Abstract
Objective
Prompt, accurate, objective assessment of concussion is crucial as delays can lead to increased short and long-term consequences. The purpose of this study was to derive an objective multimodal concussion index (CI) using EEG at its core, to identify concussion, and to assess change over time throughout recovery.
Methods
Male and female concussed (N = 232) and control (N = 206) subjects 13–25 years were enrolled at 12 US colleges and high schools. Evaluations occurred within 72 h of injury, 5 days post-injury, at return-to-play (RTP), 45 days after RTP (RTP + 45); and included EEG, neurocognitive performance, and standard concussion assessments. Concussed subjects had a witnessed head impact, were removed from play for ≥ 5 days using site guidelines, and were divided into those with RTP < 14 or ≥14 days. Part 1 describes the derivation and efficacy of the machine learning derived classifier as a marker of concussion. Part 2 describes significance of differences in CI between groups at each time point and within each group across time points.
Results
Sensitivity = 84.9%, specificity = 76.0%, and AUC = 0.89 were obtained on a test Hold-Out group representing 20% of the total dataset. EEG features reflecting connectivity between brain regions contributed most to the CI. CI was stable over time in controls. Significant differences in CI between controls and concussed subjects were found at time of injury, with no significant differences at RTP and RTP + 45. Within the concussed, differences in rate of recovery were seen.
Conclusions
The CI was shown to have high accuracy as a marker of likelihood of concussion. Stability of CI in controls supports reliable interpretation of CI change in concussed subjects. Objective identification of the presence of concussion and assessment of readiness to return to normal activity can be aided by use of the CI, a rapidly obtained, point of care assessment tool.
Keywords
Introduction
The Center for Disease Control (CDC) estimates that mild Traumatic Brain Injury (mTBI/concussion) accounts for 1.5 million Emergency Department (ED) visits annually in the United States, with an additional 1.6–3.8 million concussions occurring in sports and recreational activities annually 1 (among which, 1.1–1.9 million concussions occur in children under 18 2 ). These figures are likely to be significant underestimates since it has been reported that a large percentage of patients with mild head injury/concussion may not seek medical care.2–6 Despite the tendency to under-report, ED visits for mild head injury have significantly increased, while overall visits to the ED remained relatively unchanged, 7 likely due to increased media attention raising awareness of the short and long-term effects of TBI and its sequelae. Children/adolescents and young adults are particularly vulnerable as significant brain development continues throughout these years. The lack of, or delay, in concussion diagnosis and/or lack of appropriate concussion management have been shown to lead to much slower recovery from concussion,8,9 and may affect academic/cognitive and emotional functioning.10–12 Sustaining a concussive injury before age 25 has been shown to be associated with impaired adult functioning, psychiatric disorders, low education level, welfare, and disability. 13
Although there is currently no “gold standard” for the diagnosis and management of concussion, numerous consensus and position statements emphasize the importance of multimodal assessments of concussion, including: self-reported symptoms, motor control, mental status testing, and neurocognitive testing.14–18 Likewise, numerous societal bodies (e.g. the American Academy of Neurology (AAN), the National Athletic Trainers’ Association (NATA)) advocate a multidimensional approach to the management of SRC consisting of computerized neurocognitive testing (CNT), assessment of postural stability (balance), vestibular and ocular assessment, and self-reported symptoms. 19 In addition, the literature supporting multimodal assessments for the evaluation of mTBI/concussion has pointed to the limitations of most current, largely subjective assessment tools14,19,20
An extensive literature demonstrates that changes in brain electrical activity occur in mTBI, as reflected in measures of connectivity (disruption in relationships between brain regions), changes in complexity of the signal (disorganization of neural networks), and changes in the frequency spectra (changes in oxygen utilization, glucose metabolism and neurochemistry)21–24 Researchers have reported extremely high accuracy in the objective identification of traumatic structural brain injury (bleed of 1 cc or greater) 25 and brain function impairment (concussion)26,27 using quantitative EEG (qEEG) as input to classifier algorithms built using machine learning (ML) methods, 28 in patients seen within 72 h of closed head injury (i.e. blunt or non-penetrating head trauma that does not include a break in the skull), with Glasgow Coma Scale (GCS) 13–15. Following a similar approach, Part 1 of this paper describes the derivation and performance of a quantitative, multimodal, objective Concussion Index (CI), obtained using machine learning, which includes EEG at its core as well as neurocognitive performance measures and selected symptoms. The CI is summarized into a single multivariate index. Part 2 examines clinical implications of the CI as an objective tool to aid in the assessment of concussion from time of injury through the recovery phase, and the relationship between the CI and severity as reflected in rapid or protracted recovery and total symptom burden.
Part 1: Derivation of the concussion index algorithm
This section describes in detail the Algorithm Development study for the derivation of the Concussion Index (CI). It should be emphasized that the aim of this work was to derive such an index from a set of features which can potentially lead to a rapid, accurate assessment of concussion in a hand-held device, at the point of care.
Methods
Patient population
Data was collected from August 2015 to January 2019 using BrainScope a investigational devices (investigators were blinded to output) at 12 college and high school sites that recruited from 60 locations across the US, with approval from primary sites’ Institutional Review Boards (IRBs). b The study included concussed subjects and controls (matched primarily for age, sex and type of sport played), as well as healthy volunteers, all between the ages of 13 and 25, who met inclusion/exclusion criteria. Injured subjects were assessed within 72 h of time of injury (Day 0), 5 days after injury (Day 5), at their clinicalsitedetermined eturn-o-lay date (RTP), and 45 days after RTP (RTP + 45). Each matched control subject was assessed at time intervals corresponding to those of their injured counterpart. In addition, in order to expand the controls, a group of non-injured healthy controls were recruited from the same population as the injured and matched controls, but were only tested at pre-season. All subjects signed written informed consent and for minors recruited to participate, parental written informed consent and adolescent assent was also obtained.
Inclusion criteria for “concussed”: Males and females between the ages of 13 and 25 having sustained a traumatic closed head injury within 72 h (3 days) from time of injury to time of BrainScope assessment, with a GCS 29 score ≥ 13 and no hospital admission due to either head injury or collateral injuries for more than 24 h.
Inclusion criteria for “controls”: Males and females between the ages of 13 and 25, GCS of 15 at time of assessment and no history of concussion or TBI within the past year, or Motor Vehicle Accident (MVA) requiring Emergency Department visit within the last year.
Exclusion criteria for all subjects: Subjects were excluded who had forehead, scalp or skull abnormalities or whose clinical condition, such as head trauma, would not allow placement of the electrodes; current Central Nervous System (CNS) active prescription medications taken daily (with the exception of medications being taken for Attention Deficit Disorder (ADD) or Attention Deficit Hyperactivity Disorder (ADHD); history of brain surgery or neurological disease; pregnant women; acute intoxication or evidence of illicit drug usage; previous enrollment in BrainScope studies; loss of consciousness (LOC) of 20 min or more related to the concussion injury; active fever defined as greater than 100°F or 37.78°C at time of assessment; evidence of abnormality visible on CT of the head related to the traumatic event; and patients who do not speak or read English.
Additional exclusion criteria for “controls”: Any focal neurological signs including aphasia, apraxia, diplopia, facial droop, dysarthria/slurred speech.
With regard to factors such as drugs or alcohol, fatigue, pain, and other factors which may be co-morbid with head injury, the rule followed in this investigation was to not exclude them from any subject groups (controls and head-injured patients), except as defined by exclusion criteria. By doing this, they are eliminated as differentiating factors between groups, so that features that might be sensitive to these factors do not impact the ML algorithm development task.
Definition of concussion (“Clinical Truth”) and RTP: In this study, concussed subjects were defined as those who had a witnessed head impact and who, by local site guidelines, were removed from play for 5 or more days (as subjects who return to play within 4 days of injury are not likely to have been concussed). The use of site guidelines assured broader applicability of the results to the general population of interest. RTP determination for each subject (reported as the number of days from injury date to “cleared to play” date) was made in accordance with site RTP protocols. All the sites used a gradual/graded RTP protocol at the end of which a subject was cleared to play. Furthermore, for all the colleges this protocol conformed to NCAA policy guidelines. 30 These guidelines included the following steps once the subject was declared symptom-free: (1) Light aerobic exercise; (2) Sport-specific activity with no head impact; (3) Non-contact sport drills and resumption of progressive resistance training; (4) Unrestricted training; (5) Return to competition. If at any point the student/athlete became symptomatic, they were returned to the previous stage.
Subject subgroups: For the purpose of further assessing performance of the CI, subjects were subdivided into the following subgroups: (1) Category 1: Controls (CNT) which included non-injured subjects who were functionally asymptomatic; (2) Category 2 (Cx2) injured subjects with RTP between 5 and 13 days (“normal/rapid recovery” group); and Category 3 (Cx3) which included injured subjects with RTP of 14 days or more (“protracted recovery” group). It is noted that McCrory et al. reported that 80% of concussed subjects recover within 14 days. 14 The age, sex and race distributions of the sample were determined by the representation of each in the populations served by the participating sites involved.
Clinical assessments
All study subjects were evaluated with the following symptombased scales or assessment tools: Subsections of the Sports Concussion Assessment Tool – 3rd Edition (SCAT-3 31 ) or 5th Edition (SCAT-5), including: GCS (Section 1), Symptom Evaluation (SCAT-3 Section 3 and SCAT-5 Section 2, which assesses presence and severity of 22 common concussion symptoms on a Likert scale (symptom severity self-rated from 0–6 per item, with a total score range of 0–132), Standard Assessment of Concussion (SAC, SCAT-3 Sections 4 and 8, SCAT-5 Sections 3 and 5): a brief cognitive screening tool which includes subtests of orientation, immediate memory, concentration, and delayed recall (total score in range 0–30). 32 Subjects in both control and injured groups were queried regarding history of head injury and concussion.
Section 2 of the SCAT contains the Concussion Symptom Inventory 33 (CSI), the standard tool used for the quantitative assessment of presence and self-reported severity of concussion symptoms (“symptom burden”) at time of injury and for monitoring recovery from SRC. The total CSI score is constructed by adding clinical scores for the 22 symptoms (“CSI22”) queried, where the severity of each symptom is self-reported (by the patient) on a Likert (0–6) scale, with zero indicating absence of symptom. In this study, as frequently done in the literature, the total score on the 22-item CSI was used as an estimate of the total symptom burden.33,34
Neurocognitive performance tests
Two of the neurocognitive performance tests which have been demonstrated in the literature to be affected by concussion35,36 were included in the data reported on in this study: Simple Reaction Time (SRT; evaluates simple motor speed, information processing speed and attention), and Procedural Reaction Time (PRO; evaluates higher-order rapid responding, including visuomotor reaction time and simple decision making). While additional cognitive performance tests were included in the neurocognitive test battery for a portion of the population (Match to Sample and Go/No-Go), they were not included in the final analyses and are not reported on herein, as preliminary analyses revealed a high level of correlation between the tasks. Moreover, as inputs to the ML algorithms, only the “throughput” feature values of SRT and PRO were used, which are composite scores which jointly reflect both speed of execution and accuracy of the subject’s response. The raw neurocognitive features were age- and sex-normed and z-transformed to standard scores using a large normative database of subjects aged 13–80 collected on the device. This accounts for any effects that might be due to sex or age. These features have been shown to be both valid for use in concussion and reliable.37–40
EEG data acquisition
BrainScope handheld investigational devices c were used to collect 10 min of EEG data acquired under an eyes closed resting condition.25,26 A technician observed the subjects throughout data acquisition for vigilance and alerted them if necessary, for example if they appeared drowsy. The EEG data was recorded from the limited frontal electrode montage including Fp1, Fp2, F7, F8, AFz, A1 and A2 locations of the expanded International 10–20 Electrode Placement System, and was re-referenced to linked ears. The EEG data was acquired at a sampling rate of 1 kHz and all electrode impedances were below 10 kΩ. Amplifiers had a band-pass filter from 0.3 to 250 Hz (3 dB points) and EEG data was downsampled to 100 Hz for feature extraction.
EEG data processing and quantitative EEG feature extraction
The EEG signals were processed using BrainScope’s on device real-time suite of algorithms for artifact detection, 41 which identify for removal any physiologic and non-physiologic contamination (e.g. including lateral and horizontal eye movement, muscle activity), assuring quality of the EEG data. Only artifact-free data (1–2 min) was submitted to all further analyses. A large set of quantitative EEG (qEEG) features were then computed and z-transformed relative to age expected normal values, effectively removing any potential effect of age on these features. These steps are described in detail elsewhere.27,41 The feature sets characterizing the EEG signal include measures of power (absolute and relative), mean frequency, measures of connectivity (asymmetry, coherence, phase lag, phase synchrony), measures of complexity (fractal dimension and scale-free activity), and information theory based features (entropy), across and within frequency bands. The resulting database of qEEG feature z-scores served as inputs to the derivation of the classifiers.
Multimodal classifier development
All extracted EEG features were subjected to an informed data reduction step 41 as a first level of feature selection. In Machine Learning, overtraining is known to lead to poor performance on independent populations and this step reduces this risk. The resulting reduced feature set became the candidate feature set for classifier building.
The multimodality of the input features was represented by three broad types of quantitative features: electrophysiological (qEEG) feature z-scores, neurocognitive throughput feature z-scores, and selected clinical sign/symptom features. Internal and external cross-validation paradigms were used, involving multiple train & test splits of the dataset, in order to further avoid overtraining when building linear discriminant function classifiers that separate the two main classes (Control vs. Concussed), and to maximize the probability of validation in prospective independent data (previously unseen). The final classifier features and weights were derived from a training set consisting of 80% of the total dataset, while its performance was tested and reported on the remaining 20% (the Hold-Out dataset).
Classifier development involved searching the reduced dimensionality multimodal feature space using advanced ML-based techniques (e.g. Genetic Algorithms and LASSO Logistic Regression methods) in order to obtain classifier candidates that could optimally separate the two categories while avoiding overtraining. This methodology was described in detail previously. 27 The metrics used for evaluating classifier performance during training were the area under the curve (AUC) of the Receiver Operating Characteristic (ROC) as well as sensitivity and specificity of the classifiers at their operating point.
Statistical analysis
Demographics of the study population, including prevalence of typical concussion symptoms in the Control and Injured groups will be described. CI performance will be assessed as sensitivity and specificity of the classification algorithm at Day 0 on the independent Test and Hold-Out datasets. In addition, sensitivity will also be assessed for two distinct groups of concussed subjects. The area nder the ROC urve (AUC) will also be computed as a measure of accuracy of the classifier function.
Results
Subject demographics and symptoms prevalence at time of injury
The study population at Day 0 (time of injury) consisted of 232 Injured subjects (67.2% males, median age = 19.0 (13.8–25.0)) and 206 controls (66% males, median age = 19.0 (13.7–24.6)). Additionally, 85 non-injured healthy controls (51.8% male, median age = 17.8 (13.2–21.4)) at pre-season baseline were enrolled. The Cx group included the two subtypes described in the Patient population section above, with 93 normal recovery subjects (Cx2), and 139 protracted recovery subjects (Cx3) based on the RTP cut-off (14 days).
Table 1 shows differences in prevalence at intake for several clinical symptoms of concussion for the concussed compared with controls. As expected, concussed subjects were characterized by a significantly higher incidence of these symptoms. The median RTP for the Cx group was 15 days (range 5–70), which is consistent with the results reported by Kerr et al. in a sample of high school and college American football athletes. 42
Prevalence of common concussion signs/symptoms (Sx) at Day 0 in controls/CNT vs injured/Cx are shown in the first two rows of the Table.
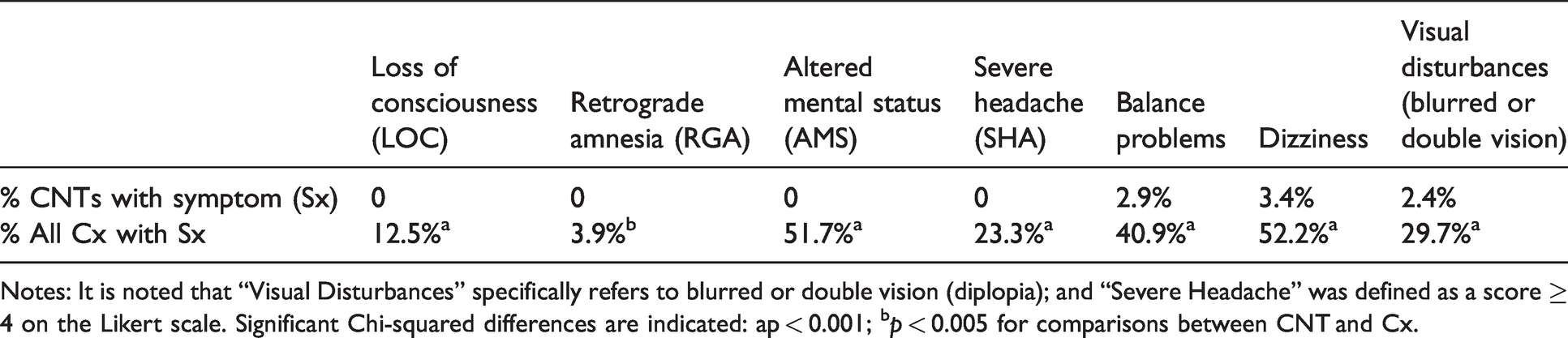
Notes: It is noted that “Visual Disturbances” specifically refers to blurred or double vision (diplopia); and “Severe Headache” was defined as a score ≥ 4 on the Likert scale. Significant Chi-squared differences are indicated: ap < 0.001; bp < 0.005 for comparisons between CNT and Cx.
Classifier performance at time of injury
Results reported in this section are based on the estimate of performance on multiple unseen datasets (Test and Hold Out), referred to as “external cross-validation.” Table 2 summarizes performance (sensitivity, specificity, and rea nder the ROC urve) of the linear classifier algorithms in an external cross-validation context (average sensitivity/specificity on the test portions of 10 train/test splits), with tandard eviation and 95% confidence intervals: sensitivity = 84.9% (95% CI: 81.6–88.2%)/specificity = 76% (95% CI: 71.7–80.3%); as well as sensitivity/specificity of the final/“All In” classifier on the unseen Hold Out group (N = 105; CNTs: N = 58; Cx: N = 47): sensitivity/specificity = 91.5%/67.2%. In addition, this table shows the average sensitivity obtained on the two injured sub-groups (“Prolonged RTP”/Cx3, “Rapid RTP”/Cx2). On the Hold Out data, the Negative Predictive Value (NPV) was 90.7% and the Positive Predictive value (PPV) was 69.4%. It is important to note that the final algorithm built using the “All In” dataset (i.e. entire dataset minus a random Hold Out group representing close to 20% of the data) was locked prior to its prospective independent validation described in Bazarian et al., 2020. 43
Summary performance table for the classifiers developed on the 10 Train/Test splits (external cross-validation) and for the “All In” classifier (performance on independent 20% Hold-Out dataset (HO)).
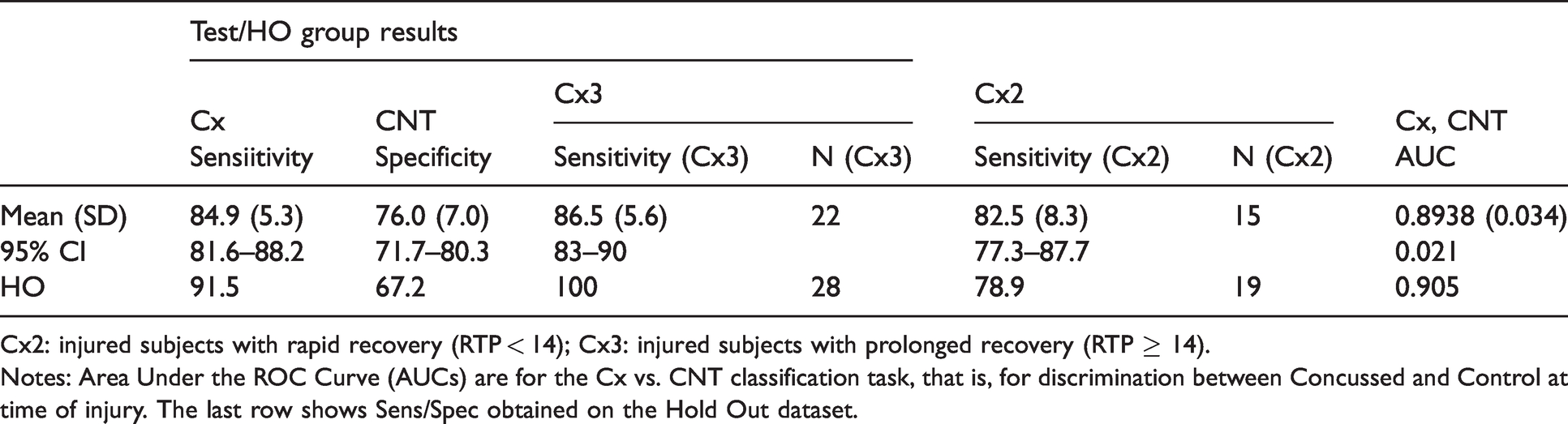
Cx2: injured subjects with rapid recovery (RTP < 14); Cx3: injured subjects with prolonged recovery (RTP ≥ 14).
Notes: Area Under the ROC Curve (AUCs) are for the Cx vs. CNT classification task, that is, for discrimination between Concussed and Control at time of injury. The last row shows Sens/Spec obtained on the Hold Out dataset.
To aid in interpretability, the weighted sum output y of the linear discriminant function classifier was mapped to a continuous 0–100 range using the scaled logistic mapping (“inverted S curve”) given in equation (1) which produces a mapped classification threshold of 70 for subjects classified in the “positive class” (i.e. disease present/concussed) if CI ≤ 70.
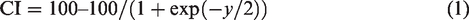
Four of the top-five contributors to the classification were qEEG features. It is also noted that the top 5 contributors to separation between the groups did not include neurocognitive features. The qEEG features contributing most to classification were those measuring connectivity between brain regions, including: phase, coherence, and power asymmetry/gradient features, especially in frequency bands that included alpha, beta and total power. It is of note that all but one of the qEEG features in the algorithm were from the connectivity measures, suggesting reflection of white matter tract integrity. 44
Part 2: Demonstration of CI as an objective tool to aid in assessment of concussion
This section of the paper examines clinical implications of the CI as an objective tool to aid in assessment of concussion from time of injury through the recovery phase, and demonstrates the relationship between the CI and severity as reflected in rapid or protracted recovery and total symptom burden.
Statistical analyses
Applying the CI derived in Part 1, this section of the manuscript demonstrates the clinical application of the CI to track recovery. Since the CI was derived from Time of Injury data only, the data presented in this section including Day 5 and RTP can be considered independent applications of the CI.
Statistical analyses
The change in CI values over time for controls and concussed subjects was evaluated within each group using the non-parametric Wilcoxon rank tests between time points. Results were also graphically displayed as boxplots for each group and each time point. The correlation between change in symptom burden and change in CI as reflected at the Day 5 intermediate time point, was computed for the full study population.
Results
Analysis of longitudinal change in the CI
Box plots showing the distribution of the CI at baseline/pre-season (when available), Day 0, and at subsequent evaluation time points (Day 5, RTP, RTP + 45, post-season), for the controls and the injured subjects are shown in Figure 1. Importantly, since the CI was developed using Day 0 data only, CI values for controls and injured at Day 5 and at the other follow-up time points can be considered partially independent test results. At baseline and all subsequent measurements, the controls have a median CI which is above threshold (CI ∼75) and a lower (first) quartile which is above the binary classification threshold (CI = 70). At Day 0, the injured Cx groups (Cx2 + Cx3 combined) have a median CI which is well below the threshold and which can be seen to gradually rise to a level which is on par with that of the control group at the same time point at RTP and RTP + 45. In addition, it can be seen that at Day 0 those injured subjects with “normal recovery” (rapid RTP) have a higher median CI than those with protracted recovery (prolonged RTP), suggesting that the CI is reflective of severity of concussion. The CI at Day 5 also shows very different patterns of recovery in the two subgroups of injured subjects, with the median CI of the normal recovery group at threshold, while approximately 75% of the protracted recovery group is still below threshold.
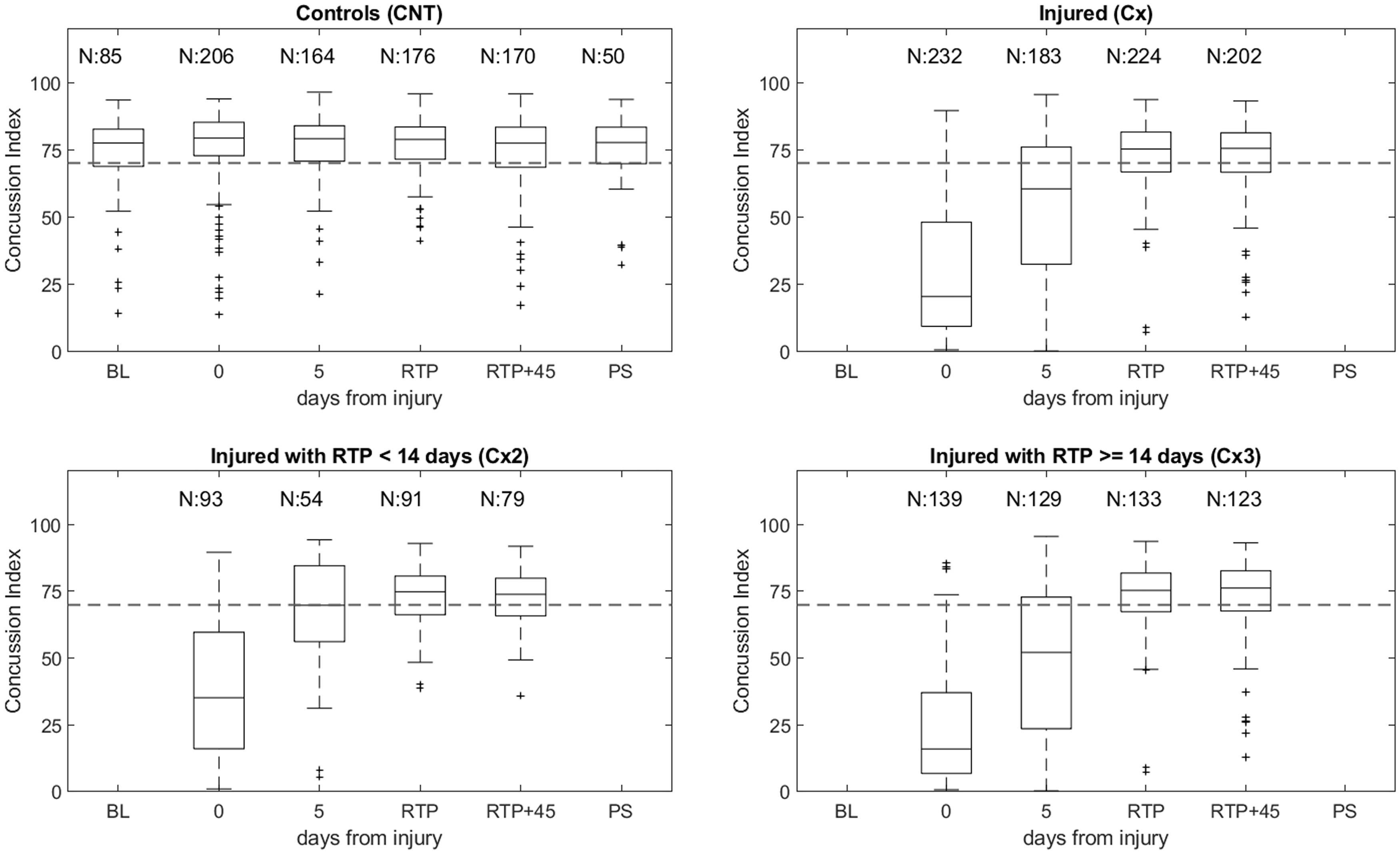
Boxplot of Concussion Index (CI) for controls and injured groups for the full algorithm development dataset. Top-left: Controls, top-right: All injured subjects, bottom-left: Injured subjects with 5 ≤ RTP <14 days (Cx2), and bottom-right: Injured subjects with RTP ≥ 14 days (Cx3).
The significance of the differences between the CI scores of the controls and the injured, were assessed at each time point using the Wilcoxon rank test. The first-order statistics of the CI for Controls and Injured subjects at the various time points are reported in Table 3. The significance levels (p-values, with Bonferroni correction for multiple tests) are reported in Table 4 for the Hold-Out (Testing) Algorithm Development dataset. At Day 0 (day of injury) the CI of the injured was significantly lower than that of the controls (HO: p < 0.001) and this significance was present when considering the Cx2 and Cx3 groups separately. At Day 5, the CI of the injured was still significantly lower than that of the controls (HO: p < 0.001). At RTP, the CI of the concussed group was no longer significantly lower than that of the controls (p = 1).
Statistics of CI for control and injured subjects at Day 0 (time of injury), Day 5, RTP, RTP + 45 for the Hold-Out dataset.
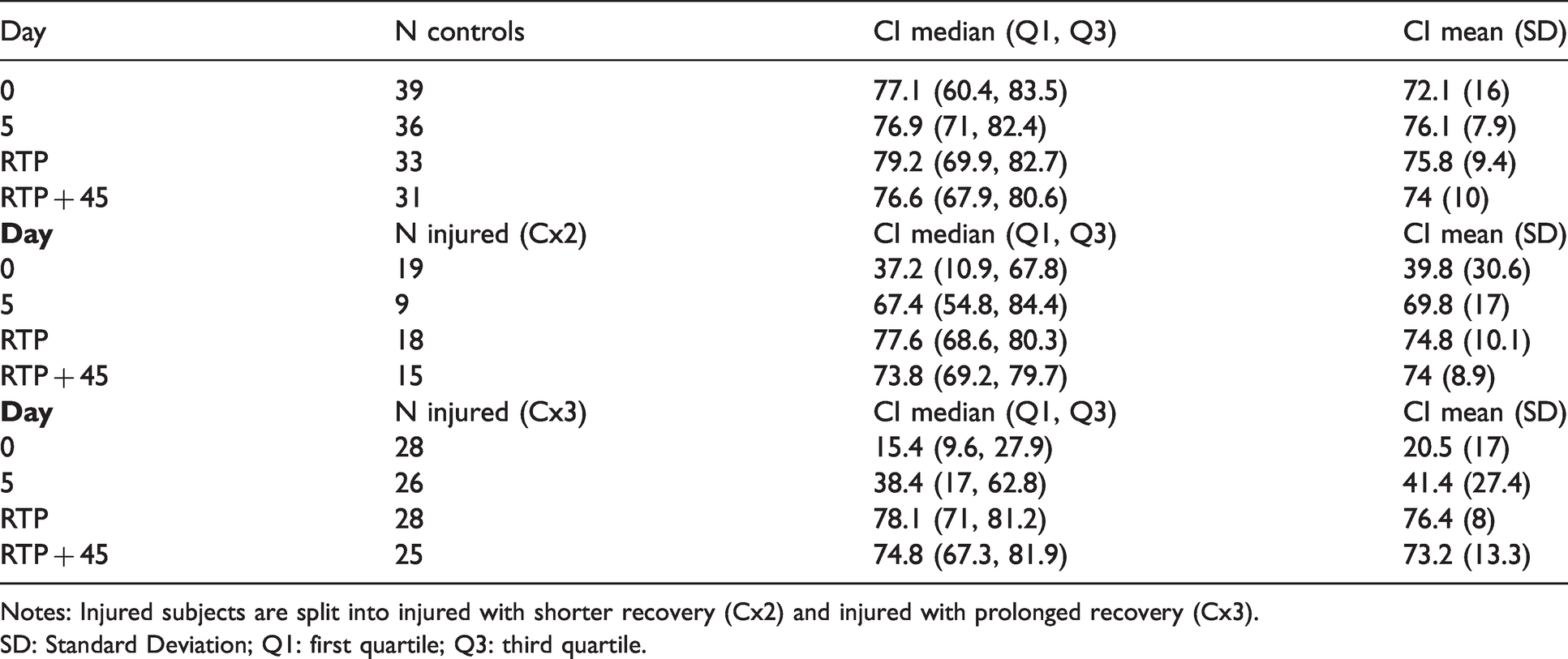
Notes: Injured subjects are split into injured with shorter recovery (Cx2) and injured with prolonged recovery (Cx3).
SD: Standard Deviation; Q1: first quartile; Q3: third quartile.
Number of control and injured subjects at Day 0 (time of injury), Day 5, RTP, RTP + 45 for the Hold-Out dataset and p-values for the significance of the differences in the CI between controls and injured (all injured (Cx = Cx2 + Cx3), and separately for controls vs. injured with normal recovery (Cx2), controls vs. injured with protracted recovery (Cx3)).
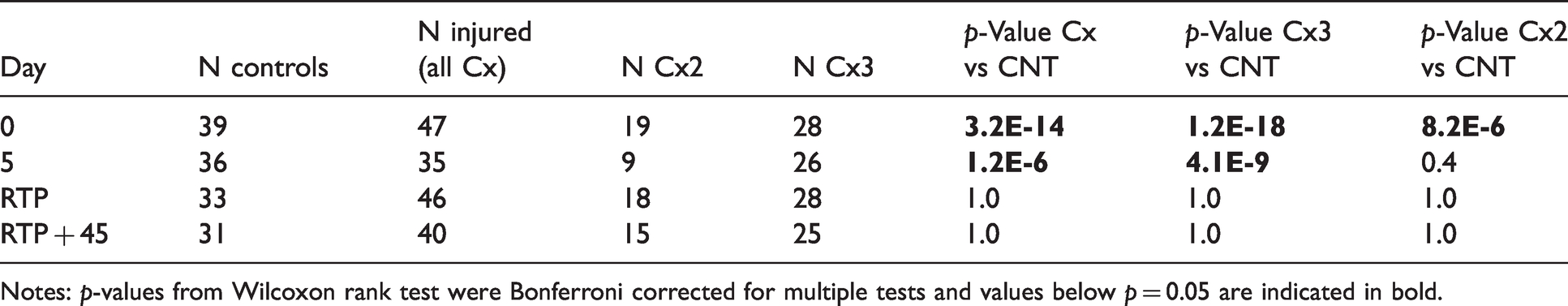
Notes: p-values from Wilcoxon rank test were Bonferroni corrected for multiple tests and values below p = 0.05 are indicated in bold.
In order to validly interpret change over time in the concussed population, it was important to demonstrate stability of the CI in the controls over the same time period. As can be seen in Table 5, there were no significant differences between CI scores of the controls at Day 0 and at any of the subsequent time points (Day 5, RTP, or RTP + 45), which demonstrates stability of the CI over time.
Number of control subjects at Day 0, Day 5, RTP, RTP + 45 and p-values for the Wilcoxon rank test for a median comparing CI scores at Day 0 vs. CI scores at subsequent days (Day 5, RTP, RTP + 45).
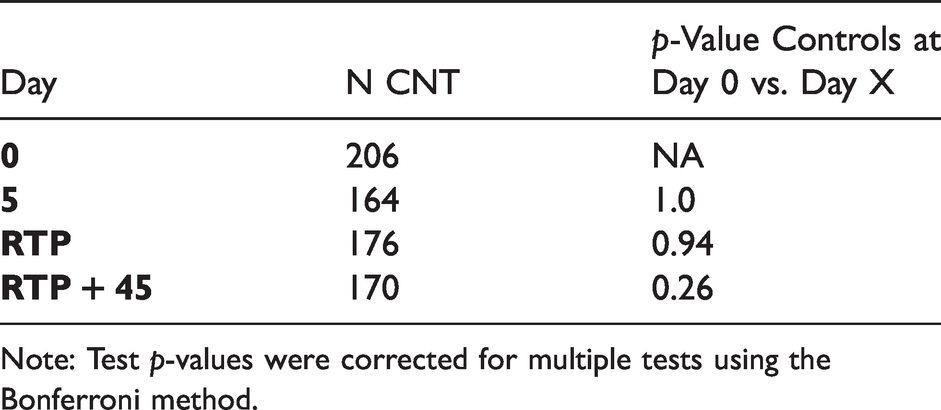
Note: Test p-values were corrected for multiple tests using the Bonferroni method.
Correlation between CI and clinical “symptom burden” during recovery (Day 5)
The different patterns of recovery of the normal and prolonged recovery groups, as noted above, suggests a relationship between CI and clinical symptom severity as measured at the intermediate time point. To further explore this relationship, the correlation between quantitative measures of clinical symptom burden (measured as total CSI score) and the CI at Day 5 was evaluated at this independent intermediate time point. Although not a measure of recovery, an association between the two metrics would support a clinical manifestation of change reported by CI.
Figure 2 shows the relationship between CI and CSI at Day 5 (this time point was used to demonstrate relationship at an intermediate time point during the recovery phase) for four different subgroups of concussed subjects based on time to RTP (RTP < 14, 14 ≤ RTP < 21, 21 ≤ RTP < 28, RTP ≥ 28). It should be noted that the Pearson correlation between CI at Day 5 and CSI was highly significant (r = –0.7, p = 5.5E-31), as were the correlations between those same features when restricting to any of the four subgroups (with respective values of: r = –0.72, p < 0.001; r = –0.65, p < 0.001; r = –0.48, p < 0.01; r = –0.79, p < 0.001). Further, the figure shows that injured subjects with low CI scores tend to have high CSI scores (top left quadrant of the figure), and that most of the subjects with RTP <14 days showed this relationship. On the other hand, subjects with a relatively high CSI at Day 5 (CSI ≥ 20) had lower CI scores, most falling below CI threshold (<70) and that most of those with RTP >14 days showed this relationship.
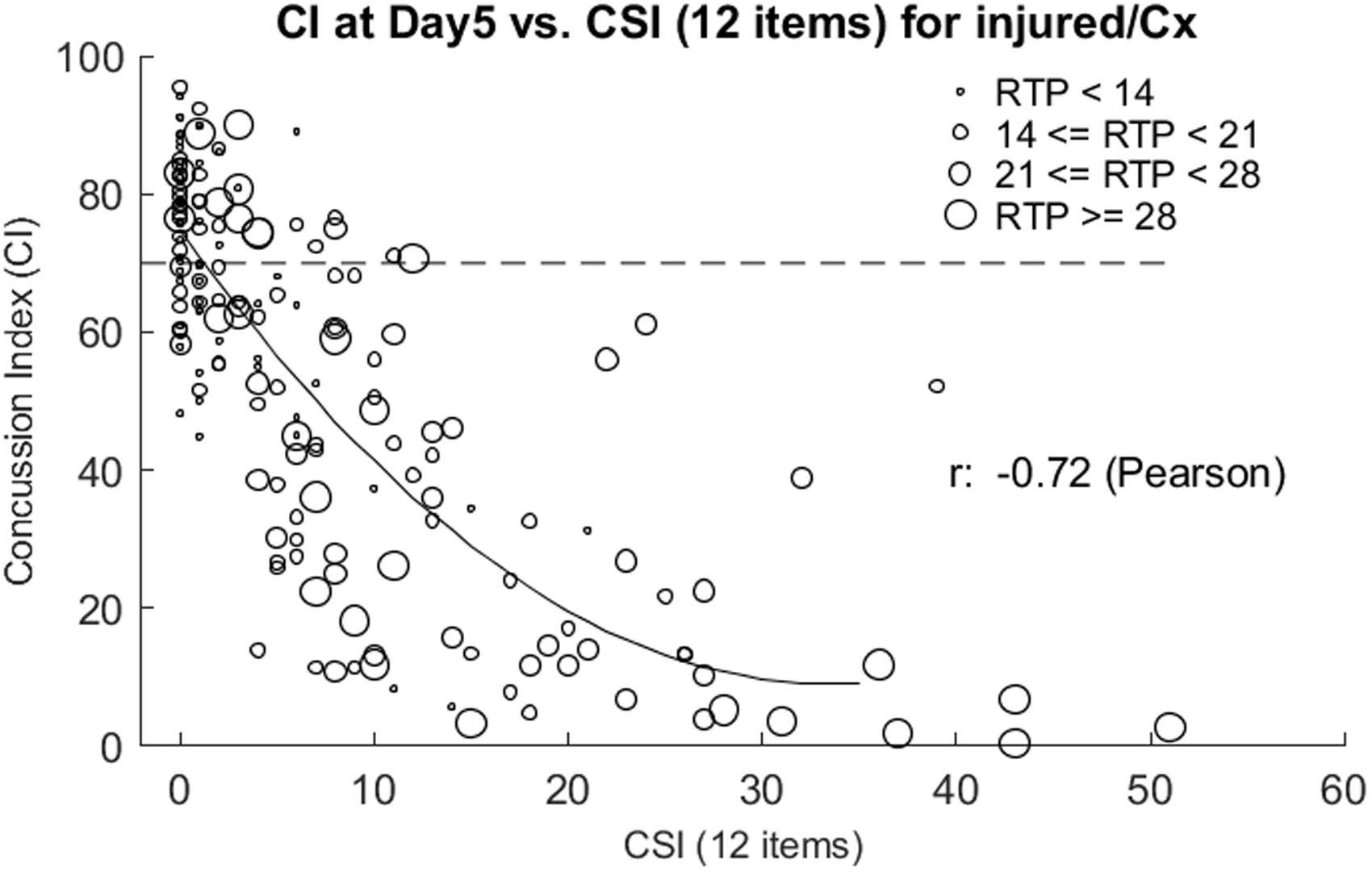
Scattergram of Concussion Index (CI) at Day 5 versus CSI (22 items), with second-order polynomial fit, for injured subjects within the full dataset. Note: circle size from smallest to largest represents increasing RTP.
Discussion
The need for objective identification of the presence of concussion and assessment of readiness to return to normal activity represents an important health care need addressed in Part 1 of this study which describes the derivation of the Concussion Index in subjects 13–25 years old. The CI is a multimodal, multivariate index with EEG at its core, integrating quantitative information about brain function impairment in concussion. Features which reflect changes in connectivity between brain regions contribute most to the index. Results of this study demonstrate high sensitivity, specificity and accuracy (AUC) of the CI in identifying the likelihood of concussion following head injury (within 72 h). Importantly, common comorbidities (e.g. ADHD, LD, headache, etc.) are not excluded from the head-injured or non-head-injured groups, such that separation between the groups are not influenced by the presence of such factors.
Although advanced neuroimaging can be used to distinguish between groups with concussive brain injury and controls, 45 such technologies are not readily available either at the sidelines, in urgent care centers, concussion clinics, or in Emergency Departments. However, studies have demonstrated significant correlation between disturbances in white matter integrity seen on DTI and the CI studied in the same subjects. These results suggest that the CI d can act as a surrogate for other neuroimaging tools, providing many advantages in sideline testing (in locker room or nearby venue) at the time of injury. 44
Part 2 of the paper demonstrates that progressive recovery was reflected in significant changes observed on the median CI values between time of injury and Day 5. While overall the CI of the injured athletes was significantly lower than that of the controls, the CI also points to the heterogeneity within the concussed population, showing different rates of recovery at Day 5 (an independent time point not used to derive the CI algorithm). At Day 5, clear differences were seen between the median CI of those with normal and those with protracted recovery. In fact, while ∼75% of the protracted group were still below concussion threshold at Day 5, ∼50% of those in the normal recovery group were already above threshold. In addition, at clinically determined RTP (largely based on self-report of symptom persistence), there were subjects whose CI was still below threshold, indicating that some subjects may have been returned to play prior to resolution of brain function abnormalities. Similar findings have been reported in the literature attesting to the persistence of brain function abnormalities in sport-related concussions beyond the point when clinical symptoms have resolved.46–50
Further, the demonstration that concussed subjects show no significant differences from the controls at RTP + 45 suggests that CI scores for injured subjects at RTP + 45 have returned to the point where they fall within the baseline range of a non-injured population, suggesting that brain function impairment was resolved. The reliability of such an interpretation is supported by the demonstrations of the lack of significant differences in the controls between Day 0 (treated as their baseline measurement) and RTP + 45, in conjunction with the lack of significant differences between controls and injured at RTP + 45. These findings taken together suggest that RTP + 45 in the concussed population can be considered as their “baseline,” and supports the demonstration of a “return to baseline” for the concussed subject at RTP.
It is of note that the presence and severity of concussion symptoms (“symptom burden”) as reflected in the total CSI score was significantly correlated with CI throughout the period of observation, from time of injury through RTP. While highly correlated, the CI is an objective measure which is less reliant on self-report, and less subject to under- or over-reporting or learning effects over time, adding confidence to interpretation of change over time.
Limitations of this study include the lack of other intermediate time points between 72 h of injury and RTP. Future studies should perform serial evaluations through the recovery phase to better estimate the recovery rates of individuals and the relationship to changes in CI. Another limitation is the restricted age range for which this study was performed (ages 13–25), although these ages are critical for the high school and college age student athletes Studies are currently underway to address this age limitation. In the context of the aim of constructing an objective assessment tool that could be rapidly applied on the handheld device, the inputs to the algorithm were selected with this in mind. Future studies will evaluate the contribution of other features, including oculomotor measures which are being investigated in an ongoing study.
Despite the fact that ADHD medication was not an exclusion criterion, only one injured and one control subject in our study were taking such a medication. It was therefore not possible to study the potential impact of ADHD medication on the accuracy of the CI for this subject group. Most importantly, the need for an independent validation of the CI in a prospective population is critical to validate the index. Such study has been successfully conducted 43 and was presented in support of BrainScope’s FDA clearance for the Concussion Index (September 2019).
Conclusions
In Gioia, 2015, 51 it is stated that “ultimately, the practice of concussion assessment and management will benefit from an evidence-based medicine approach […] whereby clinicians have access to statistical bases for evaluating a patient’s scores relative to those seen in subjects with known concussions versus non-injured subjects.” The Concussion Index, a multivariate, multimodal, objective, quantitative index of brain function impairment directly addresses these needs. Once independently validated, the CI can be used as an adjunct to other clinical assessment tools in the evaluation of concussion, with the potential to allow tracking of concussion recovery. The ability to obtain such an index rapidly, in a hand held device, at the point of care, and at any point along the recovery trajectory, potentially represents significant added value as an adjunct to current clinical practice to determine concussion and track recovery in a more confident and objective way.
Footnotes
Acknowledgments
The authors acknowledge the contributions of those who made this research possible, including research staff at all the clinical sites and the athletes who volunteered for the study. In addition, the contributions to the study of Dr. R. Lopez (University of South Florida, Tampa, FL), Dr. A. Meyer (University of New Mexico, Albuquerque, NM), Dr. C. Neville (SUNY Upstate Medical University) and Dr. K. Podell (Rice University, Houston, TX) are acknowledged.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr. Jacquin was employed by BrainScope Company at the time this study was conducted, but is no longer an employee. Dr. Prichep is employed by BrainScope, and is an inventor on patents licensed by BrainScope from NYU School of Medicine. All other authors were Principal Investigators at the clinical data acquisition sites participating in the Research, whose institutions received contracts to support subject recruitment, consenting and data acquisition and have no competing financial interests to disclaim.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The clinical study was funded in part by a contract to BrainScope Company Inc. from the U.S. Navy (Naval Health Research Center), contract #W911QY-14-C-0098. Disclaimer: The views, opinions and/or findings contained in this report are those of the author(s) and should not be construed as an official Department of the Navy position, policy or decision unless so designated by other documentation.
