Abstract
Introduction
Postburn scarring often presents a specific reconstructive challenge from both functional and cosmetic perspectives. The purpose of this study was to investigate whether autologous nanofat harvested from the donor site of full skin or a skin flap can be reused for the treatment of early postburn scaring.
Methods
From July 2018 to April 2022, patients with early postburn scarring underwent scar reconstruction surgery with full-thickness skin or a skin flap for a contour deformity and/or scar contracture, and autologous nanofat grafting was performed during the same operation. The Vancouver Scar Score (VSS) and the itch and pain scores were evaluated at the preoperation time point as well as at 2–3 weeks and 3-months postoperation. A comparison was made among the same patients at different time points.
Results
A total of 17 patients, aged from 18 months to 62 years old were included in this analysis. The VSS was reduced from 10.00 ± 2.12 to 7.41 ± 1.277 at the 2–3-week postoperation time point, and to 5.53 ± 1.37 at the 3-month postoperation time point. The pain and itch score were reduced from 4.65 ± 1.37 and 6.35 ± 1.27, to 3.70 ± 1.10 and 4.94 ± 1.30 at the 2–3-week postoperation time point, and to 3.00 ± 1.28 and 3.94 ± 0.97 at the 3-month postoperation time point respectively. The VSS and pain and itch scores showed a statistically significant reduction (P < 0.05) at the 2–3-week and 3-month postoperative follow-ups compared with the preoperation time point.
Conclusion
Autologous nanofat grafting from donor sites of full thickness skin or skin flap may be a promising treatment for an early postburn scaring as it promotes scar softening, improves itching and pain within the scar. However, this is a small case series with only 17 patients. Further conclusions need to be drawn through expanded samples for randomized controlled clinical trials.
Lay Summary
Hypertrophic scarring is the most common complication after partial thickness burn injury, and the complex pathogenesis and prolonged dynamic process render treatments only marginally effective. In the past few decades, with the technological advances of liposuction and fat grafting, nanofat grafting has been used in a variety of surgical fields, including wound healing, scleroderma, facial rejuvenation, and neuralgia. However, the role of nanofat grafting is not well documented in the prevention and treatment of early postburn scarring. Full-thickness skin grafting or skin flap transplantation is the most common method for the reconstruction of a hypertrophic scaring until now. In the current study, we harvested subcutaneous fat during the preparation of the full-thickness skin or skin flap, prepared nanofat and injected it in the scar located at a nonsurgical site. Comparison of the pre- and postoperation scores for scar color, scar thickness, scar stiffness, and scar regularity showed that the postoperation scores were decreased significantly and that there was a significant improvement in scar pigmentation and thickness as well astheaesthetic outcome after treatment. Most importantly, reductions in the scores for pain and itching could be assessed objectively. It seems that the nanofat grafting is a potential method for prevention and treatment for early postburn scaring.
Introduction
Hypertrophic scarring is the most common complication after partial thickness burn injury, and the incidence of hypertrophic scarring after partial thickness burn injury ranges from 0 to almost 100% depending on the time to healing.1,2 In addition to itching and pain, scars can result in severe aesthetic issues, deformities, or dysfunctions. Despite dedicated intervention for preventing scarring, the complex pathogenesis and prolonged dynamic process render treatments only marginally effective.3,4 Currently, pressure garment therapy, external use of silicone-based dressings, and fractional CO2 laser therapy5,6 are the preferred methods for the prevention or treatment of an early postburn scaring; however, for a hypertrophic scaring over the joint, which limits the range of motion, surgery is recommended at an early stage to prevent joint stiffness and natural growth restriction in children.7,8 Full-thickness skin or a skin flap has been used for reconstruction of a scar over a joint, 9 but it may be challenging if there is a shortage of donor sites. In addition, scars that are not located over joints undergo the natural remodeling process, which always lasts for at least one year. 10 As a safe and effective treatment method, fat grafting has been used in a variety of surgical fields, including breast reconstruction/contour defect augmentation, hip augmentation, and other concave defects. 11 In recent years, along with the development of liposuction technique, fat grafting has been widely used in different areas, such as wound healing, scleroderma, facial rejuvenation, and neuralgia.12–14 Further studies have revealed that fat grafting improves the scar or keloid appearance, promotes scar softening, and reduces pain and itching within the scar.15–17 However, some randomized, controlled, double-blind studies have shown that fat grafting has no significant effect on a scar.18,19 These contradictory outcomes may be due to the different characteristics, locations, and times for scar formation after wound healing in the clinical experiments. Nevertheless, to date, there are rarely reports on the nanofat grafting on the treatment of postburn scaring, especially for the early postburn scaring.
The regular donor-derived fat for grafting is obtained from the abdomen or thigh by liposuction, but when reconstructive surgeries are conducted, the subcutaneous fat is trimmed and discarded during the preparation of the full-thickness skin or skin flap. Inspired by pioneering work on fat grafting, we aimed to assess whether fat from a patient can be used to treat his/her own scar and its potential effect. The purpose of this clinical study was to review our experience on autologous nanofat grafting in the treatment of early postburn scarring.
Methods
Patients
From July 2018 to April 2022, patients diagnosed with early postburn scarring (the formation of scaring occurred less than 12 months from time point of burn injury), accompanied by joint deformities or dysfunctions, were selected for this study. There was no age limitation in inclusion criteria, but those who failed to complete their follow-up were excluded. This study was approved by the Medical and Ethics Committee relevant to the Declaration of Helsinki. All methods were carried out in accordance with the relevant guidelines and regulations. Informed consent was obtained from all subjects and/or their legal guardian(s). All patients and/or their legal guardian(s) provided written informed consent for publication and use of their photographs in the manuscript.
Surgical procedure
The scar with a deformity was released or excised, and then the secondary wound was reconstructed with full-thickness skin or a skin flap. The skin or skin flap was harvested from the abdomen, and the subcutaneous adipose tissue was collected. During the preparation process of full thickness skin donor, all subcutaneous fat from both deep fascia and shallow fascia were collected for the next step of fat grafting. For the abdominal flap, fat was harvested from the deep fascia of the flap, retaining most of the superficial fascia, and the deep fascia fat at the pedicle was also preserved to ensure the circulation of the flap. Unlike fat collected by liposuction, the fat harvested from donor site of full thickness skin or skin flap was massive. The first step of fat processing was to cut the block fat with scissors as much as possible into a chylous shape, and then nanofat processing was carried out as follows:20,21 The fat was emulsified by shifting it between two 30 mL syringes connected to each other by a female-to-female Luer-Lok connector. First, the granular fat was cut back and forth 30 times through a double-pass single-hole 2.0 mm cutter; then it was cut back and forth 30 times with an aperture of 1.6 mm, followed by cutting back and forth 30 times with a 1.2 mm fat cutter, and finally cutting back and forth 30 times with an aperture of 0.8 mm. At the end of the fragmentation process, the fat was liquefied and was ready to be injected via a 1 mL syringe. Fat grafting was performed only once, as described by Piccolo et al. 12 Using a 16-gauge needle, a perforation was created at an acute angle in the healthy skin at the periphery of the scar. A 70 mm-long cannula with a 1.8 mm outer diameter, which was already connected to a 1 mL syringe, was inserted through the needle puncture hole and was immediately forcefully injected underneath the scar. Then fat was deposited in a retrograde manner, followed by sectoral injection until the entire area was injected. The fat graft filled the layer between the scaring and deep fascia. Further, 0.2–0.5 mL fat was deposited at one injection point, and 0.5–1 mL of fat on average was injected per cm2, depending on the hardness and location of the scar.
Scoring
In all patients, the Vancouver Scar Score (VSS) was evaluated during the preoperative clinical assessment, at 2–3 weeks after the surgical procedure according to the time of the first dressing change, and at 3 months postoperation by two senior independent plastic surgeons (FH and JH). Each patient scored their scar characteristics, such as color, pliability, thickness, relief, itching, and pain, while the referring physicians scored scar vascularization, pigmentation, pliability, thickness, and relief. In the 14 adult patients, the itch and pain scores, which consist of a numerical scale ranging from 1 to 10, where 10 corresponds to the worst symptom and 1 corresponds to normal symptom, were evaluated at the same time point as the VSS evaluation.
Data analysis
All scores are expressed as the mean ± standard deviation. The paired t-test was used to evaluate the mean pre- and postoperation values of the VSS, as well as the itch and pain scores. P values less than 0.05 were considered statistically significant. All statistical analyses were performed using the Statistical Package for Social Sciences (SPSS) Version 19 for Windows (IBM Corp: Armonk, NY).
Results
A total of 17 patients were involved in this study, including 13 males and four females, and the age of the patients ranged from 18 months to 62 years. There were four skin flaps in three cases, and 14 full-thickness skins were collected for transplantation in total. The size of the skin graft or flap was between 24 cm2 to 168 cm2 depending on the recipient and the tension of the donor site to be pulled and sutured. The fat volume obtained was between 20 ml and 100 ml according to the area of the donor. The formation of all scars occurred between 2 months and 11 months from burn injury. The skin or skin flap reconstruction sites were the face and neck in three cases, the axillary area in one case, the hand and upper limb in eight cases, and the foot and lower limb in five cases. All patients received nanofat grafting over a particular scar during the same operation; the recipient sites were the face and neck in three cases, the trunk in four cases, upper limbs in two cases, and lower limbs in eight cases. The donor areas for the full-thickness skin, skin flap, and fat were located on the abdomen. The characteristics of patients are summarized in Table 1.
Characteristics of patients.

The donor areas for the full-thickness skin, skin flap, and fat were located on the abdomen.
In all 17 patients with early postburn scarring treated with nanofat grafting, the pigmentation was reduced, scar hyperplasia was inhibited, and the flexibility of the scar was increased. The VSS and pain and itch scores showed a statistically significant reduction (P < 0.05) at the 2–3-week and 3-month postoperative follow-ups compared with the preoperation time point. The VSS decreased from 10.00 ± 2.12 to 7.41 ± 1.277 at the 2–3-week postoperation time point, and to 5.53 ± 1.37 at the 3-month postoperation time point. The pain score decreased from 4.65 ± 1.37 to 3.70 ± 1.10 at the 2–3-week postoperation time point, and to 3.00 ± 1.28 at the 3-month postoperation time point. The itch score decreased from 6.35 ± 1.27 to 4.94 ± 1.30 at the 2–3-week postoperation time point, and to 3.94 ± 0.97 at the 3-month postoperation time point (Table 2).
Scar assessment of pre and post operation.

VSS, Vancouver Scar Score; *Significant difference between the postoperation and preoperation groups, P < 0.05.
A 62-year-old male recovered from burn injury on the face and developed scarring on his eyelids with lagophthalmos at 2 months from the injury. There was a scar on his left temple with ulcers and the orange peel sign. The VSS was 11 (Figure 1(a)). The patient complained of severe itching and pain in the temporal scar, with an itch score of 8 and a pain score of 6. He underwent excision of the scar on the eyelid and autologous full-thickness skin reconstruction. Nanofat grafting was conducted at the same time on the left temporal scar. When the patient underwent a dressing change for the first time at 2-week postoperation, all ulcers were healed, the VSS was reduced to 6 (Figure 1(b)), and both the itch and pain scores were reduced to 4. The scar became softer and was less visible at the 3-month postoperation follow-up, and the VSS, itch score, and pain score were reduced to 4, 2, and 2, respectively (Figure 1(c)).
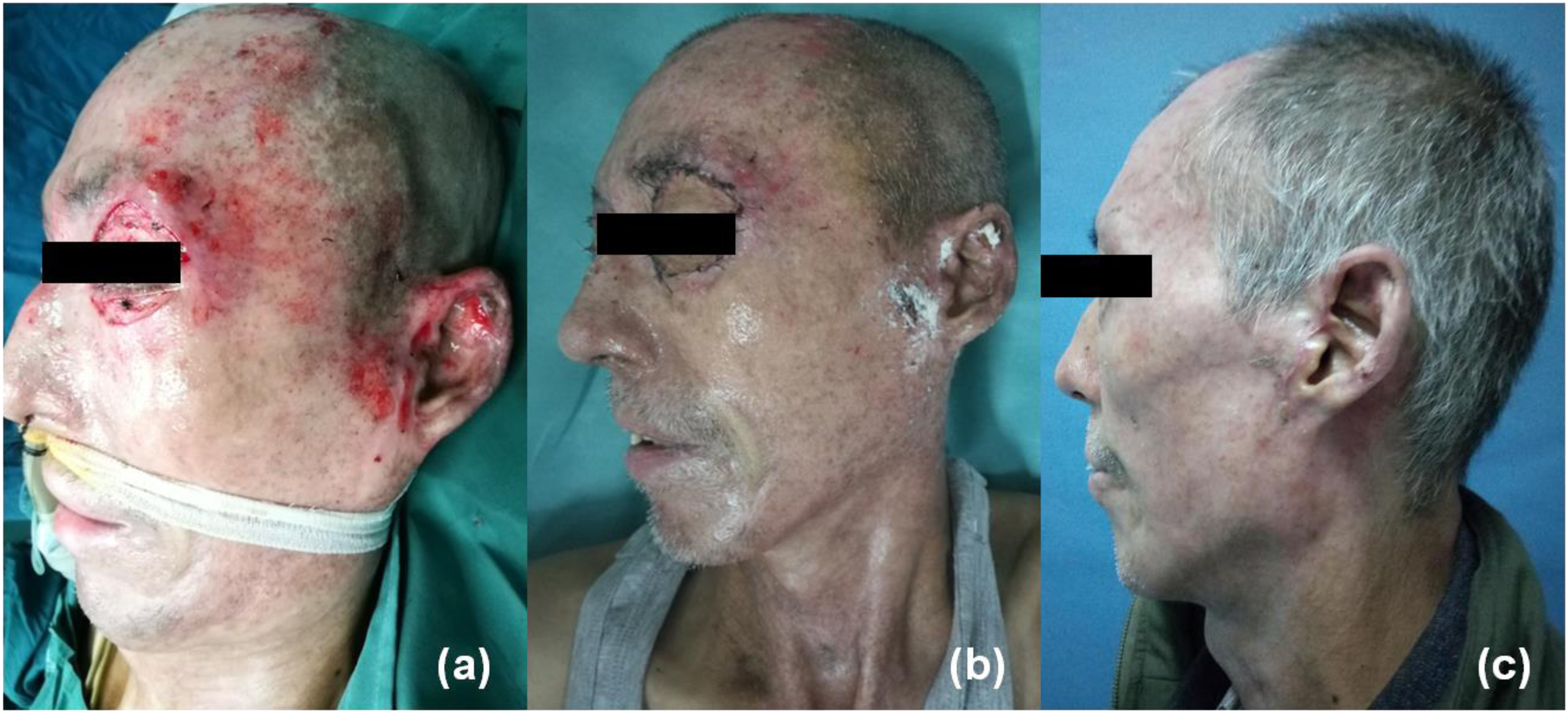
(a) A 62-year-old male suffered from postburn scarring 2 months from the burn injury with eyelid deformities, limiting the closure of the eyelids. The patient underwent excision of the scar on the eyelid and autologous full-thickness skin reconstruction. Nanofat grafting was conducted at the same time on the left temporal scar. (b) When he underwent a dressing change for the first time at 2-week postoperation, all ulcers had healed and the scar had become softer and less visible. (c) The scar was much lighter at the 3-month postoperation follow-up.
An 18-month-old boy suffered a burn injury on his right lower limb 4 months before. He was diagnosed with 12% total body surface area burned, deep second degree burn depth wound. Doctors treated his wound with silver ions dressing for the first 3 weeks, then recommended he undergo skin transplantation surgery to repair the wound and to prevent scar hyperplasia after wound healing on knee and ankle. However, his parents refused the surgery and treated the wound with traditional Chinese medicine dressing. After about another 3 weeks, the burn wound healed. Then the boy gradually developed scaring on the right lower limb. Moreover, the parents found it difficult to passively straighten his knee and ankle at 4 months from injury. The VSS was 14 at the preoperation time point (Figure 2(a)). Full-thickness skin reconstruction was conducted on his ankle and knee, and nanofat grafting was conducted on his thigh at the same time. The scar became softer, the scar color changed from red to pink at the 2-week postoperation follow-up, and the VSS was reduced to 10 (Figure 2(b)). Then the scar became much softer and lighter at the 3-month postoperation follow-up, and the VSS was reduced to 5 (Figure 2(c)).
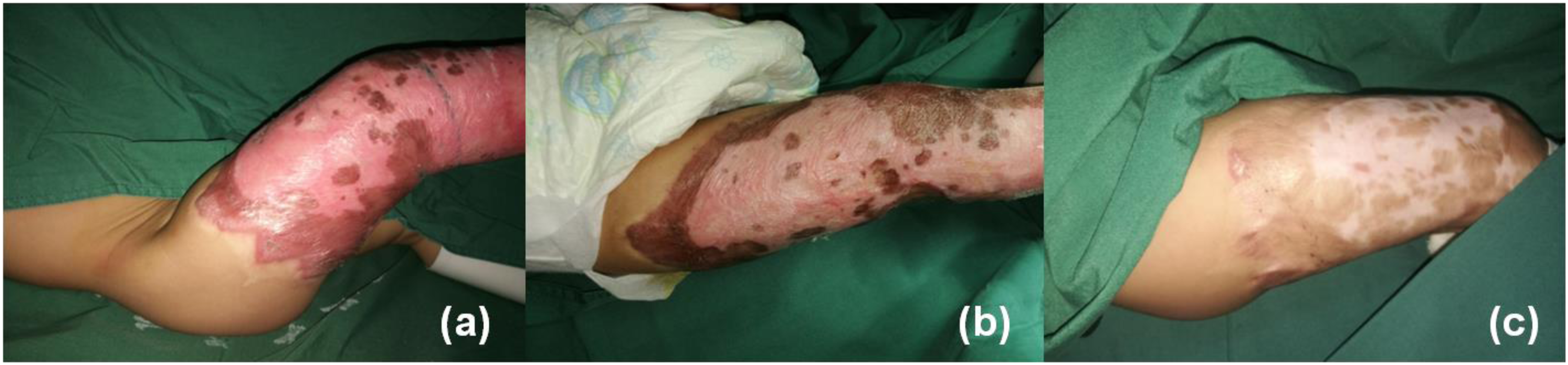
An 18-month-old boy developed a hypertrophic scaring 4 months ago on his right lower extremity, along with deformities and dysfunctions of his ankle and knee. Full-thickness skin reconstruction was conducted on his ankle and foot, and nanofat grafting was conducted on his right thigh at the same time. Images of the boy's right lower extremity at (a) preoperation, (b) 2-week postoperation, and (c) 3-month postoperation are shown.
A 28-year-old man was burned with flames on both hands 8 months before and underwent dressing change treatment with silver-containing dressing for the first 3 weeks. Then, he underwent wound excision and medium thickness skin transplantation on both hands to repair the remaining wounds, while preserving the already healed wounds, including both wrist joints. After surgery, silicone and elastic gloves were used for routine anti-scar treatment for 6 months. However, except for the area that received skin grafting, there was significant scaring on both hands, with metatarsophageal joint dorsiflexion deformity and hypertrophic scaring on bilateral wrist at the time point of 8 months from the injury. The VSS was 12 at the preoperation time point (Figure 3a and d). He underwent bilateral abdominal skin flap reconstruction on the back of both hands and fat grafting under the scar on both wrists (one was located on the palmar surface, and the other was located on the dorsum). The scars on both wrists were softer and lighter at 3 weeks after he underwent skin flap pedicle division compared to those at preoperation, the VSS was reduced to 9, the itch score was reduced from 7 to 4, and the pain score was reduced from 6 to 4 (Figure 3 b and e). The scar was much softer and lighter at the 3-month follow-up, with mild itching and pain. The VSS, itch score, and pain score were reduced to 6, 2, and 2, respectively (Figure 3c and f).
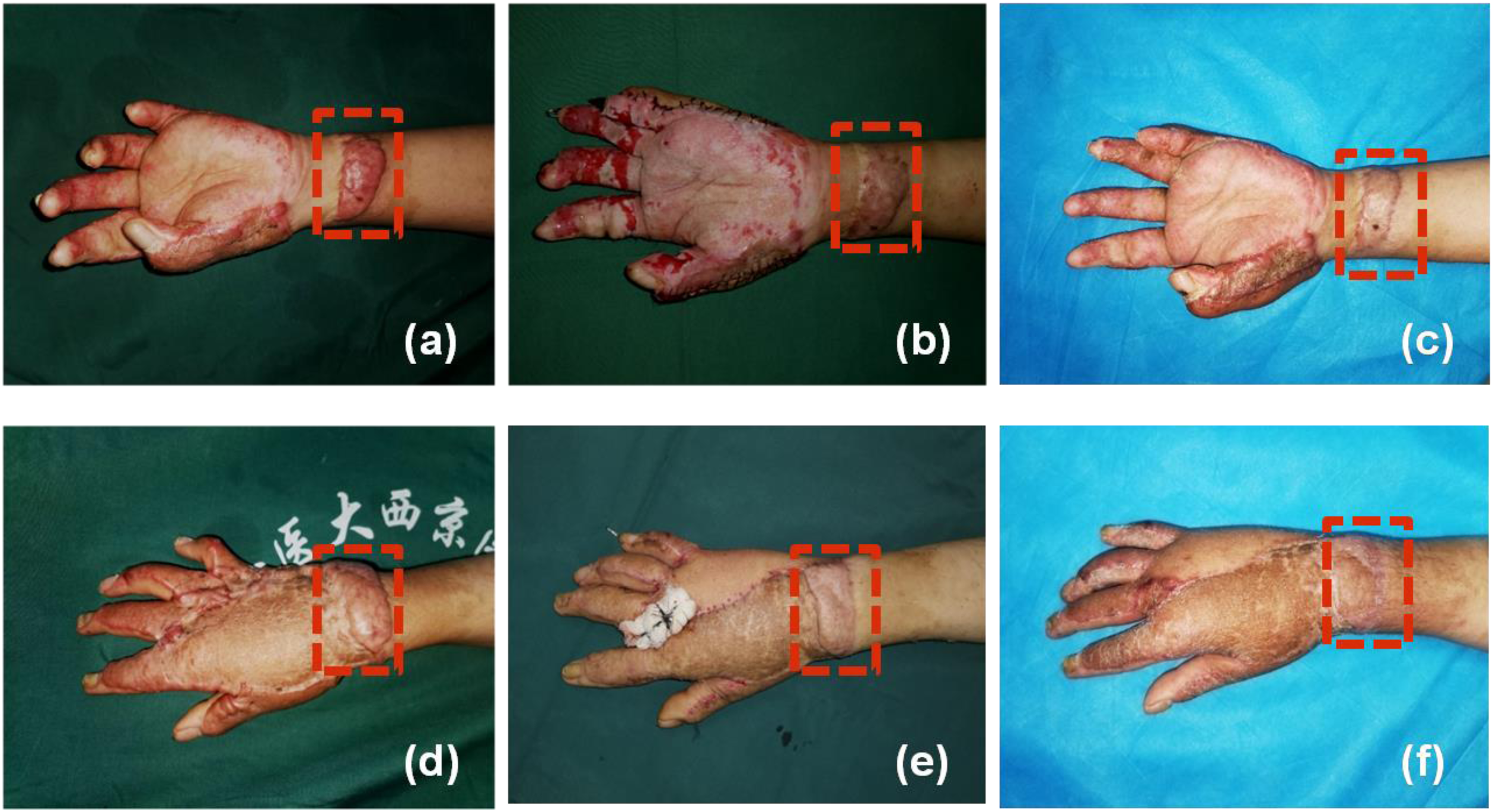
A 28-year-old male developed hypertrophic scaring on both hands over a period of 8 months. He underwent bilateral abdominal skin flap reconstruction and nanofat grafting under the scars on both wrists. The red rectangle shows the fat grafting sites. a, b, and c show the left hand, and d, e, and f show the right hand. Images were obtained at (a, d) the preoperation, (b, e) 3-week postoperation, and (c, f) 3-month postoperation time points, respectively.
Discussion
Postburn hypertrophic scarring, which is clinically characterized by a red-to-purple color, elevated height, decreased pliability, tenderness, pain, and itching, generally develops at around 2 months after burns; in addition, the remodeling phase, including hyperplasia, stabilization, and atrophy, lasts for a year or longer. 10 Researchers have found that hypertrophic scar maturation may not occur until 18 or even 24 months after a burn. 22 Many factors, such as heredity, scar location, and mechanical tension, contribute to pathological scar development, which depends on the interaction of a variety of cells, growth factors, and inflammatory factors. 23 Several lines of evidence support that a persistent, excessive inflammatory reaction in the reticular layer of the dermis disrupts the balance between collagen synthesis and degradation, resulting in typical hyperplasia of fibroblasts and excessive deposition of the extracellular matrix.24,25 Especially in the early stage of scar formation, many inflammatory cells, such as neutrophils, mast cells, macrophages, and T lymphocytes, infiltrate into the tissue, and they regulate or secrete numerous proinflammatory factors, including interleukin IL-1, IL-6, and tumor necrosis factor-α.26,27 Just as proinflammatory cytokines are linked with fibrosis, the opposite is also true about the protective activity of anti-inflammatory cytokines against fibrosis. A steroid injection is one of the most effective clinical treatments for a hypertrophic scaring or keloid because it inhibits the inflammatory reaction. 28
Over the past few decades, more attention has been paid to the role of adipose-derived stem cells (ADSCs) in diseases caused by an imbalance in complex immunoregulatory networks. Several studies have confirmed that ADSCs can reprogram the inflammatory response, and they have been suggested as an alternative to immunosuppression in enteritis, osteoarthritis, systemic sclerosis, idiopathic pneumonia, chronic liver fibrosis, renal fibrosis, and other diseases. 29 Specifically, transplanted ADSCs have been shown to reduce inflammation, promote angiogenesis, and promote the proliferation and migration of cells involved in wound healing and scar formation. 30 ADSCs can also regulate the surrounding immune microenvironment by continuously releasing extracellular components, such as miRNA and growth factors. 31 Nevertheless, to date, there is no clinical evidence for the use of ADSCs in the treatment of a hypertrophic scar. However, adipose tissue consists of several cell types, including adipocytes, immune cells, and ADSCs. 32 The therapeutic effect of fat grafting may be achieved through ADSCs that regulate the inflammatory response. 33
For more than 100 years, autologous fat has been clinically used as a substitute for tissue filling, and it is mainly used in the treatment of concave defects, breast augmentation, and hip augmentation. 11 Due to improvements in liposuction and injection technology proposed by Coleman, 34 autologous fat grafting is also widely used in wound healing, skin rejuvenation, and scar treatment. 15 Recent publications have indicated that fat grafting significantly reduces pain and itching within the scar, increases the flexibility of the scar, and improves the satisfaction of patients.12,16,17,35,36 From a histological point of view, autologous fat grafting has shown the ability to regenerate the dermis and stimulate neosynthesis of collagen fibers at the recipient site; in particular, it has the ability to improve the dermal and dermohypodermic qualities in scar areas.37,38 However, some prospective, randomized, double-blind, controlled studies have found that autologous fat grafting cannot improve the appearance and function of burn scars.18,19 The probable reason for this finding may be that mature scars (formed more than 1 year ago) were selected in these clinical studies. Most hypertrophic scars gradually improve over a period of a few years, and mature scars are characterized by a reduced proinflammatory and hypervascularized status, with elasticity and color relatively similar to normal skin. To date, no research has clarified whether the scar age and etiology have any influence on the success of nanofat treatment. Of note, all patients treated in the current study were in the early stage of scar formation after burn-wound healing and were currently experiencing dysfunction, especially with obvious red swelling, itching, pain, and other symptoms. From a clinical point of view, there was a significant improvement in both functional and aesthetic outcomes of the scar treated by fat grafting. Although the precise mechanism by which fat grafting induces tissue improvement is still unclear, its effect on mature scars may be related to the limitation of ADSCs in regulating the subinflammatory reaction.
Full-thickness skin or skin flap reconstruction is the most common method for the treatment of hypertrophic scaring. The subcutaneous fat is always trimmed and discarded during the preparation of the full-thickness skin or skin flap. In the current study, we harvested fat and injected it in the scar located at a nonsurgical site. Fat can be easily harvested without extra-surgical intervention or donor site morbidity. Comparison of the pre-and postoperation scores for scar color, scar thickness, scar stiffness, and scar regularity showed that the postoperation scores were decreased significantly and that there was a significant improvement in scar pigmentation and thickness as well as the aesthetic outcome after treatment. Most importantly, reductions in the scores for pain and itching could be assessed objectively. It seems that the fat can be reused to treat the scaring.
The limitations of this study should be acknowledged. It was a retrospective, nonrandomized case series. Patients were not compared to a control group without fat grafting or treated with other injectables. Evaluation of the results was performed clinically without any objective measurement. In addition, full thickness skin or skin flap reconstruction might also change the biomechanical direction of collagenous fiber, resulting in change in overall scar quality. In the future, long-term, separately performed nanofat grafting, randomized controlled trials with analgesic scores as the primary outcome measure are required to assess the long-term efficacy of this technique in order to obtain high-level evidence to support its standard use.
Conclusion
Autologous nanofat grafting from donor sites of full thickness skin or skin flap may be a promising treatment for an early postburn scaring as it promotes scar softening, improves itching and pain within the scar. However, this is a small case series with only 17 patients. Further conclusions need to be drawn through expanded samples for randomized controlled clinical trials.
Footnotes
Acknowledgements
We thank Medjaden Bioscience Limited Corporation for significantly improving the English language of this manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical statement
This study was approved by the Medical and Ethics Committee of Xijing Hospital, Xi’an, China. All methods were carried out in accordance with the relevant guidelines and regulations. Informed consent was obtained from all subjects and/or their legal guardian(s). All patients and/or their legal guardian(s) provided written informed consent for publication and use of their photographs in the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China, (grant number 81971835, 81901965).
How to cite this article
Wang H, Liu Y, Li Y, et al. Autologous nanofat harvested from donor site of full-thickness skin or skin flap grafting for the treatment of early postburn scarring: a case series. Scars, Burns & Healing Volume 10, 2024. DOI: 10.1177/20595131241230739.
