Abstract
The non-specificity of persisting concussion symptoms (PCS) following concussion can make treatment and rehabilitation a complex and challenging endeavour for clinicians. International studies have demonstrated that in neurologically healthy individuals similar symptoms also exist. This study aimed to examine the presence and frequency of symptoms similar to PCS in neurologically healthy adults in Aotearoa New Zealand and compare these to PCS following concussion. 252 neurologically healthy adults (34.22 years); completed the Rivermead Post-Concussion Questionnaire (RPQ). Scores on this measure were evaluated and compared with 146 individuals who were at least three months post-injury (34.62 years). 25.79% (n = 65) of the neurologically healthy individuals had scores on the RPQ of ≥16. Fatigue was the most commonly endorsed symptom (67.9%), followed by sleep disturbances (59.9%). Certain demographic factors (age, education history and gender) were associated with higher endorsement but only on specific symptoms. The concussion group had significantly higher scores on the RPQ and significantly more individuals with PCS. However, at the individual item level, just over half of PCS symptoms were significantly higher in the concussion group. These symptoms were predominately neurological, with only forgetfulness and irritability significantly different between the concussion and neurologically healthy groups. Symptoms similar to PCS are prevalent in neurologically healthy individuals in Aotearoa New Zealand. The implications of these findings on the rehabilitation and management of PCS are discussed.
The majority of people who experience a concussion have a period of days or weeks with symptoms that can be broadly classified as neurological (i.e., headache, dizziness, sensory sensitivity), cognitive (i.e., concentration and memory difficulties, slowed processing speed) and psychological (frustration, irritability, low mood, and anxiety). 1 Although it is generally assumed that these symptoms will resolve over time, a considerable minority will experience prolonged symptoms, commonly referred to as persisting concussion symptoms (PCS). 2 There is a discrepancy within the field regarding the exact length of time that constitutes “persisting”. 3 According to the International Classification of Diseases and Related Health Problems, Tenth Revision (ICD-10), 4 persisting symptoms are defined as those lasting for more than four weeks after injury. However, other definitions have stipulated that persisting symptoms are evident from three months after the injury.5–7 Despite these differences, it is evident that PCS constitutes a continuation of symptoms after a concussion that can significantly impact functioning. 2 Recent, research studies have revealed that the incidence and burden of PCS are far greater than historically believed.8–12 For example, a community-based longitudinal study in New Zealand aimed to identify all incidences of TBI in the Waikato region (BIONIC). One year following concussion (n = 341), nearly half reported experiencing four or more post-concussion symptoms. 8 The implications of PCS can be significant for both the individual and their family/whānau, as well as burdening health services. 13
The concept of PCS has been debated in abundance in the literature and amongst clinicians in the field. 14 This is predominately driven by the non-specificity of PCS, as well as concussion symptoms that don’t persist. 15 That is, the constellation of symptoms associated with a concussion exists in healthy populations (herein referred to as ‘ symptoms similar to PCS’) 16 (i.e., migraine, chronic dizziness, fatigue). This phenomenon is not unique to concussion and occurs in a range of neurological conditions such as brain tumours, stroke and epilepsy.17,18 Recent research has aimed to ascertain the incidence of symptoms similar to PCS in neurologically healthy individuals. For example, Wang, Chan and Deng 19 administered the Rivermead Post-Concussion Symptoms Questionnaire (RPQ) to a sample of 124 university students. A relatively high base rate of symptoms similar to PCS was found. The most frequently endorsed item was fatigue (76.9%), longer time to think (60.3%), poor concentration (58.7%), sleep disturbance (50.4%), and frustration (46.3%). Looking more at the severity of symptoms, Iverson and Lange 20 differentiated their sample of healthy participants (n = 104) into mild (symptom endorsement of 2 or less) and moderate-severe groups (symptom endorsement of greater than 2) on the RPQ. A similar frequency of symptom endorsement was identified in the mild group (i.e., 75.7% “fatigue”, 71.8% “irritable”, 63.1% “nervous or tense”, 62.1% “poor sleep”). However, surprisingly a substantial minority of the healthy sample endorsed symptoms similar to PCS at the moderate-severe level (11.7% to 15.5%), despite the absence of a head injury. Noting some of the limitations in the sample of these studies (i.e., small numbers, recruited from one country and predominantly in North America) Voormolen and colleagues 14 explored the prevalence of symptoms similar to PCS in the general population of Italy, The Netherlands and the United Kingdom (n = 11,759) using the RPQ. The most frequently reported symptom was fatigue (49.9%), followed by sleep disturbance (42.4%); the least reported symptom was double vision (10.7%). 45.1% of the sample had a rating of 2 or greater on at least three of the following symptoms: headaches, dizziness, fatigue, irritability, impaired memory, impaired concentration and insomnia; whereas 17.5% of the sample had a rating of 3 or greater.
Additionally, research has also explored the factors that increase the risk of experiencing symptoms similar to PCS in healthy populations. In their population-based study, Voormolen et al., 14 found that lower age, female gender, low education, unable to work and low-income level were all significantly associated with these symptoms. Interestingly, Zakzanis and Yeung 21 explored differences in base rates of symptoms similar to PCS based on cultural identity and language. The study consisted of 153 participants that were Caucasian, Chinese, Filipino, Southeast Asian, Arab and African. No differences in overall symptoms were found when considering culture and language-based groups. However, there were differences at the individual symptom level. Specifically, a higher proportion of participants from the African group reported headaches (88.5%) and poor concentration (76.9%), whereas a higher proportion of Chinese, Filipino, and Southeast Asian participants endorsed dizziness. The rate of sleep disturbance was also higher in the Caucasian participant group (72.7%).
The presence of symptoms similar to PCS in neurologically healthy individuals can make the management and treatment of concussion challenging. Further complicating matters, despite extensive research concerning the aetiology of PCS, prognostic models that aim to predict individual outcomes following concussion continue to be inconclusive. 15 Research into identifying biomarkers for a concussion is promising; however, at present clinicians do not have a biofluid or imaging biomarker of high specificity or sensitivity to assist in clinical decision-making. 22 Thus, the management of concussion tends to focus on symptom monitoring to track recovery. Recovery is therefore deemed to be achieved when the individual's symptoms have returned to baseline. However, establishing what constitutes “baseline” is challenging, particularly if symptoms similar to PCS were already present before the injury. Additionally, well-validated phenomena such as the “good-old day bias”, which is the tendency to view oneself as healthier in the past and to underestimate past problems, add additional layers of complexity. 15 It can be understood that following a concussion the individual and clinicians want to strive for symptom resolution but this may not be achieved if accuracy pertaining to pre-injury functioning is not clearly established. It can also make the provision of services extremely challenging with individuals continuing to engage in concussion treatment programmes when this may no longer be indicated. Thus, having updated and population-specific data on the incidence of PCS in Aotearoa New Zealand can help inform clinicians when faced with this dilemma.
Thus, the aims of this study was to (a) evaluate the frequency and prevalence of symptoms similar to PCS in the general healthy population in Aotearoa New Zealand, (b) assess demographic risk factors for symptoms similar to PCS and (c), compare the incidence of PCS between neurologically healthy individuals and those with concussion who are at least three months post-injury.
Method
Participants and procedure
Neurologically healthy participants were recruited through advertisements posted throughout the community and through social media sites (Reddit, Facebook, Twitter). All of the participants met the criteria of not having any identifiable history of concussion or neurological disease. Interested individuals provided their contact details via an online survey. They were then provided with a description of the general purpose of the study. Consenting participants were either emailed a link to complete the questionnaires online or met with a researcher in person. This part of the study was approved by the Victoria University ethics committee (ref 30287).
Concussion participants were recruited from outpatient concussion clinics throughout the North Island of New Zealand between March 2020 and September 2020. Eligibility criteria for participants for both studies were: 1) aged 18 years or older; 2) sustained a concussion which was defined as an acute neurophysiological event related to blunt impact or other mechanical energy applied to the head, neck or body which results in a transient disturbance of neurological function , 23 3) were less than three months post-injury at enrolment, 4) were fluent in English, and 5) had no prior neurological condition or severe unstable medical condition. Eligible and consenting participants completed questionnaires via REDCap 24 at enrollment and then followed up three months later. In this study, only the three-month follow-up sample was used. The Auckland University of Technology ethics committee provided ethical approval for this study (ref 20/32).
Materials
Statistical analysis
To examine the incidence of symptoms similar PCS in neurologically healthy individuals three approaches were taken. First, descriptive statistics were used to examine overall scores on the RPQ. Second, the percentage of scores at the individual item level on the RPQ, as well as overall positive symptom endorsement, were calculated. Finally, the number of individuals with an RPQ score of ≥16 was calculated as this has been a cut-off score used in previous research. 26 This approach was taken because, in clinical settings, clinicians tend to interpret overall scores on the RPQ rather than consider individual item endorsement. Next, the influence of demographic factors on each specific PCS on the RPQ was calculated. Bivariate correlation was used for continuous variables (i.e., age) and independent sample T-Test and One-Way ANOVA for categorical variables (i.e., ethnicity). The incidence of PCS in the concussion group was calculated using the same process as outlined above. Finally, differences in each of the PCS indices (mean total RPQ, percentage of scores at the individual item level, and percentage of individuals with an RPQ score of ≥16) between the neurologically healthy and concussion groups were calculated using independent sample t-tests and Chi-Square statistics. In the neurologically healthy sample, 1.12% of the data was missing; the mean substitution method was used to address this. 27
Results
Participants
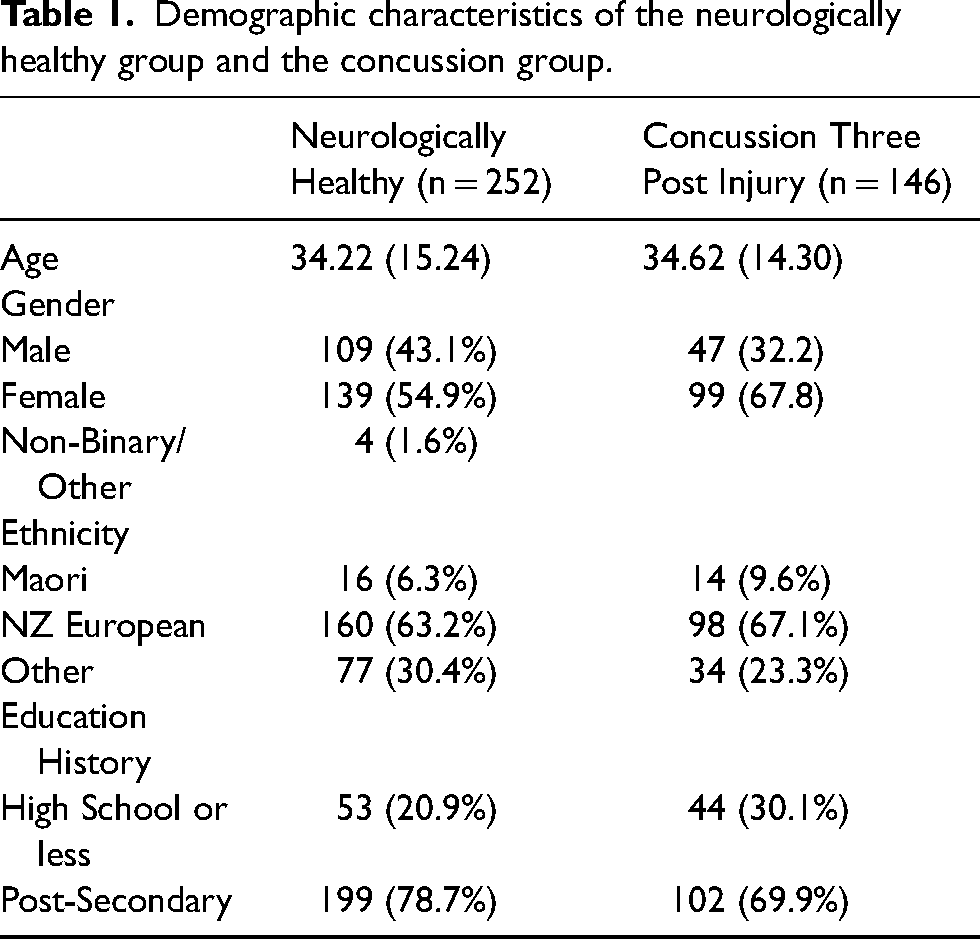
Two hundred and fifty-two neurologically healthy individuals participated in the study. The concussion group consisted of one hundred and forty-six individuals who were at least three months post-injury (mean time post-injury = 22.91 weeks). Experiencing a concussion as a result of hitting an object (42.5%) and a fall (41.1%) were the most common causes of injury in this sample; a concussion due to a motor vehicle accident (8.2%) or an assault (8.2%) was the least common. A summary of the demographic characteristics of the two groups is presented in Table 1.
Demographic characteristics of the neurologically healthy group and the concussion group.
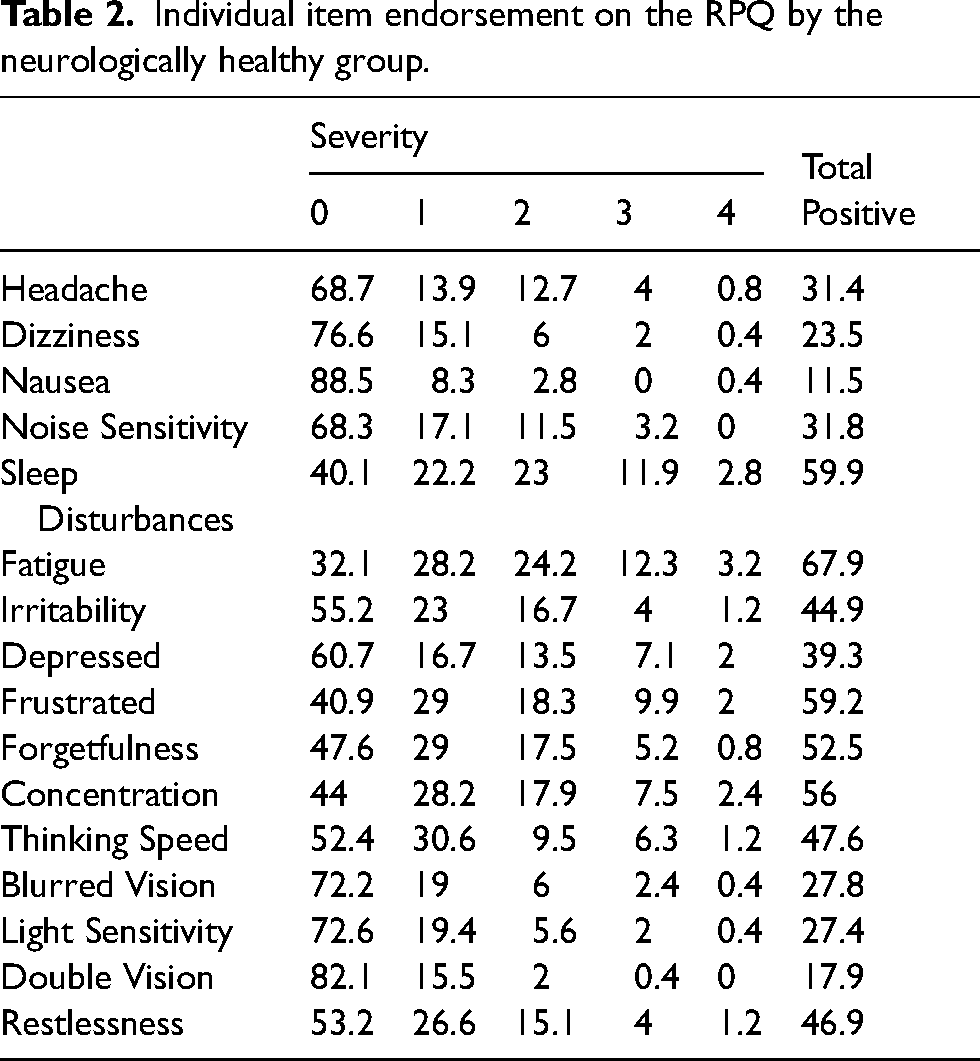
Neurologically healthy individuals’ mean total score on the RPQ was 10.61 (SD = 9.15). Table 2 summarises the frequency and percentage of post-concussion-like symptoms in the neurologically healthy group. Fatigue was the most commonly endorsed symptom (67.9%), followed by sleep disturbances (59.9%). Frustration was positively endorsed by 59.2% of the sample and was the most common affective symptom. The constellation of cognitive symptoms was also commonly endorsed; concentration difficulties (56.0%), forgetfulness (52.%) and thinking speed (47.6%). Nausea (11.5%) and double vision (17.9%) were the least commonly endorsed symptoms. 25.79% (n = 65) had scores on the RPQ of ≥16.
Individual item endorsement on the RPQ by the neurologically healthy group.
Next, the impact of demographic variables on PCS in neurologically healthy individuals was examined. Dizziness was the only symptom significantly associated with gender (t(245) = -1.31, p = .042), with females (M = 0.74) having significantly higher levels of this symptom than males (M = 0.68). Education history was significantly associated with noise sensitivity (t(245) = 2.40, p = .013), sleep disturbances (t(245) = 2.50, p = .003), and fatigue (t(245) = 2.00, p = .047). Secondary school education or less was significantly higher across all of these symptoms. There was no significant effect of ethnicity on each of the PCS. There was a significant negative correlation between age and each of the following PCS: headaches (r = -.143, p = .024), dizziness (r = -.195, p = .002), depressed (r = -.149, p = .018), frustration (r = -.174, p = .006), and concentration difficulties (-.216, p < .001).
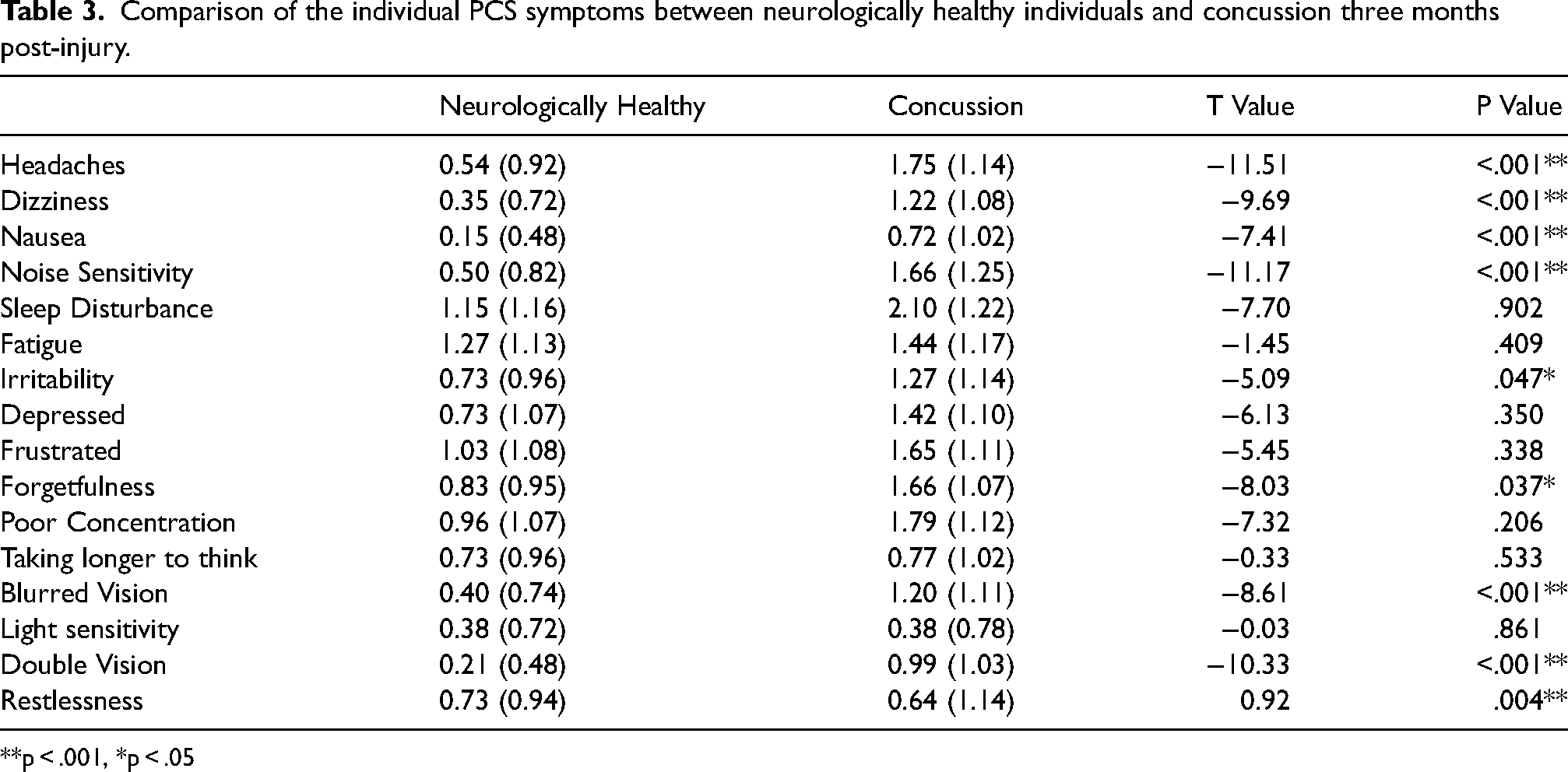
The concussion group had a mean RPQ total score of 17.00 (SD = 10.31). This was significantly higher than the neurologically healthy group (mean difference = 6.35; t(396) = -6.36, p < 0.001). 51.34% (n = 75) of the sample had a total RPQ score of ≥16 which was significantly higher than the neurologically healthy group (χ = 26.24, p < .001). As shown in Table 3, several PCS items were significantly higher in the concussion group than in the neurologically healthy group. These symptoms were predominately physical and included headaches, dizziness, nausea, noise sensitivity, and blurred vision. The only cognitive symptom significantly higher in the concussion group was forgetfulness and the only psychological symptom higher in this group was irritability.
Comparison of the individual PCS symptoms between neurologically healthy individuals and concussion three months post-injury.
**p < .001, *p < .05
Discussion
This study aimed to identify the frequency and prevalence of symptoms similar to PCS in a neurologically healthy sample of individuals from Aotearoa New Zealand. We found that one-quarter (25.79%) of the neurologically healthy individuals had scores of ≥16 on the RPQ, a score that has been used to classify PCS. 26 Fatigue was the most commonly endorsed individual symptom, followed by sleep disturbances. This finding is consistent with international studies, which also found that fatigue has the highest endorsement rate.14,19,20 Consistent with these studies, our sample also had high positive symptom endorsement in cognitive symptoms such as concentration and memory difficulties, taking longer to think, and psychological symptoms such as frustration. Thus, symptoms similar to PCS are present within the general population of Aotearoa New Zealand and occur in patterns consistent with international studies.
The second aim of this study was to investigate demographic risk factors for symptoms similar PCS in neurologically healthy individuals. Interestingly, we found no significant differences between demographic factors when considering overall scores on the RPQ. However, there were significant differences at the item level on the RPQ. This suggests that specific symptoms similar to PCS may be more prevalent within demographic groups. More specifically, we found that females had a higher incidence of dizziness. Additionally, lower education history was associated with greater noise sensitivity, sleep disturbances and fatigue; and being younger was associated with higher levels of headaches, dizziness, depression and concentration difficulties. Interestingly, this constellation of symptoms is similar to that of vestibular migraine which is common in this demographic 28 and thus may be one possible explanation. This latter finding is also consistent with the population-based study of Voormolen et al., 14 and is somewhat surprising as it would be predicted that older adults may have more difficulties in areas assessed due to ageing effects. However, the younger group did endorse higher levels of affective symptoms (depression and concentration difficulties) with the experiences of headaches and dizziness potentially contributing to this.29,30 Further exploration is needed to test this hypothesis, but it is consistent with findings pertaining to the high incidence of mental health difficulties in younger adults. 31 Interestingly we found no effect of ethnicity on symptoms similar to PCS a finding inconsistent with that reported by Zakzanis and Yeung. 21 However, our findings should be treated cautiously as ethnicity was classified into three very broad groups.
The final aim of this study was to compare differences in PCS between neurologically healthy individuals and those with a concussion. The concussion group was at least three months post-injury and thus consistent with PCS. The concussion group had significantly higher levels of PCS than the neurologically healthy group, and they also more likey to have scores on the RPQ of ≥16 (51% vs. 26%). Interestingly, at the individual symptom level more nuanced findings emerged. That is, just over half of the items on the RPQ captured a difference between the neurologically healthy and concussion groups. Additionally, of these items, all but two are classified as neurological symptoms. The only cognitive and psychological symptoms that differed between the groups were forgetfulness and irritability. These results suggest that the total score level differences between neurologically healthy individuals and concussion may be driven by predominately elevated neurological symptoms in the concussion group. These results also provide interesting insights regarding recovery from a concussion. Individuals who are experiencing PCS don’t just report neurological symptoms. Long-term fatigue, cognitive difficulties and mental distress are also very common.8,32,33 However, our results suggest that these reported persistent difficulties may be occurring at a level similar in neurologically healthy individuals. It is therefore important that clinicians consider all symptoms of PCS when monitoring concussion recovery and our findings suggest that considering neurological symptoms may be particularly important in this endeavour.
A particular challenge faced by clinicians in concussion rehabilitation is establishing an individual's pre-injury functioning which is then used as a marker for recovery. The temporal relationship between a concussion and pre-injury functioning can for some individuals exacerbate pre-injury difficulties. Importantly, the increase in symptoms can assist with diagnosis and guide clinicians in determining what symptoms are triggered by a concussion and which are not. Importantly, this can support the implementation of specific interventions as it may be that the treatment adopted is the same approach used in the general population. However, clinicians are often reliant on individuals’ self-report to determine their pre-injury functioning. An approach that is challenged by several factors. For example, immediately following a concussion, in some individuals, pre-injury difficulties (i.e., fatigue) are exacerbated by the physiological consequences of the injury. It is therefore understandable that individuals will attribute the symptom to being caused by the injury. However, if these difficulties were present beforehand, their ongoing presence can be misattributed to the injury and imply incomplete recovery. It can then set up a belief that these symptoms have to be resolved and not achieving this may fuel ongoing psychological distress i.e., “why am I still feeling fatigued? Have I not recovered yet?” Psychological factors are well known to contribute to and maintain PCS following an acute trigger.34,35 Thus, addressing the potential contributors of mental distress in concussion may be beneficial for recovery; consistent with the holistic rehabilitation approach to concussion that is now widely advocated for. 36 The use of population-based findings, such as the one presented here, can assist with this. Presenting individuals with the percentage of symptom endorsement in neurologically healthy individuals could be a beneficial rehabilitation strategy and is one commonly used when working with cognitive lapses in older adults. It may help challenge misattributions of the cause of symptoms and normalise and validate an individual's current experience. Uncertainty and confusion regarding PCS contributes to poorer long-term outcomes. 37 Providing this education by presenting individuals with a frequency of symptoms similar to PCS in neurologically healthy individuals has the potential to mitigate this. Future research may consider gathering more extensive and representative data that could be used to make a more formal normative-based process to statistically determine symptom elevation, a process used in neuropsychological tests.
It is, however, important to note that the concussion sample recruited for this study was engaged in a rehabilitation programme for the treatment and management of their symptoms. The concussion sample is therefore not representative of this population and these findings can therefore not be generalised to all concussions. Replication of this study using a more diverse sample of individuals after a concussion i.e., community population, as well as those who present to medical services without receiving rehabilitation, will be an important next step for future research. Additionally, we only assessed individuals after a concussion at one-time point. Replication of the study using a prospective design to determine how trajectories of recovery compared with symptoms similar to PCS in the general population would be another important area of future research.
In conclusion, this study showed that symptoms similar to PCS are prevalent in neurologically healthy individuals in Aotearoa New Zealand. Certain demographic factors (age, education history and gender) were associated with higher endorsement but only on specific symptoms. The concussion group, who were at least three months post-injury, had significantly higher rates of PCS than the neurologically healthy group; however, this was predominately driven by neurological symptoms with no significant differences in the majority of cognitive and psychological symptoms assessed on the RPQ. These findings can be used to inform rehabilitation services and the treatment of a concussion in Aotearoa New Zealand.
Footnotes
Acknowledgements
Concussion data was extracted from a concussion research project that was funded by a Health Research Council of New Zealand Foxley Clinical Fellowship (20/041). Thank you also to Matthew Lamb for his role in the data collection of neurologically healthy individuals.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Health Research Council of New Zealand, (grant number 20/041)
