Abstract
Establishing a diagnosis of concussion within the context of competitive sport is frequently difficult due to the heterogeneity of presentation. Over the years, many endogenous proteins, including the recent Food and Drug Administration approved for mild-to-moderate traumatic brain injury, glial fibrillary acid protein and ubiquitin carboxy-terminal hydrolase, have been studied as potential biomarkers for the diagnosis of mild traumatic brain injury. Recently, a new class of potential biomarkers, the microRNAs, has shown promise as indicators of traumatic brain injury. In this pilot study, we have analysed the ability of pre-validated serum microRNAs (mi-425-5p and miR-502) to diagnose concussion, in cases without structural pathology. Their performance has been assessed alongside a set of identified protein biomarkers for traumatic brain injury in cohort of 41 concussed athletes. Athletes with a confirmed concussion underwent blood sampling after 48 h from concussion along with magnetic resonance imaging. Serum mi-425-5p and miR-502 were analysed by quantitative reverse transcription polymerase chain reaction, and digital immunoassay was used to determine serum concentrations of ubiquitin carboxy-terminal hydrolase, glial fibrillary acid protein, neurofilament light and Tau. Results were matched with 15 healthy volunteers. No structural/haemorrhagic pathology was identified. Protein biomarkers demonstrated variability among groups reflecting previous performance in the literature. Neurofilament light was the only marker to positively correlate with symptoms reported and SCAT5 scores. Despite the sub optimal timing of sampling beyond the optimal window for many of the protein biomarkers measured, miR-502 was significantly downregulated at all time points within a week form concussion ictus, showing a diagnostic sensitivity in cases beyond 48 h and without structural pathology.
Introduction
Concussion or mild traumatic brain injury (mTBI) represents a complex biochemical and physiological pathology triggered by the absorption of mechanical forces by the brain.1–5 Understanding the significance and consequences of mTBI inside and outside of competitive (particularly contact) sport has evolved markedly in recent years. It is now accepted that recurrent or multiple mTBIs represent a significant risk to the health of an individual in terms of direct risk to life6,7 and potential long-term neurodegenerative illness. 8
The presentation of mTBI is often subtle, and frequently affected individuals do not meet diagnostic criteria from computerised tomographic scanning (CT) of the brain.2,9
The vast majority of people who sustain single or multiple concussions in sports such as rugby, hockey, soccer, American football, lacrosse and boxing show no gross anatomical or structural pathology on CT or magnetic resonance imaging (MRI). 10 The diagnosis or exclusion of concussion is therefore markedly more complicated than simply determining if an individual requires a CT scan of the brain to exclude anatomical/haemorrhagic pathology.
Within sport, the recognition of this has led to the development and implementation of activity-specific guidelines 11 and research into more effective methods of diagnosing concussion where its occurrence may not be clear or forthcoming.
Currently, mainstream assessment of mTBI both in sports and in the primary/secondary healthcare setting involves the functional and symptomatic assessment of an individual using neurocognitive tests such as the immediate post-concussion assessment and cognitive testing, King–Devick concussion screening test and other similar tools. 12 These tests have significant limitations, particularly without baseline/premorbid measurement in terms of sensitivity/specificity13,14 and are also susceptible to multiple confounding factors such as musculoskeletal injury or pre-morbid disability.
In order to improve objectivity in the diagnosis of concussion, a number of approaches have been established such as multi-modal magnetic resonance imaging (MRI) featuring diffusion and spectroscopic examination;10,15–17 however, there are considerable practical implications with this approach along with a degree of debate as to the sensitivity of this technology in excluding injury. 15
Circulating biomarkers are also an area of growing interest, as they have the potential to provide a practical objective measure of brain injury burden. Recent FDA approval has been granted to the measurement of the serum peptides glial fibrillary acid protein (GFAP) and ubiquitin carboxy-terminal hydrolase (UCH-L1) for use in mTBI;18,19 specifically, the approval was regarding the use of negative UCH-L1-GFAP assay results to rule out intracranial lesions within 12 h of head injury. GFAP represents a cytoskeletal intermediate protein that plays a critical role in the blood–brain barrier and cell signalling. 20 It has been shown to be highly correlated with TBI; however, it is not found exclusively in the central nervous system (CNS), 21 and raised levels have been shown not to be sufficiently specific to confirm or exclude TBI in the context of polytrauma. 22 UCH-L1, however, is a deubiquitylating enzyme expressed by neuronal cells exclusively and has been proposed as a more specific biomarker of TBI.23,24
Current evidence suggests that these biomarkers are potentially sensitive enough to exclude significant anatomical injury/brain haemorrhage. This information mitigates the need for a CT scan 19 and potentially provides quantitative information regarding the size of injury burden and its prognosis. 22 However, it is reasonable to suggest that they have yet to demonstrate sufficient sensitivity and specificity to positively exclude mTBI entirely in circumstances when national guidance does not indicate the requirement for CT imaging, 25 or when structural pathology has been excluded by imaging yet concussion is still suspected.
Beside UCHL1 and GFAP, other circulating proteins have been identified as potential biomarkers of TBI, and among them, neurofilament light (NF-L) and Tau protein. NF-L is a CNS-enriched heteropolymeric component of the axonal cytoskeleton predominantly expressed in the long myelinated subcortical axons. 26 In the context of TBI, for example, elevated levels of NF-L within the CSF of amateur boxers correlated strongly with the severity of brain injury sustained during bouts.27,28 Recent publications have also demonstrated the ability of variations in serum NF-L concentrations to assess the severity of neuronal injury following severe TBI if measured between 12 and 144 h. 29
Tau is another protein that has demonstrated utility as a TBI biomarker. It is primarily localized in the axonal compartment of neurons and is involved in maintaining the integrity of neuronal micro tubules. Recently, developed assays were able to identify elevated Tau concentrations in the plasma of boxers even in the absence of symptoms of concussion. 30 The biodynamics of plasma Tau in these cases demonstrated a clear biphasic release between 1 and 36 h 31 after TBI.
Serum microRNAs (miRNAs or miRs) are a potential next generation of biomarkers. They are non-coding RNA molecules involved in the modulation and regulation of gene expression, 32 and the majority are intracellular; however, a number are found within multiple bodily fluids including the blood stream. They are implemented in a variety of pathologies and have demonstrated utility as biomarkers in the specific diagnosis of mTBI.33–40 Previously, the authors demonstrated two miRNAs (miR-425-5p and miR-502) that are significantly downregulated (p < 0.05) in mTBI patients if compared to healthy volunteers (HV) and at very early time points. 34 For this reason, these biomarkers were chosen in this study to be validated in a cohort of professional contact sports athletes who have sustained a concussive injury without structural traumatic pathology as identified by MRI and to be compared to the most commonly utilised protein biomarkers used to help the management of TBI.
Materials and methods
Study approval
Study participants were recruited through the Surgical Reconstruction and Microbiology Research Centre (SRMRC), based at Queen Elizabeth Hospital of Birmingham (UK), as part of the The REpetitive COncussion in Sport (ReCoS) (Ethics Ref. 11-0429AP28). Written informed consent was received from participants prior to inclusion in the study.
Recruitment
Professional and semi-professional contact sport athletes aged 16–40 years diagnosed with concussion by their respective enhanced care settings but not requiring hospital admission were referred to the study via the SRMRC research on call services. Concussion would be diagnosed if competing individuals had sustained an impact assessed by the observing clinical team (attending doctor or qualified physiotherapist, directly or via video reply) as representing a mTBI and unsatisfactory performance in sports specific concussion assessment tests (Rugby Football Union Head Injury Assessment (HIA), 41 SCAT-5. 42 The HIA assessment is a nationally adopted HIA protocol devised by the English rugby football union. It comprises a clear three-stage schematic assessment protocol for cases of suspected concussion for both the adult and under 19-year-old game.
Specific inclusion criteria were:
A diagnosis of concussion confirmed by the observing sports specific enhanced care team; Documented mechanism of injury; No requirement for immediate assessment or transfer to secondary care; Capacity to give informed consent.
Specific exclusion criteria were:
When the severity of injury on assessment indicated the requirement for a CT scan. A history of neurodegenerative or neoplastic pathology including Parkinson’s disease, multiple sclerosis, hydrocephalus, epilepsy, primary or secondary brain tumours. But not limited to these conditions. Past cranial neurosurgical procedures, including in-situ cerebra-spinal fluid diversion devices (shunts). Any contra indication to MRI examination including claustrophobia. Any contra indication to venipuncture (blood draw) including needle phobia. Previous concussion within three months of presentation.
A target/optimal recruitment window (for assessment) of 48–72 h was initially discussed with all participating contact sports institutions; however, these institutions were not directly involved in the execution of the study, and research personnel was not in direct contact at all times with them. Facilities and expertise in sample acquisition were not available at the respective clubs, and therefore sampling was undertaken at first contact with the ReCoS research team-delegated members of each enhanced care team (club staff) would contact the research team directly when a recruitable participant was available.
Imaging
MRI acquisition was performed on either one of the two dedicated 3 T MR scanners within the SRMRC institutional complex depending on availability. These are Philips Achieva 3.0 T (32 channel SENSE coil) and Siemens Magnetom Skyra 3.0 T (32 channel SENSE coil). Structural imaging sequences were acquired, specifically T1-weighted magnetisation prepared rapid gradient echo (base resolution 526 matrix pixels, 176 slice slab, repetition time 1660 ms, echo time 2.4 ms, field of view 240 mm) and 2 D axial T2-weighted sequences (base resolution 512 matrix pixels, 27 slice slab, repetition time 6000 ms, echo time 100 ms, field of view 250 mm). Susceptibility weighted imaging (a sequence particularly sensitive to blood products within the field of MR acquisition) was also obtained in order to determine with a greater degree of sensitivity if any haemorrhagic pathology was related to the mTBI (base resolution 256 matrix pixels, 64 slice slab, repetition time 27 ms, echo time 20 ms, field of view 214 mm). All images underwent specialist radiological review, for consistency, the same consultant neuro-radiologist reviewed all the images. These sequences are selected to provide a realistic analogy to a plain brain CT scan that would be undertaken in the emergency care situation (but without the radiation exposure).
Sample collection and processing
Serum samples were obtained anywhere between 2 and 30 days postconcussion ictus. The selection of this time frame was formulated specifically due to the practicalities of recruitment and assessment of participants from a broad regional network. This extends beyond the specified window of utility for GFAP and UCH-L1 by the FDA; however, within the secondary care environment (e.g. emergency rooms and specialist head injury clinics), biomarkers that have usefulness beyond the first 12 h after injury (as blood samples at these early time points are frequently not available) and outside the context of structural pathology (imaging may frequently be negative) may provide a valuable aid to diagnosis.
The blood samples were processed for serum isolation within 2 h after they were taken. Whole blood was left to stand for approximately 30' at room temperature before being centrifuged at 3000 r/min for 10' at 4°C. Serum was divided into aliquots and stored at –80°C for different periods before the analysis and ranging from 0 to 36 months.
Simoa Human Neurology 4-plex A assay
The Simoa Human Neurology 4-plex A assay (Quanterix, Lexington, MA, USA. Product code: 10215), a two-step digital immunoassay, 43 was used to quantitatively determine the levels of UCH-L1, GFAP, NF-L and Tau, and in serum. Although this assay allows the measurement of two of the FDA-approved proteins, this is not the specific assay licenced by the FDA;18,19 hence the results must be considered within this context. The sample duplicate measurements were performed on the fully automated Simoa HD-1 analyser (Quanterix, Lexington, MA, USA) in the Immunoassay Biomarker Core Laboratory, University of Dundee. This is a magnetic bead-based digital ELISA that allows detection of proteins at subfemtomolar concentrations. 44 For UCH-L1, GFAP, NF-L and Tau assay, the lower limit of quantification was 0.467, 0.241, 0.053 and 5.450 pg/ml, respectively, and the lower limit of detection (LLOD) was 0.221, 0.104, 0.024 and 1.740 pg/ml, respectively. Any samples yielding a signal over the quantification or calibrator range were diluted and re-assayed. Any samples yielding a signal below the LLOD were excluded from the analysis. Number of samples assessed for each assay is reported in Table 2. Average intra-assay duplicate coefficient of variation for the samples was 25.3, 5.3, 13.5 and 16.8 for UCH-L1, GFAP, NF-L and Tau, respectively.
RNA isolation and miRNA analysis
MiRNAs were extracted from 400 μl of serum samples by using Qiagen miRNeasy mini kit (Qiagen, GmbH, Hilden, Germany), according to Qiagen supplementary protocol for purification of small RNAs from serum and plasma and finally eluted in 30 μl volume of RNase free water. The concentration and purity of the resulting RNA were determined with an ND-1000 UV–vis spectrophotometer (NanoDrop). Serum RNAs (20 ng) were retrotranscribed and pre-amplified, using the single TaqMan assay (AppliedBiosystems, Life Technologies™) according to the manufacturer’s instructions, and RT-qPCR analysis was performed in a Bio-Rad iQ5 real-time PCR detection system (Bio-Rad, CA, USA). Expression fold changes were calculated according to the conventional 2
Statistical analysis
The data was checked for normal distribution characteristics and transformed into natural logarithm to perform parametric tests. Comparisons across the groups at each time and within the groups over time were performed by the one-way analysis of variance and Tukey’s post-hoc test on transformed data.
In addition, a receiver operating characteristic (ROC) analysis was employed. This is a statistical method to assess the diagnostic accuracy of each biomarker at specific time points in diagnosing concussion, expressed as area under the curve (AUC). The ROC curve is a graphical display of the trade-offs of the true-positive rate (sensitivity) and false-positive rate (1-specificity) corresponding to all possible binary tests that can be formed from this continuous biomarker. Each classification rule, or cut-off level, generates a point on the graph. The closer the curve follows the left-hand border and then the top-border of the ROC space, the more accurate the test. 46
Pearson correlations were also analysed between the levels of biomarkers and SCAT5 scores and symptoms. All analyses were carried on SPSS v.22 (IBM).
Results
Samples recruitment
A total of 41 (40 M/1F) concussed athletes were recruited for this study. Serum was collected at a variety of time points from concussion. In particular, serum was collected at day 2, day 3, days 4–7 and days 8–30 from concussion in a total of 9, 14, 10 and 8 injured athletes, respectively. All patients were symptomatic at the time of assessment. Serum samples were also collected from a total of 15 age-matched HV. The demographic information regarding both populations and mechanism of injury is summarised below (Table 1, Panel A and Panel B). The vast majority (n = 38) of players referred were rugby players; however, a number of other sports were represented (soccer n = 2, ice hockey n = 1).
(a) Summary of participants, (b) Mechanism of injury sustained.
aHead of concussed player sustained contact with limb or trunk of other player.
bHeavy tackle or diffused contact/fall sustained without clear history of head contact.
cPost, stick or floor contact with head.
Imaging
All participants underwent MRI scanning, and no traumatic structural pathology was reported. SWI imaging did not highlight an evidence of haemorrhagic pathology. One individual was found to have sustained an Anderson–Alonzo type 1 odontoid peg (C2) fracture; however, no accompanying intra cranial pathology was identified, and appropriate fat suppression images were not obtained at the time to determine fracture age and relationship to the concussive event (this case was referred to local tertiary services for further management and data retained for the study). All heathy individuals underwent identical imaging protocols and no evidence of any traumatic/haemorrhagic pathology. One individual was found to have a small incidental cavernous haemangioma, and data from this individual was retained as this was accepted by the authors to represent a true random incidental factor reflective of any normally sampled population.
UCH-L1, GFAP, NF-L and Tau
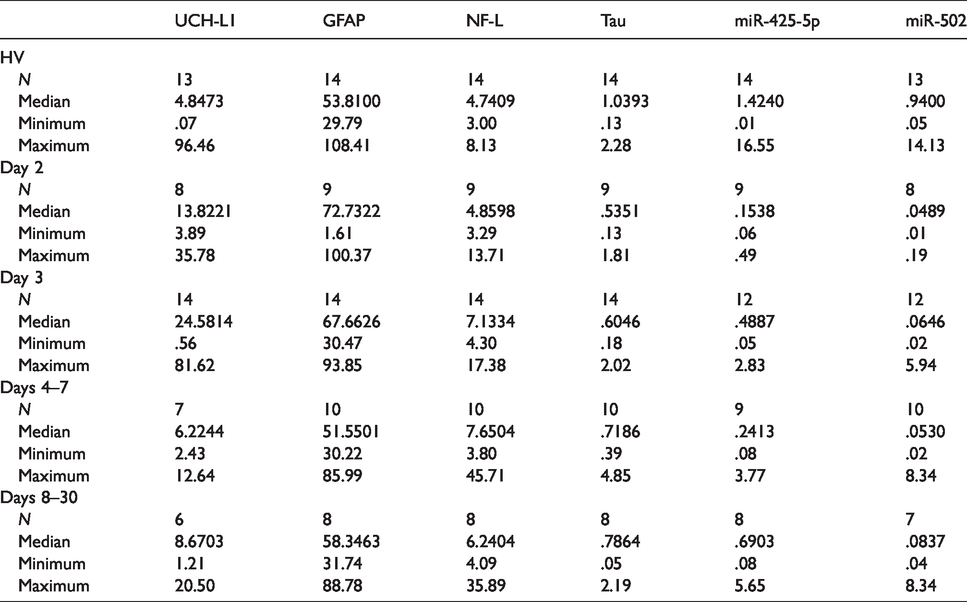
UCH-L1, GFAP, NF-L and Tau and concentrations were measured in serum of 41 concussed athletes and compared to 15 HV. The levels of these proteins did not show significant variations among the groups and at different time points. Median and interquartile ranges are reported in Table 2.
Descriptive statistics.
MiR-425-5p and miR-502 expression
The expression level of miR-425-5p and miR-502 was measured by RT-qPCR in concussed athletes and again compared to 15 HV. Specifically, miR-502 showed a significant downregulation at day 2 (p = 0.002), day 3 (p = 0.018) and day 4–7 (p = 0.025) when compared to HV (Figure 1). MiR-425-5p did not show any significant results among the time points and compared to the HVs.
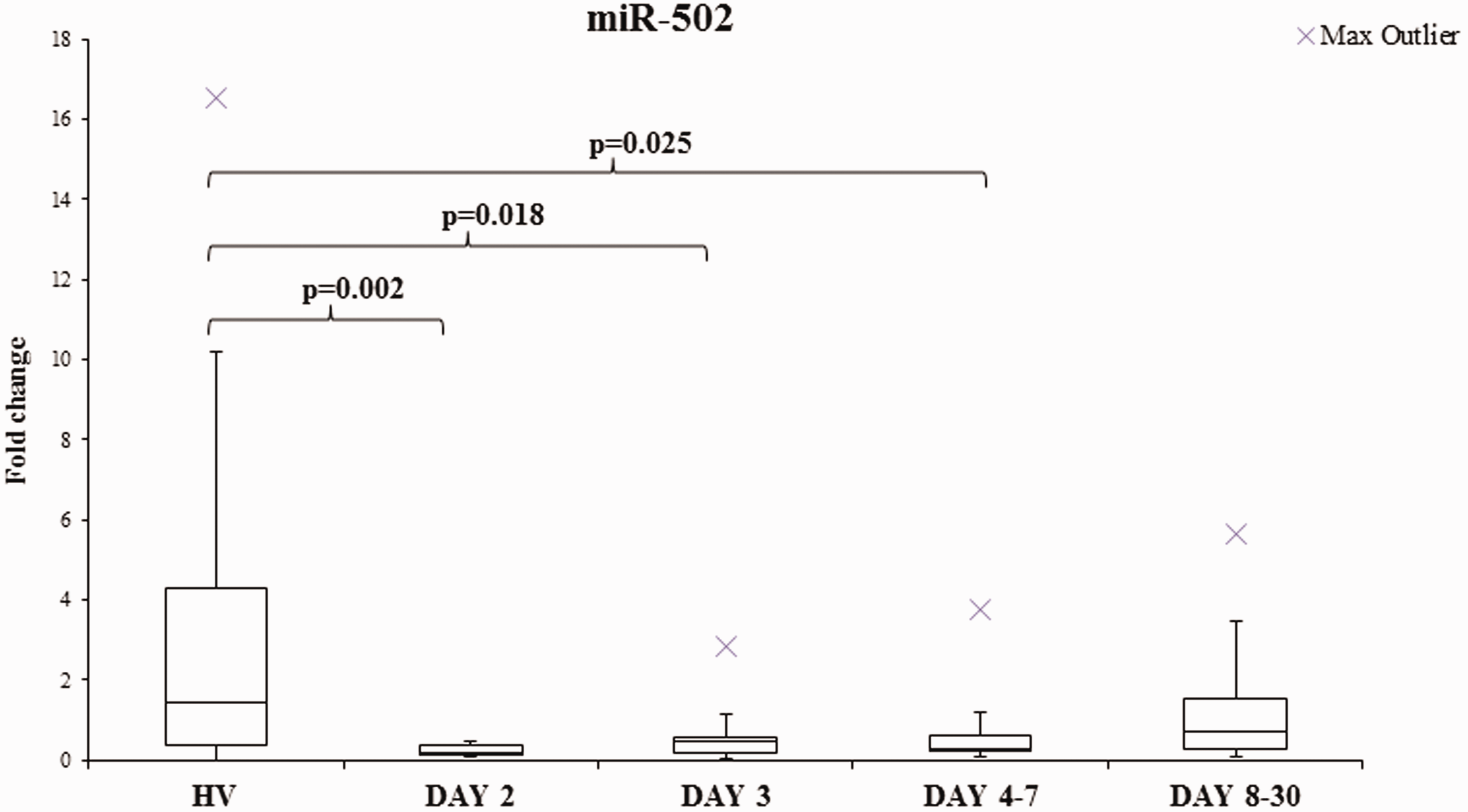
Boxplots of the fold changes of miR-502 at different time points and compared to HV. Specifically, miR-502 showed a significant downregulation at day 2 (p = 0.002), day 3 (p = 0.018) and day 4–7 (p = 0.025) when compared to HV.
ROC analysis
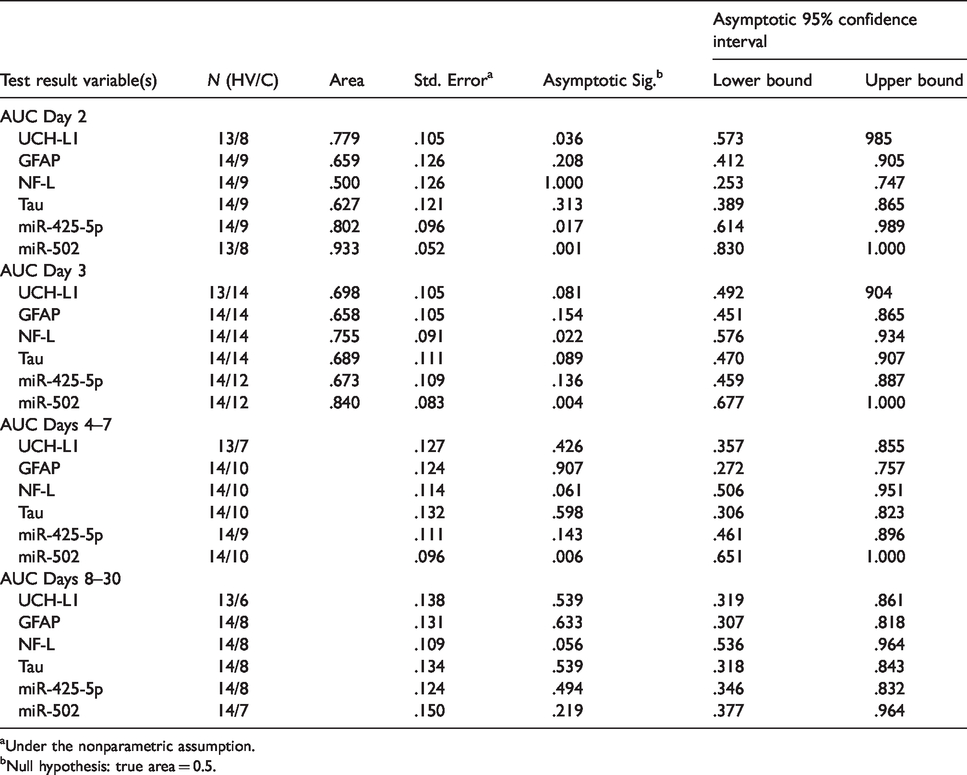
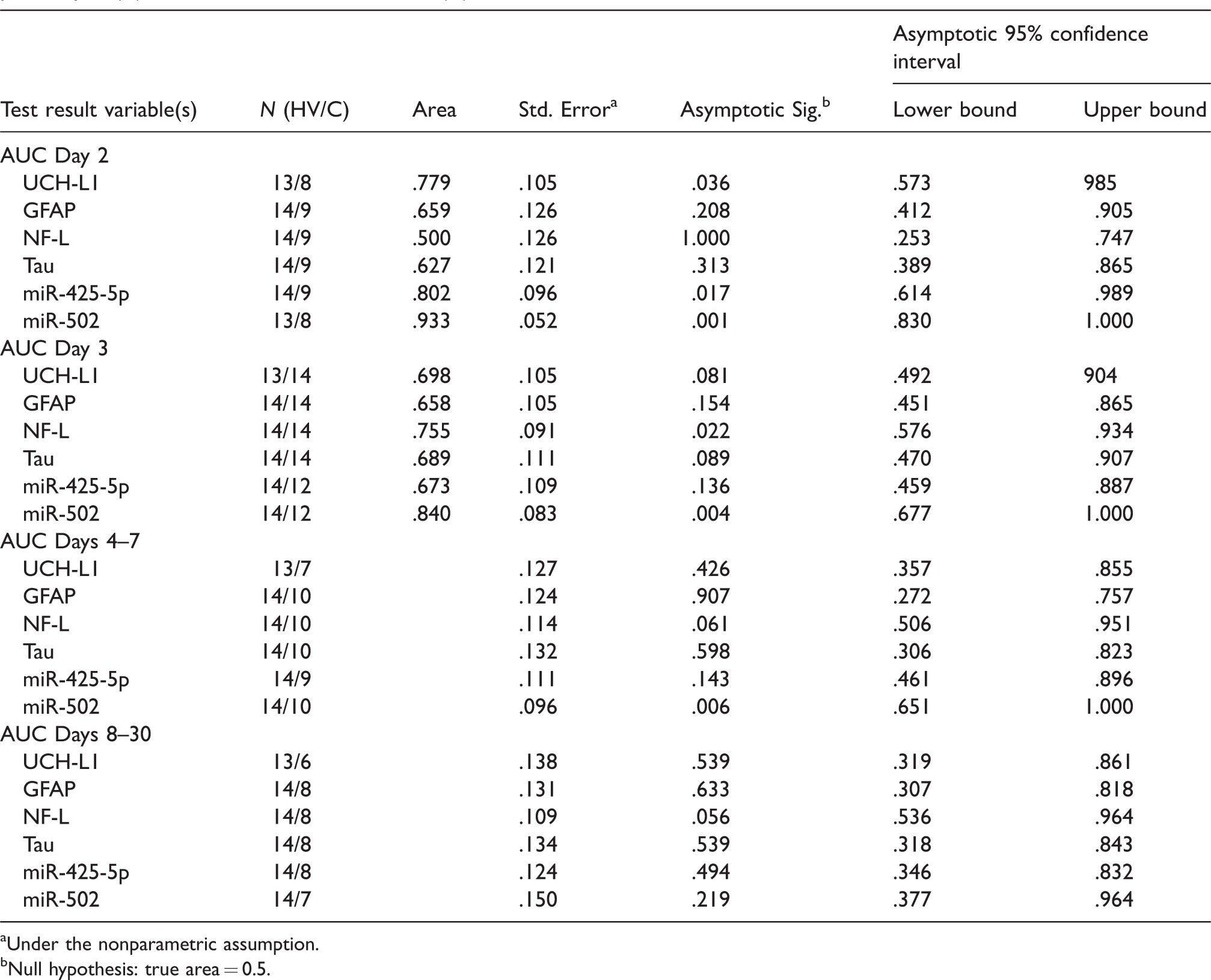
AUCs for these biomarkers at specific time points are also reported in Table 3. In particular, miR-502 showed greater accuracy (>0.80) compared to the other biomarkers analysed within a week from concussion in patients presenting symptoms at the time of assessment.
AUC and confidence interval for the serum proteins and miRNAs analysed at different time points and number of subjects per analysis (N), HV versus concussed athletes (C).
aUnder the nonparametric assumption.
bNull hypothesis: true area = 0.5.
Pearson correlations
Correlative relationships between serum biomarkers and a number of SCAT5 sub section scores are presented in Table 4. Here, we see that NF-L is the only marker with a statistically significant correlation with two of the SCAT5 sub sections (number of symptoms and balance) at day 2. No correlation was observed at later time points.
Person correlations between biomarkers and SCAT5 scores.
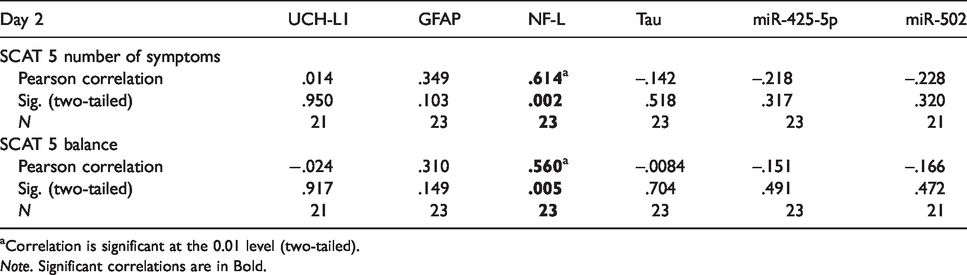
aCorrelation is significant at the 0.01 level (two-tailed).
Note. Significant correlations are in Bold.
Symptoms were also correlated with levels of biomarkers analysed at all time points and presented in Table 5. Thirteen different symptoms correlated with NF-L at days 2 and 3 with GFAP at the same time point.
Pearson correlation of SCAT 5 symptom scores versus serum biomarkers.
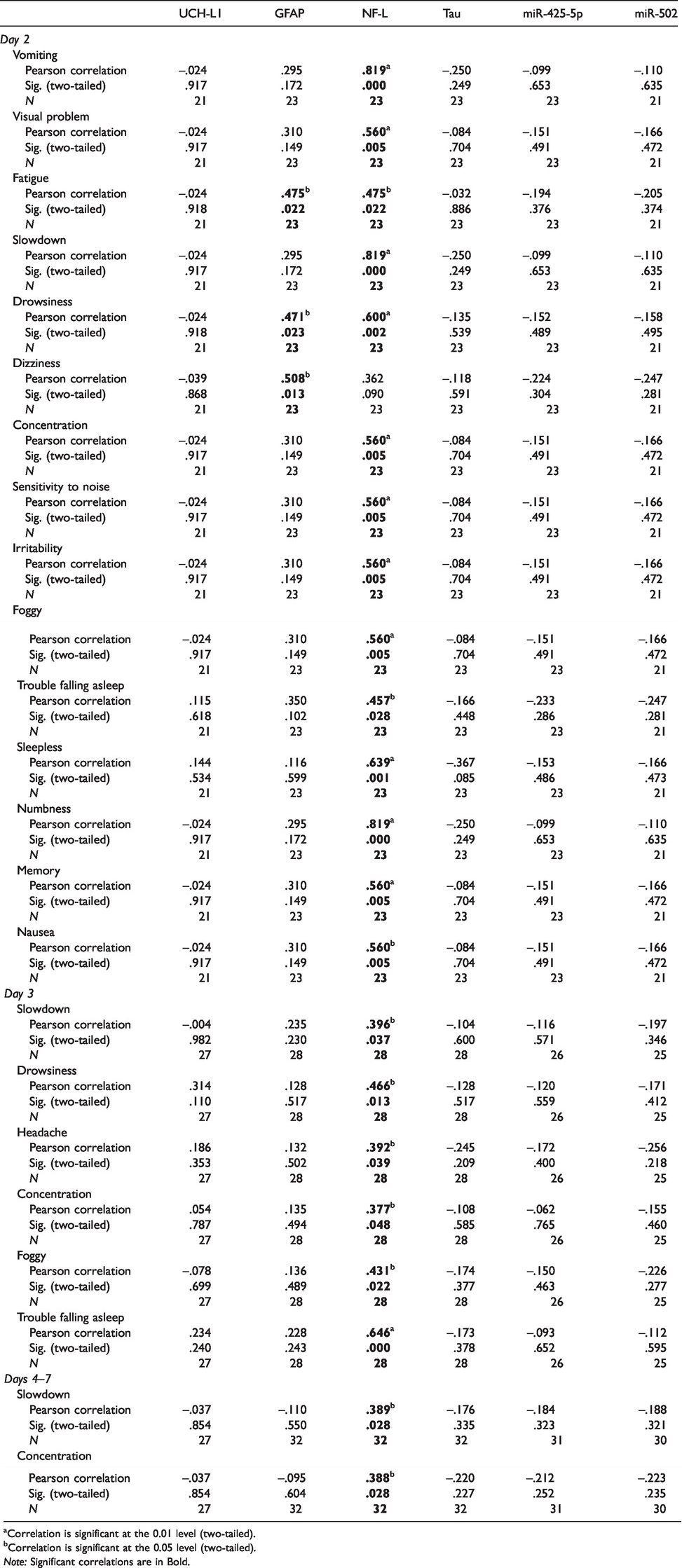
aCorrelation is significant at the 0.01 level (two-tailed).
bCorrelation is significant at the 0.05 level (two-tailed).Note: Significant correlations are in Bold.
Five of the 13 symptoms were still reported at day 3; 2 at days 4–7 and they correlate with statistical significance to NF-L. No correlation between symptoms and biomarkers was detected at 8–30 days.
Note. Significant correlations are in Bold
Discussion
Various proteomics studies have been undertaken in both animal models and clinical studies to identify potential TBI-specific biomarkers31,47,48 and to determine the distinct pathophysiological conditions following TBI. In particular, two serum biomarkers, GFAP and UCH-L1, were approved by the FDA in February 2018 as a blood test for use within the context of mTBI within the first 12 h post injury. As previously discussed, both biomarkers are detectable in serum within 1 h and are reported as being able to distinguish between mTBI patients with structural pathology and those without,23,49,50 in both children and adults. 51 Therefore, the intended use is to exclude or confirm the requirement for axial imaging where there is equipoise or uncertainty as to the requirement. This investigation does not constitute a direct comparison of any biomarker within the licensed confines of the FDA approval for GFAP and UCH-L1; moreover, it represents a comparison of performance within a secondary care (later) setting and certainly represents a new clinical context for the use.
A time course profile of these two biomarkers was described in mild-to-moderate TBI (MMTBI), showing a peak of GFAP level at 20 h after injury, and a steady decrease over 72 h. However, this protein was still detectable at 7 days. In contrast, UCH-L1 increased more rapidly after injury than GFAP, reaching a peak at 8 h and decreasing steadily over the following 48 h. 50
Our results showed no significant difference in GFAP concentration between the groups analysed, although a modest increase can be detected at days 2 and 3 as shown in Table 2. The same can be stated for UCH-L1, where a pick is present at day 2. This is consistent with the previously reported performance of these markers in the subject literature.
Tau peptide has been described as showing a peak in detectable serum levels within the first 36 h after injury; however, we did not observe this. NF-L showed a modest increase between days 4–7, again consistent with the current literature in that NF-L has better resolution of injury at later time points. However, the observed concentrations and their accompanying significance may have been different should earlier sampling had taken place. This should be considered carefully and form the basis of future comparative investigations.
In recent years, the miRNAs have emerged as the potential next generation of biomarkers for many conditions including neurodegenerative diseases such as Parkinson’s and Alzheimer’s disease.52,53 This fortifies and inspires ongoing efforts to investigate their potential utility in concussion diagnosis and management. It also serves as a reminder as to the vast amount of work that lies ahead to fully understand their complex roles relationship with the physiological function and pathological processes of the CNS.
MiRs have many advantages over protein-based markers. For instance, miRNAs are stable in various bodily fluids, 54 the sequences of most miRNAs are conserved among different species, 55 expression is tissue/fluid specific and levels can be easily assessed by RT-qPCR, which allows for signal amplification. Additionally, protein-based biomarkers may have different post-translational modifications which can affect the accuracy of measurement.
In this study, we analysed the expression of two miRNAs (miR-425-5p and miR502) which were previously selected and described as candidate biomarkers of mild TBI at very early time points. 34 Previously, our group demonstrated a significant downregulation of both miRs in a cohort of 30 mTBI patients within 1 h of injury. The downregulation expression of these two biomarkers is confirmed in this new cohort. As shown in Figure 1, miR-502 fold changes (a conventional method to measure the differentially expressed genes by RT-qPCR) remain significantly downregulated, if blood samples were collected at 2, 3 or 4–7 days from concussion and if compared to HV. In addition, it showed a robust sensitivity and specificity as demonstrated by the AUC, particularly at day 2 (Table 3). Therefore, miR-502 has potentially demonstrated the ability to independently discriminate concussed athletes without structural brain injury from HV within a time frame of seven days.
However, neither of the miRs correlate with SCAT5 scores or symptoms reported. NF-L, is the only biomarker analysed, showing a significant correlation with 13 of the symptoms reported and SCAT5 scores (number of symptoms and balance) at day 2, confirming its particular utility in the management of concussion. Understanding the temporal profile of a candidate biomarker is crucial and requires rigour and the appropriate timing of measurement. This represents a challenge with regard to obtaining samples in a timely fashion from patients with less severe injury, as they often do not present to secondary care and frequently have an inconsistent pattern of post injury activity. A biomarker that remains stable for a temporal window of at least a week has a significant practical and clinical advantage over those with much shorted windows (that rise/fall rapidly after injury and reverse over 48 h). Moreover, miR-502 has the potential to improve the sensitivity in a population in which structural TBI has been excluded, which is more reflective of the sports concussion secondary care (clinic) context, where in reality, the need for a CT scan of the brain does not represent the true diagnostic dilemma.9,10,56
However, while this data is promising, the authors recognise that there are significant limitations to the observations made. This work was undertaken in a small heterogenous cohort of patients and therefore can be considered as a pilot study only. Patients were enrolled within a broad sampling tie window from 2 to 30 days. This approach may well capture differing peaks and troughs in the natural metabolism of a given molecule; therefore, this may have a significant effect on the performance of any given marker. In addition, long-term outcome data including return to play data and chronic symptomatology in these patients were not collected.
Again, the sample size of this investigation is clearly a significant limitation against making any kind of definitive deduction. However, within the current research framework, the resources available at the time to the research team and the wide geographical area of recruitment, we feel that this serves as a very useful hypothesis forming pilot investigation. The clear heterogeneity in this sample size potentially could introduce a degree of selection bias to any conclusions drawn. The principle reasons for this are the pragmatic nature of the original investigation, with referrals open from all high-level contact sports (multiple sports) persons from within the region. Consideration was given to streamlining the inclusion criteria to improve subject matching within the cohort; however, it was felt that this may limit recruitment and, from this, create a data set that was too small to be useful. However, a positive aspect of the sample population is the professional and semi-professional nature of all the participants. This infers a consistent level of performance, athletic ability, schedule of training and the intensity of participant.
As declared in the methods session, sampling was undertaken at first contact with the research team resulting in the broad window of acquisition. The practicality and logistics of presentation to research facilities led to this sub optimal situation. As a pragmatic investigation (although all patients’ assessed presented symptoms of concussion), this was a calculated limitation accepted by the team to allow progress of the study within a reasonable time frame. Future investigations should potentially involve local blood draw/sampling at specified and precise time frame. The Quanterix assay used in this investigation as mentioned is not the exact same in the FDA approval; however, this assay has been previously used in investigations into concussion.57,58
As a continuation of the pragmatic design, trial-specific criteria for the diagnosis of a concussion for the study were not proscribed in advance to participating clubs; instead, essential qualification criteria for inclusion were requested. This is a significant limitation and does open up the possibility of an unacceptable heterogeneity in diagnostic thresholds. Sports specific concussion diagnostic criteria were employed. Criteria used by the clubs that own enhanced care team (doctor or suitably trained clinical professional) were utilised (the pitch side clinicians made the initial diagnosis); however, the essential qualifiers were (i) clinical suspicion of concussion by the enhanced care team and (ii) assessment and failure to pass the RFU HIA assessment or SCAT-5 concussion assessment tests at first assessment. It should be noted that the utility of all of the markers considered is only as good at detecting a concussive event as the clinical diagnostic criteria, and the identification of a true concussive ictus is sub-optimally assessed in this investigation.
Finally, both miRNA-425-5p and miRNA-502 have been implicated in the modulation of pathology in a number of extra cranial diseases.59–62 Their potential involvement in such a diverse set of pathologies certainly opens the possibility that numerous other factors may influence levels of expression, and in as yet unforeseeable circumstances influence their ability as effective biomarkers of concussion diagnosis. This must be considered as a further limitation in the deductions made by the authors. Future work will focus on developing a more robust mechanistic hypothesis together with further validating the value of these miRNAs as biomarkers of concussion.
In conclusion, miR-502 has shown the potential to expand the window of effective sensitivity to injury, resulting particularly useful for application within the secondary care or specialist clinic setting where sampling immediately after injury is not readily available. However, significant confirmatory work is required due to a number of significant limitations in this study.
Footnotes
Acknowledgements
The views expressed are those of the author(s) and not necessarily those of the NHS, the NIHR or the Department of Health. We acknowledge the contribution of Dr Gwen Kennedy for laboratory analyses performed in the University of Dundee’s Immunoassay Biomarker Core Laboratory.
Authors’ contribution
DD: Study design and execution, data compilation and curation, patient management, imaging data acquisition. Manuscript writing and compilation.
KMY: Data production, patient management, manuscript review
US: Laboratory processing of samples, data production and discussion
CB: Data production, patient management
MG: Study design and manuscript review
DH: Patient management, manuscript review
VS: Radiological data review
AB: Study design, data curation and manuscript review
VDP: Study design, data production, curation and review, laboratory oversight, statistical analysis and manuscript production.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The University of Birmingham has intellectual property associated with miRNA listed in this manuscript. AB and VDP have conflict of interests with micoRNAs disclosed.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the National Institute for Health Research (NIHR).
