Abstract
Previous studies have described an increased risk of developing an additional connective tissue disease (CTD) when one such ailment is present. We examine here the likelihood that individuals with systemic lupus erythematosus (SLE) screen positive for one or more of the following five autoimmune CTDs: Sjögren’s syndrome, scleroderma, rheumatoid arthritis, dermatomyositis/polymyositis, and mixed connective tissue disorder. Five hundred SLE-diagnosed subjects were asked to complete a CTD screening questionnaire (CSQ). The results were analyzed according to the set of diagnostic criteria given by the American College of Rheumatology to identify probable cases of each CTD. Significant standardized prevalence ratios and comorbidities indicate an increased risk for the other autoimmune CTDs. In all, 96% of the subjects screened positive for at least one additional CTD, and 13% screened positive for at least two additional CTDs. We see that the SLE-diagnosed population may benefit from further attention regarding the presence of additional CTDs, which may further inform treatment strategies. We also see the application of the CSQ as a potentially important tool for clinical practice, and we describe the present study’s limitations along with possible ways that these can be addressed.
Keywords
Introduction
Connective tissue diseases (CTDs) are a group of syndromes that involve inflammation of the connective tissues, which include skin, tendon, ligaments, cartilage, and bone. Autoimmunity plays a role in the etiology of CTDs, 1 and for those with one CTD there is an increased risk for another.2,3 For example, systemic lupus erythematosus (SLE) can be present in those with rheumatoid arthritis (RA) or Sjögren’s syndrome (Sjö).4-6 Further, the symptoms of different CTDs in the same individual can overlap. 7
In a study conducted in Denmark by Eaton et al 8 in 2007, associations between CTDs were found. These include associations between SLE and Sjö (odds ratio = 88) and between SLE and RA (odds ratio = 226). A further study showed that 14% of SLE patients and 11.8% of RA patients are found to have scleroderma. 9 In some CTD pairings, for example between SLE and Sjö, serologic markers suggest a subgroup that is distinct from either CTD alone. 10 The common clinical overlap, shared genetic factors, and shared immunologic pathways of CTDs has led to the grouping of some conditions as polyautoimmunity conditions or overlap syndromes. 9
The autoimmune rheumatic CTDs are thought to be underdiagnosed. 11 That may partially be due to the observation that both environmental and genetic factors are found to play a role in disease development.12,13 Overlapping multifactorial etiologies, 14 and overlapping symptoms complicate diagnoses. Screening tools may offer an effective means to identify possible cases of autoimmune CTDs and subsequently to support appropriate diagnostic and medical interventions. Here we applied the connective tissue screening questionnaire (CSQ) 3 to screen for RA, Sjö, scleroderma, dermatomyositis/polymyositis (DM/PM), and mixed connective tissue disease (MCTD) in subjects of Northeast Pennsylvania who were previously diagnosed with SLE. The intent was to estimate the presence of comorbidities of autoimmune CTDs among these individuals of the community.
Methods
Study Subjects and Data Collection
Five hundred SLE-diagnosed individuals were systematically selected from the Northeast Branch of the Lupus Foundation of Pennsylvania database of 1750 members. Surveys were administered and collected in two rounds of 250 individuals. In the first round, every fifth person in the database was selected, and in the second round every third person was selected from the remaining set. Each person was mailed a packet, which contained a cover letter that briefly described the purpose of the study, a questionnaire, an informed consent form, and a stamped return-envelope. Participants were asked to fill out the questionnaire individually as a self-administered survey. The completed surveys were to be returned within four weeks. Identification numbers were assigned to the surveys for the purpose of reporting the screening results back to the participants.
There were 35 questions in the survey. The first question identified if there had been any prior autoimmune CTD diagnoses. The next four questions collected demographic information: age, gender, race, and county of residence. The remaining 30 questions were adapted from the CSQ as developed by Karlson et al. 3 The answers to these questions were evaluated to predict the presence of five CTDs: RA, SD, Sjö, DM/PM, and MCTD. Participants were notified of their individual screening results. The study design was approved by the Scranton-Temple Residency Program Institutional Review Board.
Data Analyses
The scoring algorithms for the prediction of CTDs are based on American College of Rheumatology (ACR) classification system, 3 and we converted a corresponding scoring computer program provided by Dr Elizabeth Karlson to Microsoft Excel macros. For each CTD, participant screening results were dichotomized as a probable case (positive result) or not a case (negative result). Participants were excluded from a given CTD screening if data for that screening were not complete. Standardized prevalence ratios (SPRs) were calculated for four CTDs using their respective weighted mean prevalence rates in the United States. 15 Screening results were cross-tabulated and analyzed with χ2 tests for each CTD-CTD pair. An α value of .01 was used to ascertain a significant association as multiple tests were performed. 16
Results
A total of 167 participants with self-reported diagnosed SLE responded with a completed survey, corresponding to an average response proportion of 33%. All completed surveys indicated a screen positive for SLE. Participants were 90% Caucasian women with a mean age of 51 years. We note that the reported proportion of total Caucasians in Northeast Pennsylvania is 95%. 17
The numbers of previously diagnosed and newly screened positive participants for five CTDs are shown in Table 1. In all, 83% screened positive for an additional CTD, and 13% screened positive for at least two additional CTDs. To test for specific comorbidities, Table 2 shows the χ2 test results for each of CTD pairing. MCTD and DM/PM were associated with all CTDs except for Sjö (P < .01). An association between scleroderma with dermatomyositis is observed. A possible association between scleroderma and RA is found (P = .017). Sjö is not found to be significantly associated with any of the CTDs studied other than SLE.
Numbers of Positive Screenings and Previous Diagnoses for Each CTD.

Abbreviations: CTD, connective tissue disease; SN, screen negative; SP, screen positive; RA, rheumatoid arthritis; Sjö, Sjögren’s disease; DM/PM; dermatomyositis/polymyositis, MCTD, mixed connective tissue disease; SD, scleroderma.
Includes only those surveys with questions fully completed for the respective CTD.
Associations Between Autoimmune Connective Tissue Disorders Based on Screening Results Among SLE-Diagnosed Individuals. a
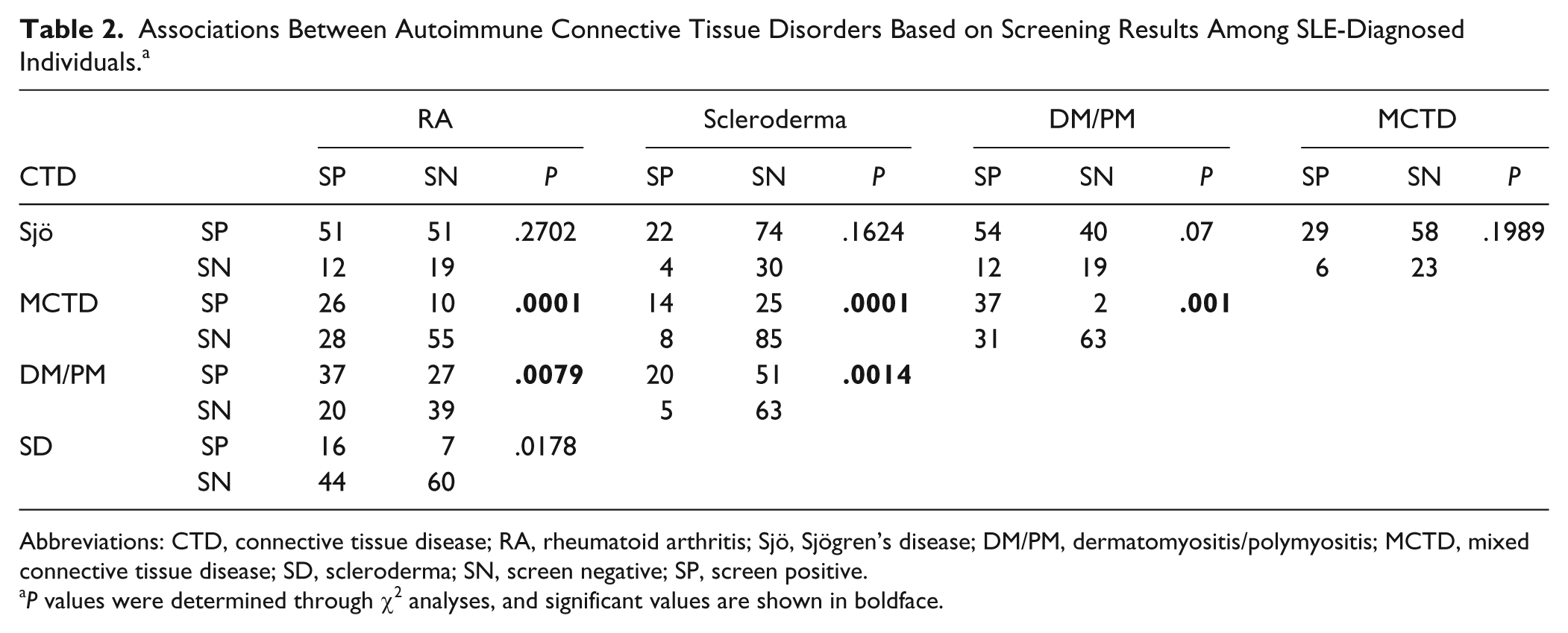
Abbreviations: CTD, connective tissue disease; RA, rheumatoid arthritis; Sjö, Sjögren’s disease; DM/PM, dermatomyositis/polymyositis; MCTD, mixed connective tissue disease; SD, scleroderma; SN, screen negative; SP, screen positive.
P values were determined through χ2 analyses, and significant values are shown in boldface.
The SPRs are elevated, which indicates the associations of the CTDs with SLE. DM/PM is the most elevated with an SPR of 1,028,383 (95% confidence interval [CI] = 841,666-1,289,110), Sjö, 163,966 (95% CI = 120,722-229,135), scleroderma, 451,535 (95% CI = 325,331-644,621), and RA, 5,602 (95% CI = 4,521-7,127).
Discussion
Connective Tissue Disease Comorbidities
Comorbidities of the CTDs can be examined in the context of previous findings. MCTD and DM/PM are both significantly associated with each CTD except Sjö. MCTD is described as an overlap syndrome, so it is expected to be associated with other CTDs. 11 Dermatomyositis has previously been shown to be associated with seropositive RA and scleroderma through a study using diagnostic testing, 6 which supports our results.
Given the relatively high positive predictive value (84.5%) of the CSQ for detecting individuals with any CTD, but relatively low positive predictive value at detecting specific CTDs (SLE, 47%; RA, 31.5%; scleroderma, 61%; DM/PM, 23.5%), 3 it is reasonable to infer that some of positive screenings are true cases. As such, these cases are going undiagnosed. Assuming all positive screenings and prior diagnoses are accurate, only 4% of individuals have only SLE, while 13% of study participants have at least two additional CTDs.
The described SPRs are highly elevated. This is to be expected as overlap syndromes are highly associated with SLE. In particular Sjögrens can be linked through common immunological pathways. 10 Although the additional CTDs studied are relatively rare in the general population, their elevated levels here suggest they are to be given further attention in individuals with SLE.
We expect that there are a significant number of false positives with the screening tool because of symptomatic overlap the diseases. For example, we found that in total 66/137 or 48% of the participants have RA. We attribute this to be due in part to the overlapping ACR criteria for SLE and RA.18,19 We also see that there is a corresponding overlap syndrome referred to as rhupus,4,14,20 and rhupus is recommended to require further attention and possibly alternative intervention strategies. 19 Stepping back, we assess that the application of the CSQ can act as a tool for decision support. That is with regard to the possible further diagnostic testing for the presence of an additional CTD and/or the presence of an overlap syndrome.
Screening Test Validity
The sensitivity and specificity estimates of the CSQ for the detection of CTDs are reported in ranges of 83% to 96% and 86% to 95%, respectively. 3 Our estimates of sensitivities of the CSQ for each CTD are based on the proportion of screen positive results among all self-reported cases. With the exception of MCTD, these sensitivities fell within the range for other control groups, 3 and we consider them as reasonable estimates. We were not able to estimate the specificities of the CSQ as we did not have the results of the corresponding diagnostic tests.
Limitations of the Study
Volunteer bias may have occurred in our sample of the SLE diagnosed population. Particularly, individuals who agreed to participate may have additional ailments and that increased their likelihood of participation. Recall bias is likely present as SLE is a chronic condition in which symptoms may fluctuate with time. As a benefit, we see the current implementation of a geographically based registry of subjects rather than a single site study.
We used here only a screening tool and no diagnostic assays. Future work may select study participants from a large clinical database and require physician records for diagnoses. From there we see the need for a more detailed look at the predictive values of the CSQ in an SLE diagnosed population. We currently see it as a supportive tool as the ACR criteria underlie its application.
Conclusion
The accuracy of the CSQ for the detection of additional CTDs among an SLE diagnosed population is yet to be fully established. Methodological studies are warranted to estimate the true operating characteristics of the CSQ for the SLE diagnosed group. Based on the current results, individuals in the study may have additional CTDs, and these CTDs may be undiagnosed. This suggests that the high-risk population of SLE diagnosed individuals should receive further attention with regard to the presence of additional CTDs and subsequently receive possibly more specific intervention strategies.
Footnotes
Acknowledgements
We thank the following for helping to facilitate in the completion of the study: The Commonwealth Medical College’s Department of Family, Community, & Rural Health and Mark V. White, MD, MPH. We thank Tyler Lambing, Darshana Patel, Maram Bishawi, Mark Esolen, Jennifer Powel, Ozo Akah, Beth Rundell, and the members and staff of the Northeast Branch of the Lupus Foundation of Pennsylvania for their help in administering the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
