Abstract
Research has improved the understanding of sports related concussion (SRC), and several classification systems and guidelines are available in the literature. The exact timing and clearing of athletes for return-to-play (RTP) is still based primarily on subjective reports of concussion symptoms, however symptoms link poorly to objective recovery. Current literature suggest that symptoms alone cannot accurately identify either all concussed athletes or their recovery. The difficult task of interpreting which symptoms are directly related to concussion, and which are related to other conditions, speaks for an increased focus on the RTP process itself. This study examines the literature on neurocognitive assessments and their importance as indicators of accurate timing of when athletes can return to either ball training with duels or the playing field. Entries in three electronic databases (PubMed, Web of Science, and SURF) were searched from January 2000 to June 2022. Search terms were concussion, mild traumatic brain injury (mTBI), sport, athlete, expert, elite, professional, diagnostic, testing, return to play, management, neurocognitive, and cognitive. Inclusion criteria comprised performance-based participation in a team sport and being in the age range of possible peak performance (18–40 years). In addition, only studies with pre-post designs were considered. The PEDro scale was used to assess methodological quality. The methodological quality of the fifteen included studies ranged from 5 (one study) to 6 (fourteen studies) from a maximum of 10. Despite being symptom-free, athletes in all fifteen studies showed lower performance compared to controls on tests of visual and verbal memory (approx. 3–5% deficit) and on processing speed (approx. 6% deficit) after mTBI. All studies report specific neurocognitive deficits after mTBI, although the athletes were declared clinically symptom-free. Therefore, the systematic consideration of neurocognitive parameters in RTP decision making is recommended, especially in light of subsequent muscular injuries of the lower musculoskeletal system, recurrence of mTBI, and residual neurodegenerative disorders.
Introduction
Sports-related concussion (SRC) is an ongoing safety issue and a common neurological injury in contact sports. 1 Athletes sustain SRC primarily through direct contact to the head or body (e.g., body checks or accidental collision) during contact sport, 1 such as ice hockey or rugby, not only in games, but also in training.2,3 Estimation suggest up to 3.8 million concussions occur annually in the United States due to sports-related activities, with up to 50% of concussions going unreported. 4 At the elite level, concussions are even more common, as evidenced by a concussion rate of 66.1 (per 10,000 athlete exposures) during the 2012–2013 and 2013–2014 National Football League (NFL) seasons. 5
SRCs are a heterogeneous condition with varied neuropathophysiology and clinical symptoms that are related to severity and location of impact, age, or sex. 1 Clinical features are broad and vary, including neurocognitive symptoms, autonomic dysfunction, and vestibular impairment. 1 In addition, impairments in cognitive abilities such as visual-motor reaction time, information processing, memory, visual tracking, problem solving, and sensorimotor balance can occur with SRC.6,7 It is critical to detect SRC in a timely manner and to safely determine when an athlete can return to play (RTP) after diagnosis. Premature RTP can put athletes at an increased risk of lower extremity injuries,2,7–9 secondary brain injury,1,6,9–11 persistent post-concussion symptoms, 12 and chronic neurologic deficits,1,3,11,12 and even cognitive problems under conditions of physical or psychological stress, even if they report full recovery.13–15 After sustaining an SRC, it is recommended that athlete's RTP follows a stepwise progression, through which the athlete should first participate in athletic activity without recurrence of symptoms before returning to competition. 1
However, determining when an athlete has fully recovered from SRC is difficult. To manage SRC, current protocols involve an initial period of rest (24–48 h) followed by a stepwise rehabilitation protocol that includes gradually increasing physical and cognitive demands.1,16,17 Each stage takes 24 h to complete and progression is dependent on meeting appropriate criteria (e.g., heart rate, duration of exercise) without recurrence of concussion-related symptoms. If symptoms do recur, the athlete should drop back to the previous asymptomatic stage for a further 24 h. Clearance for RTP is determined by completion of the graded rehabilitation protocol and passing clinical assessment at each stage. However, assessing symptoms in the recovery phase can be difficult and subject to bias. 18 For example, symptoms such as headache or difficulty concentrating do not directly belong to SRC 19 and it can be challenging to differentiate concussion-related symptoms from pre-morbid conditions such as chronic sleep dysfunction, migraines, anxiety, and attention problems.20,21 Currently, clinicians rely heavily on symptom emergence following the fundamental exercises of the protocol, which may not necessarily indicate full recovery. Relying solely on subjective self-reported symptoms of athletes may also leave them at risk of returning to play too early.
The evaluation of neuropsychological deficits after concussion has been reported throughout the literature and reviewed in depth previously.22–24 Additionally, there is increasing evidence to suggests that physiological recovery from concussion may outlast clinical recovery, even after successful completion of a graduated RTP protocol, athletes may still be at higher risk of neurological and non-neurological injury. 25 Balance assessments, which are a common tool used to assess recovery, may also not be sufficient in detecting continued impairments since cognitive deficits can persist beyond balance impairments. 26 Cognitive assessments have potential to aid in RTP decisions following SRC. Monitoring a cognitive marker during the rehabilitation phase, and even after symptoms resolve, could provide an objective measure of neurophysiological recovery from injury that could complement clinical assessment in RTP decision making. This is why standardised neurological and cognitive assessment scales have been developed to assist the sideline diagnosis of SRC and aid in RTP decision. 27 However, there is mixed evidence for cognitive testing in subacute SRC.18,28,29 Despite the focus on concussion assessment protocols and tools, most lack quantitative objectivity, which raises doubts about their ability to measure readiness for RTP following a concussion. Given the limitations of clinical assessment in determining RTP following SRC, an objective indicator of neurophysiological recovery is needed to allow clinicians to make safer RTP decisions.
Therefore, our systematic review aims to identify cognitive impairments from computerised testing batteries, paper-pencil testing, postural stability, and diverse tests that have been assessed after SRC and throughout recovery from SRC. We examine the major pathological impairments involved in SRC and highlight the neurocognitive key marker from each pathway, evaluating their time course post SRC and their relationship to clinical recovery. This might help identify objective neurophysiological recovery from SRC that could have potential use in determining RTP decisions.
Methods
Search strategies
The systematic review of the literature was conducted in accordance with the four-phase PRISMA guideline. We searched in three online databases: PubMed, Web of Science (all databases), and Federal Institute of Sports Science Germany (BISp SURF). Title, abstract, keywords, and full text were examined. In all databases we used the same strategy. A comprehensive search was performed from July 2 to July 20, 2022. The following search terms were used: (((((((concuss* OR mild traumatic brain injury)) AND (sport OR athletes OR expert* OR elite OR professional)) AND (diagnostic* OR testing)) AND (return to play)) AND (neuro* OR cogni*)) NOT children).
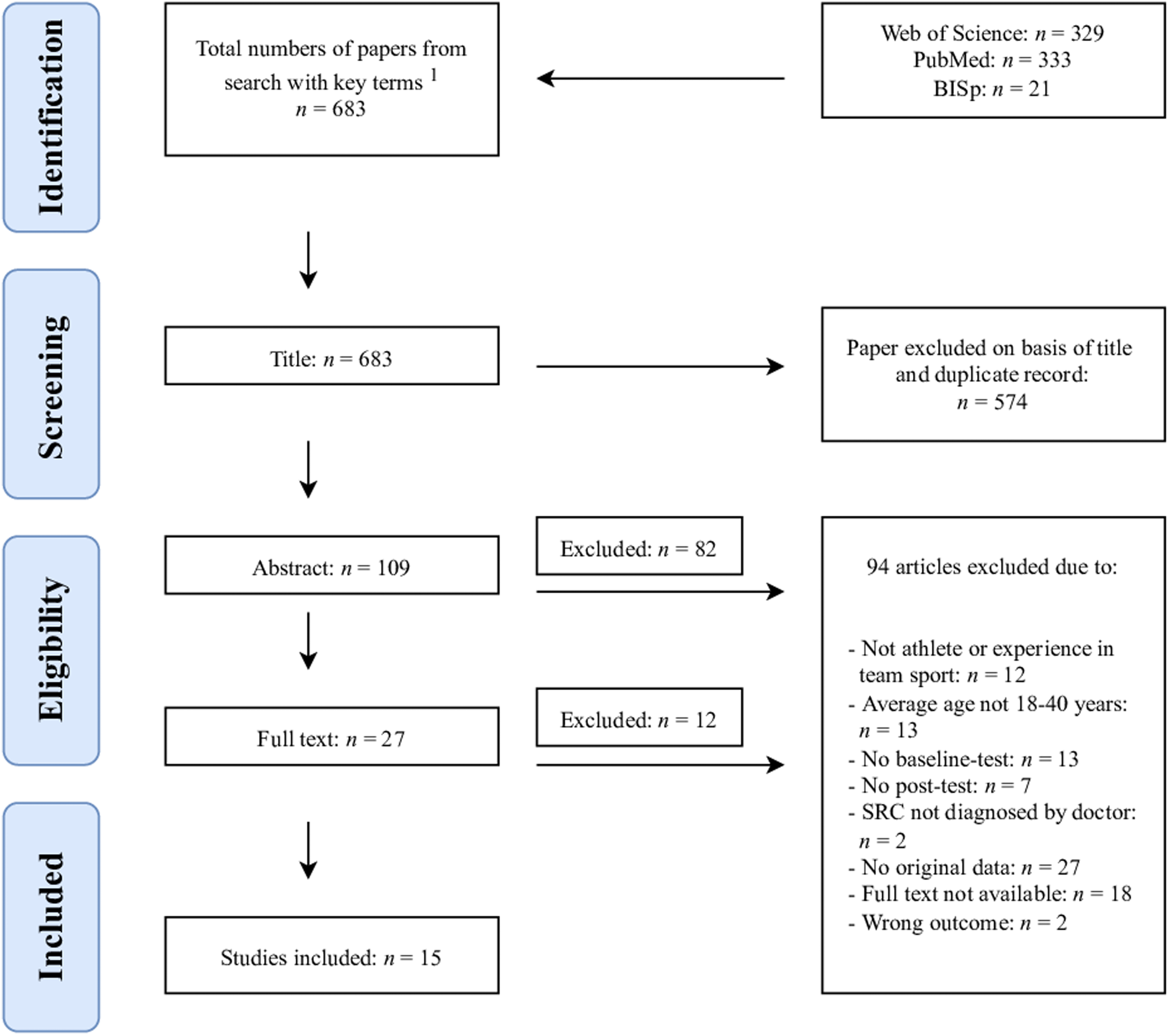
Study selection flow chart on the basis of the PRISMA statement key terms: (((((((concuss* or mild traumatic brain injury)) and (sport or athletes or expert* or elite or professional)) and (diagnostic* or testing)) and (return to play)) and (neuro* or cogni*)) NOT children).
Inclusion criteria
Studies were included if they were written in English or German and if the full text was available. We considered only original publications with human participants and publications from January 2000 up to July 2022. The athlete had to play a team/field sport and had to belong to an age range of possible top performance (18–40 years). Only experimental studies with baseline data and posttests were considered. Moreover, the SRC had to be diagnosed by medical staff (doctor, team physician, certified athletic trainer). Narrative reviews, single case reports, abstracts, and letters to the editor on the subject were excluded. Additionally, studies involving amateur athletes and studies in which there were less than two measured time points, were excluded. A screening strategy was used to identify studies for inclusion in the review. First, study titles were screened for relevance and second, for duplicates. The remaining abstracts were subsequently screened for inclusion and exclusion criteria to identify relevant articles. Articles that satisfied title and abstract review underwent an in-depth full-text screen for inclusion and exclusion criteria. Reference lists of each included study were manually reviewed to identify relevant studies.
Examination of methodological quality (PEDro scale)
The two authors (WD & ZK) rated the fifteen included articles independently for methodological quality using the Physiotherapy Evidence Database Scale (PEDro scale) based on the Delphi list developed by Verhagen et al. 30 at Maastricht University. The PEDro scale contains a validated checklist that can be used to assess the methodological quality of studies on eleven criteria. The first criterion relates to external validity (generalizability of the study). Because this is not a quality criterion, it is not included in the calculation of the PEDro scale score. Items 2–9 refer to internal validity, whereas items 10–11 provide information on the statistical interpretability of the study's results. There are two options for each item: Each “yes” scores one point and each “no” scores zero points. Points are summed up for the overall assessment. A maximum of ten points can be reached. Fifteen studies could be assigned to evidence level A with 6 points on the scale. The remaining study fulfilled the B criterion with 5 points. Item 5e of the PEDro Scale was not evaluated because it is difficult to blind participants in the RTP process (Table 1).
Included studies rated by methodological quality (PEDro scale) legend.

aInclusion and exclusion criteria have been specified (not included in the total score).
bSubjects were randomly assigned to the groups.
cAllocation to the groups was hidden.
dAt the beginning of the study, the groups were similar in terms of the main forecasting indicators.
eAll subjects were blinded.
fAll therapists who carried out therapy were blind.
gAll investigators who measured at least one central outcome were blinded.
hOf more than 85% of the subjects originally assigned to the groups at least one central outcome was measured.
iAll subjects for whom outcome measures were available received the treatment or control application as assigned, if this was not the case, data for at least one central outcome analyzed by an “intention to treat” method.
jResults of statistical group comparisons were reported for at least one central outcome.
KStudy reports both point and dispersion measures for at least one central outcome.
Results
Search result
A total of 683 papers were identified from the search with key terms in Web of Science (all databases, 329 papers), Pub Med (333 papers), and BISp SURF (21 papers). Thus, we identified a total number of 683 papers in phase 1 (identification). In phase 2 (screening), we excluded 574 papers as duplication records and on basis of titel. The remaining 109 papers were again checked for inclusion and exclusion criteria and evaluated by abstract (phase 3: eligibility). A total of 82 papers did not meet the criteria and were excluded. Upon review of full texts, 12 of these were excluded as they also did not meet the criteria. Finally, 15 key papers were included (phase 4: included).
Studies had moderate to good quality with a mean value of 5.93 ± 0.25. We present the results of the included studies by cognitive parameter in Tables 2–6
Computerised testing batteries.
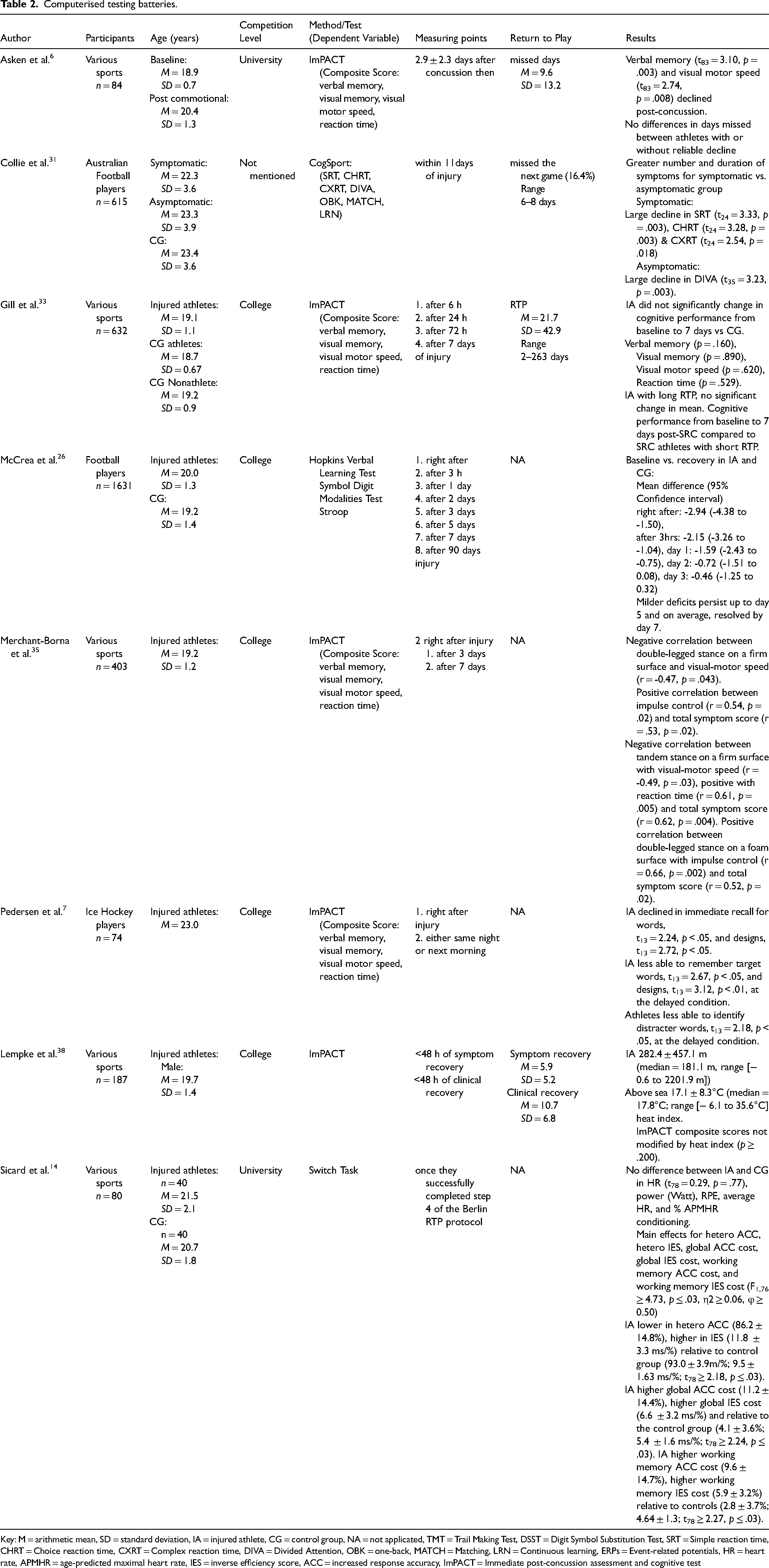
Key: M = arithmetic mean, SD = standard deviation, IA = injured athlete, CG = control group, NA = not applicated, TMT = Trail Making Test, DSST = Digit Symbol Substitution Test, SRT = Simple reaction time, CHRT = Choice reaction time, CXRT = Complex reaction time, DIVA = Divided Attention, OBK = one-back, MATCH = Matching, LRN = Continuous learning, ERPs = Event-related potentials, HR = heart rate, APMHR = age-predicted maximal heart rate, IES = inverse efficiency score, ACC = increased response accuracy, ImPACT = Immediate post-concussion assessment and cognitive test
Paper-Pencil testing.
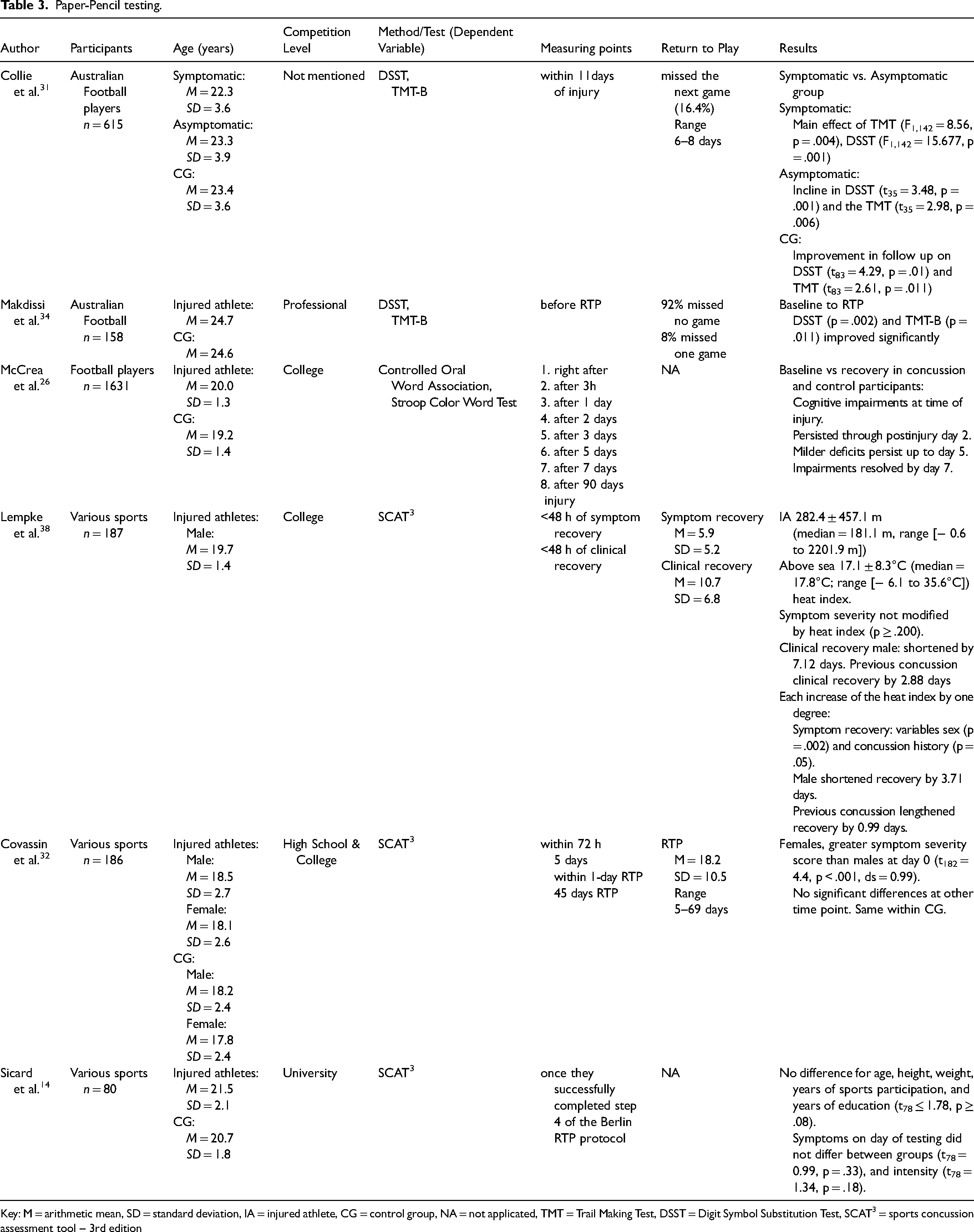
Key: M = arithmetic mean, SD = standard deviation, IA = injured athlete, CG = control group, NA = not applicated, TMT = Trail Making Test, DSST = Digit Symbol Substitution Test, SCAT3 = sports concussion assessment tool – 3rd edition
Postural stability.
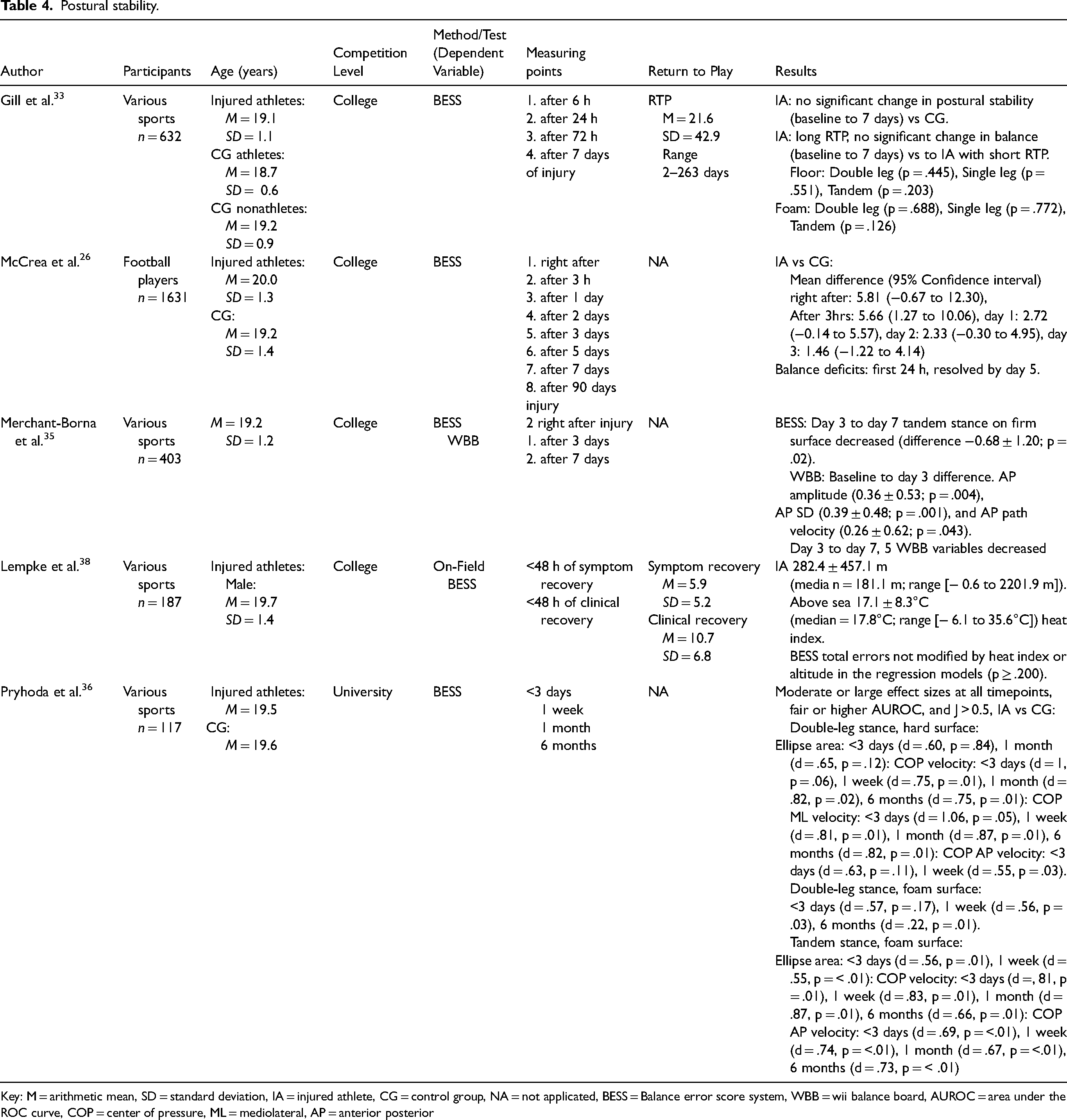
Key: M = arithmetic mean, SD = standard deviation, IA = injured athlete, CG = control group, NA = not applicated, BESS = Balance error score system, WBB = wii balance board, AUROC = area under the ROC curve, COP = center of pressure, ML = mediolateral, AP = anterior posterior
Divers.
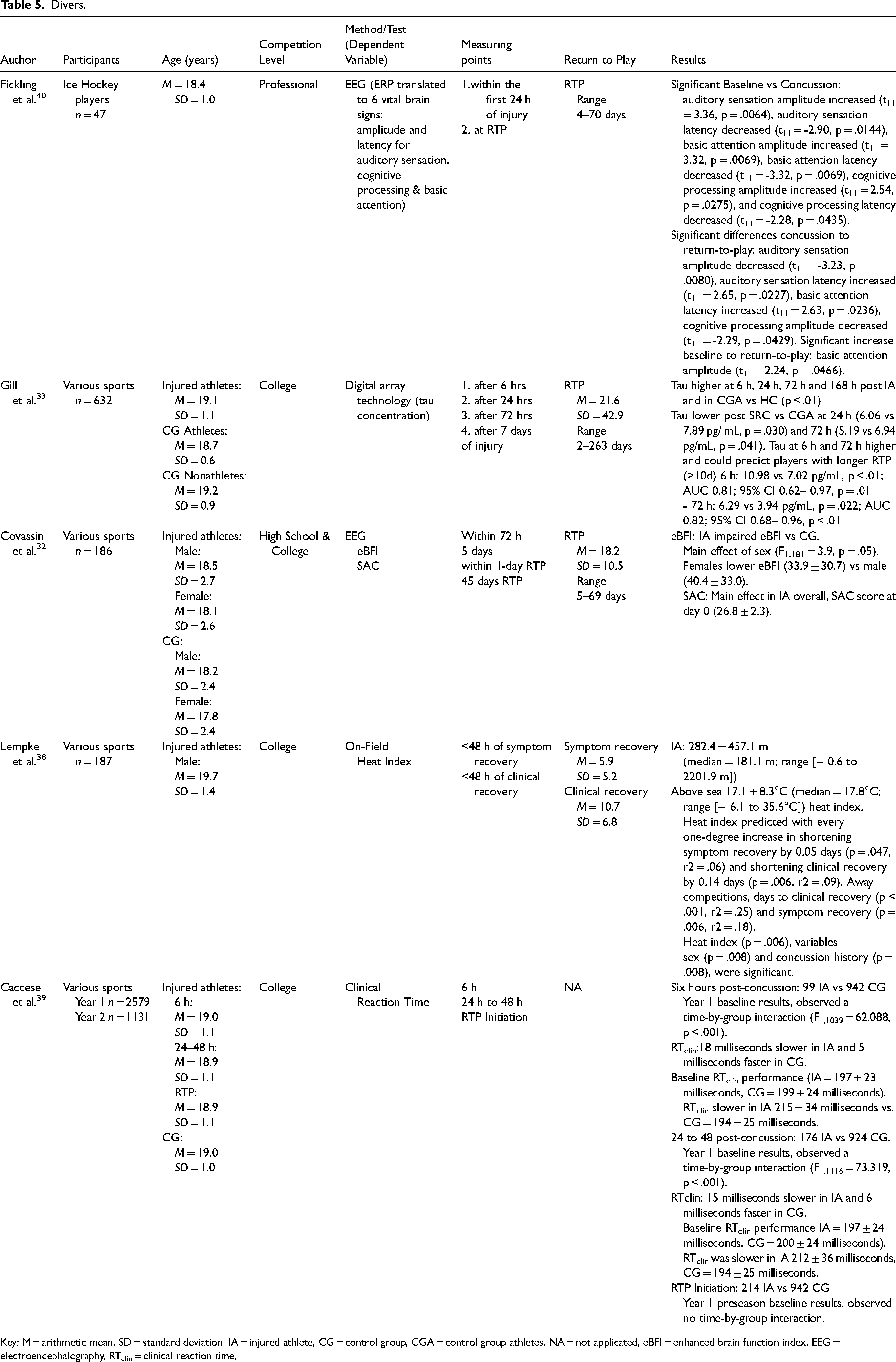
Key: M = arithmetic mean, SD = standard deviation, IA = injured athlete, CG = control group, CGA = control group athletes, NA = not applicated, eBFI = enhanced brain function index, EEG = electroencephalography, RTclin = clinical reaction time,
Protocols.

Key: M = arithmetic mean, SD = standard deviation, IA = injured athlete, CG = control group, NA = not applicated, SCAT5 = sports concussion assessment tool – 5th edition, RTclin = clinical reaction time, HIMAT = high level mobility assessment tool, NDI = neck disability index, LEFS = lower extremity function scale, IPAQ = international physical activity questionnaire – short form, DHI = dizziness handicap inventory, NII = neurosymptoms inventory index, CAP = care affiliated project, MFI = multidimensional fatigue inventory, PSQI = Pittsburg sleep quality index, I-PRRS = injury – psychological readiness to return to sport, MC = matched contro
Discussion
The purpose of this systematic review was to collate and evaluate cognitive diagnostic tools for RTP decisions after SRC. Recommended assessments like computerised tests (such as ImPACT, CogSport, Switch task) and paper-and-pencil tests (DSST, TMT-B), neuropsychological test batteries (EEG), postural stability tests (BESS), and questionnaires (SACT3) as well as other experimental tests for cognitive diagnostics, were investigated. All these approaches could show a variety of cognitive impairment and have been evaluated following SRC. Cognitive impairments including cognitive control, visual memory, verbal memory, visual motor speed, reaction time, and working memory are raised after SRC and show varying time courses of recovery post-concussion. In addition, they show a correlation with severity of concussion measured either by symptom severity or duration for RTP. 31
The current evidence base has a few limitations. These include the study design, which affects the conclusions that can be drawn from the findings. Presented studies6,7,35,38,40 did not include a matched control group without SRC (same age, gender, and sport). A lack of standardisation in the process makes comparisons more complex as the included studies varied in follow-up periods and assessments of exposures and outcomes. An explanation for this may lie in the absence of a valid indicator for cognitive recovery time after SRC in these studies. Further factors such as sleep, history of previous concussion, and attentional impairment can also affect results.42–44 Additionally, significant differences in cognitive performance between individuals may result from lifestyle factors, such as level of education, type of sport, and general cognitive ability before brain injury. 45 Consequently, studies of rehabilitation methods must carefully balance their study populations to provide usable results,34,46,47 especially with these interindividual differences. 48 The assessments help with diagnostics and RTP grading of an SRC, but it could be argued that it doesn't help players holistically. For example, for muscle or tendon injuries, there is a clear guideline that actively (isometric contraction, tens, stretching, strengthening) rehabilitees the injured tissue before returning to play. These activities are also used in the prevention of such injuries. In comparison, this form of rehabilitation is rarely used for cognition after SRC (before RTP) because of a disproportionate focus of trainers on motor and vestibular-ocular symptoms. An interesting and additional approach could be provided by Giza and Hovda 49 they showed that moderate physical activity as well as cognitive activity compared with high activity and no activity leads to optimal neurocognitive outcomes after concussion. This demonstrates that graduated guidelines are essential for returning to sport. 50
Similarly, the cognitive assessments and approach to post-injury rehabilitation must consider the individual areas of impairment, individual goals, and critical skills needed to return to activity.51,52 For example, Gavett et al. 53 and Zemper et al. 54 have shown that cognitive training can improve sport-specific performance. Gavett et al. 53 improved shooting accuracy with lacrosse players and Zemper et al. 54 on-field passing with soccer player. In both studies, the intervention was tailored to specific cognitive skills thought to support performance. Stephens et al. 55 propose that neuropsychological assessment should be combined with a motor control task to obtain more independent information about the damaged regions of the brain or all damaged areas by using dual tasks, multitasking, and virtual reality. As a result, none of these tests can currently be used on the field or sidelines for exclusion from the activity or RTP. Due to the strict rules of many sports, which do not allow for stoppages and substitutions, these tests cannot be conducted and evaluated in their entirety, that would allow safe RTP. 56
However, Sicard et al. 14 , McCrea et al. 26 , Pryhoda et al. 36 and Fickling et al. 40 could show that RTP clearance may not fully indicate cognitive recovery following SRCs, particularly for high-risk sports such as football and ice hockey. Sicard et al. 14 and McCrea et al. 26 showed strongest evidence for computer testing (cognitive control, verbal memory, visual motor speed, and reaction time), which were assessed in single cohorts with good sample sizes, multiple measuring points, and consistent use of control group, with extensive matching for history of head injury. Pryhoda et al. 51 evaluated a modified BESS (double-leg stance on hard surface and tandem stance on foam) in multiple cohorts, but in each case, a small cohort and control group were used. EEG brain vital signs (reaction time) in Sicard et al. 14 showed the weakest evidence which were from a small sample, in a design without control group. The limited evidence base suggests that more powerful and standardised studies are needed to improve our knowledge. A holistic impression is provided by the protocol of Powell et al. 41 in a repeated measures observational study using a range of multimodal assessment tools (symptom, cognitive, visual, and motor). Morris et al. 37 protocol, using the push-and-release test, attempts to examine reactive postural responses to balance restoration in athletes. Considering the statistical quality of the fifteen papers, only two papers14,33 used an a-priori power analysis. Pryhoda et al. 36 and Caccese et al. 39 reported effect sizes according to Cohen. Covassin et al. 32 provided an a-priori power analysis and effect sizes. Without information on power analysis and effect sizes, the interpretation of the results is limited, especially in studies with small sample sizes.7,35,38,40 Future studies should at least include post-hoc power analyses (better a-priori) and report the statistical effect size.
Despite these limitations there is evidence to support further research into the potential use of cognitive testings to assist clinicians to make RTP decisions. The present review identifies several cognitive impairments including cognitive control, verbal memory, visual motor speed, and reaction time that show deviations post SRC and not return to baseline by clinical recovery from injury. Sampling these impairments post-concussion and regularly throughout rehabilitation could provide valuable information on cognitive recovery.
In the future, improvement of a cognitive domain to baseline levels could be included in the criteria for RTP in addition to clinical recovery and successful completion of a stepwise rehabilitation programm. Neurophysiological recovery may differ from clinical recovery, thus leading an athlete, who has clinically recovered from SRC, being excluded from RTP due persistent cognitive abnormalities. These present findings and evaluation provide references for further exploration of the potential use of cognitive parameters to help clinicians make better RTP decisions.
Conclusion
We found that there was great variation between studies and showed a lack of standardisation in the RTP process after SRC. 15 There is currently no unified approach to cognitive diagnostic assessment including timing and frequency of measuring points after SRC, as well as for follow-ups after the RTP decision.
Furthermore, our findings reveal that some test selections are not sensitive enough. For example, while the Scat5 can be a useful tool for neurological screening but is lacking in vestibular-ocular screening. Vestibular-ocular symptoms such as dizziness and blurred vision are associated with a greater risk of protracted recovery. 57 Vestibular-cular motor screening tests evaluate vestibular-ocular dysfunction, visual motion sensitivity, rapid eye movements, and smooth visual pursuits. 58 Kontos et al. 59 found that athletes with concussions were likely to experience symptoms such as dizziness or nausea here, while this is not the case in healthy individuals. Accordingly, there is a need for sensitive and multimodal cognitive testing during the RTP protocol that is not contaminated by learning effects. Preventing this could be via a combination of different approaches. Using, for example, Powell's et al. 41 protocol, may enhance sensitivity and objectivity of measures used to make activity removal-decisions, support diagnose SRC, and make RTP decisions.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author contribution
Dennis Wellm (WD) prepared the manuscript together with Karen Zentgraf (ZK). WD performed the literature review selection.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
