Abstract
Keywords
Introduction
Directly observed treatment short course (DOTS) is the main pillar employed for the control of tuberculosis (TB) under the Revised National Tuberculosis Control Programme (RNTCP) in India. Isoniazid (INH), pyrazinamide (PZA), and rifampicin (RMP) used in DOTS as the main drugs are potentially hepatotoxic and may lead to drug-induced liver injury (DILI). 1 Ethambutol and streptomycin, although included in the DOTS regimen have different adverse effect profile other than DILI.1-3 DILI caused by anti-TB (ATT) drugs is a serious adverse effect limiting the treatment of TB as well as increasing morbidity and mortality in these patients substantially. ATT medications may give rise to severe hepatotoxicity, liver failure, and even death.2-4
The prevalence of DILI is much higher in developing countries owing to several factors such as acute or chronic liver disease, alcoholism, malnutrition, indiscriminate drug use, advanced TB, and other coexisting chronic illness. ATT drugs may cause hepatotoxicity ranging from a transient asymptomatic rise in liver enzymes to acute liver failure. The reported mortality from DILI after the development of jaundice varies from 4% to 12%. 5 It is to be noted that the frequency of DILI in different countries varies widely from 2% to 39%. 6
INH and RMP often cause DILI by diverse mechanisms. 4 In a meta-analysis, it was shown that RMP in a multidrug regimen increased the prevalence of DILI from 1.6% to 2.55% in adults. 7 PZA has also been demonstrated to increase the risk of liver injury significantly.8,9 Decreased activities of daily living and chronic heart disease have been associated with a higher prevalence of DILI with bilirubin >34 µmol/L in patients receiving ATT. 10 The reported prevalence of DILI in different anti-TB chemotherapy that included INH, RMP, and PZA ranged from 0.6% to 3%4,11,12 and among all the first-line ATT agents, PZA is the most common cause of DILI. 13
From an analysis of pooled data from 4 prospective Indian studies with DOTS,14,15 the risk of DILI was found to be 11.5%, whereas meta-analysis of 14 studies from the west found the same risk to be 4.28%. 16 With this background, this study was carried out to study the prevalence of DILI among patients who had received anti-TB medications and to study some of the factors such as alcohol consumption, hepatitis B and C, human immunodeficiency virus (HIV), doses of drugs (fixed or per kg), ATT regimen (daily or intermittent), RNTCP categories, patient age, sex, and weight responsible for causing DILI among these patients.
Materials and Methods
This study was started after obtaining approval from the Institutional Review Board, Christian Medical College, Vellore. This was a longitudinal, descriptive study, using preformatted forms, patients’ charts obtained from the Medical Records Department and patients’ register maintained in the Department of Microbiology. Patients of all ages diagnosed with smear positive pulmonary tuberculosis and treated in the Departments of Medicine–I and Pulmonary Medicine from January 1, 2008 to December 31, 2012 and those patients who came for regular follow-up after ATT was initiated were included. Patients who had no follow-up, patients with liver disease prior to initiation of ATT therapy, and patients for whom not even a single liver function test result was available during follow up period were excluded.
DILI was defined by the presence of any one of the following criteria 17 :
A rise to ≥5 times the normal serum level of transaminases (normal serum glutamate-pyruvate transaminase or SGPT: 7-41 U/L, normal serum glutamic oxaloacetate transaminase or SGOT: 12-38 U/L)
A rise in the level of serum total bilirubin > 1.47 mg/dL
Any increase in serum transaminase above pre-treatment levels together with symptoms of anorexia, nausea, vomiting, and jaundice.
Data analysis was performed using statistical program R (version 3.1.0). Multiple logistic regression analysis was performed to determine the association of different risk factors (chronic alcohol consumption, hepatitis B infection, hepatitis C infection, HIV infection, and existing chronic TB) and DILI. The confounders considered were age, sex, weight, body mass index, doses of drugs (fixed or per kg), ATT regimens (daily or intermittent), and treatment categories.
Results
A total of 787 patients’ charts were screened and only 253 were included for analysis. Out of these 253 patients, 24 (9.48%) developed drug-induced DILI. The distribution of patients receiving different treatment regimens and having different risk factors for DILI are enumerated in Table 1. According to RNTCP, patients receiving category 1 regimen are patients with new sputum smear-positive TB, new sputum smear-negative TB, and new extrapulmonary tuberculosis TB; and patients receiving category 2 regimen are patients with sputum smear-positive relapse, sputum smear-positive failure, and sputum smear-positive treatment after default. 18 Patients under DOTS regimen received intermittent dosing. 18
Distribution of Patients Based on Treatment Regimens and Presence of Risk Factors (n = 253).

Abbreviations: RNTCP, Revised National Tuberculosis Control Programme; TB, tuberculosis.
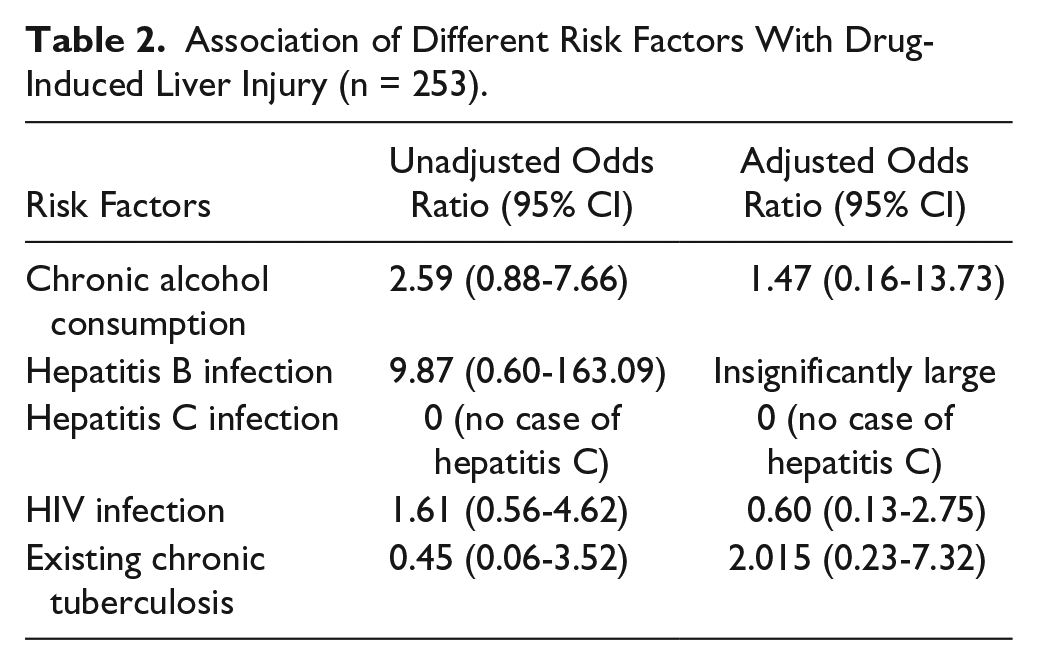
All associations of risk factors with DILI were statistically insignificant as shown in Table 2. Moreover, DILI had very poor correlations with other known confounders like age (r = −0.08), sex (r = 0.01), weight (r = −0.01), body mass index (r = −0.06), doses of drugs (fixed or per kg), ATT regimens (daily or intermittent) (r = −0.04), and RNTCP categories (r = 0.01).
Association of Different Risk Factors With Drug-Induced Liver Injury (n = 253).
Discussion
None of the known risk factors mentioned were significantly associated with DILI. This is interesting as many of these factors have been shown to be independently and significantly associated with DILI in different studies. The development of DILI during TB treatment is the most common reason leading to interruption of therapy. 19
A prevalence of DILI from TB treatment was reported from Taiwan at 12% (111/926) within a median 38 days from the initiation of treatment (with 3.5% developing severe DILI), 1 and from China at 10.4% (267/2457) within the first 2 months of treatment. 20 Independent risk factors for DILI were overweight/obesity status and smoking in a study from Peru 21 and advanced age, hypoalbuminemia, high alcohol intake, slow acetylator phenotype, and associated chronic disease in an Indian study. 22 Patients with HIV coinfection are prone to DILI possibly due to the concomitant use of protease inhibitors 23 as shown in a previous Indian study. 22 HBV coinfection is also reported to increase DILI.22,24 Our study also goes at par, showing the prevalence of DILI as 9.48% without any association of risk factors.
The therapeutic approach in DILI is quite challenging. According to the present recommendations, anti-TB drugs should be withheld until the normalization of the liver function.25,26 The American Thoracic Society recommends initiation of new treatment regimen provided that liver enzymes are below twice the upper limit of normal. 27 Reintroduction of hepatotoxic ATT drugs must be balanced carefully with their benefit by thorough supervision and close monitoring and baseline laboratory testing and monitoring should be ensured with ATT treatment. 28
Our study has some limitations. Although the odds ratio was quite high for chronic alcohol consumption, the association was insignificant owing to the small sample size, which also contributed to the broad confidence interval. The same holds true for the abnormally large adjusted odds ratio with hepatitis B infection, which was also because of the fact that there was only one positive case of hepatitis B in our sample. We could not obtain full data from case notes of some included patients such as pretreatment liver function tests, anthropometry (including body mass index), comorbidities, comedications, and so on. We also could not comment on the status of DILI among the excluded patients. We do not know why exactly many patients did not turn up for follow-up or why liver function test was not done for all patients during this period. From our usual experience in our hospital setup, we can say quite a large number of patients come from afar, who are often very irregular during follow-up. Also, affordability, poor compliance, nonspecific presentations, and subjective variation among the different treating physicians might throw some light in this regard. A prospective study in a controlled setting is much warranted.
Conclusion
The prevalence of clinically significant DILI is not much high (9.48%) in our settings. Also this was not significantly associated with known risk factors.
Footnotes
Acknowledgements
The authors are grateful to the Department of Medicine–I, Christian Medical College, Vellore, for giving access to patient data and Medical Records Department for helping with data collection.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
