination and CTD and other autoimmune diseases has been discussed in the scientific literature for decades, 5 and in the anti-vaccination movement, it has grown to the rank of an important argument for rejecting vaccination. 6
It was expected that students, particularly biomedical students, would not be susceptible to anti-vaccine rhetoric during the pandemic.7,8 However additionally, during the COVID-19 pandemic, part of the political scene in Poland, including the parliament, was openly against restrictions and presented vaccinations against SARS-CoV-2 as a threat to people, especially in pre- and reproductive age. 9 In 2021, over 22% of 793 first-degree nursing students from 12 Polish universities were not vaccinated against COVID-19, and every second participant declared that they would stick to their choice. 10 In a study conducted at the turn of December 2023 and January 2024 almost 20% of medical students from Poznań, one of the largest and most economically developed cities in Poland, believe at least 1 anti-vax conspiracy theory. 8 One of the arguments for not being vaccinated against COVID-19, even by medical students or healthcare workers, was the fear of developing autoimmune diseases.8,10,11 The dissemination of information about vaccinations inconsistent with research results on social media may be one of the most important factors in the denial of vaccination against COVID-19. 12 A recent study by Bartosiewicz et al 13 showed that sources of knowledge about vaccines influenced attitudes toward vaccination and were the main predictors of fear of the pandemic among Polish biomedical students. The absorption and processing of scientific information and news-sharing processes in social media are subjects of intensive research. 14 Appropriate emotional content can increase the willingness of social media users to share medical findings from scientific papers on vaccination and infectious diseases. 15
Thus, (I) the purpose of the study was to compare the prevalence of ANA in vaccinated against COVID-19 and unvaccinated healthy individuals, and (II) our study focused on students, with the assumption that the results obtained, apart from the expected scientific value, will inspire the students themselves and for their social media activity.
Materials and Methods
Study Design, Sample, and Setting
This single-university cross-sectional study was performed in a small academic center, educating approximately 3000 students in Biała Podlaska, Poland. The vast majority of vaccinated students received the BNT162b2 mRNA COVID-19 vaccine intramuscularly (Comirnaty, Pfizer, Inc., and BioNTech). The active substance of the preparation is the mRNA that encodes the spike protein of SARS-CoV-2 and acts as an antigen. Of the approximately 2500 students actively participating in the classes, it was intended to recruit 10%.
Basic data, data on vaccination against COVID-19, the course of COVID-19, and blood samples were collected from participants between April 2022 and December 2022.
Inclusion/Exclusion criteria
The inclusion criteria were a negative history of immunological diseases, vaccination with the Comirnaty vaccine (vaccinated group of students), and no anti-COVID-19 vaccinations (unvaccinated group of students). 2. The exclusion criteria were age <18 years, foreign students, pregnancy, and use of drugs that affect the immune system (eg, anabolic glucocorticoids, cytostatics, tacrolimus, and azithromycin).
Research Question
Are there differences in the prevalence of ANA among students vaccinated and unvaccinated against COVID-19?
Ethical Considerations
The study protocol was approved by the local ethics committee (approval number: 10/2021). The authors assert that all procedures contributing to this study are compatible with the ethical standards of the relevant national and institutional committees on human experimentation and the 1975 Helsinki Declaration, as revised in 2008. Written informed consent was obtained from all the participants. The members of the research team invited all healthy students with no history of immunological disturbances to participate in the study and those who agreed to complete the questionnaire.
Before conducting the research, the participants were introduced to the purpose of the research and their rights and obligations. They were also informed of their voluntary participation, anonymity, and ability to leave the study at any time.
Laboratory Tests
Blood samples were immediately transferred to the laboratory, where IgG antibody titres to the SARS-CoV-2 S1-protein were determined using an Anti-SARS-CoV-2 QuantiVac ELISA IgG assay (Euroimmune, Germany). The ELISA was developed on 96-well microplates coated with the SARS-CoV-2 S1 domain, which was expressed recombinantly in human embryonic kidney 293 cells. Quantification of S1-specific IgG was performed according to the manufacturer’s instructions using a 6-point calibration curve covering a range of 1 to 120 relative units (RU)/ mL. Serum samples yielding results in the analytical range were each time retested at a higher dilution. In each test run, positive and negative controls were included. Seropositivity was defined as >11 RU/mL. Serologic screening for antinuclear antibodies (ANA 1) was performed using the ANA Screen 11 ELISA IgG (Euroimmune, Germany) with a cutoff antibody index >1.45 for seropositivity. Autoantibodies in ANA 1—positive participants were confirmed by indirect immunofluorescence (IIF) on Hep-2 cells (ANA 2; Euroimmune, Germany) and by line immunoblotting (ANA 3 Profile with 15 antigens; Euroimmune, Germany) on a Euroblot Master 44 device using Euroline Scan Software (Euroimmune, Germany). The IIF cutoff was set at 1:160 dilution, which was in accordance with clinical practice. 3
Data Management and Analysis
Collected data were verified for completeness, quality, and consistency. All the data collected on the paper were transcribed into electronic databases for analysis. None of the participants dropped out of this study. Statistical analysis was performed using the Polish version of TIBCO Software Inc., Palo Alto, Santa Clara, CA, USA (2017). The compliance of the data with the normal distribution was assessed using the Shapiro-Wilk test (P > .05 for normal distribution). Categorical data were compared using the chi-squared or Fisher’s exact test based on the size of the smallest subgroup. The comparison of proportions between groups was performed using the Mann-Whitney U test. All qualitative findings are presented as n (%). Statistical significance was set at P < .05.
Results
Sample Characteristics
This study analyzed the data of 210 students, of whom 159 were vaccinated against COVID-19 and 51 were unvaccinated. The most numerous participants were nursing and emergency medical students, constituting 69.2% and 10.1% of the vaccinated group and 47.1% and 3.9% of the unvaccinated group, respectively. A comparison between the 2 groups in terms of demographics and history of COVID-19 is shown in Table 1. The age of students who rejected the offer of vaccination was statistically significantly higher than that of those who were vaccinated. More than one-third of the unvaccinated students were male, with 22% of the vaccinated students being male. Almost half of the unvaccinated students reported COVID-19 to less than 1 in 3 vaccinated. Among the nursing students, 30% of the vaccinated (33/110) and 58.3% of the unvaccinated (14/24) had COVID-19. Loss of taste and smell during the illness was reported by 70.8% of unvaccinated students (17/24) and 78.3% of vaccinated students (36/46), but a high fever above 39°C was observed in 18.5% of unvaccinated students (5/24) and only 1 vaccinated student (2.2%).
Baselin Characteristics of the Study Population.
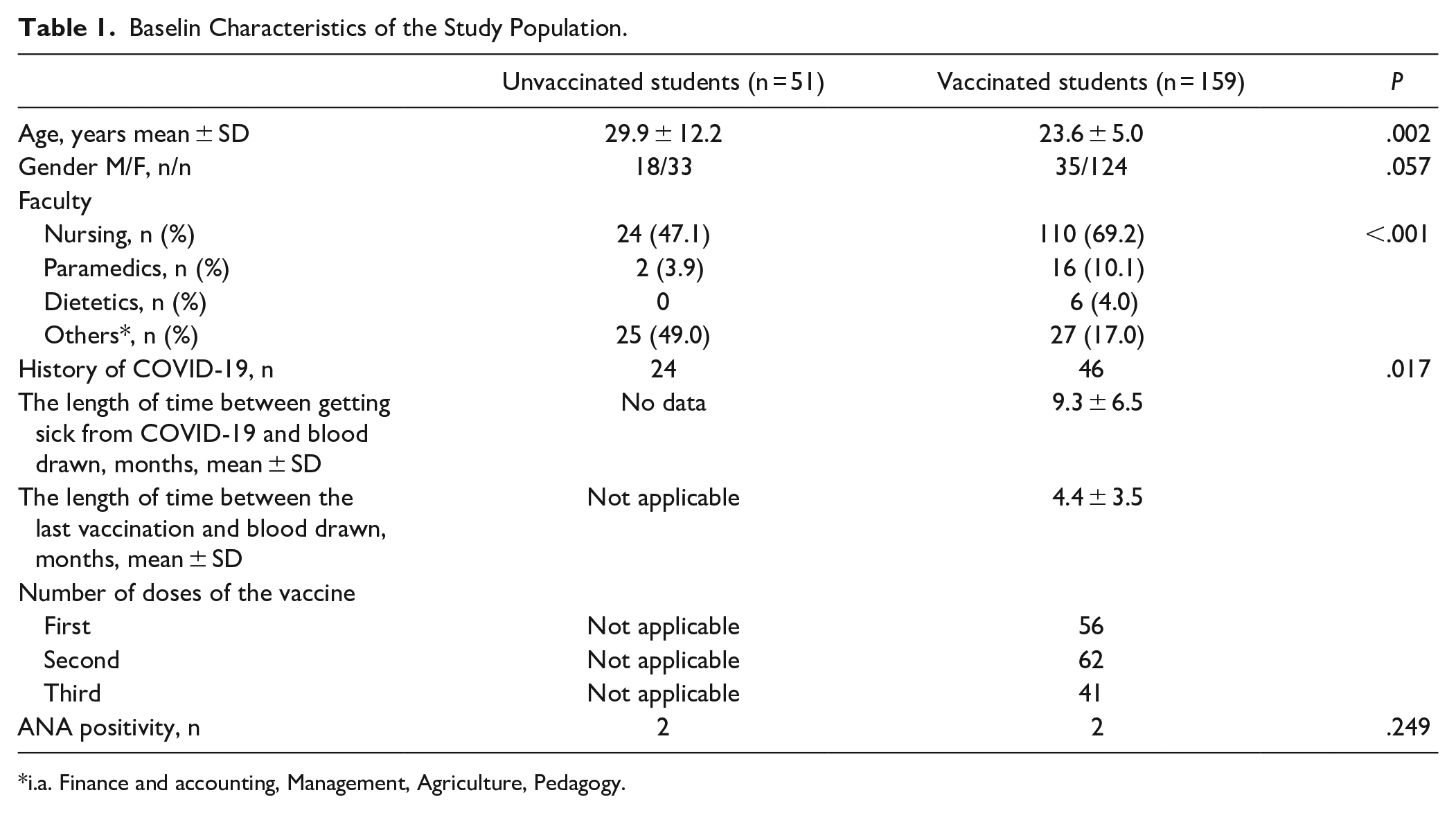
i.a. Finance and accounting, Management, Agriculture, Pedagogy.
Anti-SARS-CoV antibodies
No antibodies against SARS-CoV-2 were detected in any of the fourth unvaccinated students (Table 2). Only 1 vaccinated student did not develop any antibodies. High titres of antibodies above 2000 RU/mL were not found in unvaccinated students. There were no statistically significant differences in the levels of antibodies against SARS-CoV-2 in the unvaccinated students who thought they had not been infected with the virus and those who were sick, which was confirmed by real-time polymerase chain reaction (RT-PCR) test, which is considered the gold standard for viral detection, 16 (Table 3).
Comparison of Anti-SARS-CoV-2 IgG Antibody Levels in Unvaccinated and Vaccinated Students.

P < .001.
Comparison of Anti-SARS-CoV-2 IgG Antibody Levels in Unvaccinated Students Without Prior COVID-19 and Convalescents With SARS-CoV-2 Infection Confirmed With RT PCR Test.

P = .210.
ANA assessments
In the groups of vaccinated and unvaccinated students, 2 cases with ANA 1 were found and confirmed by IIF (1.3% vs 3.9%, P = .25). Both unvaccinated ANA-positive female students had COVID-19 and developed anti-SARS-CoV-2 antibodies (Table 4). Vaccinated female students with ANA had no COVID-19 history and titters of anti-SARS-CoV-2 antibodies below 2000 RU/mL. In 1 ANA-positive unvaccinated student, we found increased anti-Ro-52 antibodies (Table 5). Notably, no signs of new-onset inflammatory or autoimmune disease were reported by the students, regardless of ANA development.
Characteristics of ANA-Positive Students. a
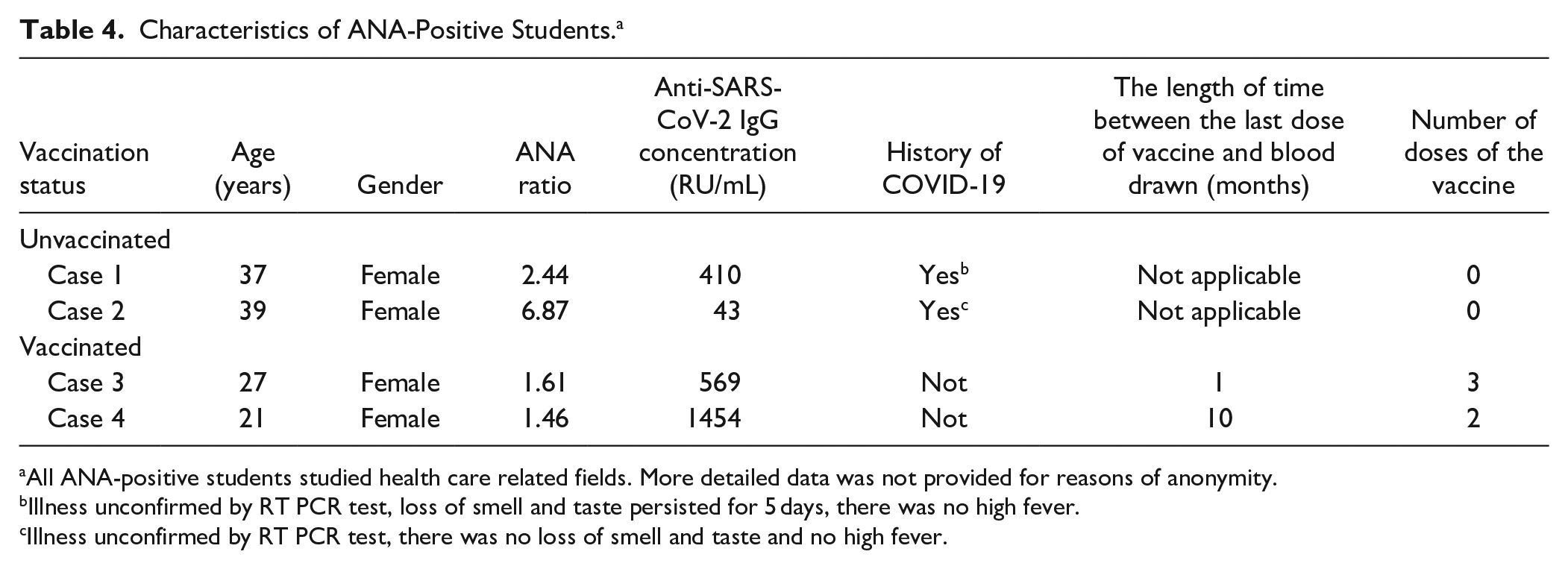
All ANA-positive students studied health care related fields. More detailed data was not provided for reasons of anonymity.
Illness unconfirmed by RT PCR test, loss of smell and taste persisted for 5 days, there was no high fever.
Illness unconfirmed by RT PCR test, there was no loss of smell and taste and no high fever.
Results of ANA-positive Sera Analysis Based on Indirect Immunofluorescence on Hep-2 Cells (ANA2) and Line Immunoblot (ANA3).

Discussion
The influential voice of the worldwide “anti-vax” movement led many people to postpone or even reject the anti-COVID vaccination in general and therefore jeopardised the efforts to slow down the pandemic. 11 In our study, ANA was 3 times less common in vaccinated students than in unvaccinated students, and 1 unvaccinated ANA-positive student developed anti-Ro-52 antibodies. To the best of our knowledge, this is the first study to compare the ANA levels between vaccinated against COVID-19 and unvaccinated healthy students. The 52 kDa protein Ro-52 (TRIM21) is mainly expressed in lymphoid tissues and serves as a shared target for autoantibodies in collagenopaties. 17 Convincing proof for the pathogenic role of anti-Ro-52 antibodies in systemic autoimmune diseases was provided by transfer experiments of anti-Ro-52 sera into New Zealand mixed mice and the onset of disease. 18
The quantitative determination of anti-SARS-CoV-2 antibodies is pivotal for estimating the humoral response to infection caused by this virus and vaccination. In this study, we used the Anti-SARS-CoV-2 Quantivac ELISA IgG assay kit which targets the S1 surface protein. This test kit had a high sensitivity for detecting anti-SARS-CoV-2 antibodies at a rate of 97.8%. 19 Three of every 4 unvaccinated students developed anti-SARS-CoV-2 antibodies. Seven out of 10 unvaccinated students who denied suffering from COVID-19 had anti-SARS-CoV-2 antibodies, which is consistent with observations about frequent clinically asymptomatic cases of COVID-19. 20 The longer the pandemic lasts, the more the interview about not getting sick from COVID-19 should be treated as not the lack of illness, but the avoidance of more severe disease. Interestingly, no antibodies were detected in the 3 unvaccinated students whose SARS-CoV-2 infection was confirmed by positive PCR test results.
SARS-CoV-2 has had a devastating impact on human health and the world in many other dimensions, such as the global economy and politics, leading to the rapid development of vaccines to prevent the spread of the disease. Currently, the vaccine is widely available worldwide and has noticeably lowered the spread of SARS-CoV-2. The pioneering use of mRNA vaccines has been accompanied by concerns regarding potential autoimmunization.4,21 The relationship between vaccination against COVID-19 and the occurrence of autoimmune disorders has been documented in numerous clinical case reports.22,23 The largest cohort to date of 11 patients with new-onset autoimmune diseases with positive ANA related to the mRNA-based COVID-19 vaccine from a single center study (Khobar, Saudi Arabia) included 4 SLE patients (22, 25, 33, and 37 years-old), 2 patients with inflammatory arthritis (26 and 27 years-old), and 1 patient each with Sjögren’s syndrome (44 years-old), dermatomyositis (46 years-old), autoimmune hepatitis (16 years-old), autoimmune pancreatitis (21 years-old), and antisynthetase syndrome (18 years-old). 24 Very recent database search in PubMed, EMBASE, Web of Science, and Scopus identified 16 new-onset cases of SLE (44% under the age of 30 years) following COVID-19 vaccination reported between 2021 and 2023 worldwide, 25 which does not change the fact that, in statistical terms, this is a marginal phenomenon. 21 Most cases were associated with mRNA-based vaccines (82.5%) and the first dose (56.2%). 25 The time elapsed from vaccine administration to the onset of symptoms of SLE was between 1 and 30 days (average 11.8 ± 8.1 days). Two-thirds of the patients who developed the disease had no family members or were immunologically predisposed. Studies based on case reports are particularly vulnerable to risks of over-interpretation and selection bias. Nevertheless, the South Korean cohort of 9.2 million individuals who had received at least 1 dose of the mRNA-based COVID-19 vaccine and had no prior diagnosis of COVID-19 in comparison to a historical control cohort was at considerably higher risk of developing SLE (adjusted hazard ratio = 1.53; 95% CI = 1.09-2.13). 26
Autoimmune disease experts are consistently convinced that in the general population, the benefits of vaccination against COVID-19 significantly outweigh the possible risks and should motivate people to be vaccinated.4,21 Even for patients with existing CTD at risk of flares with vaccination, in a “living document” the American College of Rheumatology recommends vaccination because the risk of poor outcomes from COVID-19 infection outweighs the risk of increased disease activity. 27 Several studies have reported that autoantibodies are prevalent in COVID-19 patients. COVID-19 patients who tested positive for ANA tended to have a severe condition and worse prognosis, including the prediction of a 28-day mortality period in critically ill patients. 28 Persistently positive ANA 1-year post-COVID-19 is associated with persistent symptoms and inflammation, similar to the so-called long COVID for a subset of COVID-19 survivors. 29 COVID-19 is a predisposing factor for auto-reactivity and is implicated in pathways that contribute to the onset of autoimmunity and may result in autoimmune diseases such as CTD, autoimmune hemolytic anemia, and multiple sclerosis. 30
Limitations and Recommendations
This study has some limitations. Recruitment plans were implemented by 84 % of participants. Signing up to participate in our study required blood sampling, which probably had a demotivating effect, probably less in the group of biomedical students. 31 Interestingly, in the recent study investigating vaccine hesitancy without planned blood collection only 17.7% of Poznań’s medical students decided to participate by completing an online survey. 8 The number of unvaccinated participants included in the study was low, making it difficult to draw definite conclusions regarding the development of ANA in this subgroup. Moreover, when discussing the frequency of ANA-positive results in the study group, we must emphasise that we did not know the frequency of their occurrence before the COVID-19 pandemic. Both contracting COVID-19 and vaccination against it can initiate a cascade of processes leading to the production of ANA.3,21,24,30 In the unvaccinated group, vaccination was certainly not a risk factor for developing ANA. In turn, in the group of vaccinated students, even if they do not contract COVID-19, exposure to SARS-CoV-2 is a potential risk factor for the development of ANA due to the high frequency of asymptomatic SARS-CoV-2 infections, especially among young people. In our study, the majority of the unvaccinated students with no history of COVID-19 disease developed anti-SARS-CoV-2 IgG antibodies.
The average age differed significantly statistically in the groups of vaccinated and unvaccinated students, but this can be directly explained by the specificity of the latter group and not faulty recruitment. 8 The study is also limited by the anti-SARS-CoV-2 antibody test, which was designed to detect post-vaccination antibodies and, to a lesser extent, post-infection antibodies. 32 Furthermore, some cross-reactivity between SARS-CoV-2-specific antibodies and other endemic RNA viruses, causing respiratory tract infections, has been reported and can reduce test credibility 33 ; however, the data sheet does not account for cross-reactivity with samples from patients infected with SARS-CoV-1, HCoV-OC43, or HCoV-229E (Euroimmun. Anti-SARS-CoV-2 Quantivac IgG ELISA. Package Insert). Extended serological diagnostics could enable a more complete understanding of the immune status of unvaccinated students who have had COVID-19 and do not have anti-SARS-CoV-2 antibodies as well as that of students who did not demonstrate a post-vaccination humoral response or demonstrated a modest response.
Referring the presented results to other population groups limits the specificity of the study group, which are students below 30 years of age on average.
Implications for Practice
Our study provides important information that could be valuable for healthy individuals who have difficulty deciding whether to get vaccinated because of the fear of developing autoimmune diseases. The data from our study could be used to inform communities that do not support or trust vaccination on the lack of the likelihood of auto-immunoreactivity associated with CTD after vaccination against COVID-19. Moreover, medical professionals dealing with vaccinations in the student population should direct a particularly reinforced message to older and male students, who raise concerns. Male students seem to be more susceptible to the message of anti-vaccination movements. 8
Conclusions
We did not find a presumed more frequent occurrence of ANA in healthy individuals vaccinated against COVID-19 among a group of 210 students from a small university located in the rural part of Poland. The results of our study did not confirm the rationality of rejecting vaccinations against COVID-19 for fear of developing autoimmune diseases among healthy students. It is important to emphasise the need for further studies on the connection between vaccination and autoimmune diseases.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Akademia Bialska Center of Excellence under Grants No PB/3/2021 and PB/4/2022.
Ethical Approval
Permission to perform this research was obtained from the local ethics committee (Komisja Bioetyczna przy Akademii Bialskiej im. Jana Pawła II, no. 10/2021). Before conducting the research, participants were introduced to the purpose of the research and their rights and obligations. They were also informed of their voluntary participation, anonymity, and ability to leave at any time.
Data Availability Statement
The datasets analyzed during the current study are available from the corresponding author on reasonable request.
