Abstract
Cognitive impairment is a common symptom of mild traumatic brain injury (mTBI) and can have long term cognitive and behavioral consequences. Despite this, there is no universally accepted protocol for assessment of cognition in this population. Conventional neuropsychological assessment tools rely on verbal or manual responses which lend themselves to confounding factors such as stress, intelligence, initiation, and motivation, suggesting the need for more objective tools. A scoping review was undertaken to explore the utility of eye-tracking methods for detecting cognitive impairment in mTBI patients, and to survey the kinds of tasks used in this context. Six academic databases were searched for studies related to brain injury, eye tracking, and cognition. Data from 17 articles were extracted and synthesized. In most cases, neuropsychological and eye-tracking methods were in accordance when detecting cognitive impairment. However, in many cases, eye-tracking measures detected impairments when neuropsychological tasks did not. This review suggests that eye tracking could provide an effective, objective method to measure cognitive impairment in mTBI.
Introduction
Traumatic brain injury is a serious public health issue. The Centers for Disease Control and Prevention defines a traumatic brain injury (TBI) as a bump, blow, or jolt to the head or a penetrating head injury that disrupts the normal function of the brain. 1 In the US alone, an estimated 1.7 million people sustain a TBI annually. Of those cases, 275,000 require hospitalization and 58,000 die. 2 These estimates are thought to be conservative due to the high rate of underreporting.3,4 The vast majority of TBI are mild, accounting for 70–90% of all treated brain injuries. 5 There are several differing definitions of mild traumatic brain injury in the literature. The American Congress of Rehabilitation Medicine previously defines a mild traumatic brain injury (mTBI) as post-traumatic amnesia (PTA) no greater than 24 h and loss of consciousness (LOC) no greater than 30 min. 6 More recently, they have updated the mTBI definition to be caused by biomechanically plausible mechanism of injury with one or more of the following criteria: clinical signs (i.e., LOC, amnesia), two or more acute symptoms (i.e., headache, nausea, memory problems, confusion), and/or evidence of TBI on neuroimaging. 7
There is a high degree of heterogeneity in the symptomology, recovery, and assessment of mTBI. Common symptoms include physical, ocular, psychiatric, and cognitive symptoms. Physical symptoms include headache, fatigue, sleep disturbances, vertigo, balance and gait abnormalities. Ocular problems can include cranial nerve palsies, deficits of saccades, optic nerve dysfunction, 8 accommodative and or/convergence insufficiency, photophobia and dry eyes.9,10 Psychiatric disorders include post-traumatic stress disorder (PTSD), major depressive disorder, generalized anxiety disorder, obsessive compulsive disorder and panic disorder, and increased risk of suicide.11,12
Symptoms typically occur in the early stages with resolution varying as early as 1–2 weeks 13 or up to 3 months.14,15 Cognitive impairment, however, has been reported to persist long term, sometimes greater than a year post injury. 16 This includes memory problems, trouble concentrating and increased distractibility. These symptoms can arise from deficits involving executive function resulting in difficulty with organizing thoughts and planning, which can lead to irritation. 17 Impaired judgment can also occur and manifest as inappropriate reactions to social situations, due to an inability to interpret the actions of others. Given the severe interruption to everyday functioning and therefore quality of life, cognitive deficits are one of the most important complications of brain injury. 18
The suitability of cognitive assessment tools varies with injury severity and the stage of recovery. There is a plethora of assessments within each cognitive domain ranging from screening tools in the early stages to more comprehensive neuropsychological batteries. 19 Neuropsychological testing has been a leading tool in the assessment of cognitive function in mTBI. 20 Like other kinds of assessments, there are limitations that accompany these methods. Several external factors such as age and exercise can confound the results of neuropsychological testing. 21 More complex factors include motivation and initiation, 22 fatigue, pain, and litigation status. 17 Co-morbidities such as depression, PTSD, substance abuse, premorbid intelligence, and psychiatric status can also influence the results.12,23,24 Furthermore, neuropsychological tests typically require verbal or manual responses, which can pose difficulties if the patient has comorbid language or motor deficits. There is certainly scope for improved methods of assessing cognitive function in mTBI that are less sensitive to these confounding factors. One possibility is to explore cognitive functions using eye movements as a response to visual stimuli that do not require prior knowledge or experience. By varying the properties of the visual stimuli and the kind of eye movement response that is required, one can create fairly simple tasks that engage particular cognitive functions such as executive control, memory, or attention.
Given the rate of visual impairments after mTBI, eye movement assessments are becoming increasingly more prevalent in the diagnosis and surveillance of mTBI. 25 The quick and non-invasive manner makes eye-tracking technology an attractive means to assess eye movements. There has been a movement towards multi-modal mTBI assessment tools to increase detection rate. These methods include but are not limited to oculomotor, vestibular, neuropsychological assessment and various imaging techniques.26–28 Eye tracking in these cases is one of many tools to determine whether there is a brain injury and function to detect this by the presence of abnormal oculomotor findings. However, rarely have eye-tracking tasks been used as a response modality to indicate the level of cognitive functioning. For example, one can assess muscle function for the hands to observe motor deficits, but the hands can also simply be used to indicate abilities by responding to tasks that require cognitive processing. Just as the hand can respond via a key press, the eyes can respond via a saccade.
The present scoping review was conducted to determine if eye-tracking tasks are a useful tool for detecting cognitive impairment in individuals with mTBI. This objective was addressed by reviewing studies that measured performance on conventional neuropsychological tasks in addition to cognitively demanding eye movement tasks using a case-control design. We evaluated whether the two kinds of cognitive assessments (neuropsychological test versus eye movement test) reached similar conclusions about the level of cognitive function. We also describe the kinds of cognitively demanding eye movement tasks that were used in this sample.
Materials and methods
An initial scan of the literature revealed that very few studies directly addressed the question of whether neuropsychology and eye movement tests reach similar conclusions about cognitive status in mTBI. In addition, there was little consistency in the kinds of eye movement tasks that could reasonably be construed as cognitively demanding. For these reasons, we chose a scoping review methodology. Grant and Booth 29 define a scoping review as “preliminary assessment of potential size and scope of available research literature”. This approach allowed our question to be addressed with flexibility and consideration of different methodologies and objectives from diverse research studies. Our scoping review was guided by the Arksey and O’Malley 30 framework (later revised by Levac et al., 31 ). The optional sixth stage of this framework involving stake-holder consultation was not undertaken in this review.
Step 1: Identify research question
According to Levac and colleagues, 31 stage one of a scoping review involves a broad research question but clearly articulated scope of inquiry, health outcomes, and target population. The target population for this scoping review was individuals with mTBI. The American Congress of Rehabilitation Medicine definition of brain injury was adopted for this review. The health outcome of interest was cognitive status. Thus, the question guiding this review was: can eye-tracking technology be used to effectively detect cognitive impairment in individuals with mild traumatic brain injury?
Step 2: Identify relevant studies
A systematic search of six electronic databases was initially conducted in November 2020 and subsequently updated in February 2022 and December 2022. Table 1 outlines the keywords and search terms used to explore databases for relevant articles published between 1990–2022. The six databases were chosen as they encompass peer-reviewed research in the fields of neurology, ophthalmology, neuropsychology, and cognition. The search strategy was constructed around three main concepts: brain injury, cognitive assessments, and eye-tracking technology. For each concept, both a key term search and a subject heading search was conducted. These searches were combined with the Boolean operator “OR” to ensure an exhaustive search for each concept. These searches were then combined with the Boolean operator “AND” to identity articles that included all three concepts. This process was repeated for all six databases.
Search translation table.

DE = Descriptors; MH = MeSH Headings.
Step 3: Study selection
After the electronic database search, the articles were filtered in a three-step screening process: title and abstract screening, full text screening, and extraction. This was accomplished using the citation streamlining software, Covidence. Collectively, the search yielded 1945 references; once duplicates were removed 1583 references remained to screen. After the primary exclusion criteria were applied, 219 references were assessed for full text review. Seventeen articles were included in the final review. Inclusion criteria was case control studies that included a neuropsychological and eye-tracking task to assess cognitive impairment. Figure 1 displays the PRISMA flow diagram for the article selection process with the specific exclusion criteria listed at each stage.
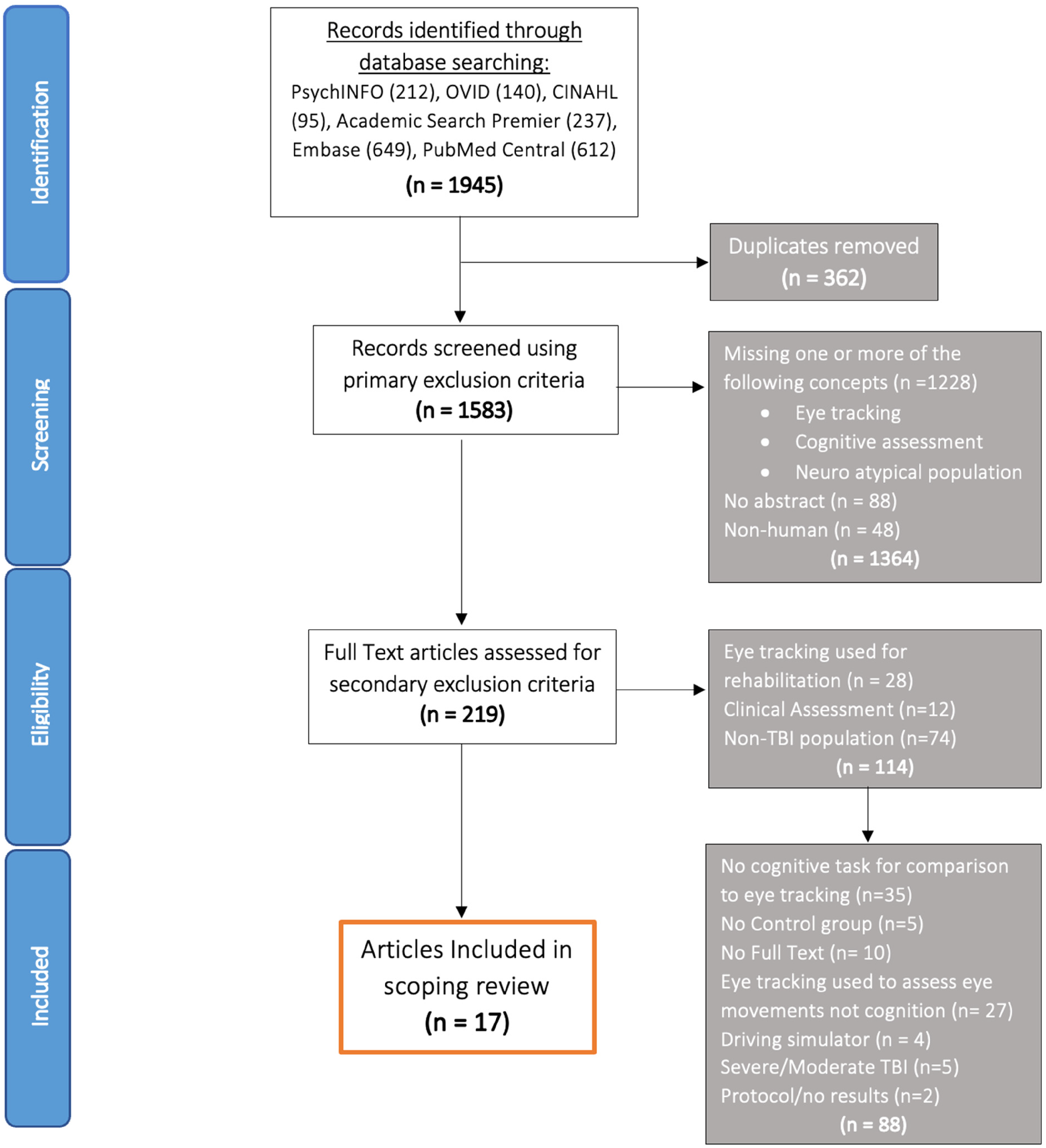
PRISMA scoping review flow diagram.
Step 4: Charting the data
A data-extraction form was developed to chart relevant data from the 17 selected articles. Data of interest that was extracted included information such as title, author, year of publication, location the study took place, number of participants, participant demographic information, inclusion/exclusion criteria, how TBI was defined, cognitive task used, eye-tracking task used, etc.
Quality assessment
The case-control Newcastle-Ottawa assessment scale was used to assess the methodological quality and risk of bias of the included articles. The scale has established content validity and has been widely used in review research. 32 Three main criteria are assessed: selection, comparability, and exposure with several subcategories. A star-rating system is used to assign a quasi-quantitative assessment of study quality. The lowest possible score is zero and maximum score is nine. The assessments were performed by two authors (HP and DTO) independently. Any discrepancy was resolved by a third independent reviewer on the author team.
Step 5: Collating, summarizing, and reporting the results
Once the data were extracted and organized, it was synthesized and interpreted for common themes and relationships to address the research question. We compared performance on conventional neuropsychological tasks and cognitively demanding eye movement tasks to determine if the two kinds of assessments reached similar conclusions about the level of function. Table 2 provides a descriptive summary of the studies included.
Summary of the included studies characteristics.
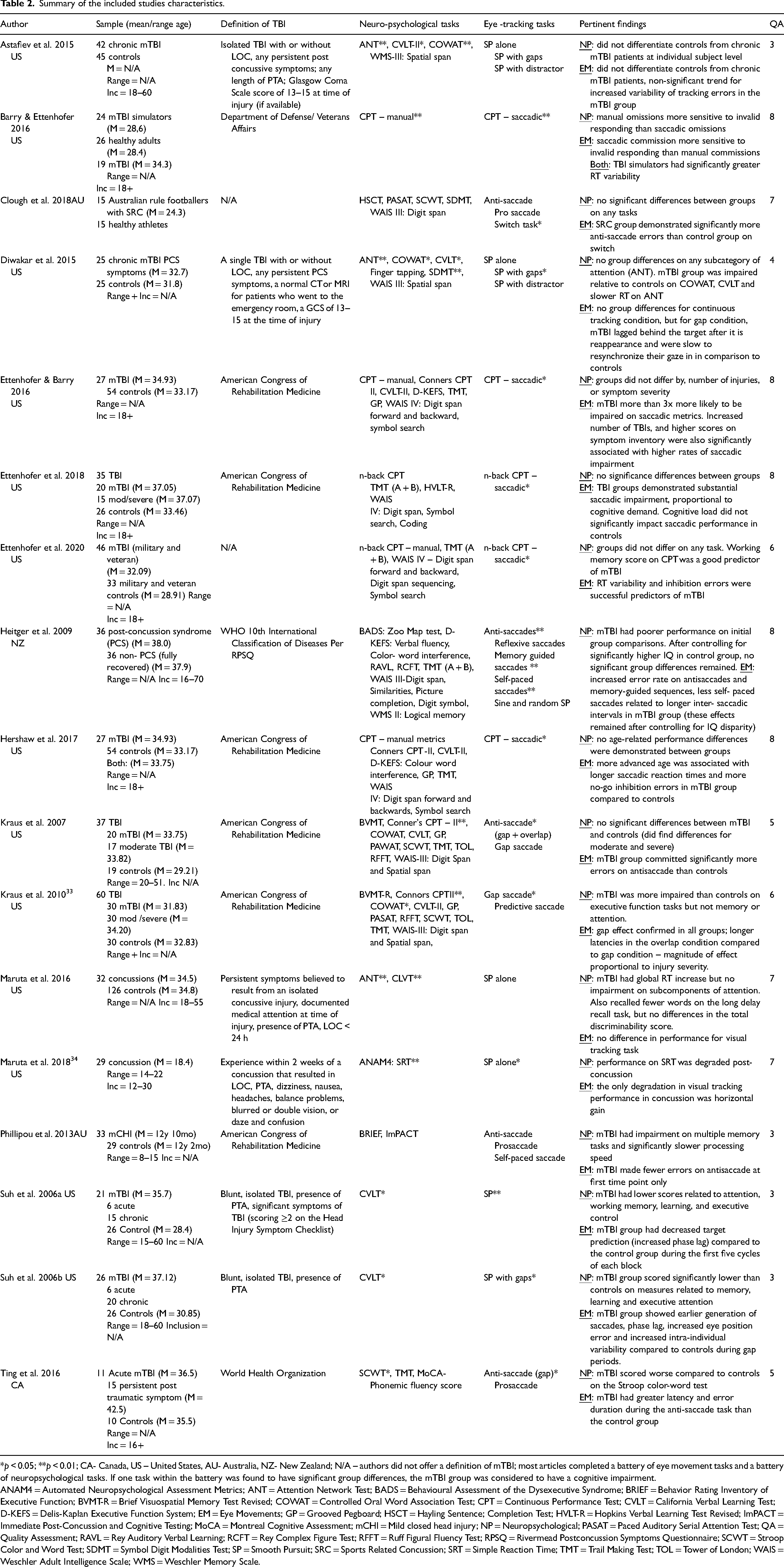
*p < 0.05; **p < 0.01; CA- Canada, US – United States, AU- Australia, NZ- New Zealand; N/A – authors did not offer a definition of mTBI; most articles completed a battery of eye movement tasks and a battery of neuropsychological tasks. If one task within the battery was found to have significant group differences, the mTBI group was considered to have a cognitive impairment.
ANAM4 = Automated Neuropsychological Assessment Metrics; ANT = Attention Network Test; BADS = Behavioural Assessment of the Dysexecutive Syndrome; BRIEF = Behavior Rating Inventory of Executive Function; BVMT-R = Brief Visuospatial Memory Test Revised; COWAT = Controlled Oral Word Association Test; CPT = Continuous Performance Test; CVLT = California Verbal Learning Test; D-KEFS = Delis-Kaplan Executive Function System; EM = Eye Movements; GP = Grooved Pegboard; HSCT = Hayling Sentence; Completion Test; HVLT-R = Hopkins Verbal Learning Test Revised; ImPACT = Immediate Post-Concussion and Cognitive Testing; MoCA = Montreal Cognitive Assessment; mCHI = Mild closed head injury; NP = Neuropsychological; PASAT = Paced Auditory Serial Attention Test; QA = Quality Assessment; RAVL = Rey Auditory Verbal Learning; RCFT = Rey Complex Figure Test; RFFT = Ruff Figural Fluency Test; RPSQ = Rivermead Postconcussion Symptoms Questionnaire; SCWT = Stroop Color and Word Test; SDMT = Symbol Digit Modalities Test; SP = Smooth Pursuit; SRC = Sports Related Concussion; SRT = Simple Reaction Time; TMT = Trail Making Test; TOL = Tower of London; WAIS = Weschler Adult Intelligence Scale; WMS = Weschler Memory Scale.
Definitions of mTBI
American Congress of Rehabilitation Medicine: a single TBI with or without LOC, any persistent PCS symptoms, a normal CT or MRI for patients who went to the emergency room, a GCS of 13–15 at the time of injury. 6
Department of Defense/ Veterans Affairs: defined as events that involved a sudden movement or a blow to the head the resulted in a loss of consciousness (LOC) ranging from 0 to 30 min or loss of memory (i.e., posttraumatic amnesia [PTA]) less than or equal to 24 h.
WHO 10th International Classification of Diseases: Glasgow Coma Scale (GCS) of between 13 and 15 on first assessment; Post-injury disturbance of consciousness (if applicable) had to be 30 min and duration of post-traumatic amnesia (PTA) 24 h.
Results
Within the 17 studies included in this review, there was a vast variability of time passed since injury and assessment, ranging from within a week to 21 years. The population was typically adult but mixed and pediatric were utilized as well. Numerous etiological mechanisms were identified with the most frequent being sport-related injuries. The most common eye-tracking technology used were EyeLink devices (SR Research, Ottawa, Canada). Most models were desktop mounted, only two included mobile (head-mounted) units. Two studies used electrooculography; the remaining studies used video-based units. Two of the video-based units used infrared illumination, while the remaining used visible light. For detailed report of cohort age, mechanism of injury, and eye tracker used, see Table 3.
Descriptive characteristics of the studies included.

Number of articles refers to the number of times this mechanism was included in the studies.
A wide array of neuropsychological tasks was used across various domains of cognition. There was an emphasis on executive functions tasks considering the common involvement of the frontal lobes in brain injury. See Figure 2 for the list of neuropsychological tasks used organized into domains of cognition and frequency of administration.
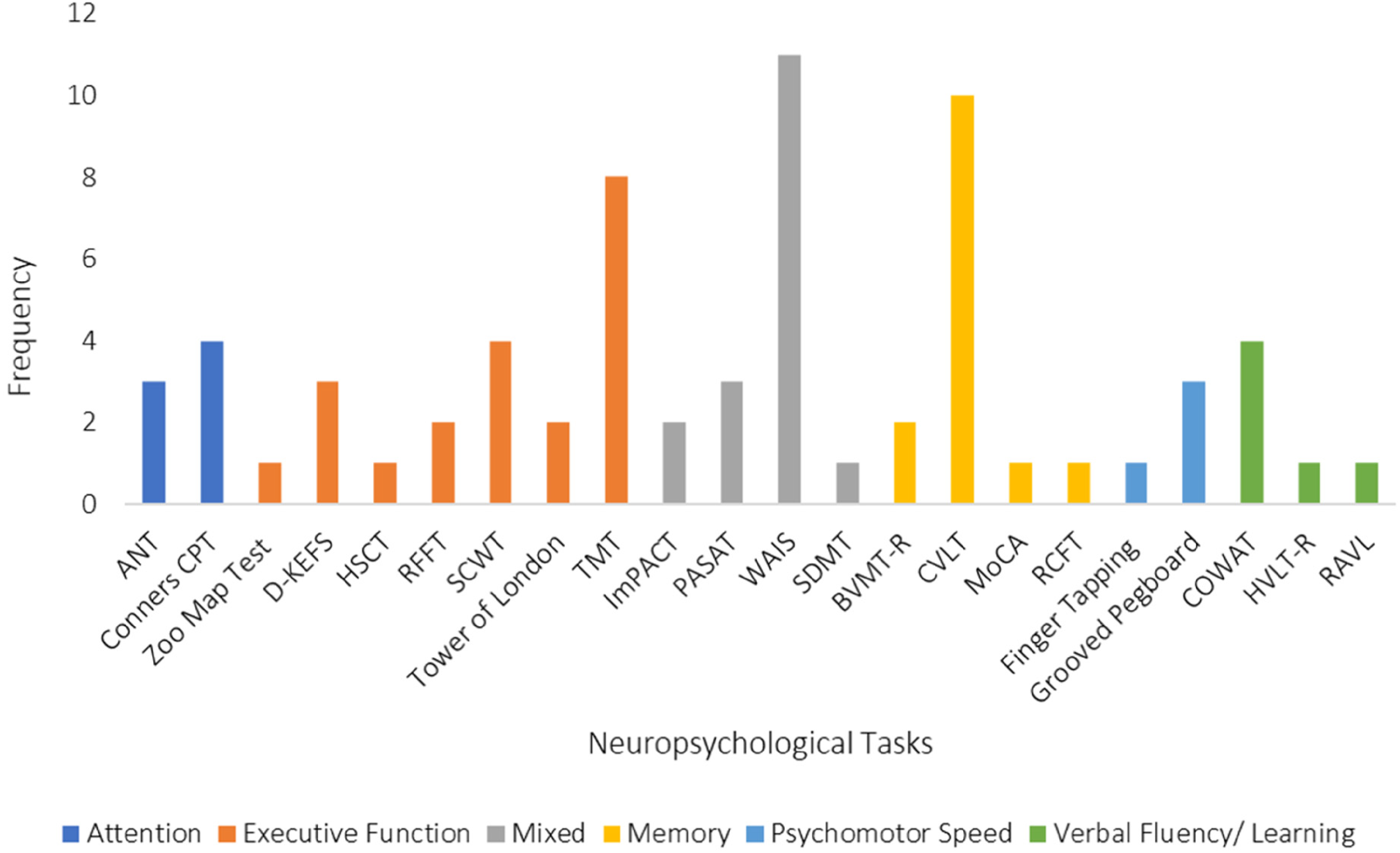
Frequency of neuropsychological tasks grouped by cognitive domain. Abbreviations: ANT = Attention Network Test; CPT = Continuous Performance Task; D-KEFS = Delis-Kaplan Executive Function System; HSCT = Hayling Sentence Completion Test; RFF = Ruff Figural Fluency Test; SCWT = Stroop Colour and Word Test; TMT = Trail Making Test; IMPACT = Immediate Post-Concussion Assessment and Cognitive Testing; PASAT = Paced Auditory Serial Addition Test; WAIS = Weschler Adult Intelligence Scale; SDMT = Symbol Digit Modalities Test; BVSM-R = Brief Visual Spatial Memory Revised; CVLT = California Verbal Learning Test; MoCA = Montreal Cognitive Assessment; RCFT = Rey Complex Figure Test; COWAT = Controlled Oral Word Association Test; HVLT-R = Hopkins Verbal Learning Test; RAVL = Rey Auditory Verbal Learning.
Comparison between neuropsychological tasks and eye movement tasks
The primary objective of this review was to determine whether eye-tracking tasks are a useful tool in detecting cognitive impairment in individuals with mTBI. By virtue of our selection criteria, each article had a sample of individuals with mTBI complete at least one neuropsychological task and at least one cognitively demanding eye movement task, and the results were compared to control participants to determine the presence of significant impairment.
Of the 17 studies, 1 1 reported significant differences between individuals with mTBI and controls on neuropsychological tasks. Of those 11 studies, 8 reported significant group differences on the cognitively demanding eye-tracking tasks. Given that neuropsychological tests are the accepted norm for assessing cognitive status in mTBI, this analysis shows that eye-tracking methods generally (8/11 times) lead to the same conclusion of impairment as reached by traditional methods; in other words, the two kinds of tests are usually in agreement about impairment. This suggests a level of concurrent validity for eye-tracking tests as a tool for assessing cognitive function.
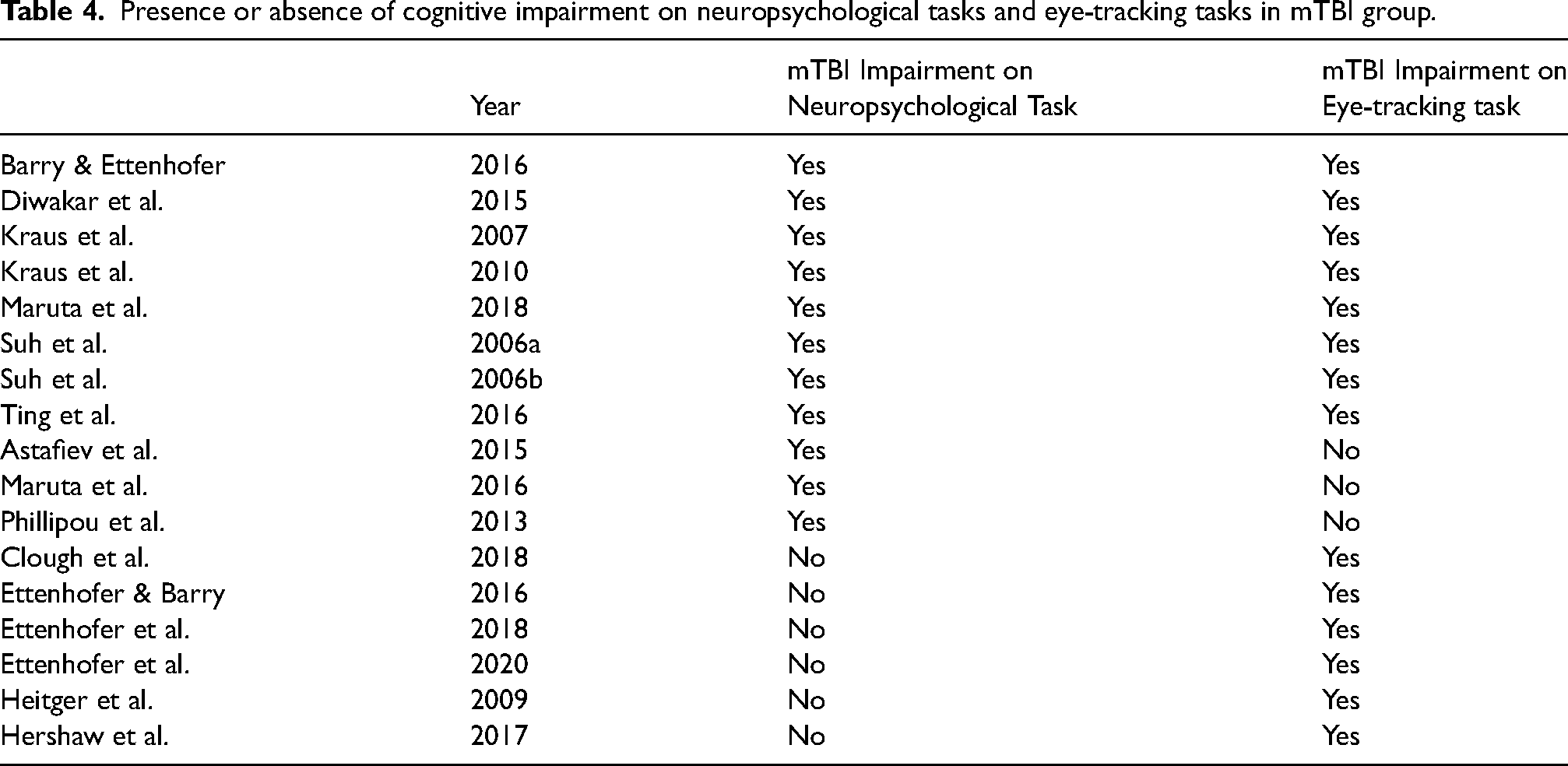
Six out of the 17 studies reported no significant differences between mTBI and controls on neuropsychological tests. Intriguingly, however, all 6 studies reported significant group differences on the eye-tracking tasks. One interpretation of this finding is that eye-tracking tasks are more sensitive to cognitive impairments than neuropsychological tasks, since impairments were revealed using eye-tracking methods that were not detected on neuropsychological tests. There are other possibilities, of course, including that the two kinds of tests are simply measuring different functions. However, the high degree of agreement between assessment methods described earlier (when neuropsychological tests reveal deficits, 8/11 times the eye-movement test agrees) argues against this interpretation. Therefore, eye movement tasks may detect impairments among individuals with mild TBI who are unimpaired on neuropsychological tasks. These findings are summarized in Table 4.
Presence or absence of cognitive impairment on neuropsychological tasks and eye-tracking tasks in mTBI group.
Types of eye movement tasks
Several different types of eye movement tasks were utilized to examine cognitive status in individuals with mTBI. Three categories of eye movement tasks were used in the included articles: smooth pursuit, saccadic, and dual tasks.
Saccadic eye movement tasks (N = 6)
Numerous saccadic eye movement tasks were utilized, ranging from low cognitive load and high cognitive load. This study considered prosaccade and self-paced saccades to be low cognitive load and gap saccade, memory guided saccades and predictive saccades to be high cognitive load. The simplest of the saccadic tasks was the prosaccade task where the participant simply saccades towards a target. The more cognitively challenging version of this task is the antisaccade task, where the participant is asked to saccade in the opposite direction of the peripheral target. This task engages inhibitory control mechanisms as it requires the participant to inhibit a reflexive saccade to the peripheral target. Some variations of this task included the gap antisaccade where on some trials the central fixation point was extinguished before the peripheral stimulus was presented (gap trial) and other trials the target appeared with the central fixation target still present (overlap). These latter trials, require the participant to disengage attention from the central target to initiate a saccade to the peripheral target. This gap paradigm was used for prosaccade tasks as well. Other types of tasks included switch tasks (antisaccade and prosaccade), memory guided saccades (performing a memorized sequence of saccades), self-paced saccades (perform as many saccades as possible between two stationary targets in a specified time frame), and predictive saccade (saccade towards a target presented sequentially between two fixed locations).
There were no significant group differences found on the simple prosaccade tasks.35–38 However, tasks that incorporated gap conditions noted some interesting differences. Longer saccade latencies were found in the overlap condition than the gap condition, a phenomenon called the gap effect. The overlap condition requires the subject to disengage visual attention from a central fixation point, prolonging saccade initiation. This effect was most pronounced in the moderate TBI group, followed by the mTBI group and finally the control group. 39 The antisaccade task consistently found impairments in the TBI groups. Participants in the mTBI group demonstrated higher error rates and higher switch cost – difficulty switching from a prosaccade trial to the antisaccade trial. 35 Additionally, some studies found specific eye movement deficits in the TBI group such as larger absolute position errors of the final eye position in antisaccades and the gain of the final eye position were hypermetric compared to controls.36,38,39 The findings for self-paced saccades were mixed. Some studies found no significant differences between groups, 37 but others found less self-paced saccades and longer intersaccadic intervals in the mTBI group. 36 One study included memory guided saccades but found that higher error rate in the mTBI group. 36 A predictive saccade paradigm was used in one study and found that controls showed a more rapid and greater decrease in response latency than the TBI groups. This deficit was proportional to injury severity. In other words, the control group was able to anticipate and catch on quicker than the TBI group. 39
Smooth pursuit tasks (N = 6)
The same task was used in the six articles investigating smooth pursuit. The task was to track a stimulus in a predictive circular trajectory. Some studies had variations of this tasks where the stimulus was continuously visible, or a more cognitively challenging version where the stimulus disappeared at random intervals (gap condition) before reappearing. The participants were asked to continue to track the trajectory in the absence of the stimulus, predicting the targets movement. Another variation of this task included a distractor stimulus moving in the opposite direction of the target stimulus. Participants were asked to ignore the distractor stimulus and track the target stimulus.
The tasks with tracking of predictive circular trajectory alone were not effective at identifying differences between mTBI participants and controls. Mean eye position error, variability of position error (radial/tangential), mean phase error, and saccade frequency were similar between the groups.26,40–42 However, if these tasks included gap conditions, there were significant differences between the groups. Participants with mTBI demonstrated phase lag, meaning they lagged behind the target after it's reappearance and were slow to resynchronize their gaze. Conversely, the control subjects demonstrated more precise tracking and, in some cases, slight phase lead, tracking ahead of the target. Therefore, the control group was able to anticipate the target speed and continuous change in position. The mTBI group also had larger average radius than controls and higher error variability.40,43
Dual tasks (N = 5)
Unlike the two preceding categories of eye movement tasks, in which participants responded solely with eye movements in cognitively demanding tasks, studies in this group included eye movement responses in addition to other forms of responding in a traditional cognitive task. These studies had participants complete a continuous performance task (CPT) with different cue conditions. These conditions included nondirectional cues (provide temporal information on the appearance of target), directional cues (pointing towards to the target), mis directional cues (pointing away from the target), un-cued (central fixation cross persists through the appearance of the target), gap cues (a blank image replacing the fixation cross), and no-go cues (provide a signal that the participant should not respond to that target). The participant was asked to both press a keyboard key and fixate a target as quickly as possible once it appears. Having both these responses allows for comparison of manual and saccadic metrics. Some studies added another level of difficulty by increasing cognitive load. Low cognitive load trials required a key press and fixation on the target. Moderate cognitive load required the participant to press the key that corresponds to the colour of the target. High cognitive load trials required the participant to press a button labelled ‘same’ or ‘different’, depending on whether the target circle was the same colour or different colour relative to the previous target.
Every study in this category found saccadic indices on a continuous performance task to enhance neurocognitive assessment. When comparing rates of impairment on the two types of metrics: saccadic vs. manual, the saccadic metrics often demonstrated more impairments. For example, impairment on individual saccadic metrics was, on average, more than three times more likely to be seen in participants with mTBI than in those in the control group. Compare this to less than one times more likely for manual metrics. Furthermore, rates of saccadic impairment were significantly associated with multiple mTBIs and higher symptomology, this finding did not extend to manual metrics. When incorporating cognitive load into the continuous performance task, there was a significant interaction between groups and load but only for saccadic metrics. The TBI group demonstrated substantial saccadic slowing proportional to cognitive load (most impairment in high load condition). Increased reaction time variability and inhibition errors served as successful predictors of mTBI. In contrast, cognitive load did not significantly impact saccadic performance among controls. For manual responses, participants across all groups demonstrated increased reaction time as cognitive load demands increased but both groups were affected equally. Eye tracking was also useful in identifying invalid responding. Saccadic commissions were more sensitive to invalid responding than manual commissions, and manual omissions were more sensitive than saccadic omissions. Therefore, incorporating saccadic indices could help identify invalid responding and avoid false positives. Additionally, saccadic measures were found to be beneficial in identifying the effect of TBI on cognitive aging. Age was found to be more strongly related to saccadic measures of visual attention in mTBI group than controls. This was demonstrated through slower saccadic response times and more difficulty inhibiting saccadic responses. No age-related performance differences were demonstrated between the mTBI and the control group on manual measures. Overall, saccadic indices yielded greater sensitivity than manual indices to detect cognitive impairment under conditions of increased cognitive demand44,45 invalid responding 46 and cognitive aging. 47 Table 5 outlines the eye-tracking tasks by cognitive involvement.
Eye-tracking tasks categorized by cognitive involvement.
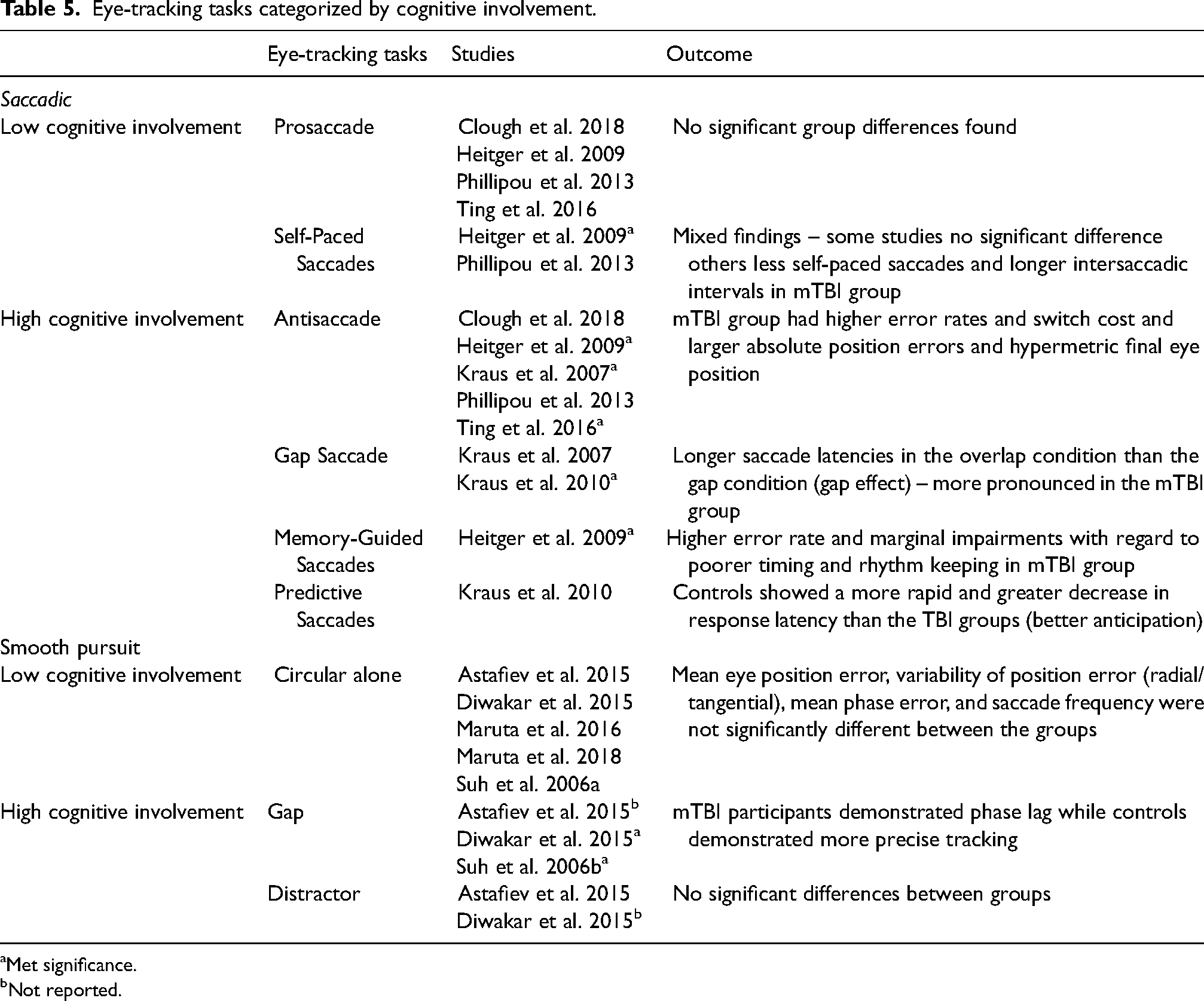
Met significance.
Not reported.
As Table 5 portrays, only the more cognitively involving tasks yielded significant group differences, whereas the simpler tasks failed to differentiate between mTBI and controls.
Discussion
The objective of this scoping review was to assess the utility of eye-tracking tasks for detecting cognitive impairment in individuals with mTBI. The majority of studies found that eye movement tasks were effective at detecting cognitive impairments in a mTBI population. In the 11 studies that reported significant impairment on neuropsychological tests, 8 of them additionally reported significant impairment on eye-tracking tasks, indicating a high level of agreement. In 6 studies, impairment was not reported on neuropsychological tests, yet all of these studies reported significant impairments on eye-tracking measures. This suggests that these eye-tracking tasks might be more sensitive to deficits. Several kinds of cognitively demanding eye movement tasks were used in these articles. Three categories of tasks emerged: saccadic, smooth pursuit and dual tasks involving both saccadic and manual responses.
As noted at the outset, traditional neuropsychological tests of cognitive function after head injury are affected by premorbid intelligence, fatigue, initiation and motivation, motor/language constraints and reliance on self-report.22,23,48 The broader context for our work in this area is to develop improved methods to assess cognitive function following mTBI that eliminate some or all these limitations. This review set out to ascertain whether there is any evidence that supports the use of eye tracking for this purpose. The current study found considerable evidence that eye movement tasks were an effective method to detect whether cognitive impairment is present, and in fact might be more sensitive to cognitive impairment than traditional neuropsychological tests in many cases.
Development of eye movement-based assessment battery
The results of this scoping review provide justification for continuing to develop tests of cognitive function that rely on eye movements as the mode of responding. Of course, much more work will be required to move from this proof-of-concept to the development of an evidence-based cognitive assessment battery based on eye movements. For instance, the range of studies identified in the current scoping review did not permit a meaningful comparison of specific cognitive processes. Future studies examining eye movement tasks and neuropsychological tasks within the same cognitive domain would be useful to determine whether the two kinds of tasks are in fact measuring the same cognitive impairment, and whether certain domains of cognitive function are more amenable to measurement via eye movements.
The subconscious control of many kinds of eye movements makes this kind of testing more robust against initiation and motivational factors that could affect task performance. For example, Barry & Ettenhofer 46 were able to accurately identify true mTBI individuals from healthy adults instructed to feign deficits on a continuous performance task using saccadic and manual response metrics. Saccadic commission errors were more sensitive to invalid responding than manual commission errors and there was increased reaction variability in the participants feigning mTBI. Kanser et al., 49 explored this concept but used eye tracking as an adjunct to assess visual behaviors during traditional neuropsychological tasks. During forced-choice trials, participants feigning mTBI showed more transitions, fixations, and time spent looking at correct and incorrect response options. These eye-tracking indices led to high accuracy in determining group status.
As outlined earlier, neuropsychological tests are notoriously sensitive to a large number of confounding factors such as premorbid intelligence, and cultural differences, which is one of the reasons that eye movement tasks could be superior. Heitger et al., 36 found that performance differences between TBI participants and controls on several neuropsychological tests were eliminated after controlling for IQ and depression, which differed between groups, whereas group differences in performance on the eye movement tasks remained despite controlling for these factors. Similarly, Ettenhofer et al., 45 found that estimated intelligence, depression, and PTSD were related to conventional neuropsychological measures and multiple manual metrics on a continuous performance task but the same was not true for any of the saccadic metrics. Therefore, there is good reason to believe that assessment of cognitive function via eye movement tasks might be less affected by confounding extraneous factors than traditional neuropsychological assessment.
Given that basic deficits in oculomotor function are associated with mTBI, it is reasonable to ask whether the impairments observed in the eye movement tasks reported here are due to the cognitive demands of the task or rather the fact that mTBI participants had underlying eye movement deficits. Consistent with our argument that the deficits are cognitive (rather than oculomotor) in nature, our analyses show that the most basic tasks such as prosaccade task and a simple smooth pursuit task did not yield significant differences between mTBI patients and controls which suggests that there were no underlying oculomotor deficits. Nevertheless, the more cognitively taxing tasks such as the antisaccade task, gap saccade, memory guided saccades, predictive saccades, and smooth pursuit tracking with gaps did yield group differences. Johnson et al., 50 paralleled the current study's findings where they had a TBI group and control group complete seven oculomotor tasks. The three tasks that reached significance on group comparisons were the antisaccade task, self-paced saccades, and memory guided saccades. While reflexive saccade, fixation, sinusoidal, and circular pursuit tasks did not. Once again, specifically the tasks with higher cognitive demand demonstrated differences. Notably, the TBI group exhibited increased fMRI activation during these tasks suggesting reduced processing efficiency and more effort required based on complexity.
Eye tracking increases the accessibility of cognitive assessment
Advancements in technology have allowed portable, light weight, and cost-effective eye trackers to be widely available. With this rise in mobile eye-tracking devices, eye tracking can be implemented in a variety of settings such as on the sidelines during athletic competition, or at the patient's bedside. Research is ongoing to validate the use of smart phones/tablets for eye tracking via the front camera. Valliappan et al., 51 replicated oculomotor findings on prosaccade, smooth pursuit and visual search tasks compared to expensive desktop eye trackers in a healthy population. Moreover, they were able to achieve comparable accuracy with their method compared to state-of-the-art eye trackers. This shows promise for implementing this method in an mTBI population. Considering the lack of hardware involved, this method could easily be administered for sideline evaluation of possible sports related brain injury. Other applications include by the bedside for individuals who are poor candidates for chinrest/forehead rest configurations due to postural restrictions. This could apply to the elderly population whose frailty does not support this constricted positioning, or mTBI patients with other co-morbidities that limit mobility such as spinal stabilization or other orthopedic injury precautions. Mobile brain injury assessment methods are emerging with a focus on eye movement deficit detection, vestibular performance, and neurocognitive tests.27,52 The neurocognitive tasks still require a motor response from the participant such as an arm movement or button press. Assessing cognition through eye movement tasks allows for hands free assessment. These methods are then accessible to a wider population of individuals such as locked in syndrome individuals who have limited to no motor function.
Clearly, assessments of cognitive function based on eye movement tasks would be inaccessible to individuals with primary oculomotor deficits. Causes include cranial nerve palsies and restrictive strabismus stemming from skull fractures. Deficits in ocular motility would limit the utility of eye-tracking tasks as these individuals would not be able to fixate a target in the field of action of the affected extraocular muscle. Another barrier for these individuals is that they would likely be diplopic thus complicating which target to fixate. However, this review suggests using eye movement tasks is useful for detection of mild traumatic brain injury where the incidence of oculomotor palsy and restrictive strabismus is rare as compared moderate and severe brain injury compared to mild. Jin et al., 53 examined 3417 individuals with TBI and found that 312 had a cranial nerve palsy (I-XII), 80 of those involved cranial nerves III-VI. Only two of the 80 were mild traumatic brain injury. Therefore, this method will exclude some individuals from its use but the prevalence of this group within this population is low.
Limitations
The exclusion criteria were curated to allow for a comparison of the two assessment methods (neuropsychological tests vs. eye movement tests) in a case-control design, which resulted in a relatively small subset of all the studies that have measured eye movements in mTBI. While this approach enabled us to conclude that eye tracking is useful for detection of cognitive impairment, further steps are necessary to validate its use as a comprehensive assessment tool. Due to the narrow selection criteria, the survey of the kinds of eye-tracking tasks being used to study cognition in mTBI is not comprehensive as many studies using innovative methods did not include neuropsychological tests for comparison.
Conclusions
In conclusion, the findings of this review indicate that cognitively challenging eye-tracking tasks could be a useful method for detecting cognitive impairment in an mTBI population. Compared to traditional neuropsychological methods of assessing cognitive function, eye-tracking tests could aid in greater sensitivity, fewer confounding extraneous influences, and greater accessibility to various populations.
Footnotes
Acknowledgements
Shelley McKibbon, Information Services Librarian at the W.K. Health Sciences library at Dalhousie University, provided guidance for refining the literature search strategy adopted in this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Mathers Trainee Scholarship in Ophthalmology.
