Abstract
Premature return to play (RTP) following sports-related concussion (SRC) is associated with significant morbidity including risk of neurological and non-neurological injury, persistent post-concussion symptoms and chronic neurological deficits. Assessing athletes for RTP is critical but these decisions are currently based on clinical assessments that are subject to bias and symptomatic reporting that rely on compliance. An objective and easily obtained biomarker that can indicate recovery following SRC would aid clinicians to make safer RTP decisions. We performed a systematic review to identify potential biomarkers from saliva, urine and blood sources that could inform the clinical RTP decision. The MEDLINE database was searched. Inclusion criteria were studies focusing on adults diagnosed with SRC, fluid biomarkers from blood, saliva or urine and clinical recovery from SRC or at RTP. We assessed each biomarker for their time course post SRC and relationship to clinical recovery. Secondary outcomes included correlation with symptom scores and predictive value for prolonged RTP. We identified 8 studies all investigating blood-based markers of diffuse axonal injury (tau, NFL, SNTF), neuroglial injury (NSE, VLP-1, UCH-L1, S100B, GFAP), inflammation and hormonal disturbances. Tau, SNTF, UCH-1, GFAP, S100B and the inflammatory cytokine MCP-4 are raised post SRC and return to baseline by RTP. Changes in tau, NFL, SNTF, GFAP and MCP-4 post SRC correlate with severity of concussion as measured by symptom severity or RTP duration. There is only preliminary case-reporting for hormonal biomarkers. The evidence is limited by a lack of highly powered studies, variation in use of athletic and Contact sport controls (CSC) and a lack of consistent sampling and assessment protocols. There is promise for biomarkers to aid RTP decisions following SRC, most notably in use alongside clinical assessment in RTP criteria to allow greater precision in identifying mild and severe concussion.
Introduction
Sports-related concussion (SRC) is a growing public health concern with significant and underestimated morbidity. SRC is a traumatic brain injury that causes transient loss of neurocognitive function and accounts for 25% of mild traumatic brain injuries (mTBI) presenting to the emergency department. 1 Clinical features of SRC are broad and varied, including neurocognitive symptoms (headache, amnesia, impaired concentration, drowsiness), autonomic dysfunction (dizziness, low blood pressure, palpitations, sweating, flushing, gastrointestinal symptoms), sleep disturbance, emotional lability and balance impairment. Any athlete with a suspected SRC must be removed from play. Safely determining when an athlete can return to play (RTP) following SRC diagnosis is a clinical priority as premature RTP puts athletes at increased risk of developing further neurological and non-neurological injuries, 2 persistent post-concussion symptoms 3 and chronic neurological deficits. 4
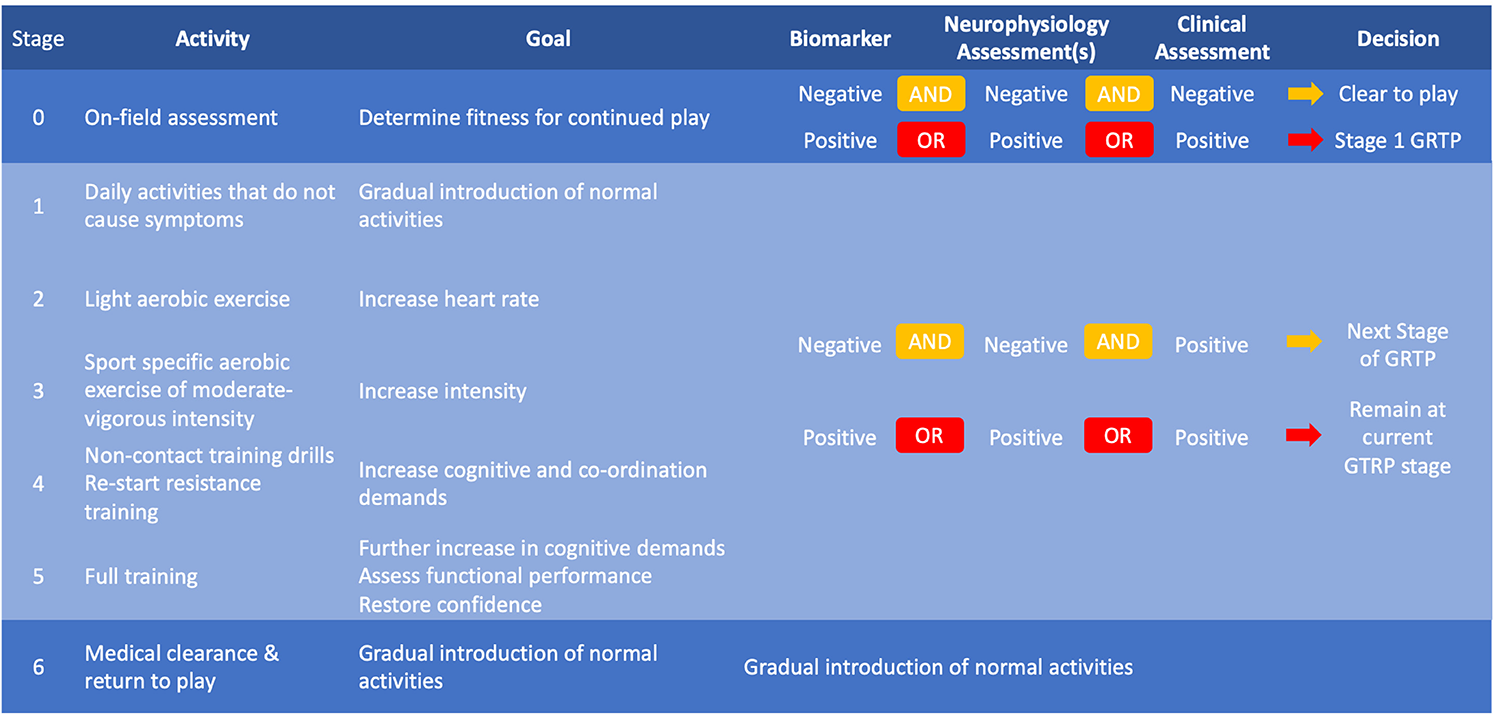
However, establishing when an athlete has recovered from an SRC is difficult. Current protocols for managing SRC involve an initial period of rest (24–48 h) followed by a stepwise rehabilitation protocol with gradually increasing physical and cognitive demands, 5 an example of which is shown in Table 1. Each stage takes 24 h with progression dependent on completion of the activity and meeting appropriate criteria (e.g. heart rate, duration of exercise) without recurrence of concussion-related symptoms. If symptoms do recur the athlete should drop back to the previous asymptomatic stage for a further 24 h. Clearance for RTP is determined by completion of the graded rehabilitation protocol and passing clinical assessment at each stage. However, symptom assessments in the recovery phase can be difficult and are subject to bias. They rely on subjective, athlete-dependent assessments of symptoms, whilst it can also be challenging to differentiate concussion-related symptoms from pre-morbid conditions such as chronic sleep dysfunction, migraines, anxiety and attention problems. Standardised neurological and cognitive assessment scales have been developed to assist the sideline diagnosis of SRC and following diagnosis, to aid the RTP decision. 6 However, these are imperfect and no single test can be used for either SRC diagnosis or RTP. Moreover, increasing evidence suggests physiological recovery from concussion may outlast clinical recovery meaning even after a successful graduated RTP protocol, athletes may still be at higher risk of neurological and non-neurological injury. 7 Given the limitations of clinical assessment in determining RTP following SRC, there is a great need for an objective indicator of neurophysiological recovery to enable clinicians to make safer RTP decisions.
A potential biomarker-informed graduated return to sport strategy following SRC.
A biomarker is an objective physiological indicator of biological disease or an injury state. 8 TBI involves a variety of pathological mechanisms and allowing a range of biomarkers to be detected in blood, saliva, urine and cerebrospinal fluid (CSF) samples following injury. During mTBI shear forces mechanically damage neuronal axons leading to release of intracellular cytoskeleton proteins such as tau, neurofilament light (NFL) and α-II spectrin N-terminal fragment (SNTF)9,10 and these biomarkers of axonal injury have been shown to accumulate following mild TBI, severe TBI and chronic repetitive head injuries.11,12 Neuronal cell bodies and astrocytes also undergo mechanical injury during mTBI releasing intracellular proteins such as neuron specific enolase (NSE), visinin-like protein-1 (VLP-1), ubiquitin carboxyl-terminal hydrolase L1 (UCH-L1), S100B and glial fibrillary acidic protein (GFAP).13–15 Growing evidence suggests secondary pathologies such as neuroinflammation and pituitary dysfunction play a role in TBI. Moderate to severe TBI has been shown to alter peripheral inflammatory cytokine profiles which negatively correlate with outcome 16 whilst repeated concussions have been linked to pituitary dysfunction 17 with cases of secondary growth hormone deficiency 18 and diabetes insipidus 19 raising the possibility that dysfunction of the hypothalamic-pituitary axis may be detectable in the acute post-concussive state.
Fluid biomarkers have potential to aid RTP decisions following SRC. Monitoring a biomarker during the rehabilitation phase post-concussion could provide an objective measure of neurophysiological recovery from injury, which could be used to complement clinical assessment in RTP decision making. Fluid sampling from CSF is invasive and carries significant procedural risks meaning only biomarkers from blood, urine and saliva samples would be feasible for serial monitoring. We performed a systematic review to identify biomarkers from blood, urine or salvia samples that have been assessed after SRC and throughout recovery from SRC. We look at the major pathological mechanisms involved in mTBI and highlight the key biomarkers from each pathway, evaluating their time course post SRC and their relationship to clinical recovery. In this way we identify objective biomarkers of neurophysiological recovery from SRC that could have potential use in RTP decisions.
Methods
A systematic review of the literature was conducted in accordance with PRISMA guidelines. 20 A sensitive MEDLINE search strategy was comprised to identify relevant studies up to August 2021 using 3 groups of MESH terms: ‘concussion’, ‘human’ and ‘fluid biomarker – blood, saliva, urine’: (("Brain Concussion"[Mesh] OR "mild traumatic brain injury"[All Fields] OR mTBI[All Fields]) AND "humans"[MeSH Terms]) AND (("serum"[MeSH Terms] OR "serum"[All Fields]) OR ("blood"[Subheading] OR "blood"[All Fields] OR "blood"[MeSH Terms]) OR ("urine"[Subheading] OR "urine"[All Fields] OR "urine"[MeSH Terms]) OR ("saliva"[MeSH Terms] OR "saliva"[All Fields])) AND English[lang]. Reference lists of included papers were also searched for relevant studies.
Eligibility criteria
Inclusion criteria
Study population consists of adults (age 18 or over) diagnosed with an SRC
Studies examining fluid biomarkers in blood, saliva and urine
Studies examining biomarkers during recovery from concussion or at RTP
Exclusion criteria
Studies assessing chronic neurological symptoms following repeated head injuries.
Studies focusing on subconcussive head injuries
Non-fluid biomarkers
Conference abstracts
Study selection
All studies identified were screened independently by 3 authors (NS, ES and AH). This involved an initial title and abstract screening followed by full text screening to assess if eligibility criteria were met. Any disagreements regarding inclusion or exclusion were resolved by consensus.
Biomarker assessment
Biomarker studies were assessed to identify time courses post-SRC and relationship with clinical recovery. Secondary outcomes included correlation with symptom severity and predictive value for athletes with prolonged RTP.
Risk of bias assessment
The risk of bias of included studies was assessed independently by three authors (NS, ES and AH) using the ROBINS-I tool 21 which explores seven bias domains: bias due to confounding, bias in selection of participants into the study, bias in classification of interventions, bias due to deviations from intended intervention, bias due to missing data, bias in measurement of outcomes, bias in selection of the reported result. Each of the seven domains was assessed for low, moderate, serious or critical risk of bias. An overall rating for risk of bias for each study based on the scores of these seven domains: low risk of bias was determined if all domains were rated low risk, moderate risk if all domains rated low or moderate risk, serious risk if at least one domain rated serious but not critical and critical risk if at least one domain was rated critical.
Results
Search results
The literature search revealed 572 potentially relevant papers (Figure 1). Title and abstract screening identified 12 potentially relevant papers. Upon review of full texts, 5 of these were excluded as they did not evaluate the biomarker in the recovery period or involved duplicate data sets leaving 8 key studies. We present the results by biomarker in Tables 2–6. Risk of bias assessments are shown in Table 7. All 7 studies investigated biomarkers from blood samples. No studies evaluating the correlation of biomarkers from urine or saliva with RTP were identified.
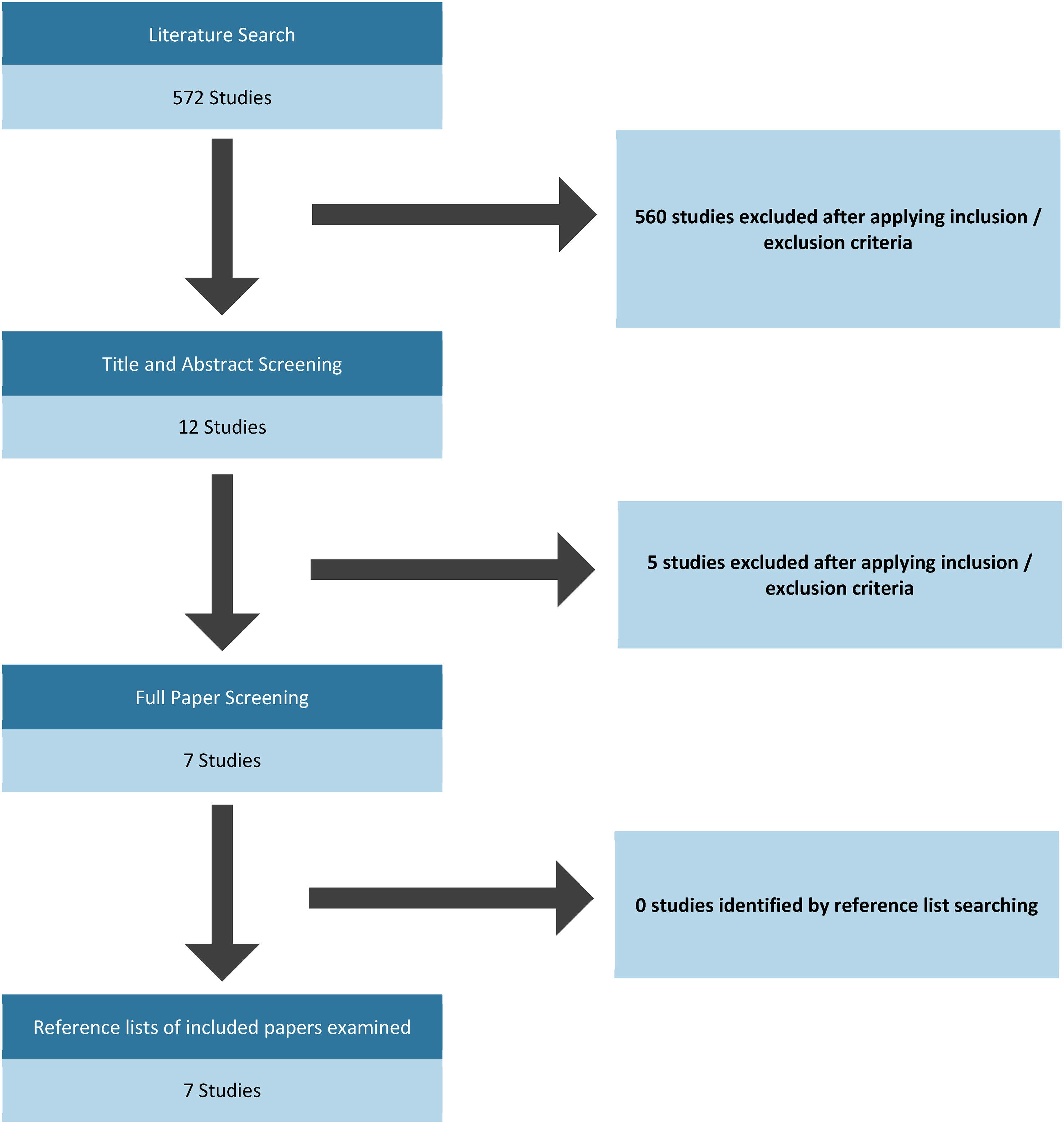
Search results.
Biomarkers of axonal injury.

Key: SRC = sports-related concussion, RTP = return to play, d = days, SEM = sports exercise medicine PLSDA = Partial Least Squares Discriminant Analysis, PLS = partial least squares, IQR = interquartile range
Biomarkers of neuronal injury.

Key: SRC = sports-related concussion, RTP = return to play, d = days, SEM = sports exercise medicine PLSDA = Partial Least Squares Discriminant Analysis, PLS = partial least squares, IQR = interquartile range
Biomarkers of astroglial injury.

Key: SRC = sports-related concussion, RTP = return to play, d = days, SEM = sports exercise medicine PLSDA = Partial Least Squares Discriminant Analysis, PLS = partial least squares, IQR = interquartile range
Biomarkers of pituitary dysfunction.
Key: SRC = sports-related concussion, RTP = return to play, d = days
Biomarkers of neuroinflammation.
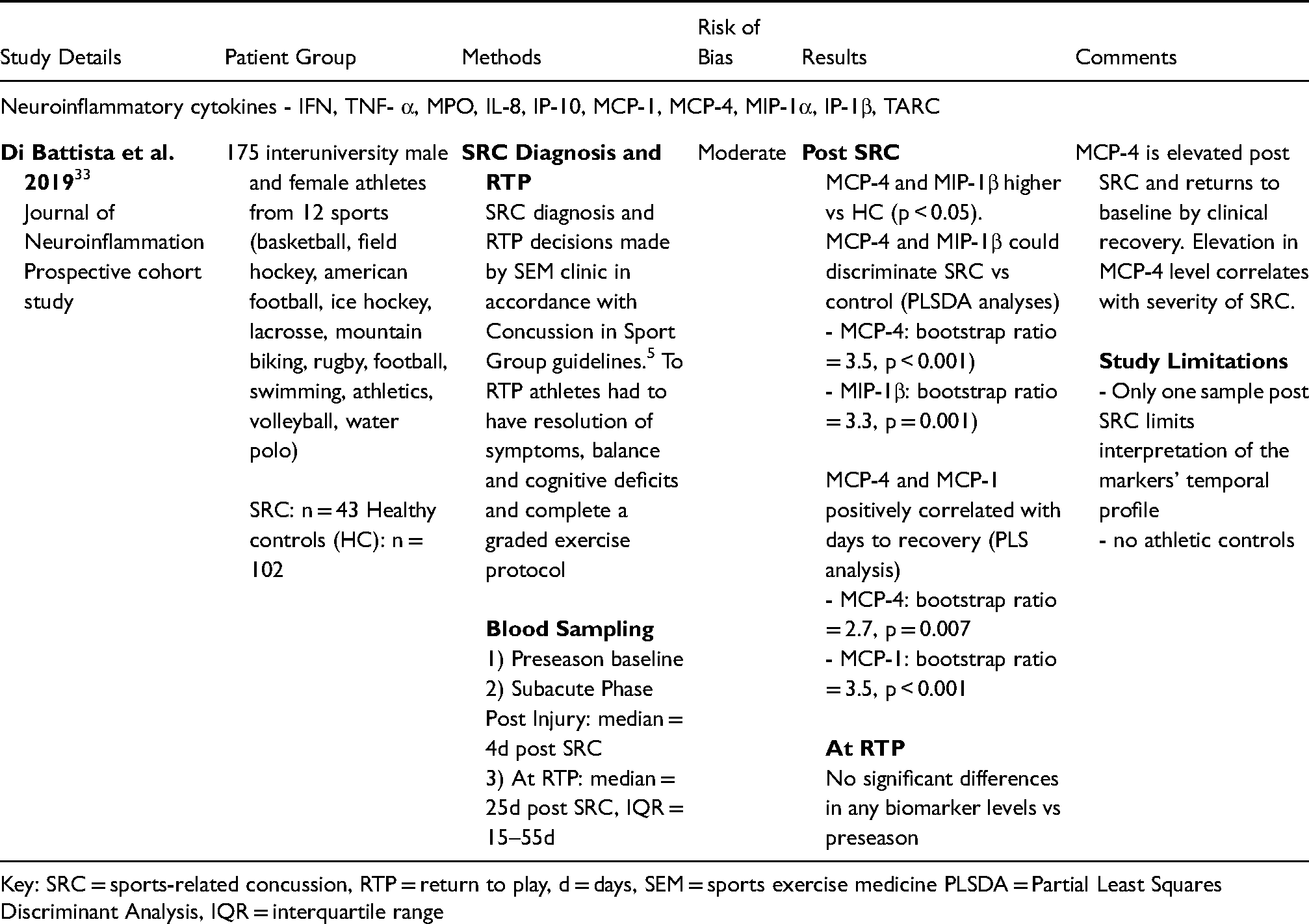
Key: SRC = sports-related concussion, RTP = return to play, d = days, SEM = sports exercise medicine PLSDA = Partial Least Squares Discriminant Analysis, IQR = interquartile range
Risk of bias assessments.

Biomarkers of axonal injury
Tau
McCrea and colleagues 22 evaluated a range of biomarkers including tau in a prospective cohort study of 1760 US collegiate athletes. 264 athletes were diagnosed with SRC in line with US Department of Defense guideline 22 using the Sport Concussion Assessment Tool 3 (SCAT-3) 23 and underwent biomarker sampling were taken at 1 h and 24–48 h post SRC, at RTP and at 7d post RTP. These were compared with preseason baseline levels, contact sport controls (CSC - matched for demographics as well as sport, position, years of participation and concussion history) and non-contact sport controls (NCSC - matched for exertional requirement and demographics). Tau was higher than preseason, CSC and NCSC at 1 h, lower than preseason or controls at 24–48 h and was no different to preseason or either control at RTP (Table 2). It showed no difference in athletes with loss of consciousness and post-traumatic amnesia (LOC-PTA), used as symptomatic markers of severe concussions.
Shahim et al. also assessed total tau 24 and tau fragments A and C 25 in a prospective cohort study of 288 professional Swedish ice hockey players between 2012–2015. They took total tau samples from 87 players at 1 h, 12 h, 36 h and 144 h following SRC and at RTP and compared biomarker levels to preseason baseline (n = 288), athletic controls (AC) (n = 12) and non-AC (n = 19). Athletes cleared for RTP had to be symptom free and complete a graduated RTP protocol with no symptoms at any stage in accordance with the latest Concussion in Sport Group guidelines. 5 49 players returned to play within 10d, 38 took longer than 10d and 7 had symptoms persisting for more than 1 year. Total tau at 1 h post SRC was higher than preseason (p = 0.05) and Healthy controls (HC) (p < 0.001) but not AC (p = 0.80) and correlated with scores on the Rivermead Post-Concussion Symptoms Questionnaire (RPQ). Tau levels then normalised by 12 h and was at baseline levels by RTP. Tau at 1 h could predict players who took longer than 10d to RTP (OR = 1.9, 95% CI 1.1–1.36, p = 0.032, AUC 0.67). A similar protocol was used for tau fragments using 28 cases of SRC. Tau C was increased in all time points vs preseason (p = 0.03) with tau A showing no change. Both normalised by RTP. Tau A at 1 h and 12 h could discriminate players with prolonged RTP > 10d (1 h: AUC 0.87, 95% CI 0.71–1, p = 0.01; 12 h: AUC 0.91, 95% CI 0.78–1, p = 0.005).
Gil et al. also investigated tau in 623 American university athletes between 2009 and 2014. 26 Samples were collected from 46 athletes at 6 h, 24 h, 72 h and 168 h post SRC diagnosed by the Sport Concussion Assessment Tool 2 (SCAT-2) 27 and compared with preseason baseline (n = 623), AC (n = 37) matched for sport, prior concussion history and demographics, and non-AC (n = 21) matched for demographics. Balance Error Scoring System (BESS) 28 + Immediate Post Concussion Assessment and Cognitive Testing (ImPACT) 29 were measured 7d post-concussion. RTP decisions were made by individual universities following national guidelines with athletes being asymptomatic at rest and at each point during a graduated RTP protocol. After SRC tau was higher at 6 h, 24 h, 72 h and 168 h vs non-AC (p < 0.01) but was lower vs AC at 24 h and 72 h (Table 2). Tau was higher in athletes with prolonged RTP (> 10d) compared with short RTP (< 10d) at 6 h (p < 0.01), 24 h (p < 0.01) and 72 h (p = 0.022) and could accurately predict athletes with prolonged recovery at 6 h and 72 h (Table 2). BESS and ImPACT scores showed no difference between long RTP and short RTP.
Overall, tau is raised acutely after SRC compared to preseason and well-matched AC, remains raised for 24–72 h and returns to baseline by RTP. Within this acute period tau shows good correlation with athletes with more severe concussions.
Neurofilament light (NFL)
In addition to tau, McCrea et al. also assessed NFL in their study of US collegiate athletes. They found that NFL was no different at any time point post SRC compared to preseason, CSC or NCSC. However, in athletes who suffered loss of consciousness or post-traumatic amnesia NFL increased over time and was higher at RTP and 7d compared to preseason and both controls (preseason: mean difference 0.498 pg/mL, 95% CI 0.295–0.701 pg/mL, p < 0.001; CSC: mean difference 0.481 pg/mL, 95% CI 0.271–0.692 pg/mL, p < 0.001; NCSC:mean difference 0.448 pg/mL 95% CI 0.228–0.668 pg/mL, p < 0.001).
Shahim et al. also investigated NFL, 24 finding that NFL was higher than preseason levels at 1 h, 12 h, 36 h and 144 h post SRC and higher than healthy and AC at 1 h. At 1 h NFL levels correlated with symptom severity scores on the RPQ (ρ = 0.41, p = 0.011). At RTP NFL levels remained raised vs preseason levels (p < 0.001). At all times post-SRC, NFL could separate players with RTP > 10d vs RTP < 10d (1 hr: OR = 8.8, 95% CI 3.0–36.0, p = 0.0006, AUC 0.82; 12 h: OR = 2.8 95% CI 1.3–7.3, p = 0.021, AUC 0.72; 36 h: OR = 3.0, 95% CI 1.4–7.8 p = 0.011, AUC 0.73; 144 h: OR = 3.30 95% CI 1.40–11.5, p = 0.025, AUC 0.73). NFL at 144 h post SRC could separate athletes with persistent PCS symptoms lasting over a year who had to retire from the game (AUC 0.89, p = 0.005).
These studies show NFL is raised after severe concussions, correlating with symptom severity scores and RTP duration. In these patients NFL does not return to baseline by clinical recovery - as measured consistently throughout being symptom and sign free on concussion assessment scales such as the SCAT 5.
α-II spectrin breakdown products
Siman et al. 30 investigated the potential of SNTF as a concussion biomarker in part of their cohort of professional ice hockey players. Serum SNTF levels at 12 h, 36 h and 144 h post SRC (n = 28) were higher than preseason, returning to baseline at RTP. SNTF at 36 h was a good predictor of prolonged RTP > 6d (AUC = 0.85; 95% CI 0.73–0.97). SNTF changes at 12–36 h post SRC correlated with tau changes at similar time points (R2 = 0.84, n = 24) and multivariate measures of tau and SNTF better correlated with RTP than tau alone but worse than SNTF alone. This study demonstrates SNTF is raised post SRC vs preseason levels, correlates with longer RTP at 36 h and returns to baseline by RTP.
Biomarkers of neuronal injury
Neuron specific enolase (NSE)
Shahim et al. 25 assessed NSE in their ice hockey study and found no change in NSE levels post SRC vs preseason baseline and no correlation with number of days taken to RTP.
Visinin-like protein-1 (VLP-1)
Shahim et al. 31 assessed serum VLP-1 in their ice hockey study and found no changes in VLP-1 post-concussion at 1 h, 12 h and 144 h compared with preseason levels, with a reduction at 36 h. There was no correlation between VLP-1 levels and RTP duration. Overall, these markers of neuronal injury do not show any change post SRC.
Ubiquitin carboxyl-terminal hydrolase L1 (UCH-L1)
McCrea et al. also assessed UCH-L1 in their US collegiate athlete study and found that UCH-L1 increased 1 h post SRC compared to preseason baselines and CSC and NCSC (preseason: mean difference 0.449 pg/mL, 95% CI 0.167–0.732 pg/mL, p < 0.001; CSC: mean difference 0.577 pg/mL, 95% CI 0.236–0.919 pg/mL, p < 0.001; NCSC: mean difference 0.463 pg/mL, 95% CI 0.088–0.839 pg/mL, p = 0.01). At the 24–48 h time point UCH-L1 was no different vs preseason or controls. At RTP UCH-L1 was lower vs preseason (mean difference − 0.321 pg/mL, 95% CI, − 0.546 to − 0.095 pg/mL, p < 0.001) and NCSC (mean difference − 0.373 pg/mL, 95% CI − 0.714 to − 0.032 pg/mL, p = 0.03) but no different to CSC. By 7d post RTP there were no differences. At neither points post RTP did UCH-L1 show any difference in athletes who experienced loss of consciousness or post-traumatic amnesia).
Biomarkers of astroglial injury
S100b
Shahim et al. also looked at S100B in their ice hockey study.24They found that S100B at 1 h post SRC was higher vs preseason samples (p < 0.001), as well as healthy (p < 0.001) and AC (p = 0.014). S100B at 1 h did not correlate with RPQ scores and could not discriminate between players with long or short RTP. At 12 h, 36 h and 144 h and RTP there was no difference between S100B post SRC and preseason samples.
Shahim and colleagues show S100B increases post SRC compared to athletic and non-AC and returns to baseline by RTP. S100B does not correlate with symptom severity or athletes with prolonged RTP.
Glial fibrillary acidic protein (GFAP)
Amongst their other biomarkers, McCrea and colleagues also investigated GFAP. They showed that GFAP is increased at 1 h post SRC and 24–48 h post SRC compared to preseason, CSC and NCSC (Table 4). At RTP and 7d post RTP GFAP was higher than preseason but no different to either athletic control group (Table 4). At RTP athletes who suffered loss of consciousness or post-traumatic amnesia had higher GFAP levels compared to those who didn’t and compared to both groups of AC (Table 4).
Biomarkers of pituitary dysfunction
Prolactin (PRL)
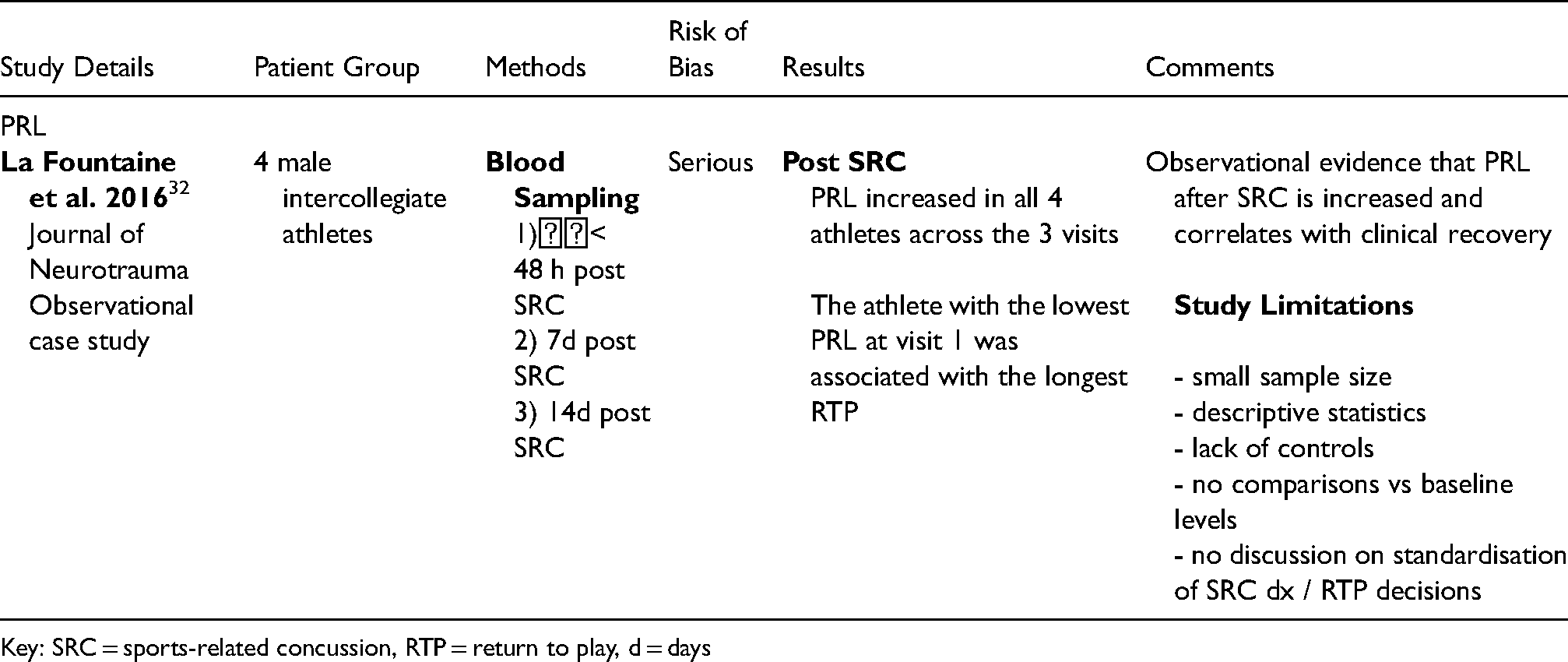
La Fountaine et al. 32 investigated PRL in 4 intercollegiate athletes who suffered SRC taking samples at 3 time-points post SRC (within 48 h, after 7d and after 14d). PRL levels increased across the 3 visits in all 4 athletes but the athlete with the lowest initial PRL level was associated with the longest RTP.
This study provides preliminary observational evidence that PRL levels may correlate with clinical recovery.
Biomarkers of neuroinflammation
Di Battista et al. assessed a range of inflammatory biomarkers following SRC in interuniversity athletes. 33 In athletes who suffered SRCs (n = 43) or MSK injuries (n = 30) they assessed a panel of cytokines (IFN-γ, TNF-α, MPO, IL-8, eotaxin, IP-10, MCP-1, MCP-4, MIP-1α, MIP-1β and TARC) at preseason, within 8d of injury and within 14d of RTP and compared these with HC (n = 102). SRC diagnosis and RTP decisions were made at a single sports medicine clinic in accordance with the most recent Concussion in Sport Group Guidelines. 5 Two biomarkers (MCP-4 and MIP-1β) were raised post SRC and could discriminate between SRC and healthy athletes (p < 0.001 and p = 0.001 respectively). These differences resolved by RTP. MCP-4 and MCP-1 correlated with days to RTP (p = 0.007 and p < 0.001). These changes were not seen in the MSK-injury patients. The inflammatory cytokine MCP-4 is elevated post SRC compared to HC, returns to baseline by RTP and correlates with length of recovery.
Discussion
A variety of concussion biomarkers taken from blood samples have been evaluated following SRC. Markers of axonal injury including tau, NFL and SNTF are consistently raised after SRC and show varying time courses of recovery post-concussion. In addition, they show a correlation with severity of concussion measured either by symptom severity or duration for RTP. Amongst other classes of biomarkers, the neuronal marker UCH-L1, astroglial proteins GFAP and S100B and the neuroinflammatory cytokine MCP-4 are also raised post SRC and return to baseline at RTP with GFAP and MCP-4 also showing a correlation with severity of injury. Other markers of neuronal injury such as NSE and VLP-1 are not affected by SRC and markers of pituitary dysfunction such as PRL have only been described in case studies. Our review identified no studies evaluating the utility of urine and saliva biomarkers for RTP following SRC which reflects their more relatively recent recognition as potentially useful concussion biomarkers. Identifying changes in microRNA expression in saliva has recently been recognised as a potential diagnostic marker for concussed athletes34,35 and similar changes are also seen in athletes following recurrent concussive episodes. 36 Exploration of the diagnostic and prognostic utility of microRNAs in saliva and urine for SRC is ongoing 37 and given the ease of access and non-invasiveness of these tests in comparison to blood sampling, the results are highly anticipated.
The current evidence base has a number of limitations. Only a small number of studies have correlated concussion biomarkers with clinical recovery each with relatively poor sample sizes. Study protocols varied in their sampling time-points, use of controls, clinical assessments of SRC and had a range of severities of concussion. In particular, the lack of widespread use of AC, particularly matched for contact history, limits inferences given these markers are influenced by physical activity and previous history of head trauma. The body of evidence is strongest for tau and NFL which are the only markers to be assessed in multiple cohorts with good sample sizes and consistent use of AC some with extensive matching for prior history of head injury. NSE, UCH-L1, GFAP and S100B have only been evaluated in single cohorts but on each occasion this did utilise a large cohort and AC. The evidence is weakest for SNTF, VLP-1 and inflammatory cytokines, coming from a single small sample with no AC whilst PRL has only been investigated in a small case series. Overall, these do reflect significant limitations in the evidence base and further higher-powered and standardised studies are needed to strengthen our knowledge.
Despite these limitations there is evidence to support further research into the potential use of biomarkers to assist clinicians to make RTP decisions. The present review identifies several biomarkers including tau, SNTF, UCH-L1, S100B, GFAP and MCP-4 that show deviations post SRC and return to baseline by clinical recovery from injury. Sampling these markers post-concussion and regularly throughout rehabilitation could provide valuable information on neuropathological recovery. In the future, alongside clinical recovery and successful completion of a graduated rehabilitation programme, improvement of a biomarker to baseline level could be included in criteria for RTP. Given neurophysiological and clinical recoveries may be distinct, this could mean an athlete who has clinically recovered from SRC could be precluded from RTP due to persisting biomarker abnormalities (eg Table 1). However, use of these markers in this way is not without its own risks. Currently the safety implications of these biomarkers are poorly understood. For example, in severe concussions NFL is raised at clearance for RTP suggesting some neurophysiological markers lag clinical recovery. It is unclear currently if returning to sport with incomplete neurophysiological recovery from SRC carries with it further neurological or non-neurological risks. Unnecessarily precluding athletes from returning to sport can be harmful for the individual athlete and team perspective and many sports currently operate mandatory lock out periods preventing return to training or match play for a period after injury. In athletes with normal clinical examinations biomarker data supporting full neurophysiological recovery could support a more individualised decision-making process that could prevent players being kept out of play longer than required (eg Table 1). Future work to establish the clinical significance and risk profiles of biomarkers is needed to allow their safe and effective use.
Several markers including tau, NFL, SNTF, GFAP and MCP-4 show correlations with severe concussions with more pronounced symptoms or longer RTP duration and tau was shown to outperform the commonly used clinical assessments (the BESS and ImPACT tools) in severe cases. 24 As such a secondary use for these biomarkers may be in the early detection of severe SRCs. This would help to highlight athletes whose injury may be more severe than indicated by clinical assessment alone and who need a longer and more closely observed rehabilitation period. To facilitate this further elucidation of the temporal and prognostic profiles of these biomarkers is needed to identify the most clinically useful and practical sampling time points post SRC.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.