Abstract
Background:
Telemonitoring for COVID-19 has gained much attention due to its potential in reducing morbidity, healthcare utilization, and costs. However, its benefit with regard to economic outcomes has yet to be clearly demonstrated.
Objective:
To analyze the costs associated with the use of the Opal portal to monitor COVID-19 patients during their 14-day confinement in Quebec and compare them to those of non-users of any home telemonitoring technology.
Methods:
A cost analysis was conducted through a cross-sectional study between COVID-19 patients who used (PU) the Opal platform during their 14-day confinement at home and those who did not use (PNU) any home remote monitoring technology. Data was collected between June 2021 to April 2022. An individual interview with each participant using an adapted questionnaire was conducted by telephone or online using a teleconferencing platform. A micro-costing approach was undertaken using a dual patient and Quebec’s health-care system perspective.
Results:
27 telemonitoring participants, 29 non-users, 8 clinicians, and 4 managers were included. Telemonitoring reduced the average total costs incurred by PU by 82% ($537.3CAD) between PU ($117.2CAD) and PNU ($654.5CAD). Telemonitoring enrollees used healthcare less intensely with fewer emergency room visits (1 PU compared to 6 PNU), which translated to an average savings of $253.3CAD per patient.
Conclusion:
This is the first study to demonstrate that telemonitoring through the Opal platform is a viable strategy to reduce healthcare costs and utilization for patients and the healthcare system. The evidence provides strong support for introducing telemonitoring as a component of case management.
Background
COVID-19 has had a significant impact on hospital and emergency care functioning. Most hospitals have experienced periods of operating below capacity with physical distancing restrictions followed by intermittent increases in patient volume. 1 Physical distancing and quarantine measures have been adopted and generalized to minimize the impact and slow the spread of the COVID-19 pandemic.2 -5 These measures have required new strategies to maintain contact between, on the 1 hand, patients and their relatives, and, on the other hand, healthcare professionals. In addition, the pandemic and the mitigation measures put in place have led to several challenges in terms of quality and safety of care and the establishment of partnerships between patients and professionals.2 -6 In this setting, people diagnosed with COVID-19 had to be isolated at home for a period of about 14 days.
Mobile health (mHealth) technologies are seen as promising alternative strategies. This technology has been defined by the World Health Organization as “the use of mobile and wireless technologies to support the achievement of health objectives.” 7 The use of mHealth has been growing in Canada and in several countries since well before the COVID-19 pandemic. 8 More than 76% of Canadians own a smartphone, and older people are increasingly using this type of device. 9 mHealth technologies have various benefits for patients and professionals, including immediate access to data, alerts, or notifications in case of immediate needs, and facilitating the exchange of important information between patients and healthcare professionals.6,10,11 These technologies also allow the implementation of care adapted to patients with special needs or a low level of literacy. Several means of communication and access to health professionals are possible via these technologies (eg, SMS, e-mail, teleconference, or videoconference).6,10,11 They therefore have the potential to increase productivity and support decision-making processes for complex patients.6,10,11 In response to the challenges posed by the COVID-19 pandemic, a “Blinded Institution” worked on using a patient portal called Opal to remotely monitor at home self-isolated COVID-19 patients with daily self-report symptoms, vital signs, and mental health reviewed by healthcare professionals (https://beta.clinicaltrials.gov/study/NCT04978233).12,13 Patient portals are health-care related applications allowing patients to securely access their personal health information, 14 manage aspects of their own health care, and to interact and communicate with healthcare providers.15,16 Currently used by over 5000 patients, Opal is an award-winning patient portal first implemented in the Division of Radiation Oncology at the “Blinded Institution” in 2018 (https://www.opalmedapps.com/). Opal includes a smartphone app that allows patients to access their appointment schedule, medical records (including radiation oncology clinical notes, lab test results, and radiation treatments), personalized educational materials tailored to diagnosis and treatment, a remote recording tool, and patient-reported outcome measures (PROMs). The data is contextualized and explained to ensure it is useful and reward for patients (https://www.Opalmedapps.com/fr-selected). Physicians and nurses use Opal to remotely administer the vital signs through a desktop dashboard.
Opal has been used with COVID-19 patients to home monitor them, and informative and educational content has been designed for this purpose. 12 The care pathway for Opal for COVID-19 patients included daily self-questionnaires for 14 days on symptoms and physical signs, including body temperature, oxygen level rate, and mental and psychological health such as anxiety levels. Participants’ answers to these self-questionnaires were automatically transferred to a nurse who reviewed them using a dashboard (Opal’s “nurse” interface). If necessary, patients were contacted by telephone or teleconsultation. A transfer to the emergency room can be proposed if the clinical situation of a patient required it. A COVID box containing a pulse oximeter and thermometer has been sent to patients who have been trained to use them to monitor vital signs at home.
In its current state, the Opal portal has been evaluated on the clinical and technological efficiency and feasibility aspects. 17 Through its ease of use, accessibility, security, adaptability to meet every patient needs and close follow-up by healthcare professionals, Opal portal has helped provide patients with emotional support and reduce stress associated with their health status. 17 However, the organizational and economic impacts of the use of Opal by healthcare professionals and COVID-19 patients have not yet been evaluated. Given the dearth of much-needed evidence, this study aimed to analyze costs associated with the use of the patient portal to remotely monitor COVID-19 patients during their 14-day confinement and compare them to those of non-users of any home telemonitoring technology. It included effects on total costs incurred by COVID-19 patients and healthcare resource utilization.
Methods
A cost analysis was carried out through a cross-sectional study between COVID-19 patients who used the patient portal during their 14-day confinement at home and those who did not use any home remote monitoring technology. The intervention consisted of a telemonitoring set for transmitting vital parameters, clinical support, and patient education.
Setting and Participants
This study was embedded in the large Opal-COVID study conducted at a “Blinded Institution” and aimed to assess the implementation of the Opal patient portal for distance monitoring of self-isolating patients with COVID-19, identify influences on the intervention’s implementation, and describe service and patient outcomes of this intervention. 12 This study was approved by the “Blinded Institution” Research Ethics Board (REB) (approval # 2021-7632) and registered in the ClinicalTrials.gov database (NCT04978233). A total of 51 patient participants who used the Opal patient portal and consented to participate in the study were enrolled between December 2020 and March 2021. Of these, 2 withdrew voluntarily before Day 14, 45 completed the 14-day follow-up, and 4 were extended to 21 days since they were still symptomatic at Day 14. All participants who completed at least 14 days of follow-up (N = 49) were included in the analysis. During the Opal-COVID study consent process, participants were asked whether they agreed to be contacted to participate in subsequent studies.
Participants Recruitment and Selection
Four types of population were recruited in this study including:
- Patients diagnosed positive for COVID-19 at the “Blinded Institution” and users of the Opal patient portal (P1);
- Clinicians (doctors and nurses) who were involved in the use of Opal patient portal to take care and home monitor COVID-19 patients (P2);
- Managers/administrators (P3); and
- Patients diagnosed positive for COVID-19 who did not use Opal or any other telemonitoring or remote monitoring technology (P4) (a comparator group to the population P1).
Inclusion and Exclusion Criteria
All patients’ users of Opal aged 18 and above who have received a positive diagnosis of COVID- 19 by PCR test, have participated in the Opal-COVID study and agreed to be contacted again for further research studies, who speak French and/or English, and are resident of Quebec were included in the study.
Healthcare professionals who practice at the “Blinded Institution” as a nurse, doctor, social worker, or any other health profession, who have been assigned to remote monitoring by Opal of COVID-19 patients during the Opal-COVID Study were included in this study.
Implementation managers were also included in this study. This population consisted of individuals who, for the Opal-COVID Study, were involved in study design, data collection, training, development of educational materials to be used in Opal, coordination, data analysis, and/or piloting or implementation of the intervention were included in this study.
A control group of 29 patients’ non-users of the patient portal who have received a positive diagnosis of COVID-19 by PCR (self-reported), aged 18 years or older, speak French and/or English, and are resident of Quebec were recruited in this study.
Participants from the 4 populations were excluded if they didn’t comply with the inclusion criteria, or for any other reason justified by the investigator and the research team, and which would make the respondent unsuitable for participation (eg, cognitive impairment, medical instability, schedule that does not allow participation, health condition, other problems, or obstacles). Additionally, participants non-users of Opal were also excluded if they have used home remote monitoring technology for the management of COVID-19 and isolation at home or have participated in a clinical study protocol on COVID-19.
Recruitment and Sampling
Out of the total of 49 patients Opal users, 27 patients who were available and willing to participate in the study were selected. A member of the research team (AR) contacted those who have agreed to be contacted again for further research by telephone and explained them the objectives, procedures, risks, and benefits of this study, verified their willingness to participate and scheduled a time to conduct informed consent and interview for data collection. For this purpose, a script has been formulated. P1 didn’t receive financial compensation as they had already been compensated for their participation in the Opal-COVID study.
Also, 7 physicians, 4 clinical nurses, and 4 implementation managers were recruited using a purposive sampling technique. Participants have confirmed with the principal investigator (BL) their agreement to be contacted for additional studies on the use of Opal for the follow-up of the COVID-19 patients during study implementation. Four physicians, 4 nurses, and 4 managers participated in the study. A member of the research staff contacted these participants by phone or email. The phone call and/or email script informed them of the study goals, procedures, and inclusion criteria. The research team member verified their willingness to participate in the research. For those interested in participating, an information and consent document was sent by email or mail, and a time and mode (phone call or videoconference by Zoom©, Teams©, etc.) was determined for obtaining verbal consent. Participants had the right to refuse to participate in the study by telephone or by answering “No” to the email. P2 and P3 didn’t receive any financial compensation.
As for patients’ non-users of Opal (P4), 29 were online recruited via Facebook and Instagram by a network (snowball) sampling technique. To generate results comparable to those of Opal users while ensuring the feasibility of the study, a sample of 30 patients was targeted. According to the central limit theorem, a sample size of 30 participants is sufficient for field studies.18 -22 A flyer was shared on Facebook and Instagram, inviting people who have been tested positive for COVID-19 and who did not use Opal or any other telemonitoring or remote monitoring technology to participate in an online survey. This leaflet summarized the purpose of the study, the procedures, the inclusion criteria, and the compensation of $20CAD in the form of gift cards to encourage them to participate and to compensate them for the time and effort put into this study. Also, the brochure invited people interested in participating to contact the research coordinator by phone or email. When contacting the research coordinator, a time was scheduled to proceed with the informed consent process and the data collection interview with the participant.
Data Collection
Data was collected between June 2021 to April 2022 during an individual interview with each participant of the 4 participating populations (P1, P2, P3, P4) using an adapted questionnaire which was tested before administration for readability, simplicity, comprehensibility, and completion time. For this, 2 COVID-19 patients (P1, P4), 1 professional (P2), and 1 manager (P3) meeting the inclusion criteria participated in the pilot study. This resulted in no changes or modifications. The individual interviews took place by telephone or online on a teleconferencing platform (Zoom©, Teams©), and lasted approximately 10 to 15 min to complete the questionnaire. They were conducted by a trained and experienced research assistant (RA) who began by explaining the purpose of the study and then conducted the interview. The questionnaire was also self-administered to those who wished to complete it themselves.
Regarding Opal user patients, individual interviews were conducted 1 to 3 months after their recruitment into the Opal-COVID Study.
A questionnaire adapted from the Costs for Patients Questionnaire (CoPaQ) was used for P1 and P4.23,24 As for P2 and P3, another questionnaire was prepared based on the CoPaQ questionnaire. This validated questionnaire covered questions on sociodemographic characteristics and on the costs and out-of-pocket expenses, direct and indirect, related to health for any type of patient. Regarding the costs for the healthcare institution, additional questions were added to the adapted CoPaQ questionnaire. The adaptation of this questionnaire included the deletion of some questions because of their non-applicability to the context of the study or the “Blinded Institution” and the reformulation of a few questions for suitability. The answers to the questions were noted directly by the interviewer on the questionnaire during the interview. All completed questionnaires were then manually entered and stored into the secure REDCap database.
Study Outcomes
The selection of the study outcomes was based on the most commonly used outcomes in the literature and can be subdivided into (1) costs of the use of Opal platform for COVID-19, (2) out-of-pocket costs for patients, including direct medical and non-medical costs, indirect costs and patients’ caregivers’ costs and, (3) health-care services utilization costs including hospitalization (resources used, duration, remuneration of physicians and nursing staff) and emergency/clinic visit. All outcomes represent the average values over the 14-day follow-up isolation period for COVID-19 patients enrolled between December 2020 to March 2021.
Data analysis
Statistical Analysis
Descriptive (mean, median, and standard deviation) and comparative cost analyses (out-of-pocket costs for patients including direct medical and non-medical costs, indirect costs and patients’ caregivers’ costs, and healthcare services utilization costs) for patients using Opal (P1) from the questionnaires collected from P1, P2, and P3 versus non-users collected from P4 were performed. These analyzes were performed using Excel software.
Cost Analysis
A micro-costing approach was undertaken using a patient and Quebec’s healthcare system perspective (ie, dual perspective). This analysis aimed to list all costs associated to the use of Opal for both patients and the Quebec healthcare system. In this study, we assessed the net costs or savings incurred in the healthcare organization and for patients associated with the remote follow-up of COVID-19 patients users of Opal over 14 days of isolation (P1), compared with non-users of Opal or any other virtual technology (P4). We estimated the mean cost per Opal user by use of data on costs for deployment and use of Opal (administrative costs, personnel costs, logistical costs, resource use), and costs (direct and indirect) incurred by Opal users and the healthcare system.
The total costs related to the COVID-19 patient’s health state represented the net amounts remaining payable by the patient, that is, those not reimbursed by insurers, that is out-of-pocket costs. These costs must result directly from their use of the health care and services necessary for their treatment or must be linked to the repercussions of their health state on their daily life during their 14-day isolation at home. 24 Two categories of costs were distinguished here: direct costs (medical and non-medical), and indirect costs represented by the economic aspect of the illness for the individual (eg, work absenteeism, isolation, sick leave, lost leisure time) and for their caregivers. Moreover, costs related to the healthcare system in Quebec were calculated. More specifically, the costs for hospitalization and emergency/clinic visits during the 14-day follow-up period were considered. All costs were reported in 2021 Canadian dollars (ie, year of the study). No discount rate was used given the short timeframe.
Emergency Costs
Given the absence of data on hospital spending in Quebec on the Canadian Institute for Health Information (CIHI) website, 25 we considered the costs of $425CAD for an emergency visit for the year 2015 to 2016, as stated by Poder et al. 26 and calculated following a micro-costing method. This includes the operating cost of $257CAD and the medical fees of $168CAD drawn from the billing manuals for general practitioners and medical specialists produced by the RAMQ. 26 The costs were then discounted using the Consumer Price index for 2021 provided by Statistics Canada, generating an estimated cost of $463.3CAD (425 × 1.09) for an emergency visit (https://bdso.gouv.qc.ca/pls/ken/Ken213_Afich_Tabl.page_tabl?p_id_raprt=3877).
Results
Description of Study Population
In total, 27 of the 49 COVID-19 patients’ users of Opal (PU), 29 COVID-19 patients’ non-users of Opal (PNU) or any other telemonitoring technology, 8 clinicians (4 nurses, 4 physicians), and 4 managers participated in the study, including the pilot study participants. A total of 22 (45%) patients’ users of Opal declined to participate in the study.
Most of the PU were female (59.3%). The majority (33.3%) were aged between 31 to 40 years old followed by 22.2% and 18.6% aged 41 to 50 and 18 to 30 years old, respectively. As for PNU, a majority of female (62%) participated in the study. Most of the respondents (44.8%) were aged 41 to 50 with 24.1% and 13.8% aged 18 to 30 and 31 to 40 years, respectively. Results showed that most of the respondents were married and/or in a couple with 29.6% and 26% for PU and 37.9% and 27.6% for PNU, respectively. Additionally, 48.15% of PU were white compared to 82.76% of PNU. Most respondents lived in urban areas with 88.9% for PU and 89.7% for PNU (Table 1).
Description of the Sociodemographic Characteristics for Patients Opal Users (P1) and Non-Users (P4).

Patients’ Perspective—Costs of Opal Platform Utilization for COVID-19
Out of 27, 11% PU incurred expenses for the purchase of oximeter and 14.8% for a thermometer. However, 48.3% (n = 14) PNU of Opal have also incurred expenses for the purchase of thermometer, 20.7% (n = 6) for arterial blood pressure equipment and 10.3% (n = 3) for an oximeter.
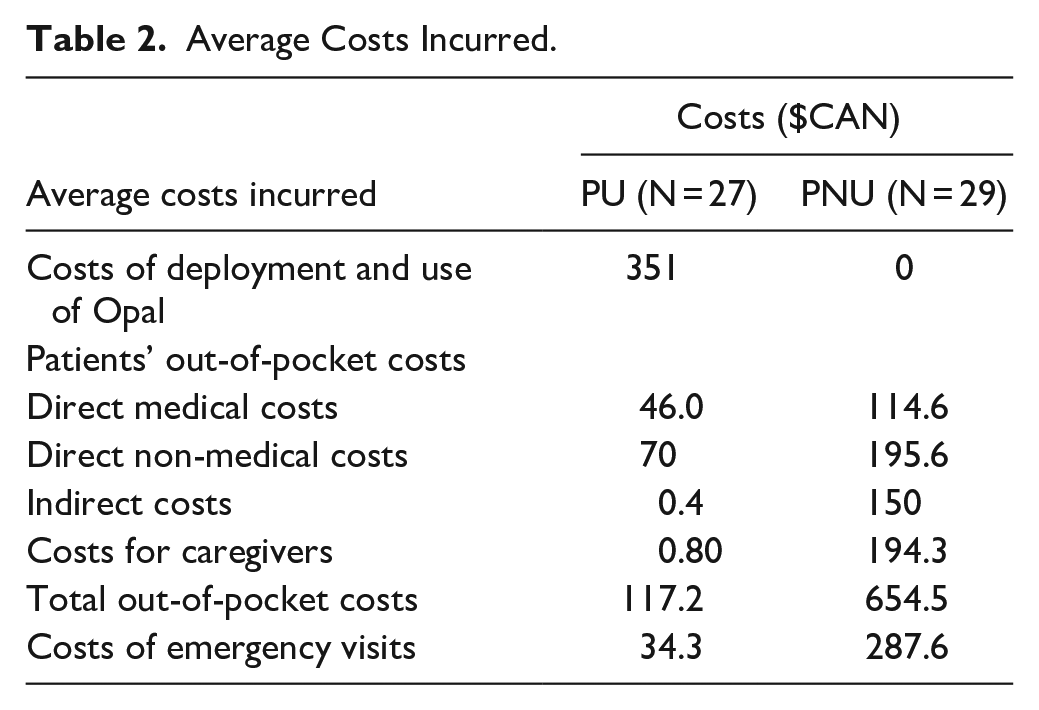
The average total cost of Opal utilization incurred by PU was $117.2CAD compared to $654.5CAD incurred by PNU. Specifically, PU paid an average of $4CAD for direct medical costs (eg, medical devices, medical examinations, medication, hospitalization), $70CAD for direct non-medical costs (eg, travel and parking fees to go to medical appointments, fees of transfer to health services, non-medical devices and health services, income loss) and $0.4CAD for indirect costs (eg, waiting time in the emergency, travel time to health services) compared to $114.6CAD, $195.6CAD, and $150CAD paid by PNU, respectively. Also, the average total costs incurred by PU caregivers was $0.803CAD compared to $194.3CAD incurred by PNU (Table 2).
Average Costs Incurred.
Hospital Perspectives—Home Monitor or Follow-Up Visits
The results showed that 44.4% PU used the tele-follow-up visits offered by the Opal platform from which 29.6% used the video call and 14.8% the phone call.
Over the span of the 14-day telemonitoring period for COVID-19 patients’ users of Opal, a total of 26 teleconsultations for PU using a videoconference and/or a phone call were carried out by 4 clinicians. The average time for a video call consultation was 18.5 and 25 min for a phone call. According to the Canadian Medical Association (CMA), the cost of a teleconsultation in Canada was $49CAD (AMC, 2019) compared to in-person visit of $51CAD 27 resulting in a cost saving of $2CAD per patient.
Hospital Perspective—Opal Platform Operating Costs
The average cost of deploying and using the Opal platform per patient was $ 351CAD. The operational costs included the costs of medical equipment (oximeter, thermometer) provided to each followed patient, the software, the educational material for patients, the technical and clinical personnel assigned to work with the platform, etc.
Hospital Perspective—Emergency Visits
No one among Opal users and non-users was hospitalized during the 14-day period of isolation. However, 1 out of 27 PU was transferred to the ER twice during this period. As for the PNU, 6 of the 29 had unscheduled ER visits with a total of 18 visits varying between 1 to 6 visits. Since the actualized cost per ER visit was set at $463.3CAD (425 × 1.09) for a standard consultation in 2021, our results showed that the total costs of the ER visits incurred by Opal users were $926.6CAD compared to $8339CAD for PNU. The average cost per ER visit was $34.3CAD per PU compared to $287.6CAD for PNU (Table 2). Thus, the use of Opal for COVID-19 avoided ED visits which translated to an average savings of $253.3CAD per patient.
Discussion
Economic evaluation studies are of central importance to assess the extent to which remote monitoring provide good value for money compared with alternative options. This cost analysis adds new knowledge to the costs involved to implement a platform in hospital setting to home monitor COVID-19 patients or other patients with acute or chronic illness compared to non-users of any home monitor technology. To date, research has demonstrated that telemonitoring via telephone or video can be a rapidly deployed, safe, and cost-effective mode of chronic disease management. 28 Additionally, telemedicine has been shown to reduce health care expenditures, both monetarily and in ED visits and hospital visits, across a range of specialties. 28 Our study also demonstrated that the home monitoring with Opal was cost beneficial for the acute care of patients with COVID-19 with savings for the patient and the healthcare sector. The results showed that the use of Opal avoided costs associated with COVID-19 illness for patients who, according to clinicians, did not have health condition, cognitive impairment, medical instability or other problems. The savings associated with Opal platform utilization were at the level of all types of costs incurred by PU ($117.2CAD) which were almost 6 times lesser than those incurred by PNU ($654CAD) which respected the same inclusion criteria in terms of health conditions and were not closely monitored by healthcare professionals. The results showed that telemonitoring simply shifted the burden and costs of care away not only from the PU themselves but also from their caregivers who incurred an average total cost of $0.803CAD compared to $194.37CAD incurred by PNU’s caregivers.
The results showed that another key source of telemonitoring savings was averted ER visits. 28 In fact, Opal platform allowed for triaging patients at high risk for health complications with teleconsultations, resulting in a low rate of ER visits by 1 PU versus 6 PNU with an average cost saving of $253.3CAD per patient. The availability of next-day follow-up using Opal by physicians or nurses afforded ED physicians a safe discharge plan instead of electing for overnight hospitalizations for patient observation. Moreover, a small cost savings of $2CAD per patient consultation was attributed to the use of teleconsultations ($49CAD) services compared to the in-person clinic visit ($51CAD). The 26 teleconsultations encounter resulted in a savings of $52CAD compared to the equivalent in-person clinic visits, meaning that using the Opal platform by a larger population size would result in additional savings.
Despite the fixed costs of supplying at-home pulse oximeters and thermometers for patients and the assignment of administrative, logistics, and informatics staff to maintain and support the platform, and of clinical staff to monitor and follow-up with COVID-19 patients, these costs were outweighed by savings for patients and from averted unnecessary ER visits. The use of teleconsultations/follow-ups services avoided equivalent in-person clinic visits with average cost savings of $253.6CAD per patient. Our results are in line with those of Dcruz et al. 28 who also demonstrated financial benefits of home telemonitoring and follow-ups services for COVID-19 patients. This helped avoid unnecessary ER visits and hospital admissions with significant costs savings for the hospital and the patients. Also, Padula et al., 29 in their study, found that remote home monitoring of COVID-19 patients was a cost-effective alternative for decreasing ER visits and admissions.
This study fills the gap of a thorough long-term economic assessment of costs and consequences which often has been cited as a hurdle towards reimbursement. In addition to cost savings, other benefits included an improved patient experience 17 and enhanced safety for staff and other patients. For example, continuous monitoring via Opal provided reassurance to often anxious patients and empowered them to manage their symptoms and reduced their stress. This was also supported by Cheng et al. who demonstrated that home telemonitoring helped COVID-19 patients feel less lonely and less anxious during their isolation. Moreover, the provision of pulse oximeters and thermometers reassured and comforted to dyspneic and often anxious patients.28,29 Overall, Opal demonstrated that a small, fixed investment can potentially save significant resources when deployed safely and efficiently.
eResearch on cost-utility and cost-effectiveness of telemedicine, electronic health, and mobile health systems concluded that although telemedicine is cost-effective, results are dependent on the administrative costs for establishing and maintaining screening. 30 In the case of Opal, infrastructure costs for deployment (eg, software, website, education material, maintenance, human resources) were established using existing hospital platforms; the sole purchases during deployment were pulse oximeters and thermometers), and operational costs were limited to few people salary including clinical and management/administrative staff.
Limitations
This study helped address the lack of a long-term economic assessment of costs and consequences which often has been cited as a hurdle towards reimbursement. However, the interpretation of our results must consider certain methodological and data-related limitations. First, the sample size was small for both groups. Moreover, there was an overrepresentation of people aged 41 to 50 years old and white and in the P4 compared to those older than 70 years. Of the 27 patient users and 29 non-users, only 2 aged 61 to 70 years from each group participated in the study, compared to 23 aged between 31 to 60. Additionally, recall bias is another limitation to consider. Opal users were asked to remember the costs of their 14-day home isolation 2 to 6 months after using the platform. Not to mention that the accuracy of our calculated net savings depended on the validity of each of the individual activity-based cost; thus, the calculated average costs may be an over- or underestimate, depending on errors made by patients when reporting the costs. In addition, patients were not asked about their comorbidities which might could have led to the use of healthcare services and generated costs not directly associated with COVID-19. We would also highlight that the need for additional personal protective equipment and cleaning equipment during in-person appointments raises the costs of ambulatory patient care. However, these additional costs were not included in the expenses and, thus, we assert that the saving per visit is an underestimate of costs. Our cost savings are therefore higher. Another limitation that needs to be raised is the unavailability of actual data on hospital spending for Quebec province which might bias the increase in savings per ER visit. Future prospective studies directly comparing in-person care to virtual care would also confirm that the avoided resource use observed in this study is actually avoided, allowing for more accurate cost analyses.
Conclusion
The economic analysis of the Opal platform for home remote monitoring demonstrates the financial benefits of this innovative care model. This platform was developed as a person-centered patient portal providing timely medical care to isolated patients diagnosed with COVID-19. Opal has proven to be a leading example of the ability to rapidly deploy low-cost virtual clinics, with significant savings to the hospital. Overall, this analysis demonstrated that a small, fixed investment, safely and efficiently deployed, can potentially save significant resources. In addition to cost savings, other benefits including an improved patient experience and an enhanced safety for staff and other patients have been identified.
Footnotes
Acknowledgements
We are grateful for the “Blinded Institution” for providing the patients and costs data. The authors also thank the patients, clinicians, nurses, and managers, for their time and effort to participate in this study. Special thanks to AR and DL for their support and help in data collection and for their availability.
Abbreviations
CoPaQ: Costs for Patients Questionnaire
COVID-19: coronavirus disease-19
PU: patient user
PNU: patient non-user
P1-4: populations 1 to 4
Author Contribution
Guarantor: RA
Conceptualization, M-P.P., B.L., R.A.
Methodology, R.A, T.G.P.
Validation, R.A., M-P.P., B.L., T.G.P.
Formal analysis, R.A., T.G.P.
Investigation, R.A.
Resources, M-P.P., B.L.
Data curation, R.A., T.G.P.
Writing—Original Draft preparation, R.A.
Writing—Review and Editing, T.G.P., M-P.P., B.L.
Visualization, R.A., T.G.P, M-P.P., B.L.;
Supervision, T.G.P, M.-P.P. and B.L.;
Project administrator, M.-P.P.;
Funding Acquisition, M.-P.P., B.L.
All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: BL has received research support, consulting fees and speaker fees from ViiV Healthcare, Merck, and Gilead. The other authors declare no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Canadian Institutes of Health Research (CIHR), Strategy for Patient-Oriented Research (CIHR Funding Reference Number: VR4−172769). M.-P.P. has a Senior Career Award financed by the Quebec Health Research Fund (FRQS), the Centre de Recherche du Centre Hospitalier de l’Université de Montréal and the ministère de la Santé et des Services sociaux du Québec,
Opal-COVID study secured funding from the “Blinded Institution” Interdisciplinary Initiative in Infection and Immunity (M[i]4) Emergency COVID-19 Research Funding (grant ECRF-R2-44, Principal Investigator: BL) and from the Canadian Institutes of Health Research Strategy for Patient-Oriented Research Québec Support Unit-Methodological Developments (grant M006, PI: BL). MPP and BL received CIHR Operating Grant: COVID-19 Rapid Research FO—Clinical Management/Health System Interventions “Real time evaluation of the deployment of connected technologies and of the partnership of services and care during the COVID-19 sanitary crisis -the Techno-COVID-Partnership program,” in 2020 to 2021 to support the evaluation of this study.
YM is supported by the Postgraduate Scholarship—Doctoral program (PGS D) from the Natural Sciences and Engineering Research Council (NSERC) and a doctoral research award from the Fonds de recherche Nature et Technologies (FRQNT).
MPP is supported by a Senior Career Award financed by the Fonds de recherche du Québec–Santé (FRQS), the Centre de Recherche du Centre Hospitalier de l’Université de Montréal and the Ministère de la Santé et des Services sociaux du Québec.
BL is supported by 2 career awards: a Senior Salary Award from Fonds de recherche du Québec–Santé (FRQS) (#311200) and the LE 250 (from the Québec Ministry of Health for researchers in Family Medicine), from the Quebec’s Ministry of Health for researchers in Family Medicine and holds a Canadian Institutes for Health Research, Strategy for Patient-Oriented Research Mentorship Chair in Innovative Clinical Trials for HIV Care.The authors acknowledge the development and operation of Opal as an in-kind support provided by the Opal Health Informatics Group at the Research Institute of the “Blinded Institution.” Opal’s development and operation at the “Blinded Institution” was facilitated by a MEDTEQ+ FSISSS grant (PI: JK) well as by the generous support of the Montreal General Hospital Foundation, the Cedars Cancer Foundation, the “Blinded Institution” Foundation, and the Montreal Children’s Hospital Foundation. The authors also acknowledge helpful conversations with JK and TH during the configuration of Opal and preparation of the M[i]4 grant application.
The authors thank the chronic Viral Illness Service research team, particularly, the nurses of the clinical monitoring team, LD-B, GTh, and NP. The authors also thank the laboratory research personnel responsible for shipping the medical equipment to our study participants.
TGP is member of the FRQS-funded Centre de recherche de l’IUSMM.
Ethical Review Statement
The study was approved by the Institutional Ethics Committee of the “Blinded Institution” (approval # 2021-7632), and a signed consent was obtained from each participant. The study was conducted according to the guidelines of the Declaration of Helsinski and approved by the Ethics Committee of the research center of a “Blinded institution” (protocol code CER-CHUM: 20.040, 23 April 2020). An informed consent was obtained from all subjects involved in the study.
