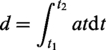Abstract
When the human head is subjected to blunt force impact, there are several mechanical responses that may result from the forces involved, including absorption of impact forces through the various layers of the head. The purpose of this study was to develop an anatomical head model to measure force transfer through the various head layers and their displacement when subject to short-duration high-velocity impacts. An anatomical head model was constructed using previously validated simulant materials: epoxy resin (skull), polyvinyl siloxane (scalp), agar/glycerol/water (brain) and modified intravenous fluid for the cerebrospinal fluid. An array of accelerometers (4 mm × 4 mm × 1.45 mm) was incorporated into the various layers of the head to measure forces in x- (anterior/posterior), y- (left/right) and z- (up/down) axis. All sensors were connected to a signal conditioning board and USB powered data loggers. The head model was placed into a rigid metal stand with an optical sensor to trigger data capturing. A weight (750 g) was dropped from a height of 0.5 m (n= 20). Impact forces (z-axis) of 1107.05 N were recorded on top of the skin, with decreasing values through the different layers (bottom of skin 78.48 N, top of skull 319.82 N, bottom of skull 87.30 N, top and centre of brain 47.09 N and base of brain 78.41 N. Forces in the x- and y-axes were similar to those of the z-axis. With the base of the brain still receiving 78.41 N, this highlights the potential danger of repetitive impact forces to the head. Upon impact the layers of the head are displaced in the x-, y- and z-direction, with the highest values shown in the z-axis. In conclusion, this study identified the importance of considering short-duration high-intensity impacts to the head and their effect on underlying tissues.
Introduction
When the human head is subjected to impact forces, there are several mechanical responses that may result, including the absorption of the impact through the various layers of the head, leading to varying degrees of structural damage. 1 Blunt force head injuries remain poorly understood, especially repetitive short duration high intensity, which may occur in concussion and subconcussion. The absorption of impact force and subsequent transmission to the brain is of particular importance given the significance and increased awareness around sports-related concussion.
Head injury models using animals have been previously used; however, their main disadvantage is that the head anatomy of animals is incomparable to humans. 2 Obtaining in vivo data from living animal or human cranial impact studies is clearly unacceptable. Therefore, having an accurate biomedical model of the human head would be highly desirable for impact testing. In order for it to be as anatomically correct as possible, the model would need to include a scalp and skull layer, a brain and cerebrospinal fluid (CSF). The advantages to researchers of such a head model include the ease of fabrication, decreased costs and the elimination of ethical issues that are involved with human subjects in research of this type.3,4
Previous literature has described various head models that involved the use of simulant materials for the scalp, skull and brain. Thali et al.3,4 were the first to develop a spherical skin–skull–brain model for ballistic and blunt force wounding. A silicone cap was used to emulate the human skin, which contained synthetic fibres to mimic the collagen and fat of the scalp. Layered polyurethane was used to simulate the human skull and the brain simulant was made out of ordnance gelatine (10% gelatine). Within the limitations of their study, the authors concluded that these materials resembled and behaved in similar ways to human tissues and that the use of these simulant materials produced similar results comparable to that seen in real cases.3,4
Another human head model reported by Merkle et al., 5 included skull, brain, facial structure and skin to measure the response to overpressure loading in protected and unprotected conditions. Dielectric silicone gel (Sylgard 527) was used for the brain, glass/epoxy mixture for the skull and syntactic foam for the facial structure. Pressure sensors were positioned in the anterior and posterior region of the brain simulant. The authors concluded that this head model could be successfully used for future research. 5
Freitas et al. 6 used perma-gel to simulate the soft tissues, brain and skin for a head model to use in behind helmet blunt trauma experiments. The brain simulant also included iron powder, which was uniformly mixed into the perma-gel, to increase the brain mass to 1.4 kg. In this human head model water pressurized at 0.29 psi was included in the system to simulate CSF. This study also included sensors to measure the intracranial pressure and cranial strain and reported successful use of the head model as well as its great potential to provide insights into high impact injury mechanics. 6
Even without the use of a sophisticated head model, Tong et al. 7 were able to quantify impact forces using a quartz impact sensor to determine values that may be associated with subconcussion. This study found that although impact peak intensity had been reduced through a protective material layer and silicone scalp layer the overall total energy might still impart forces to the underlying brain. In other words, in addition to the peak intensity, repetitive low peak, high impact trauma over time may be contributory to subconcussive head injury. 7
Physical activities, especially contact sports such as boxing, American football and rugby, have been associated with high rates of concussive and subconcussive trauma, especially in younger generations. 8 Symptoms may include temporary loss of consciousness and forces being transmitted to the brain resulting in neurological changes and deterioration. Determining the mechanisms and forces that produce concussive and subconcussive injuries presents a challenge, 9 which adds weight to the argument for a more accurate skin–skull–brain model.
The limitations of the previous studies included the shortage of validating the mechanical properties of materials used in the studies, the use of a non-anatomical model and use of simulant materials that are not suitable, such as 10% gelatine, which the authors of this study found to be not brain like.10,11
The purpose of this study was to first develop a more anatomical skin–skull–brain model in a realistic manner for the purposes of impact head injury research. Second, to measure the impact force and displacement (observation of trends) through the various layers of the head by incorporating an array of accelerometers in between the layers and subjecting the head model to impacts using a drop tube system which has not been previously reported in the literature.
Methods and materials
Human skull simulant
A split mould technique was used to construct an anatomical skull, using a solid foam adult anatomical skull model (Sawbone model 1345, USA) and duplicating silicone (Duosil H, Shera Werkstoff Technologie, Germany), contained within a plastic container. Undercuts were blocked out, especially around the zygomatic arches and eye sockets, to allow easier removal of the model from the mould. Plasticine (modelling clay) was added to the base to later mimic the neck. Two smaller spheres were included laterally to act as mechanical location for accurate relocation of the halves (mould ‘A’ and ‘B’) (Figure 1(a)).
(a) and (b) Skull simulant mould fabrication.
The next stage involved creating a space for the desired 7 mm skull thickness of the top skull half (mould ‘A’). This was done by rolling out plasticine to the desired thickness and carefully placing it inside the mould halve of the top skull. Vents were added to allow for injection of the skull simulant material and to let air escape during this process. Duplicating silicone was poured in and mould ‘C’ created (Figure 1(b)).
By using mould ‘B’ and by assembling mould ‘A’ and ‘C, the simulant material (Masterflow 622, Degussa, Germany), an epoxy resin, was mixed with the ratio of 5:1 (Part A:B) and subsequently injected via the vents (for the assembly of mould ‘A’ and ‘C’) or directly poured into the mould (‘B’), to create the skull simulant halves. After setting these were trimmed to ensure a perfect fit of the skull halves. Note, the model in this study was a homogenous structure and did not include sutures.
Human scalp simulant
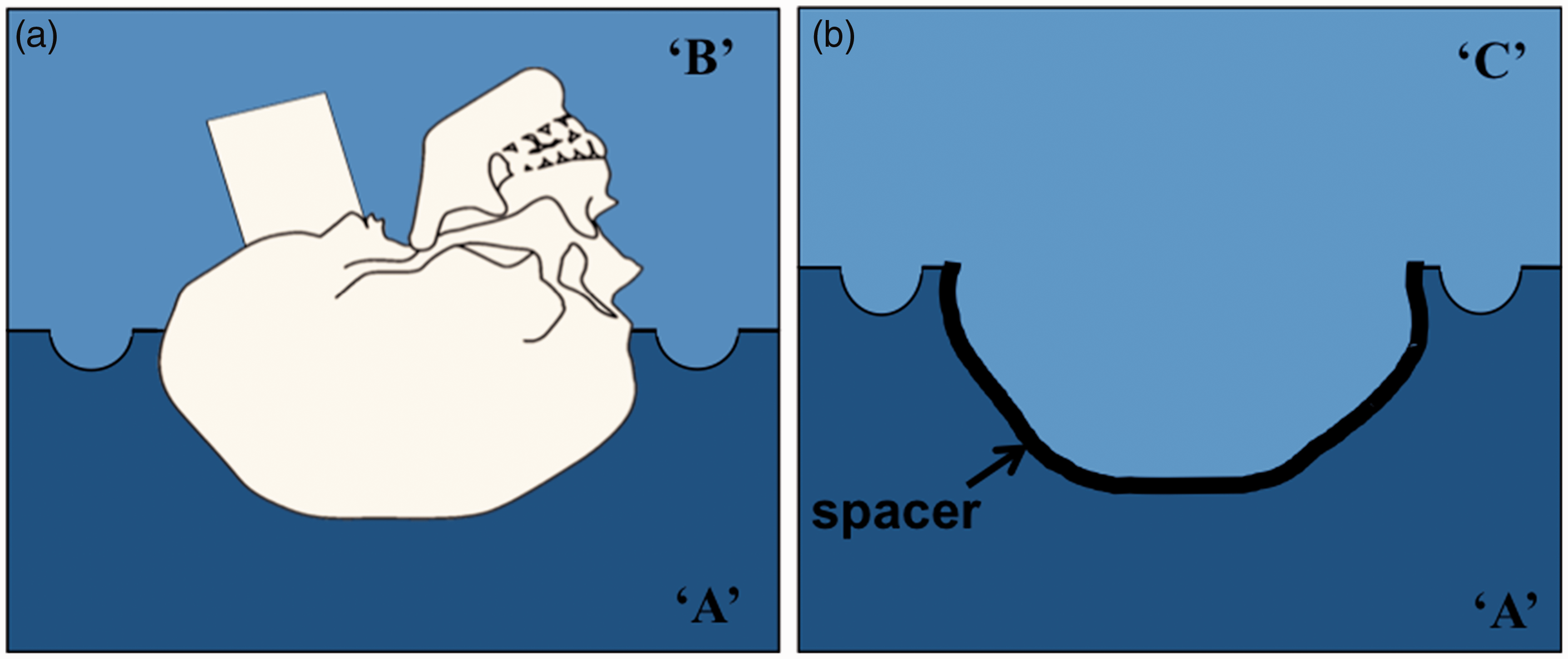
The top half of the skull simulant was placed back on its corresponding mould (‘C’), then plasticine rolled out to 6 mm thickness and carefully placed on top. Again air vents were added to allow for injection of the skin simulant material and to let air escape during this process. Duplicating silicone was then used to create mould ‘D’ (Figure 2).
Scalp simulant mould fabrication.
By combining moulds ‘D’ with ‘C’ (top skull simulant half in place) the skin simulant material (Exahiflex, regular type, GC corporation, Japan), a polyvinyl siloxane, was injected via the vents.
Human brain simulant
The top half of the skull simulant was lined with 3 mm plasticine (mimicking subarachnoid space for CSF) and filled with duplication silicone, which was then used to make a mould to create the brain simulant (Figure 3). The mould was lined with a thin plastic bag, mimicking the dura mater, electronic sensors (see ‘Electronic data capture system and testing set-up’ section) positioned at the base, centre and top of the mould, and an agar/glycerol/water mixture 11 with a ratio of 40:50:10% (by volume), poured in to create the brain.

Brain simulant mould fabrication.
Human CSF simulant
For the CSF an intravenous fluid (Hartmann’s solution, Baxter Viaflex, UK) was used as its constituents closely matched those of the human CSF12,13 apart from a deficiency of glucose, which is present in human CSF. To account for this, 0.6 g of glucose was added to the solution using a syringe. Adding the glucose would increase the osmolality of the solution, thereby mimicking human CSF composition more closely.
Electronic data capture system and testing set-up
An array of accelerometers (Analog Devices Ltd, USA, accelerometer’s dimension 4 mm × 4 mm × 1.45 mm) with different acceleration ranges was incorporated into the various layers of the head (three per layer, except top of skin which had an impact plate (40 mm × 40 mm) mounted to it that included one accelerometer): skin top (±70 gn), skin bottom (±16 gn), skull top (±16 gn), skull bottom (±5 gn), brain top (±3 gn), brain centre (±3 gn) and brain base (±5 gn) (Figure 4). All sensors, except for the top impact plate (z-axis only), were capable of an output voltage for three axes (x – front to back movement, y – left to right movement, z – up and down movement). Different ranges of accelerometers were used due to initial trials showing that lower ranges were inadequate for different parts of the model that had different ranges of force/acceleration. One accelerometer was used on the top of the skin in the strike plate due to the rigidity of the metallic plate material producing the same results as if all three accelerometers were fitted. Three accelerometers were placed in the subsequent layers to capture the impact effect on the area directly below that of the impact plate. Supply voltage to the sensors was 3.0 V, giving a resting output of 1.5 V. This also meant that each sensor with its cable carried three signals and required three separate channels of voltage conversion. The sensors were mounted to RJ45 insulation displacement connectors with each one providing signals for each axis. The cable also carried the 3 V supply voltage to the sensor and the signal ground. To ensure the fragile connection of the wires attached to the accelerometers does not get damaged upon impact and that they were waterproof, they were embedded in a thin layer of resin. The sensors were then connected to a signal conditioning board and to USB powered data loggers (PicoLog 1000 series, Pico Technology Ltd, USA), with each logger providing channels of analogue to digital conversion (Figure 4). One channel of each logger was connected to the trigger signal.
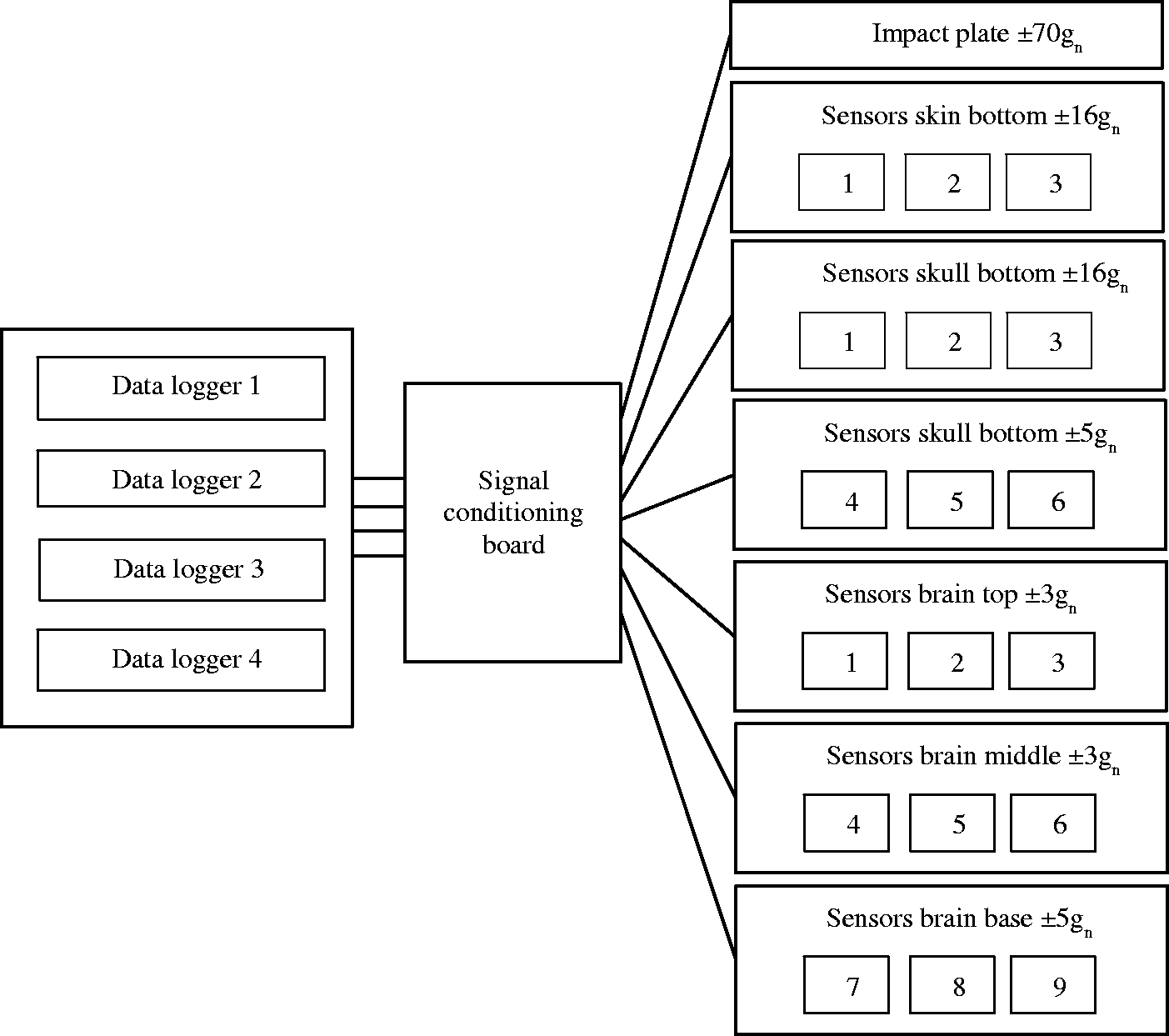
Simplified diagram illustrating the electronic data capture system.
A rigid metal stand into which the head model was placed upon testing had an optical sensor incorporated 50 mm above the head to detect the passing of the drop weight (750 g) just prior to impact, at which point the data capture started. The data capture system was connected to a laptop and the associated software (PicoLog, Pico Technology, USA) used to record the data of n= 20 impacts using a drop tube rail system (Figure 5(a) and (b)). A weight was dropped from a height of 0.5 m.

(a) Testing set-up and (b) drop tube rail system.
Recorded voltage values from the impacts were converted to Newton values using (current voltage − resting voltage)/voltage per gn × acceleration of gravity × mass (mass of object linked to accelerometer). The impact forces on top of the head were confirmed using a load cell set-up, made up of a piezoelectric quartz force transducer to capture the impact data mounted in a metal frame structure as per Tong et al.,
7
resulting in a value of 1107.05 N
The displacement (d) in the x-, y- and z-axis, was calculated by the sum of the individual displacement at each time interval with the measured acceleration, using the above equation, where a was the acceleration measured at each 160 ms interval and t1/t2 was the current time duration in seconds of the impact. The measurement interval of 160 ms was arbitrarily determined based on how many data points were recorded per second, as well as the limitations of the response time of the device. For values of t1 and t2, they were determined by the time interval of 160 ms (e.g. t1= 0, t2= 160…. t1= 160, t2= 320, and so forth). The instantaneous acceleration was used per time interval with the total distance summed together. Because the force values on the top of the skin were recorded only in the z-axis, the displacement values were only calculated for the subsequent layers.
Model assembly
Grooves were cut into the skull simulant to allow embedding of the sensors and cables, using epoxy resin (Masterflow 622, Degussa, Germany). Part of the cable was not embedded to allow movement and thus accurate measurement of the impacts. The sensors for the bottom of the scalp were embedded using the simulant material. To minimize leakage of the brain simulant the cables were joined using a copolymer sealant (Selleys, New Zealand), prior to pouring the brain simulant into the prepared mould.
A hole was cut into the bottom half of the skull to allow exiting of the wires (sensors) and a tube embedded that would later deliver the CSF. For the final assembly, the brain was placed inside the top skull half and joined with the bottom skull half using the simulant epoxy resin and the hole/cables sealed using epoxy resin and sealant. Modelling clay (FIMO, Steadtler Mars GmbH & Co company, Germany) was attached around the neck to mimic the elasticity of the spine (as per Thali et al.3,4) and the head model placed into a stand to keep the model stable during testing (Figure 6).
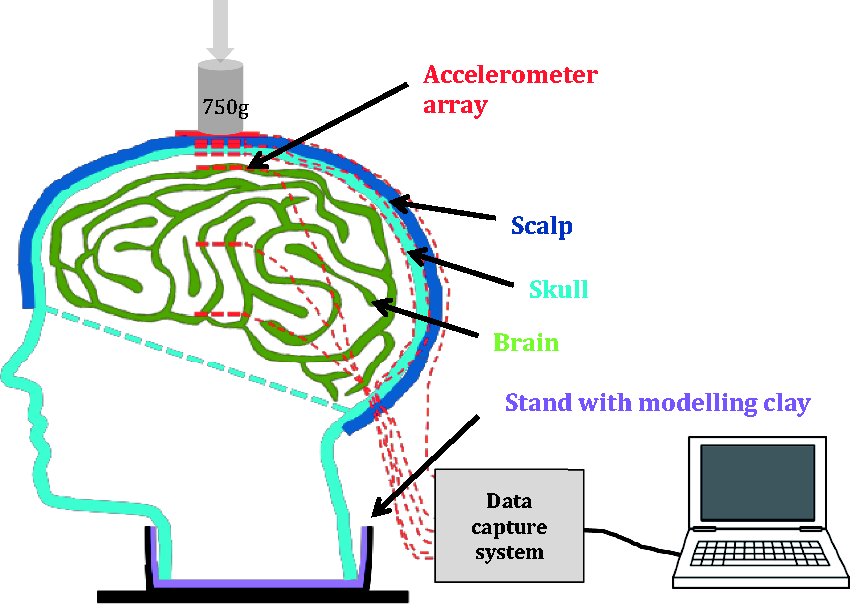
Diagram of anatomical head model testing set-up.
Statistical analysis
The statistical analysis was conducted using SPSS (Version 24 14 ) to examine group differences between the different layers in regards to the dependent variable (impact and displacement). Post hoc tests (Wilcoxon signed rank) for the impact values were carried out to conduct multiple comparisons between specimen groups in pairs.
Results
Impact forces and displacement
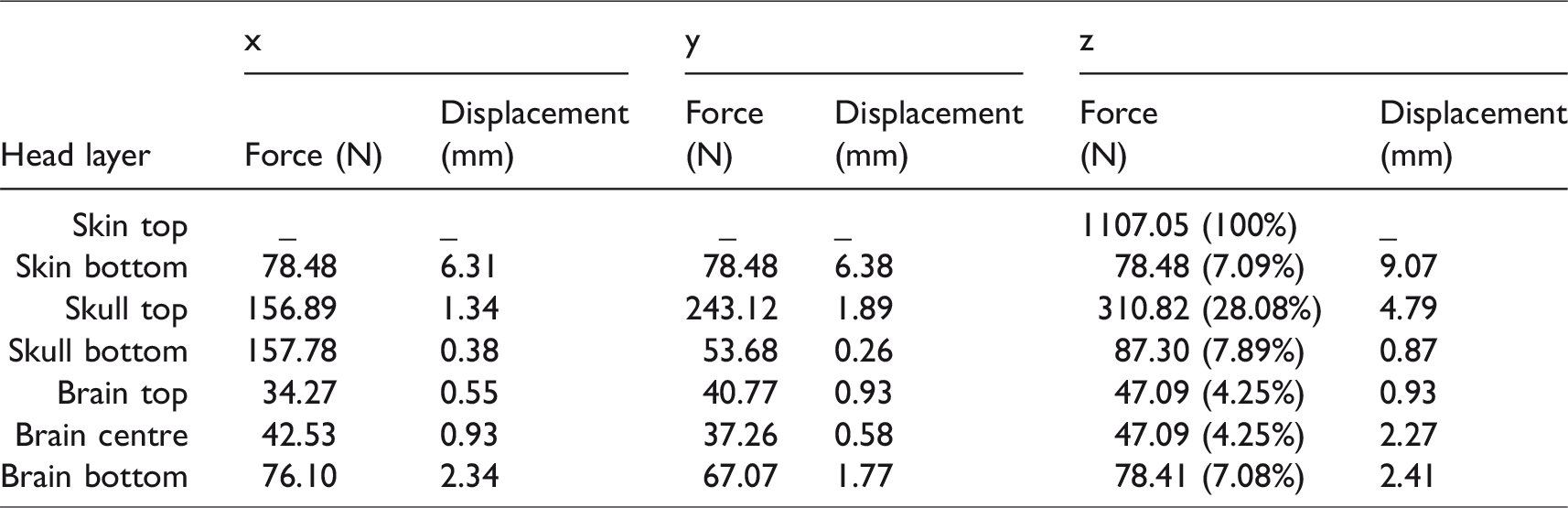
Mean impact forces (z-axis) of 1107.05 N were recorded (up to 121 µs) on top of the skin, with decreasing values through the different layers (Table 1, Figures 7 and 8). The bottom of the skin received 7.09% of the initial impact force (78.48 N), the top of the skull received 28.08% (310.82 N), the bottom of the skull received 7.89% (87.30 N), the top of the brain received 4.25% (47.09 N), the middle of the brain received 4.25% (47.09 N) and the base of the brain received 7.08% (78.41 N). Similar observations were made in the x- and y-directions as seen in Table 1.
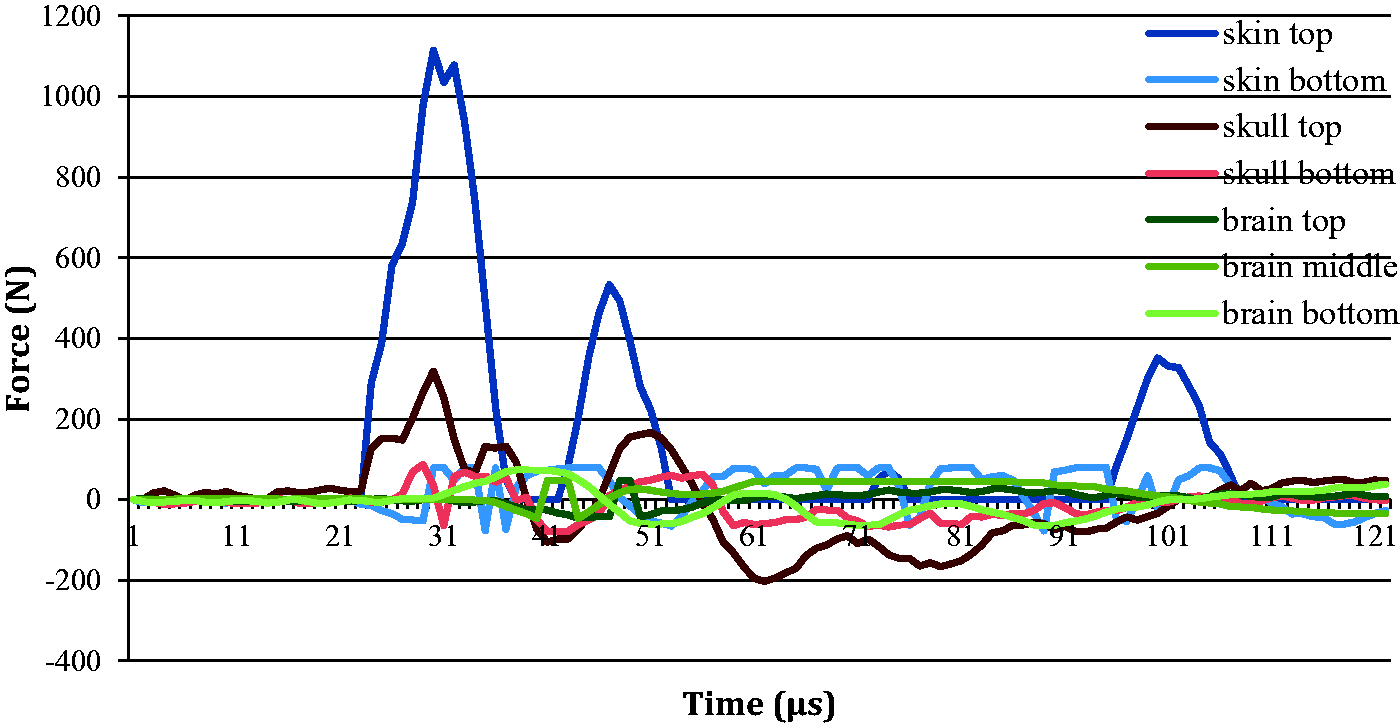
Showing a typical example of force versus time for the z-axis from one impact.
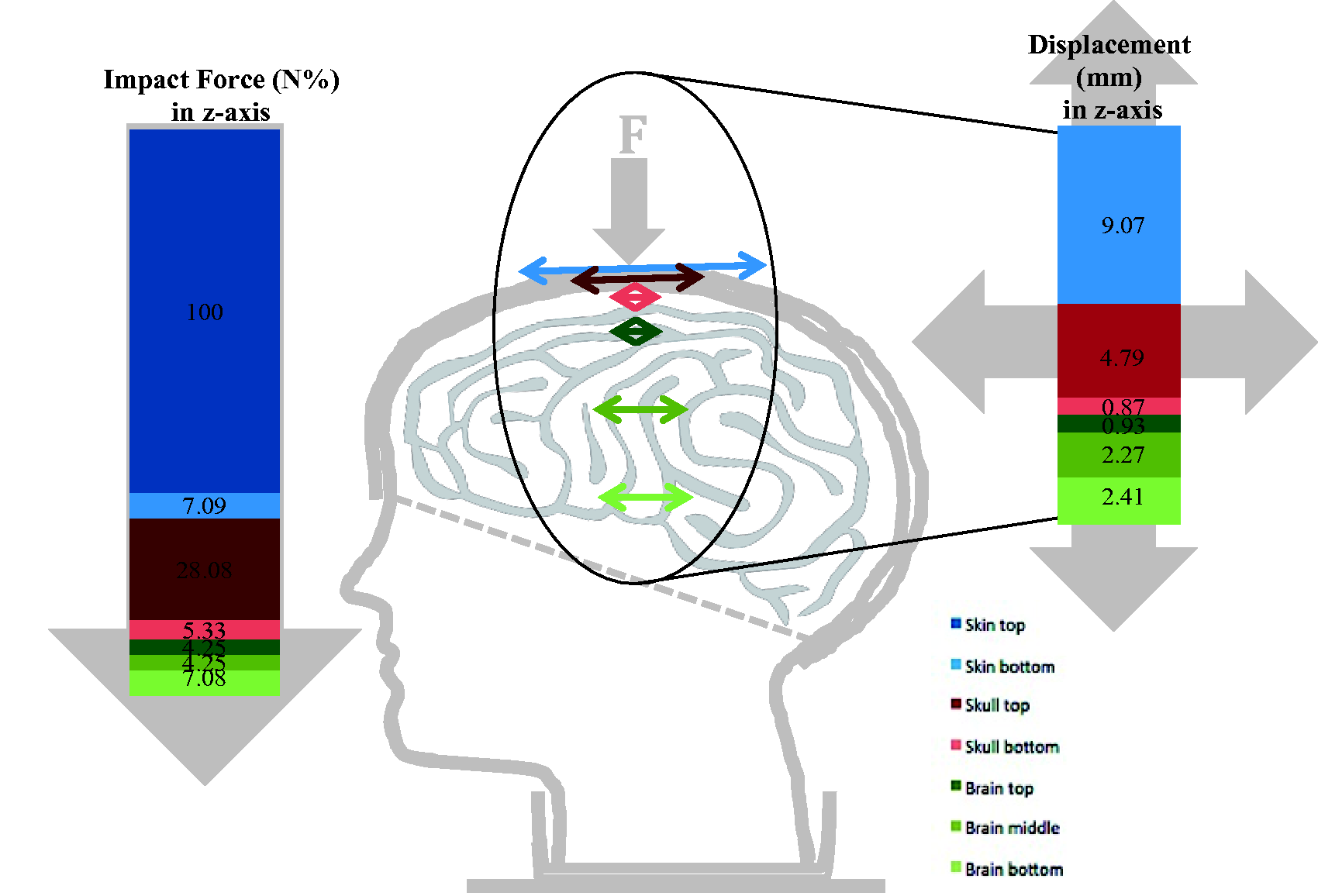
Impact force (N%) and displacement (mm) in the z-axis for the layers of the head.
Impact force (in N) and displacement (in cm) in x-, y- and z-direction. The decrease in force at each level, as a % of the total force measured on the top of the skin, is included in brackets in the z-axis column only.
The displacement results show displacement in all directions, with the highest ones in the z-axis (Table 1, Figure 8).
Statistical analysis
For the statistical analysis, the differences between the different layers (bottom skin; top and bottom of skull; top, middle and base of brain) were examined using normality tests (Shapiro–Wilk) for impact transfer in x-, y-, z-direction. An individual normality test was carried out for the top skin layer in the z-direction (no x and y recorded). The results showed that the data sets for impact of the different layers in the x-, y- and z-direction only fit the assumption of normality (p > 0.05) for some layers (skull bottom (x, y, z), brain top (y, z), brain middle (y, z) and brain base (y)), with violation of normality (p < 0.05) for others (skull top (x, y, z), brain top (x), brain middle (x) and brain base (x,z)). A non-parametric Friedman test of differences among repeated measures was conducted and rendered a Chi-square of 268.02, which was statistically significant (p < 0.05). A post hoc test (Wilcoxon signed rank) with an adjusted alpha level (α = 0.017) was carried and showed a statistical significant difference between impact and all the layers (p < 0.017), except the top of the brain and middle of the brain (p > 0.017).
A further Friedman test of differences between the impact and axes was conducted and rendered a Chi-square of 150.72, which was statistically significant (p < 0.05). A post hoc test (Wilcoxon signed rank) with an adjusted alpha level (α = 0.017) was carried out and showed a statistical significant difference between the z-axis and both the y-axis and the x-axis, with no statistical significant difference between the x-axis and the y-axis for the investigated layers.
The results of the data set for the top skin resulted in violation of the assumption of normality (p < 0.05). A non-parametric Friedman test of differences between impact and layer among repeated measures was conducted and rendered a Chi-square of 114.30, which was statistically significant (p < 0.05). A post hoc test (Wilcoxon signed rank) with an adjusted alpha level (α = 0.0024) was carried out and showed a statistical significance (p < 0.0024) difference between all the different layers, except between the bottom of the skin and the base of the brain and between the top of the brain and the middle of the brain (p > 0.0024).
In addition, the results of the displacement data fit the assumption of normality (p > 0.05), thus paired sample t-tests were carried out, resulting in no statistical significant difference between the axes and the layers with the exception of the bottom of the skin and the top of the skull/middle of the brain/base of the brain (p < 0.05).
Discussion
The results of this study show that short-duration high-intensity impact forces at the top of the head transfer through the various levels of the head to the base of the brain, with various amounts of displacement exhibited by each layer.
The results of Pearce and Young 15 highlighted the importance of repetitive short duration high impacts, which were also observed in our study, suggesting the head to be susceptible to critical intercranial pressure transients upon impact. In our study, the base of the brain still received 78.41 N (7.08% of the initial impact force), which indicates that the brain is still subject to inertial forces and highlights the potential danger of repetitive impact forces to the head. Nusholz et al. 16 used an accelerometer array attached to the head (live anaesthetized and post-mortem Rhesus monkeys as well as re-pressurized human cadavers) to measure head motion. They observed skull deformation and angular acceleration due to the impact. Skull movement due to impact resulted in energy transmission to the brain via the dura matter. Further skull acceleration was reported due to forces generated from the brain and neck subsequent to the impact and after impact load removal. The authors concluded the skull deformation from the impact possibly having important implications on injury produced with blunt force head impacts, noting that the skull geometry of Rhesus monkeys and humans differ significantly. Mass movement of the brain, skull deformation and acceleration upon impact increased cranial pressure at time of impact and the transmission of impact to the brain has been linked to brain damage and concussion as reported by Hodgson et al. 17
Melvin and Evans 18 reported that upon impact, the forces produce local deformations of the skull at the impact site with stress waves travelling away from the impact site throughout the skull to the underlying structures – likened to ripples in water. The combination of forces transmitted to the skull and subsequent deformation creates energy distribution and displacement throughout the materials. Leestma and Thibault 19 found that impact to the head results in disruption and haemorrhage in some areas of the brain, which is less obvious if the impact causes injury that does not disrupt tissue or is physically visible. Furthermore, multiple neuropathological cascades are triggered from the point at which biomechanical forces are transmitted to the brain. They concluded that injury to the brain upon impact not only resulted in immediate injury but also undergoes a prolonged process that can evolve subsequently after impact. The first visible trauma tends to be vascular (in the form of haemorrhage) even without any physical disruption, followed by neurological trauma. The brain also undergoes considerable contusion (bruising) directly beneath the impact site with immediate brain injuries (coup contusion) while some bruising occurs on the opposite site of impact (contrecoup) 19 as the brain undergoes displacement as it compresses against the hard surface of the skull and then rebounces. 20 This was also seen in our study with higher impact forces and displacement recorded at the base of the brain, which may indicate that the brain is subject to inertial forces, highlighting the potential danger of repetitive impact forces to the head. Although the embedding of the sensors and connecting wires in the skull may have had a small influence on the physical response of the material to impact and subsequent force transfer, this was necessary to maintain them in place and not affect the behaviour of the adjacent skin/brain layers. The authors made the assumption that this would be minimal as the sensors and connecting wires were embedded in the corresponding simulant material.
Leestma 21 reported that upon impact, movements of the brain may be due to CSF redistribution within the intracranial space. However, to maintain ambient hydrostatic pressure upon severe impacts and the addition of significant intracranial volume, compensatory mechanisms must exist as the tissues are displaced. While the sutures in children can expand due to their increased elasticity, this does not apply in adults. Therefore, as the CSF is driven out due to pressure-induced absorption in an effort to maintain normal intracranial pressure, increased volume changes and intracranial pressures occur, the brain moves away from the impact site, resulting in various injury symptoms (e.g. headaches, nausea and in the worst case loss of consciousness and respiration). Thus, the inclusion of a CSF simulant in the head model is an important consideration, which was considered in this study.
In our study, the impact site was the top of the head, the area where injury to the temporal lobe, which is responsible for processing sensory input, such as retention of auditory memory, language comprehension and behaviour association, is located.22,23 Damage in this area can result in short- and long-term memory loss, as well as speech difficulties and the inability to regulate emotion. These can also be seen with patients suffering from mild traumatic brain injury.21,24 While this study did not look at possible damage to the brain simulant post testing, the measurement of forces in this area could potentially be of use in further studies.
Kinematics involved with head impacts are translational (linear three-dimensional movement) and rotational (angular movement around inertial centre) in nature, usually involving a combination of the two as the head is connected to the torso via the neck. Upon impact to the front of the head, the momentum of the impactor will be transferred to the head, which in turn experiences acceleration. Initially the head will be displaced backwards, due to the impulsive loading of the impactor, resulting in a backward flexion and rotation as the neck constrains the pure linear head motion. The head will be temporarily subject to a change in the angular velocity and as a result experiences angular acceleration. The consideration of both translational and rotational movements is important as trauma to the head and its central nervous system is dependent on the kinematic parameters of the head, apart from physical parameters that determine the injury mechanism; trauma occurring due to linear and angular head rotation is related to the structural and mechanical properties of the head. 19 To partially accommodate this aspect, this study used modelling clay wrapped around the neck of the head model where it was inserted into the stand to mimic the elasticity of the spine (Figure 6), which had previously been used by Thali et al.3,4
The head model used in this study differed to previously reported physical head models in the literature3–7 in that, overall the head was more anatomically correct and provided additional important anatomical features which previous models were lacking. In addition, this study used simulant materials that were more similar to the properties of humans; thereby offering results that would be closer to force impact transfer through the human head. Further, this model has the potential to measure the effectiveness of head protection systems such as helmets and measurement of force and energy transfer from impacts associated with soccer (repeated heading of the ball), rugby, American football and boxing, in an effort to minimize traumatic brain injury due to accidental blows to the head. In addition to the forgoing, further research on the horizontal impact effects is also recommended to provide greater clarity to the damage/force transfer mechanisms of brain injury.
Conclusion
In conclusion, our study identified that short-duration high-intensity impact forces to the top of the head transfer through the various layers and they undergo various amounts of displacement. This suggests that the brain is subject to traumatic forces that are likely to lead to vascular and neurological injuries. However, further research is needed to establish the degree to which these forces can cause damage to the brain.
Footnotes
Acknowledgements
The authors would also like to thank Bruce Partridge from Biotronics Otago for the extensive help with the electronic data capture system.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by Maurice and Phyllis Paykel Trust (grant number 8.1.30) and the Neurological Foundation New Zealand (grant number 1624-SPG).