Abstract
Introduction:
COVID-19 vaccines became available after being carefully monitored in clinical trials with safety and efficacy on the human body. However, a few recipients developed unusual side effects, including cerebral venous sinus thrombosis (CVST). We aim to systematically review the baseline features, clinical characteristics, treatment, and outcomes in patients developing CVST post-COVID-19 vaccination.
Methods:
This study was conducted according to the PRISMA (Preferred Reporting Items for Systematic Review and Meta-Analysis) 2020 guideline. Investigators independently searched PubMed, Embase, and Google Scholar for English language articles published from inception up until September 10, 2021, reporting the incidence of CVST post-COVID-19 vaccines. We analyzed CVST patients’ baseline data, type of vaccines, clinical findings, treatment, and outcomes. Our systematic review process yielded patient-level data.
Result:
The final analysis included 25 studies that identified 80 patients who developed CVST after the COVID-19 vaccination. Of the 80 CVST cases, 31 (39.24%) patients died. There was no significant relationship between mortality and age (P = .733), sex (P = .095), vaccine type (P = .798), platelet count (P = .93), and comorbidities such as hypertension (P = .734) and diabetes mellitus (P = .758). However, mortality was associated with the duration of onset of CVST symptoms after vaccination (P = .022). Patients with CVST post-COVID-19 vaccination were more likely to survive if treated with an anticoagulant (P = .039). Patients who developed intracranial hemorrhage (P = .012) or thrombosis in the cortical vein (P = .021) were more likely to die.
Conclusion:
COVID-19 vaccine-associated CVST is associated with high mortality rate. Timely diagnosis and management can be lifesaving for patients.
Introduction
Vaccines were introduced as the most important countermeasure against the COVID-19 pandemic. At present, the leading vaccines across the world are of 2 types according to composition. Two messenger RNA (mRNA) based vaccines, which are encapsulated in lipid nanoparticles and encode the spike (S) protein antigen of SARS-CoV-2, are BNT162b2 (Pfizer–BioNTech) and mRNA-1273 (Moderna).1,2 The second type is the recombinant adenovirus type; among them, one is a recombinant chimpanzee adenoviral vector that encodes the SARS-CoV-2 S glycoprotein named ChAdOx1 nCov-19 (AstraZeneca); and the other one is recombinant adenovirus type 26 vector that encodes S glycoprotein of SARS-CoV-2 (Johnson and Johnson/Janssen). 1 After the initiation of the vaccines, there have been many reports of cerebral venous sinus thrombosis (CVST) and thrombosis of other unusual sites. 2 After analysis of these clinical features, it showed a striking resemblance to heparin-induced thrombocytopenia (HIT). Heparin-induced thrombocytopenia is a prothrombotic disorder caused by platelet-activating immunoglobulin (Ig) G reacting against platelet factor 4 (PF4), which is a self-protein bound to heparin. 3 However, these patients did not have prior heparin exposure, and antibodies to PF4 binding occurred without heparin. 3 Therefore, it was called vaccine-induced immune thrombotic thrombocytopenia (VITT). In VITT, serological findings reflect the findings of the condition, where thrombi develop without prior exposure to heparin, known as “autoimmune” or “atypical” heparin-induced thrombocytopenia, but thrombi in VITT form in locations different from HIT.4,5 According to the American Hematologic Society, “definitive” VITT is defined as a clinical syndrome having the following criteria. 6
The onset of symptoms after 4 to 42 days after COVID-19 vaccination;
Venous or arterial thrombosis at any sites, especially uncommon sites like cerebral venous sinus thrombosis (CVST);
Mild to moderate decrease in platelet count (less than 150 000 mm3);
Positive PF4 antibodies detected by ELISA;
D-dimer elevation to more than 4000 FEU or equivalent
However, not all patients will meet all criteria; thus, any concerning post-vaccination symptoms should be considered.5,6 We conducted a systematic review to describe various presentations, risk factors, treatments regimens, and outcomes in patients of post-COVID-19 vaccine-induced CVST, a VITT, based on the available published articles.
Methods
This systematic review was conducted and reported following the Cochrane and PRISMA (Preferred reporting items for systematic review and Meta-analysis) 2020 guidelines. 7 The pre-specified study protocol has been registered with PROSPERO (CRD42021278094).
Search Strategy
A systematic literature search of electronic databases (PubMed, Embase, and Google Scholar) for peer-reviewed articles conducted in humans and published in the English language from inception up until September 10, 2021. Boolean logic was used for conducting a database search, and Boolean search operators “AND” and “OR” were used to link search terms. The following search terms were used: “SARS-CoV-2” OR “COVID-19” AND “Vaccine” AND “Cerebral venous sinus thrombosis” OR “CVST” OR “CVT” OR “Cerebral venous thrombosis.” The medical subject headings (MeSH) database was used for advanced PubMed search to find MeSH terms for the search terms mentioned earlier. Similarly, for advanced Embase search, Emtree terms were used for the search terms mentioned above. The search was also expanded to include preprint servers and thesis repositories, and any additional references were found by hand-searching the reference lists from the selected articles.
Study Selection
The studies that had been shortlisted were then imported into the EndNote software (Clarivate), where duplicates were removed. A manual check was then performed to remove any remaining duplicates. Two reviewers first independently reviewed papers based on title, keywords, and abstract and any conflicts were resolved by a third reviewer. Two reviewers thoroughly reviewed articles that passed the initial screening to determine their suitability for inclusion in the systematic review. We resolved differences in the final study selection between the 2 primary reviewers by consulting a third reviewer. Population overlap was assessed based on authorship, hospital setting, and recruitment period. Higher-quality studies or studies with larger sample sizes were included when there was overlap.
Inclusion and Exclusion Criteria
The systematically searched studies were read in their entirety to determine their suitability for inclusion in the systematic review. The following criteria were used for inclusion: Studies published in English, either case reports or case series/observational studies; studies including subjects of age group >18 years who developed CVST after receiving COVID-19 vaccine. We excluded all review articles, papers reporting CVST following COVID-19 infection, letters to the editor, short commentaries, and animal studies.
Data Extraction
The following data were extorted from the included studies: demographic data (study design, country, gender, and age), timing from vaccination to symptoms onset, comorbidities, presenting symptoms, anti-PF4/heparin antibodies, thrombosis risk factors, platelet nadir, medications used, arterial or venous thrombosis, diagnosis, and mortality. All available information was assembled in a shared spreadsheet by 4 authors. If any required data was missing, was not reported in the paper, or was written in an unusual format, the corresponding authors of the respective papers were contacted via email for clarification. Supplementary material related to the main article was also investigated in such cases.
Statistical Analysis
Our systematic review process yielded patient-level data. Of the 25 case studies and case series identified, 80 patient-level data were available. The published outcome data (mortality) for each case were dichotomized as dead or alive. This variable was set as a dependent variable for analysis. Other aforementioned variables extracted were set as independent variables. Descriptive statistics were tabulated for the analytic cohort. Continuous data were reported as mean with standard deviation (SD) and compared using the independent t-test. Categorical data were expressed as frequency and percentage and compared using the χ2 test. All the statistical analyses were performed using SPSS 25 (IBM Corp. Released 2017. IBM SPSS Statistics for Windows, Version 25.0. Armonk, NY: IBM Corp.). A two-tailed P-value <.05 was considered statistically significant.
Results
Study Selection and Characteristics
After a rigorous search, we found 597 articles through different electronic databases. After manual removal of duplicates, 130 articles were initially screened by title and abstracts against the eligibility criteria. After removing 73 articles from screening, 57 full-text articles were selected. Thirty-two did not meet the inclusion criteria and outcome of interest and were thus excluded. Finally, 25 articles were included in our review. Figure 1 depicts the PRISMA diagram illustrating the study selection process. Four studies were from the USA, 19 from Europe, and the rest were 2 studies from Asia. Twenty-two studies were case reports, and 3 studies were case series. The detailed methodological features and patient characteristics of 25 included studies are provided in Supplemental Table 1.
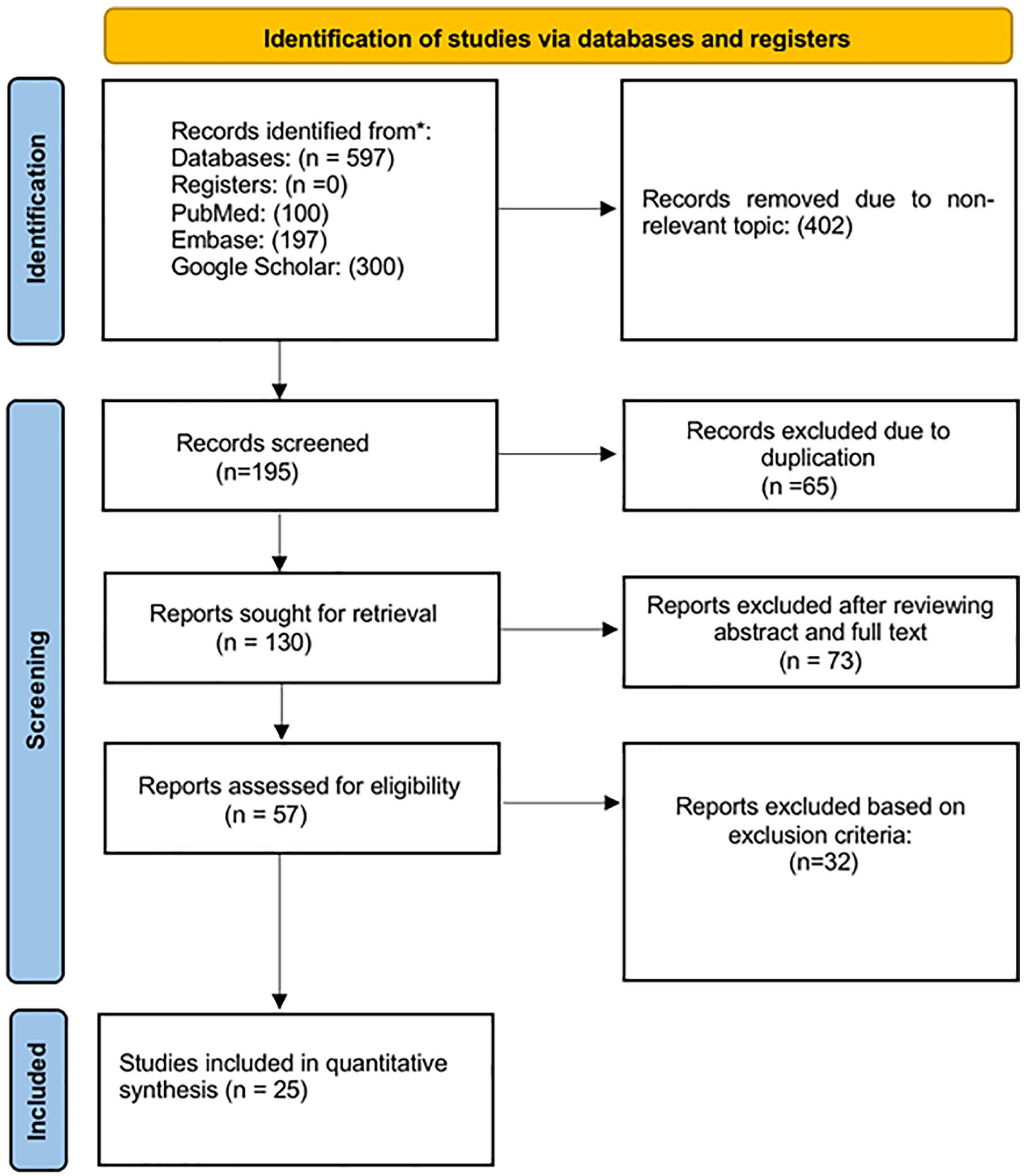
PRISMA flow diagram depicting the flow of information through the different phases of our study.
Baseline Characteristics of Included Patients
Of the 25 case studies and case series identified, 80 patients were identified to have developed CVST after being vaccinated against COVID-19. Of the 80 patients, 21 (26.3%) were male, and 59 (73.8%) were female. The mean age of patients was 42.68 ± 13.968 years. Mean platelet count was 46 113 ± 57 670/mm3. Four different vaccines were identified: 54 (66.3%) AstraZeneca (ChAdOx1 nCoV-19), 16 (20.0%) Johnson & Johnson/Janssen (Ad26.COV2.S), 7 (8.0%) Pfizer BioNTech (BNT162b2 mRNA), and 4 (5.0%) Moderna (mRNA-1273). The mean time for onset of symptoms after vaccination was 11.10 ± 5.34 days. Lab results for antibodies against platelet factor-4 (anti-PF4) were found in 45 (56.3%) subjects but were negative in 35 (43.8%) subjects. The comorbidities and other risk factors found among the patients of this study are as follows: hypertension (n = 9, 11.3%), diabetes mellitus (n = 7, 8.8%), dyslipidemia (n = 5, 6.3%), thyroid disorder (n = 5, 6.3%), clotting factor disorder (n = 4, 5.0%), allergy (n = 2, 2.5%), obesity (n = 1, 1.3%), Meniere’s Disease (n = 1, 1.3%), cholangitis (n = 1, 1.3%), pre-eclampsia (n = 1, 1.3%), use of hormonal contraceptives (n = 8, 10.0%), previous thrombotic event (n = 3, 3.8%), corticosteroid use (n = 1, 1.3%), and Tricyclic Antidepressants (TCA) use (n = 1, 1.3%) (Table 1).
Demographic Characteristics of Patients With CVST Events After COVID-19 Vaccination.

Tricyclic antidepressant. **Antibody against platelet factor 4.
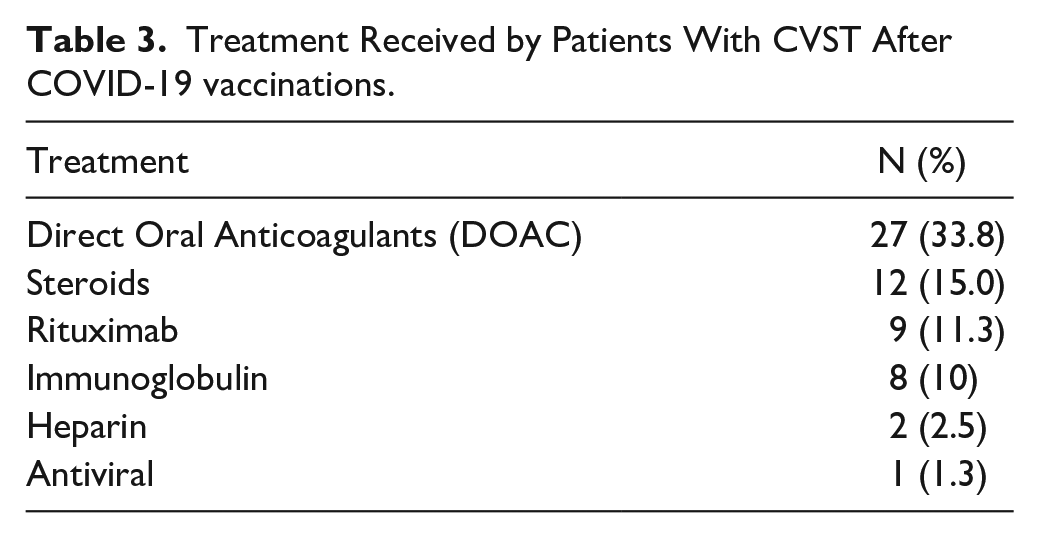
Table 2 shows the veins involved in CVST, complications associated with CVST and other non-CVST thrombotic conditions. Sigmoid sinus (n = 13, 16.3%) was the most commonly involved sinus followed by superior sagittal sinus (n = 9, 11.3%) and right transverse sinus (n = 8, 10.0%). Other thrombotic events associated were deep vein thrombosis (n = 7, 8.8%), pulmonary embolism (n = 11, 13.8%), and other systemic venous thrombosis (n = 20, 25.0%). Intracerebral hemorrhage (ICH) occurred in 35 out of 80 patients (43.75 %), thrombocytopenia occurred in 10 (12.5%) patients and thrombotic microangiopathy occurred in 1 (1.3%) patient. Among 80 CVST patients, 27 (33.8 %) CVST patients were treated with DOAC, 12 (15.0 %) with steroids, 9 (11.3%) with rituximab, 8 (10%) with IVIG, 2 (2.5 %) with heparin, and 1 (1.3%) received anti-viral (Table 3).
Veins Involved in CVST, Complications, and Other Thrombotic Events.

Unspecified location.
Treatment Received by Patients With CVST After COVID-19 vaccinations.
Mortality
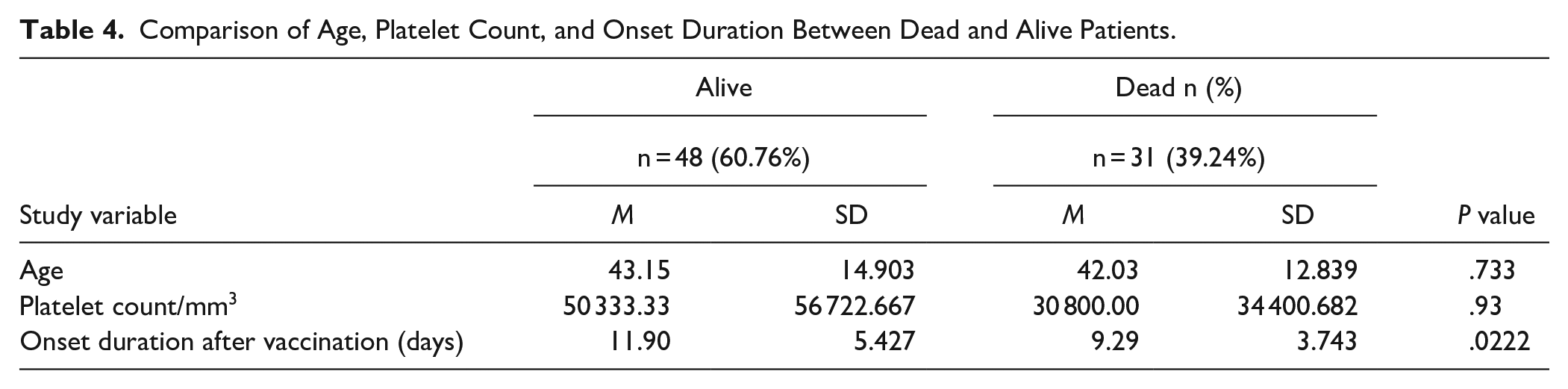
Out of 80 CVST cases, 31 (39.24%) patients died. Univariate analysis was performed to see the association of independent variables with mortality. Age and baseline platelet count were not significantly different between dead and alive patients. However, dead patients had early onset of CVST symptoms after vaccination compared to living patients (Table 4).
Comparison of Age, Platelet Count, and Onset Duration Between Dead and Alive Patients.
There was no significant relationship between mortality and sex (P = .095), vaccine type (P = .798), hypertension (P = .734), diabetes mellitus (P = .758), dyslipidemia (P = .651), thyroid disorder (P = .971), coagulopathy (P = .651), hormonal contraceptive use (P = .915), previous thrombotic event (P = .321), and anti-PF4 positivity (P = .440) (Table 3). Similarly, mortality was not associated with any of the following factors: allergy, hepatitis B infection, obesity, Meniere’s disease, cholangitis, corticosteroid use, and TCA use (Table 5).
Comparison of Various Categorical Variables Between Dead and Alive Patients.

Patients with CVST after having a COVID-19 vaccination were more likely to survive if they were treated with an anticoagulant (P = .039). Steroids, IVIG, rituximab, and heparin did not influence mortality (Table 5). There was a significant relationship between having intracranial hemorrhage and mortality (P = .012). Similarly, there was a significant relationship between having a thrombosis in the cortical vein and mortality (P = .021). There was no significant association of other venous thrombosis and complications with mortality (Table 6).
Site of CVST, Complications, and Other Thrombotic Events in Alive and Dead Patients.

Discussion
Our findings indicate that the majority of CVST cases post COVID-19 vaccination occurred in patients administered AstraZeneca vaccination (74%) and Johnson & Johnson/Janssen (8%). This is in line with the previous reports by the Centers for Disease Control and Prevention (CDC) highlighting this rare side effect among both vaccines. 8 Other reports have indicated that there was an increase in CVST incidence following the post-COVID-19 AstraZeneca and Johnson & Johnson/Janssen vaccination when compared to pre-pandemic incidence.2,9
The increase in incidence could be explained by pathophysiological changes thought to occur in VITT. CVST post-COVID-19 vaccination arises as a consequence of VITT. 10 This is corroborated by the presence of anti-PF4 antibodies and the low mean platelet count (46 113 mm3) in the reported cases in this review. It is hypothesized that the free DNA in both adenovirus-based vaccines (AstraZeneca and Johnson & Johnson/Janssen) bind PF4, provoking the formation of PF4-reactive antibodies.1,11
Our findings also indicated that there is a female gender predilection in the reported CVST cases, which is consistent with previous reports that indicate that there is an increased prevalence of CVST amongst the female gender.12,13 This predilection is thought to be attributed to several risk factors that include oral contraceptive use and hormonal replacement therapy. 14 This is in line with our findings that showed 16% of identified cases of CVST in female patients reported the use of oral contraceptives. It is important to note that our findings, however, showed that the presence of the various risk factors that increase the risk for CVST had no statistically significant effect on the mortality rate.
Large cohort studies conducted pre-pandemic indicate the mortality associated with other causes of CVST is quite low. A cohort by Haghighi et al 15 revealed that among 3488 patients with CVST, the overall mortality rate was 4.39%. Additionally, a study by Nasr et al 16 have shown that among 11 400 hospitalized CVST patients, only 232 (2.0%) suffered from in-hospital mortality. A notable finding in our review is the high mortality rate (39%) associated with CVST which could indicate that mortality rate of vaccination induced CVST differs from other causes of CVST. This is further corroborated by a study by Kryzwicka et al 17 which showed that among 117 patients with CVST following AstraZeneca vaccination, the mortality rate was 38%. However, this systematic review had a small sample size, 80 patients, and larger observational studies would be needed to address the mortality rate of CVST associated with COVID-19 vaccinations.
Our results indicate that the only treatment modality that was associated with a statistically significant reduced mortality risk was non-heparin anticoagulation as recommended by the current guidelines. 18 This finding is of clinical importance as it confirms the hypothesis that VITT and HIT share similar pathophysiology and respond to similar treatment regimens. Other treatment modalities that could be used in VITT that showed promise in HIT include corticosteroids and intravenous Ig (IVIG). 18 IVIG could aid in interrupting prothrombotic mechanisms in VITT and also help in improving platelet count.19,20 Administering corticosteroids as a second-line therapy to patients with CVST may lead to an increase in platelet count and hence decrease the risk of hemorrhagic events. 3 Further research is required in this area to improve recognition and determine the optimal management of CVST.
It is important to note that COVID-19 infection is also of itself a risk factor for developing CVST and is more likely to cause CVST in comparison to COVID-19 mRNA vaccines (Pfizer and Moderna). 21 A previous systematic review analyzing VITT and CVST post-COVID-19 vaccination showed that no cases were reported following Pfizer or Moderna. 22 However, our review identified only 11 cases of CVST that occurred following mRNA vaccinations. A recent cohort study also revealed that there is an increased risk of acquiring CVST following Pfizer vaccination, however, the risk remained higher following AstraZeneca. 23
Limitations of this review include missing information in the case description of some of the case reports and case series. Secondly, the type of studies reviewed (case reports and case series) are inherently limited and considered to possess a lower level of evidence compared to those acquired from larger clinical studies, limiting the inferences that could be gathered from such studies.24,25
Conclusion
COVID-19 vaccine induced CVST can occur at any age and gender with any vaccine type. Even though it is more common in adenoviral vaccines, mRNA vaccines are not devoid of such complications. Clinicians should remain vigilant not to overlook COVID-19 vaccine induced CVST, as our findings indicate CVST is still associated with a high mortality rate despite the small sample size. Timely diagnosis and management can be lifesaving for patients.
Supplemental Material
sj-docx-1-jpc-10.1177_21501319221074450 – Supplemental material for Cerebral Venous Sinus Thrombosis Following COVID-19 Vaccination: A Systematic Review
Supplemental material, sj-docx-1-jpc-10.1177_21501319221074450 for Cerebral Venous Sinus Thrombosis Following COVID-19 Vaccination: A Systematic Review by Vikash Jaiswal, Gaurav Nepal, Patricia Dijamco, Angela Ishak, Mehak Dagar, Zouina Sarfraz, Nishat Shama, Azza Sarfraz, Kriti Lnu, Saloni Mitra, Preeti Agarwala, Sidra Naz, David Song and Akash Jaiswal in Journal of Primary Care & Community Health
Footnotes
Acknowledgements
We want to thank Siena Placino, Dattatreya Mukherjee, and Nikkita Choudhary for their immense help in preparing this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
Data Availability Statement
All data relevant to the study are included in the article or uploaded as supplemental information.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
