Abstract
Background:
In the US 48% of adults have hypertension, with direct costs in excess of $130 billion per year. Remote patient monitoring (RPM) has been discussed as a useful tool in the treatment of hypertension, but few studies evaluate its cost effectiveness or efficacy in minority, lower socio-economic (SES) populations. Our study aims to evaluate the clinical and financial outcomes of RPM in hypertension management in a primarily minority, low-SES population.
Methods:
In this prospective cohort pilot study, patients with uncontrolled primary hypertension (defined via Joint National Committee 8 guidelines) were randomly selected from a single academically affiliated primary care clinic. Patients were enrolled on a rolling basis for 90 days. Patients were given blood pressure cuffs and transmission hubs and asked to transmit daily blood pressure readings. Patients were called weekly by research assistants and concerns were escalated to the primary care physician. The control group was the remaining 299 uncontrolled hypertensive patients from the same clinic population analyzed via retrospective chart records at the conclusion of the interventional study period. The primary outcome was blood pressure control. Secondary outcomes were relative improvement in systolic pressure and direct costs.
Results:
A total of 13 patients were enrolled into the RPM intervention; these patients were 54% female, 100% African American, and 77% Medicaid. When assessed via intention-to-treat analysis, patients in the intervention group had non-inferior blood pressure control at 90 days (46% experimental vs 31% control, P = .33) and average change in systolic blood pressure at 90 days (13.5 vs 3.7 mmHg, P = .174) while experiencing a significant reduction in office-based visits at 90 days (1.5 vs 5.9, P < .001) as compared to control. Results on per-protocol analysis also showed non-inferior BP control (63% vs 31%, P = .135). Financially, the program generated margins of $29 per patient at 90 days.
Conclusions:
Patients in our minority- and Medicaid-predominant cohort achieved noninferior blood pressure control as compared to retrospective control at 90 days and a significant reduction in all-cause clinic visits at 90 days. The program generated little revenue per patient, with main barriers to implementation including patient compliance and payor denial.
Keywords
Background
Primary hypertension is a widespread public health issue, with a prevalence of 48.1% in adults, and $130 billion per year in direct costs.1,2 Hypertension tends to disproportionately affect minority patients and those with lower socioeconomic status.3,4
Remote patient monitoring (RPM) of blood pressure (BP) by physicians has been proposed as a novel intervention to improve hypertension management. RPM can increase compliance, BP measurement accuracy, and medication dose optimization without relying on office visits and the associated risks of being lost-to-follow-up. International trials have demonstrated that RPM is accessible to a variety of patient populations,5,6 can be non-inferior 7 or even superior in controlling blood pressure as compared to in-office care,8,9 and can often lead to cost savings for patients and healthcare systems.9,10 Though some US-based implementations exist, 11 the cost-effectiveness and sustainability of this approach, especially in populations with lower socio-economic status, has not been well-explored.
Objectives
The objectives of this study were to measure the efficacy of RPM in improving blood pressure control and the financial feasibility of RPM in a largely minority and Medicaid outpatient clinic population. Our primary outcome was the proportion of patients who achieved blood pressure control at 90 days, as per the Joint National Committee 8 (JNC8) guidelines, loosely defined as a blood pressure of less than 140/90 mm Hg. 12 Secondary outcomes included change in systolic blood pressure from baseline at 90 days and cost-efficiency, which includes both financial and operational assessments.
Methods
Study Design
A prospective cohort study was conducted to assess the effectiveness of a digital health intervention for hypertension management. The study followed an a priori hypothesis proposing that by combining weekly check-ins and RPM, hypertension control at 90 days would be improved as compared to a non-RPM control. We also hypothesized that the program costs would be lower than reimbursement.
Participants
Participants were identified through an electronic medical record (EMR) review. Inclusion criteria for the intervention group were patients over 18 years old with a diagnosis of hypertension and at least 2 most recent clinic readings of uncontrolled hypertension within the last 6 months. Exclusion criteria included patients under 18 years old, with an initial home blood pressure less than 140/90 mmHg, or not receiving primary care through the clinic. The hypertension definitions were based on JNC 8 guidelines. 12 A total of 374 patients meeting inclusion and exclusion criteria were selected based on EMR review of patients seen in the outpatient clinic from Dec 2019 to May 2021. About 75 of these patients were randomly selected via computer algorithm for potential inclusion in the intervention group and 299 were assigned to the comparison group. The original power calculation assumed a difference of 15% in the primary outcome between the 2 groups, a 50% rate of primary outcome achievement in the usual care group, and an intervention group of 25 participants, leading to a power of 0.86. The 75 patients randomized to the intervention group were called via telephone and, if amenable to participating in the study, had in-office appointments made between June and September 2021 to sign consent forms and receive instruction on setup and use of the home blood pressure monitoring devices. Of the 75 patients contacted for enrollment, 18 were consented and instructed on BP cuff use. All 18 patients transmitted at least 1 blood pressure reading and were therefore analyzed as part of the study.
Intervention
Participants in the intervention group were given BP cuffs and data transmission hubs to measure blood pressure daily (the BP cuffs transmit over Bluetooth to the hubs, which transmit to a tracking software using cellular networks). Patients were instructed to use blood pressure cuffs once daily while in a relaxed setting and initially given in-person operation instructions for 15 min after the consenting process. Blood pressure readings were then automatically uploaded to a tracking dashboard accessible to the study team. The study team included medical students that were trained in BP management. Medical student team members called patients weekly to check on efficacy, compliance, and any adverse events. To minimize the impact of individual team members on patient results, students rotated assigned patients weekly. Any significant issues were escalated to the primary care physician. BP readings, contact notes, and care plan modifications were kept in a password-protected HIPAA-compliant online repository. Patients were asked to return the devices upon completion of the program, and reminded to do so by phone and physical mail up to 5 times.
Outcomes
The primary outcome measure was the proportion of patients who achieved blood pressure control at 90 days per JNC8 guidelines, which was defined in this study as blood pressure of less than 140/90 mmHg on final reading. Secondary outcomes included change in systolic blood pressure, number of ambulatory care visits in study period, and a cost-efficacy analysis. Office visits included all in-system ambulatory visits and did not differentiate between primary, urgent, or specialty care. Outcome variables were measured by comparing the initial versus final home blood pressure readings for the intervention group and initial versus final office-based blood pressure readings for the control cohort.
Statistical Analysis
Descriptive statistics were used to summarize demographic data, clinical comorbidities, and the number of in-system visits over the 90-day period. An intention-to-treat analysis was conducted using Python statistical software. Univariate outcomes were compared using Welch’s T-tests for continuous data and chi-square analysis for categorical variables.
Financial Analysis
The market price of the BP cuffs and hubs were recorded ($43.54 and $154.39, respectively), along with time spent during clinical management and amount of data transmitted. Staff time costs were estimated using $21/h. The cost of the dashboard used by the clinic to track patient blood pressures was included because the home health hubs were not integrated directly into the EHR. Patient insurance was billed using CPT codes 99453 (RPM setup), 99457 (RPM data transmission), and 99454 (RPM team management time). All program costs were summed up and compared to insurance reimbursement for patients in the RPM program. Fixed clinic costs that preceded the trial (physician salary, billing services, office upkeep costs) were not included in this analysis.
Ethics Approval
The study was approved by the Institutional Review Board at the George Washington University School of Medicine and Health Sciences in 2021 (NCR203075).
Results
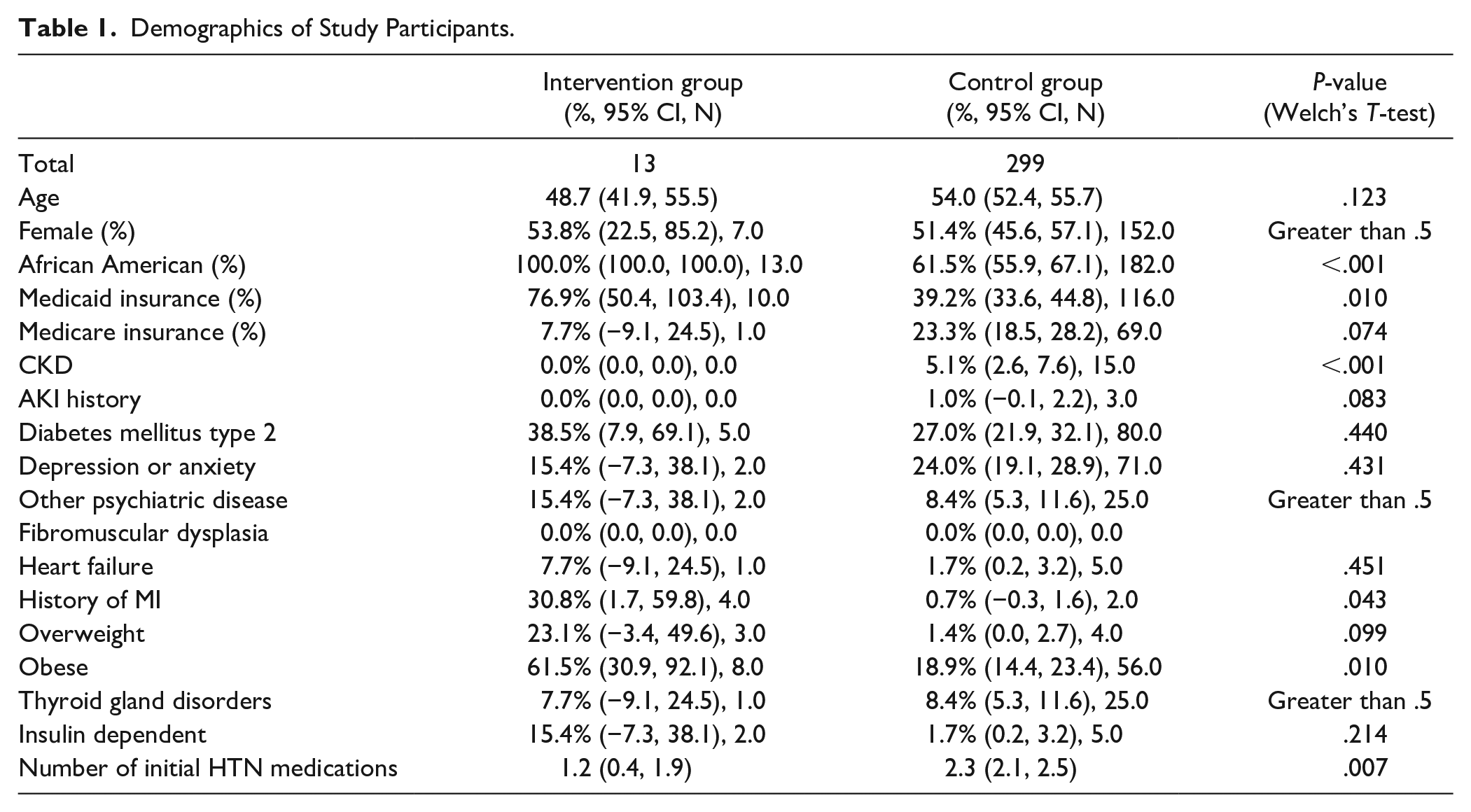
Of the 18 patients consented for the study based on inclusion criteria, 1 was dropped due to subsequent refusal to participate and 4 were dropped due to initial home blood pressure readings of less than 140/90 mmHg. The remaining 13 patients were 54% female, 100% African American, 62% obese, with 77% Medicaid, 8% Medicare Advantage, and 15% privately insured. The intervention group had an average age of 49 (95% CI: 42-56) and took an average of 1.2 (95% CI: 0.4-1.9) antihypertensives at enrollment. Control group patients were taking a larger number of unique antihypertensive medications per patient (2.3, 95% CI: 2.1-2.5) on enrollment with a significantly lower proportion of Medicaid (39%), African American (62%), and obese (20%) patients than the intervention group on univariate demographic analysis (Table 1).
Demographics of Study Participants.
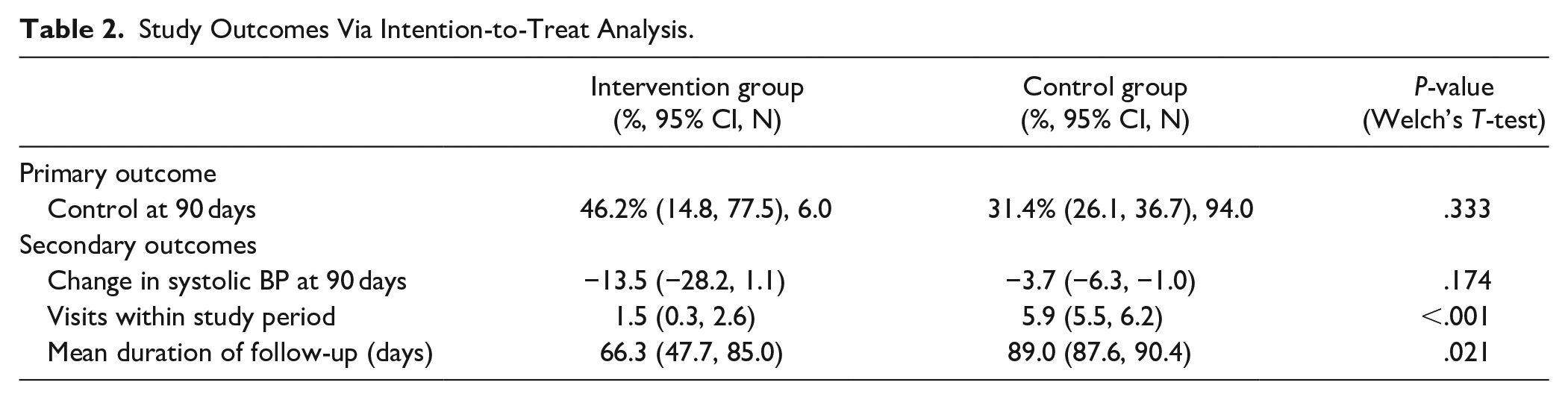
After 90 days, there was no difference in blood pressure control between the intervention and control groups (46%, 95% CI: 15-78 vs 31%, 95% CI: 26-37; P = .33) or average change in systolic BP at 90 days (13.5, 95% CI: −1.1 to 28.8 vs 3.7, 95% CI: 1.0-6.3; P = .174). However, there was a significant difference in the number of office visits within 90 days (1.5, 95% CI: 0.3-2.6 vs 5.9, 95% CI: 5.5-6.2; P < .001; Table 2). In the intervention group, 12 (92%) of patients had medication adjustments over the course of the follow-up period. None of the patient clinic visits in the intervention group were for blood pressure management, and all medication adjustments occurred without office visits.
Study Outcomes Via Intention-to-Treat Analysis.
Mean length of follow-up for the intervention group was 72 days (standard deviation 29 days), and the average number of home BP readings was 63 (standard deviation 61 readings). Of the 13 patients analyzed, 8 patients in the intervention group had a length of follow-up greater than 60 days. At 90 days, BP control in these patients was 63% (95 CI 19-105) versus 31% (95% CI: 26-37) in the control cohort, with P = .135. Systolic blood pressure change was also not statistically significant between the groups (19.5, 95% CI: −0.7 to 39.7 vs 3.7, 95% CI: 1.0-6.3; P = .108), but there continued to be a significant reduction of office visits for enrolled patients (1.2, 95% CI: −0.2 to 2.9 vs 5.9, 95% CI: 5.5-6.2; P < .001; Table 3).
Study Outcomes for Patients Completing the Protocol.

On financial analysis, average cost per patient included $77 for cuff and home health hub (given return rate of 39%), $30 for hub cellular network access, and $31 for clinical follow-up time (at a rate of $21/h for 29 min of time/patient/month), and $300 total for access to the dashboard over 3 months. Physician time and clinic maintenance costs were not accounted for. Revenues included an average reimbursement of $190 (maximum $528) per patient per 90 days, which factored in a 30% denial rate and an additional 27% patient non-adherence rate. Gross margin was $29 per patient.
Limitations
This study was limited by its small sample size, short follow-up period, and single-center design, which may limit the generalizability of the findings. Additionally, the study comparison group was analyzed retrospectively and significantly differed from the intervention group in several tracked features including insurance status and obesity rates, which may introduce bias in the comparison between the remote blood pressure monitoring program and the comparison group. Patients in the intervention group all self-identified as African American, potentially limiting generalizability to other populations. Patients in the intervention group knew they were participating in an intervention, leading to possible participation bias.
Discussion
This study evaluated the effectiveness of an RPM program in controlling blood pressure at a single academically affiliated clinic that serves a primarily low-income, minority population. Our study showed that the implementation of an RPM program was financially feasible and resulted in a statistically significant reduction in office-based visits. Our study also found that the RPM intervention was effective at increasing blood pressure control and decreasing systolic blood pressure at 90 days post-intervention; however, our study was under-powered to detect a statistically significant difference in BP control.
Several factors may have reduced the measured efficacy and power of RPM in improving BP control. Firstly, 4 patients were dropped from the intervention group due to non-hypertensive home blood pressures on initial measurement, which can be explained by white coat hypertension. Additionally, lack of adherence to regular BP measurement over 90 days adversely impacted RPM efficacy, since when the analysis was restricted to the patients with BP measurements spanning over 60 days, BP control rates doubled within the intervention group. It should additionally be acknowledged that significant univariate differences were present in demographic and comorbidity rates between intervention and control groups, which may have led to some confounding of the results.
This study was not designed as a non-inferiority study a priori, but the results support the non-inferiority of RPM programs as compared to an office-based model at controlling blood pressure. Our findings agree with prior literature supporting the efficacy of RPM in leading to BP control compared to office-based care.7 -9 Our study importantly adds to the literature by showing the efficacy of this intervention in a minority and primarily Medicaid population, which tends to have poorer outcomes in a traditional office-based setting, have higher rates of uncontrolled blood pressure, have worse BP-related outcomes, and are historically underrepresented in the literature.3,4,13 Our implementation of RPM also demonstrates the feasibility of starting a financially viable RPM program at a primary care clinic, which has not been fully elucidated for this patient population in the literature.
Barriers to effective implementation of RPM remain. Up-front program costs can be substantial and may vary. These costs include both medical devices (such as blood pressure monitors) and transmission hubs. Given interoperability constraints, more costs are incurred securing software and dashboards that syncs with the medical devices and/or the EHR.
While third party vendors that handle data management and clinical follow up have appeared to improve reimbursement consistency, they still require significant physician review, and the contracting fees are likely to consume any meaningful revenue. Furthermore, these companies fail to provide one of the fundamental aspects that could make RPM a success—increased touches with a trusted practice and physician. A significant number of office visits were prevented in the intervention group. While the sample size may be too small to generalize, the importance and effect of increased touches within primary care has been well studied.
To simulate a leaner clinical operation, we did not contact any payers to discuss RPM prior to program commencement, nor did we appeal any denials. As a result, 30% of claims were denied by payers. Another significant barrier to the financial viability of our implementation was variable reimbursement by insurance for RPM that meets documentation requirements. In fact, most of the variability and initial claims denials that our clinic encountered were from managed Medicaid plans. These plans insure many of the patients who have the worst outcomes in traditional office-based BP management and would therefore benefit most from RPM.
We hope that this implementation guides future integration of RPM into the outpatient setting. Though there is literature beyond our implementation showing the efficacy of RPM as a modality compared to office-based visits in improving blood pressure control in a controlled research setting,7 -9 this implementation provides evidence for non-inferiority of this methodology in a real-world setting, which is not well-explored in the literature. Additionally, this methodology drastically reduces the need for physical infrastructure and office-based visits, potentially allowing for the creation of blood pressure management clinics and primary care practices with little start-up cost and increased flexibility of office space and staffing. Additional study is necessary to confirm the non-inferiority and financial feasibility of this intervention on larger and more heterogenous datasets before additional extension, however.
Conclusions
This study demonstrates that minority and primarily Medicaid patients managed with RPM can have non-inferior BP control at 90 days compared with traditional office-based management, though the study was underpowered to demonstrate efficacy in BP control and there were differences in some demographic variables between groups. RPM was able to significantly reduce office-based visits during the study period and was financially feasible and cost-effective. However, significant administrative, compliance, and payor denial barriers remain to sustainable implementation. More research is required to assess efficacy of RPM in a minority population and to overcome the significant barriers to implementing RPM programs.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The study was approved by the Institutional Review Board at the George Washington University School of Medicine and Health Sciences in 2021 (NCR203075).
