Abstract
Lemierre syndrome develops in healthy young patients as a result of bacteremia after oral cavity infection. It causes thrombophlebitis in the internal jugular vein. Infection can easily occur during immunosuppressive treatment in patients with systemic lupus erythematosus and become severe. We present a case of Lemierre syndrome in a patient with systemic lupus erythematosus. A 56-year-old woman presented with fever, left lower toothache, and skin symptoms from the left neck to the anterior chest. Clinical presentation and laboratory investigations revealed Lemierre syndrome. The inflammation and thrombus disappeared with antibiotic and anticoagulant therapies. However, transient hypocomplementemia and elevated antinuclear antibody levels were observed during treatment; therefore, a concomitant systemic lupus erythematosus flare was considered. In systemic lupus erythematosus patients with Lemierre syndrome, complement and antinuclear antibody levels are modified, so other indicators should be precisely evaluated, such as levels of urinary protein, sediment, serum creatinine and anti-dsDNA antibody, and systemic lupus erythematosus disease activity index.
Keywords
Introduction
Lemierre syndrome is a septic thrombophlebitis of the internal jugular vein that can develop as a severe complication of oropharyngeal infection. Most cases result from bacteremia in otherwise healthy young patients after oral cavity infection, and the disease is still associated with high mortality.1,2 Infection easily occurs during immunosuppressive treatment in patients with systemic lupus erythematosus (SLE) and can become severe. 3 In this regard, hypocomplementemia and elevated autoantibody concentration have been reported during infections in patients with SLE. 4 Nonetheless, Lemierre syndrome is rare in these patients, and the changes in disease markers associated with infection in SLE are unclear. Herein, we describe a case of Lemierre syndrome in a patient with SLE. During treatment, hypocomplementemia and elevation of antinuclear antibody (ANA) concentration occurred, so an SLE flare was suspected. Complement and ANA levels are modified in SLE combined with Lemierre syndrome, so other indicators should be evaluated precisely, such as urinary protein, sediment, serum creatinine and anti-dsDNA antibody concentration, and SLE disease activity index (SLEDAI).
Case report
The patient was a 56-year-old woman who developed lupus nephritis (International Society of Nephrology/Renal Pathology Society class V) around 20 years previously and had been treated with 10 mg/day prednisolone (PSL). Although she had exhibited a stable clinical course since her diagnosis, she developed malaise and exhibited urinary protein, hypocomplementemia, and fever. We supposed that she was experiencing an SLE flare and increased her PSL dosage to 20 mg/day. Two months later, she complained of a left lower toothache and then developed skin symptoms such as reddening, swelling, pain, and warmth from the left neck to the anterior chest and was admitted to our department. Upon admission, her body temperature was 37.2°C, blood pressure, 114/60 mmHg, pulse rate, 82/min, and oxygen saturation level, 98%. She had no signs of infection in her oral cavity and no pitting edema.
Table 1 shows the results of urine and blood tests performed upon admission. In a routine urine examination, protein (2+) and blood (1+) were positive, the pus cell count was 1–4 per high power field, and the 24-h urine protein level was 0.48 g/day. The patient’s laboratory findings were as follows: white blood cells, 14,980/µL (95.7% segmented neutrophils); hemoglobin, 14.4 g/dL; platelets, 24.9 × 104/µL; C-reactive protein (CRP), 14.9 mg/dL; ANA (×2560), homogeneous pattern; anti-dsDNA antibody, 21 IU (international units)/mL; complement C3, 76.8 mg/dL; complement C4, 17.5 mg/dL; and CH50, 51.2 U/mL. Protein C, protein S, anti-cardiolipin immunoglobulin G, or lupus anticoagulant was negative. Blood cultures were collected three times but were negative. In a jugular vein echo, vasodilatation was observed from the left subclavian vein to the internal jugular vein, and a thrombus without blood flow was confirmed using Doppler examination. Contrast-enhanced computed tomography (CT; Figure 1) revealed a thrombus in the internal jugular vein and swelling of the subclavian and left axillary lymph nodes. No signs of distant metastasis were observed. The CT did not show any findings that suggested mandibular cellulitis or Ludwig’s angina, such as abscess formation in the lower jaw.
Laboratory tests for blood and urine.
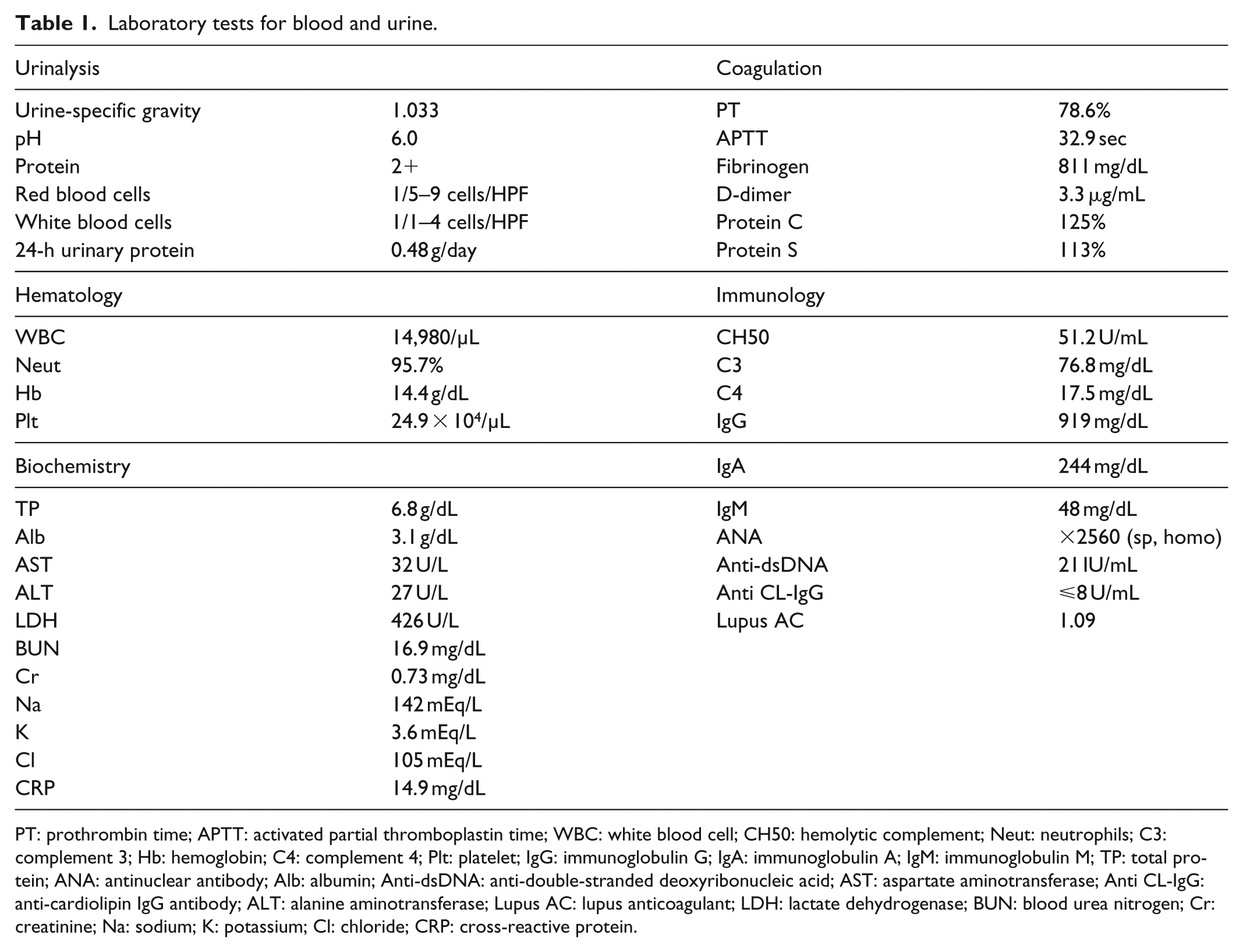
PT: prothrombin time; APTT: activated partial thromboplastin time; WBC: white blood cell; CH50: hemolytic complement; Neut: neutrophils; C3: complement 3; Hb: hemoglobin; C4: complement 4; Plt: platelet; IgG: immunoglobulin G; IgA: immunoglobulin A; IgM: immunoglobulin M; TP: total protein; ANA: antinuclear antibody; Alb: albumin; Anti-dsDNA: anti-double-stranded deoxyribonucleic acid; AST: aspartate aminotransferase; Anti CL-IgG: anti-cardiolipin IgG antibody; ALT: alanine aminotransferase; Lupus AC: lupus anticoagulant; LDH: lactate dehydrogenase; BUN: blood urea nitrogen; Cr: creatinine; Na: sodium; K: potassium; Cl: chloride; CRP: cross-reactive protein.

Contrast-enhanced computed tomography (CT) image showing a thrombus in the left internal jugular vein. The contrast-enhanced axial CT image demonstrates thrombophlebitis in the left subclavian vein (arrows).
Upon admission, we doubted that the patient had pulpitis, but she was diagnosed as having Lemierre syndrome on the basis of the left toothache, inflammatory findings, and thrombus from the left subclavian vein to the internal jugular vein. We began treatment using antibiotics and anticoagulants, subsequently changing the antibiotics from ampicillin/sulbactam and clindamycin phosphate to levofloxacin and metronidazole because she developed chemical neutropenia and drug eruption. The antibiotics were administered for around 6 weeks. Similarly, the anticoagulant therapy was switched from warfarin to apixaban (10 mg/day) because the patient developed drug hepatopathy (Figure 2). After the antibiotic and anticoagulant therapies, the thrombus gradually regressed. During treatment, elevation of ANA concentration (day 40: ×1280 to day 0: ×2560) and hypocomplementemia occurred (day 0: C3, 76.8 mg/dL; C4, 17.5 mg/dL; CH50, 51.2 U/mL to day 26: C3, 25.1 mg/dL; C4, 4.7 mg/dL; CH50, 14 U/mL), so the possibility of an SLE flare was considered. In as much as the patient’s SLEDAI, urinary protein level and anti-dsDNA antibody level were stable, and the SLE flare was not complicated. Thus, immunosuppressant therapy was considered unnecessary. Antibiotic and anticoagulation therapies improved the patient’s complement and ANA levels.
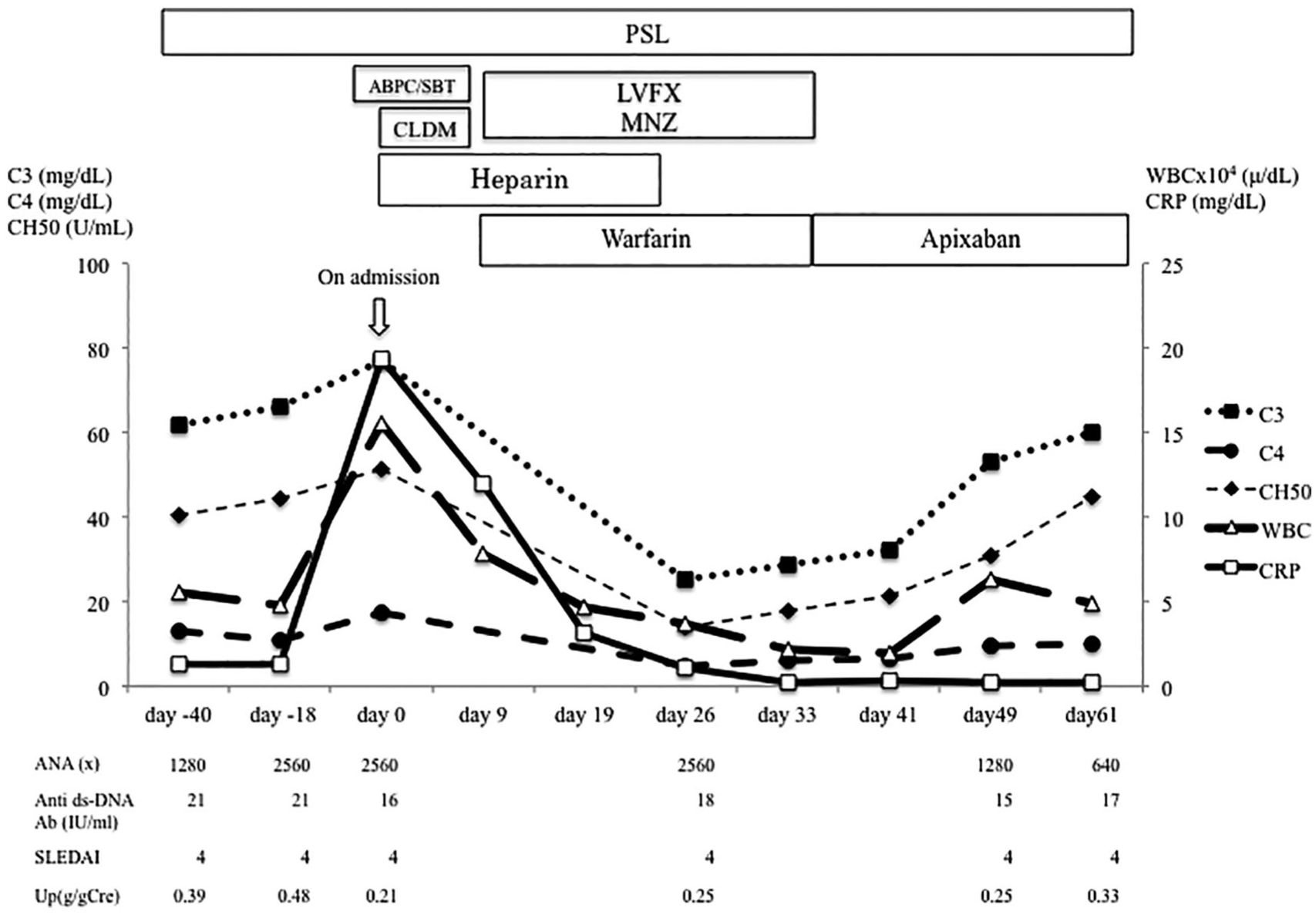
Clinical course of and changes in SLE disease markers during antibiotic and anticoagulation therapies.
Discussion
Lemierre syndrome commonly occurs in healthy young patients owing to infection by anaerobic bacteria in the oral cavity. 1 The morbidity and mortality of the disease is 3.6 per 1 million people and 4%–22%, respectively.2,5 Around 70% of cases involve pharyngitis or tonsillitis as a prior infection. Dental infection or sinusitis may also be a precursor. Treatment of Lemierre syndrome consists of antibiotic and anticoagulation therapies. Indeed, prolonged antibiotic therapy constitutes the mainstay of Lemierre syndrome treatment. However, as no controlled clinical trials have identified any optimal antibiotic regimen, decisions must be based on known in vitro studies and anecdotal clinical evidence. The duration of antibiotic therapy should be 2–6 weeks. 6 The indications for anticoagulation are controversial. Several reports have recommended treatment using anticoagulation if the thrombosis extends into the cerebral sinuses or if symptoms have not improved with antibiotic therapy alone. 7
Our patient had Lemierre syndrome with SLE, and transient hypocomplementemia and elevated ANA concentration during treatment. To our knowledge, only four cases of SLE with Lemierre syndrome have been reported (Table 2),8–10 all in women of varying ages. No characteristic age of onset or therapeutic effects were observed, and all the patients improved with antibiotic and anticoagulant therapies. Immunosuppressants were used in all the cases, but only one patient received an increased immunosuppressive dose prompted by disease progression prior to the onset of Lemierre syndrome. All reports were inconclusive regarding the relationship between Lemierre syndrome and SLE flare.
Summary of case reports of Lemierre syndrome with SLE from a literature review.
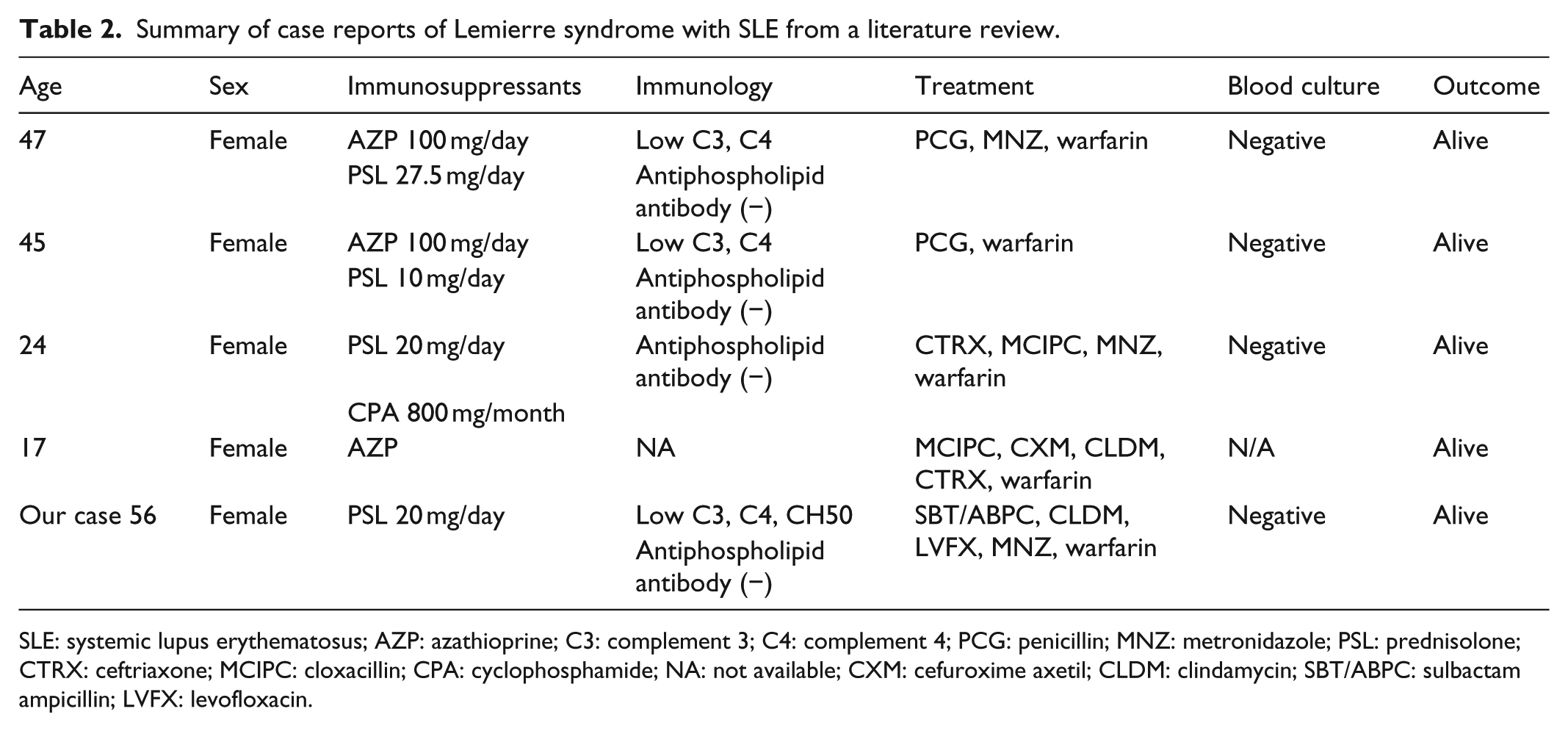
SLE: systemic lupus erythematosus; AZP: azathioprine; C3: complement 3; C4: complement 4; PCG: penicillin; MNZ: metronidazole; PSL: prednisolone; CTRX: ceftriaxone; MCIPC: cloxacillin; CPA: cyclophosphamide; NA: not available; CXM: cefuroxime axetil; CLDM: clindamycin; SBT/ABPC: sulbactam ampicillin; LVFX: levofloxacin.
In the present case, Lemierre syndrome developed in a patient with SLE. Infection is a leading cause of mortality in SLE patients, with bacterial infections being the most frequent. The PSL amount must not exceed 7.5 mg/day and 5 mg/day for chronic treatment. 11 In this case, PSL was originally taken internally at 10 mg/day, which was increased to PSL 20 mg/day, which may have contributed to the infection. As the patient had transient hypocomplementemia and was positive for ANA, we struggled to distinguish this condition from a coincident SLE flare. Indeed, patients with SLE can develop autoantibodies as a result of infection 12 and are less responsive to immune reactions against infections such as bacteria and viruses. 12 As bacterial DNA stimulates the immune system non-specifically, autoantibody production is likely to occur. Patients with SLE can also develop hypocomplementemia because of bacterial and viral infections. 13 In our case, the causative bacteria were not identified in blood culture. However, Fusobacterium necrophorum, a causative agent of Lemierre syndrome, is known to decrease C3 by binding to the inhibiting factor H. 14 In the present case and two of the four previously reported cases, C3 and C4 levels were decreased at the onset of Lemierre syndrome. Conversely, the increase in autoantibody levels and hypocomplementemia were thought to be caused by the infection.
CRP level plays a major role in differentiating SLE-related disease conditions, 15 and an elevated CRP level suggests a concomitant infection in patients with SLE. Similarly, the anti-dsDNA antibody titer and SLEDAI are altered during SLE flares, and relapse of lupus nephritis is indicated by an increase in proteinuria, serum creatinine concentration, and/or abnormal urine sediment. 16 As Lemierre syndrome can easily become severe, it may be fatal to increase a patient’s PSL dosage on the basis of the false conclusion that transient hypocomplementemia and elevated autoantibody level reflect an SLE flare. In the present case, although an SLE flare was suspected, the SLEDAI, urinary protein and sediment levels, serum creatinine concentration, and anti-dsDNA antibody level were maintained, and the patient’s elevated CRP level suggested a possible cause other than an SLE flare. When Lemierre syndrome occurs in patients with SLE, the complement and ANA levels might be modified by the infection, and evaluation of the patient’s general condition and symptoms is necessary.
Conclusion
Infection is a leading cause of mortality among patients with SLE. However, infection and SLE are similar in that they both increase the disease activity markers indicative of SLE. Although Lemierre syndrome is rare in patients with SLE, it can modify complement and ANA levels, so other indicators should be precisely evaluated, such as urinary protein, sediment, serum creatinine and anti-dsDNA antibody concentrations, and SLEDAI.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
