Abstract
Introduction
Reactivation of pulmonary tuberculosis is a prevalent concomitant infection and cause for mortality in burns patients in Sub-Saharan Africa. First line laboratory diagnostic studies for pulmonary tuberculosis are often negative in these patients and if relied on, result in high mortality. The purpose of this report is to share our experience with a novel protocol of empiric treatment of clinically suspected pulmonary tuberculosis in severely burned patients with negative GeneXpert tests in a tertiary burns centre and to present a brief literature review on the topic.
Methods
A retrospective chart review of all patients, who sustained thermal injury with an inhalation component, with negative GeneXpert tests who were treated empirically for pulmonary tuberculosis over a five-year period (2015–2020) was performed. Additionally, a literature search was performed on Medline (PubMed), Cochrane and Google Scholar databases.
Results
Over the five-year period, 20 patients with suspected pulmonary tuberculosis and severe burns requiring ventilation were managed according to the protocol and all survived to discharge. The literature search identified six factors that explain the consequence of pulmonary tuberculosis in severely burned patients and provide a hypothesis for the negative laboratory studies encountered.
Conclusion
There was an improved outcome for patients with the clinical diagnosis of reactivation of pulmonary tuberculosis when they were started on empirical pulmonary tuberculosis treatment. There are several potential mechanisms that can contribute to reactivation of pulmonary tuberculosis in susceptible severely burned patients. The GeneXpert test should not be relied upon in these patients for a diagnosis, but rather all other clinical evidence should inform management.
Lay Summary
From the current literature evidence most patients who have severe burns complicated by a secondary infection known as pulmonary (lung) tuberculosis, die as a result. The purpose of this report is to share our experience with treatment of pulmonary tuberculosis during a five-year period, from 2015 to 2020, in a specialized adult tertiary burn center. Severely burned patients who were suspected of having pulmonary tuberculosis and received treatment despite having negative laboratory tests for pulmonary tuberculosis, had a better survival rate than expected. A brief literature review on the topic of pulmonary tuberculosis and severe burns was done to investigate causes for reactivation of pulmonary tuberculosis and negative laboratory studies in these patients.
The literature search identified the following factors that can potentially affect the reactivation of pulmonary tuberculosis in severely burned patients: decreased immune system; secondary infections; low blood albumin(protein) levels; decreased clearance of bacteria from the airways, the development of pulmonary tuberculosis biofilms (capsules that protect the bacteria from chemicals and antibiotics) and the role of a fat molecule called phosphatidylinositol mannoside in pulmonary tuberculosis.
In conclusion, the pulmonary tuberculosis test should not be relied upon in these patients, but rather all the clinical evidence such X-ray changes in association with difficulty to wean the patient off the ventilator should be used to inform treatment choice.
Introduction
The Western Cape Provincial Adult Tertiary Burn Centre is located at Tygerberg Hospital in Cape Town South Africa. It is the only Burn Centre dedicated to the management of severe burns for the 7.2 million people of the Western Cape Province. All patients older than 13 years old are eligible for admission. Every year, approximately 110 patients are admitted to the Intensive Care Unit. The average length of stay of patients is 2.1 days per percent total body surface area (TBSA) burnt. The average TBSA burn injury seen in our department is 22.5%. The incidence of HIV among the patients is about 12%. 1 Pulmonary tuberculosis (PTB) is a prevalent concomitant infection and cause for mortality in the Western Cape population in South Africa. 2 Patients with severe immunosuppression are at risk of reactivation of PTB. Thermal injuries are widely known to cause severe immunosuppression and consequently, patients with thermal injury with an inhalation component requiring ventilation are at risk of reactivation of PTB. Thermal injury patients who are human immunodeficiency virus (HIV) positive are known to have a high mortality rate secondary to concomitant infection.1,3,6
There are several tests available for the diagnosis of PTB. Sputum sent for a PTB culture is generally considered the gold standard diagnostic test for PTB, however, it typically takes six weeks before the result is obtained. By that time most patients will have passed away due to the severity of their injuries and subsequent infections. Microscopy is commonly used but has low sensitivity in comparison to GeneXpert, which is a quick and sensitive tool for early diagnosis of PTB. In a retrospective review study by Agrawal et al. 7 they found that GeneXpert had a higher sensitivity than smear microscopy (79.1%) in respiratory samples. The overall sensitivity, specificity, positive predictive value (PPV) and negative predictive value (NPV) of GeneXpert were 86.8%, 93.1%, 78.5% and 96% respectively in this study. 7 In addition GeneXpert testing is more cost effective than Line probe assays and is recommended by the current South African Guidelines as first line in the diagnosis of PTB in symptomatic patients. 8 GeneXpert studies are therefore a valuable tool, commonly used to guide diagnosis and treatment for PTB in non-burns patients. However, in our Tertiary Adult Burns Centre, we were faced with the dilemma of having negative GeneXpert tests for PTB in severe burned, immunosuppressed patients with inhalational injury where, based on clinical evidence, reactivation of PTB was suspected.
This led us to adopt a policy of bedside diagnosis of the reactivation of PTB and the immediate initiation of empiric treatment, despite a negative GeneXpert. Classic clinical diagnostic features of TB include persistent cough, night sweats, weight loss, loss of appetite, fatigue and malaise. 8 Unfortunately, these features are difficult to observe in intubated patients with inhalational burns and it was necessary that other clinical criteria be considered to inform diagnosis in this group when developing the protocol. In addition to clinical features, X-ray was a helpful tool in this cohort. Upper lung cavitation is a classic sign of reactivation PTB and remains a useful diagnostic tool in ventilated burns patients to distinguish PTB from Acute respiratory distress syndrome (ARDS) and community acquired pneumonia. 9 While, it is important that other causes for cavitation should be considered, PTB's classic upper lobe location and high prevalence in South Africa make it the most likely diagnosis in this group. The differential diagnosis for lung cavitation is summarized in Table 1.10,12 In addition, miliary TB has a different radiological presentation, typically featuring numerous small nodules throughout both lung fields.9,13 While TB's classical X-ray presentation is valuable in the clinical diagnosis of TB, lung damage in ventilated patients with inhalational burn injury can obscure features and make interpretation difficult.
Lung cavitation differential diagnosis
Given the challenges in establishing diagnosis of PTB in ventilated patients with inhalational injury it is important that a combination of clinical and radiological features be considered and a high index of suspicion maintained. In our experience, GeneXpert's are often negative in patients with severe burns and if relied on in this patient group, may lead to under treatment in this patient population and higher mortality. For this reason, a protocol to aid in diagnosis and treatment was developed based on the literature search in conjunction with expert opinion and multi-disciplinary team (MDT) discussion.
The purpose of this report is to share observations and experience with a novel protocol of PTB diagnosis in ventilated patients with inhalational injury looking at outcomes following empiric treatment of PTB in a Tertiary Adult Burns Centre in South Africa and to present a brief literature review that explores the factors leading to reactivation of PTB with negative laboratory findings in burns patients.
Methods
A retrospective chart review of all patients, who sustained thermal and inhalation injury, requiring ventilation, with negative GeneXpert tests who were treated empirically for PTB over a five-year period (2015–2020) was performed.
Data on clinical presentation and treatment outcome were collected. A descriptive summary of findings is presented. A literature search combining variations of the terms “tuberculosis” and “burns” was performed on Medline (PubMed), Cochrane and Google Scholar databases, with the findings qualitatively assessed.
Results
Over the five-year period, 20 patients with a clinical diagnosis of PTB, despite a negative GeneXpert screening test, and severe burns requiring ventilation survived and were discharged. This included HIV positive patients with CD4 counts as low as 20. All patients had burns that were > 35% total body surface area (TBSA) and all were managed according to the protocol and treated empirically for PTB based on clinically diagnostic features described in the protocol. Eight of these patients were later found to have laboratory evidence of PTB on culture. In the same five-year period, five patients who tested positive on GeneXpert were treated for PTB. Thus, the ratio of positive GeneXpert to negative GeneXpert for patients with a clinical diagnosis of reactivation PTB in critically ill burn patients was 1:4 (5:20).
Our observations revealed that patients with negative screening tests (microscopy for acid-fast bacilli and GeneXpert tests) for PTB, but who had chest X-ray features suspicious of reactivation of PTB in conjunction with ventilatory difficulty (including difficulty weaning off ventilators, failed extubation attempts, requiring re-intubation, or just prolonged ventilation) highly likely have reactivation of PTB. Figures 1 and 2 have been included as examples from our patient cohort of X-ray features suspicious of TB observed in severe burns patients with negative GeneXpert studies.

Chest X-ray of miliary tuberculosis.

Chest X-ray showing upper lung cavitation.
This common clinical pattern observed in severe inhalational burns patients with suspected reactivation PTB formed the basis of the clinical diagnosis in the protocol (Table 2). In those patients, with the common pattern of failed ventilator weaning and negative TB tests, that were then started on empiric TB treatment, rapid improvement was noted. It is important to note in these patients, that other opportunistic infections such as herpes virus and fungal infections must also be excluded when treating severely immunosuppressed patients.
The common pattern of reactivation PTB in severe burned patients on ventilators.

The accompanying literature search identified 39 applicable articles that helped explain the prevalence and effect of PTB in severely burned patients and provided a hypothesis for the negative laboratory studies for PTB in patients with severe burns. Six factors were identified through this search are summarized in Table 3.
Factors predisposing to reactivation PTB and negative laboratory evidence in patients with severe burn injuries.

Discussion
Due to the significant mortality associated with inhalational burns observed in our institution and others, as well as the observed survival benefit of empiric pulmonary tuberculosis treatment in selected patients, it was decided to do a literature review to see whether there was any evidence to explain our findings and rational for initiating treatment despite negative laboratory evidence in patients with severe burn injuries. Six factors were identified as possible explanations for these findings and are described below.
Immunology in severe burns
It is well known that severe burn injury is associated with massive immunosuppression.14,17
A study using mice conducted by Parry et al. illustrated that traumatic injury decreases glutamine concentrations which, in turn, decrease human lymphocyte proliferation and macrophage phagocytosis. 14 Additionally, following burn injury, macrophages increase their production of inflammatory mediators such as nitric oxide, prostaglandins, tissue necrosis factor (TNF) alpha and interleukin (IL) 6 leading to further immune suppression. The result of this in major burn wounds is a systemic inflammatory response syndrome (SIRS), which is an excessive inflammatory response that predisposes to sepsis, further tissue damage and multiple organ failure. 15
In PTB, the cellular immune system acts to block disease extent to less than four to six weeks by the formation of granulomas consisting of macrophages and giant epithelioid cells. This leads to a delayed hypersensitivity response where cytotoxic T cells (CD8 cells) cause degeneration of the granuloma centre. CD8 cells are sparser than CD4 (T helper) cells, which dominate granulomas, and tend to lie more on the periphery and serve an immunosurveillance function.18,19 CD4 cells, and to a lesser extent CD8 cells and natural killer cells, function by producing cytokines interferon (IFN) gamma and alpha that in turn recruit and activate innate immune cells (monocytes and granulocytes). 18 CD8 cells possess antimicrobial mechanisms including release of cytokines and cytotoxic molecules leading to apoptosis of target cells. 19 Figure 3 summarizes the cellular immune response to mycobacterium tuberculosis.
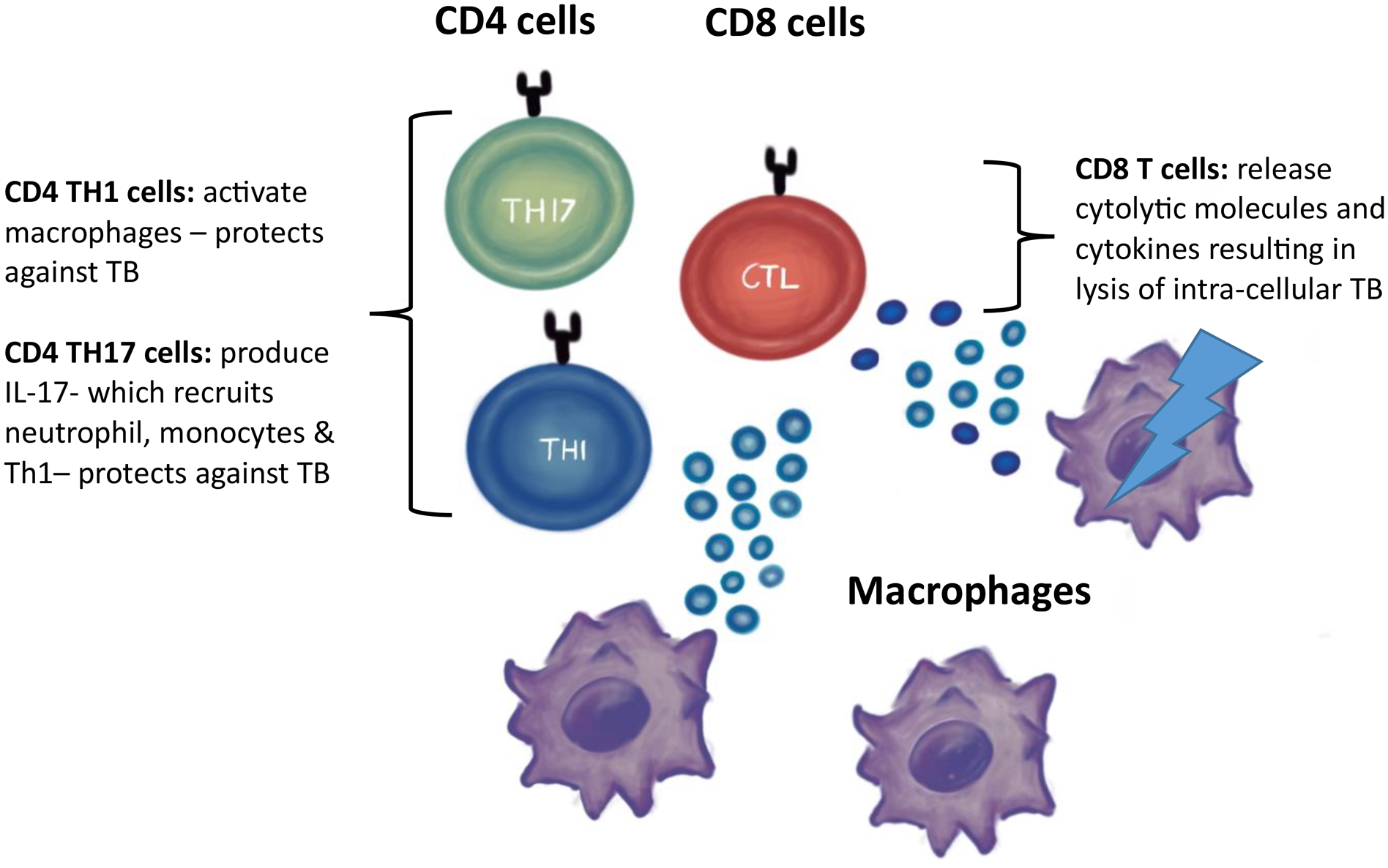
Conventional T cells involved in the immune response to mycobacterium TB.
Watanabe et al. describe another mechanism by which chronic inflammation may lead to compromised immune function due to impaired T-cell response, via various inflammatory proteins, including sIL-2R (soluble interleukin 2 receptor), VEGF (Vascular endothelial growth factor) and IL-17. 20 Watanabe et al. continue to surmise that the main mechanism leading to cachexia in cancer patients appears to be primarily due to the immune system reaction to chronic inflammation. 20
We postulate that it is the immunosuppression from the burn injury and its sequelae that leads to the reactivation of the PTB in susceptible patients.
Infections contribute to mortality in severe burns
Because of this impairment in function and proliferation of CD4 cells in the acute phases of severe burn injury, the practice of stopping HIV medication in severely burned patients coinfected with HIV has been adopted.1,21 In our unit it has been observed in this patient population that CD4 counts can drop to below 20 from the burn injury alone.
Chalya et al. found that mortality rate was independently and significantly associated with CD4 count, patient age, presence of AIDS, inhalation injury, % TBSA (Total body surface area) and severity of burn. 1 James et al. further stipulated that HIV status and TBSA were independent predictors of CD4 count. 4
However, the outcome in HIV positive and HIV negative severe burn patients has been proven to be more or less the same.21,23 The only exception here is in patients who have AIDS and severe burns where the reported mortality was 100%.3,22
The current understanding with infection control specialists in our hospital is that the patients should restart their HIV medication at their local clinic when discharged. This is done to avoid patients having to change medications because clinics do not have the same medication as the provincial hospitals and this protocol hopes to ensure better compliance.
The role of hypoalbuminemia
Albumin is the body's most abundant serum-binding protein. It contributes 75–80% of normal plasma colloid oncotic pressure and 50% of protein content. Hypoalbuminemia leads to oedema as the body is no longer able to sustain sufficient colloid osmotic pressure to counterbalance hydrostatic pressure.
Another consequence of hypoalbuminemia is that drugs that are usually protein bound are free in the plasma, allowing for higher blood drug levels or more rapid hepatic metabolism. Additionally, alterations in albumin level affect platelet function. 24
It is likely that the immunosuppression, in combination with hypoalbuminemia/ hypoproteinaemia, plays a role in the negative laboratory results seen in patients tested for PTB, however more research is needed on this topic.
Bacterial clearance in severe burns
Clearance of bacteria from the lungs in ARDS could also play a role in PTB reactivation and negative PTB laboratory tests.25,27
ARDS in severe thermal injury developed as a result of inhalation injury leading to diffuse alveolar damage. The injury causes release of proinflammatory cytokines (TNF and interleukin 1, 6 and 8)28,29 and recruitment of neutrophils which in turn releases proteases and reactive oxygen species that damage alveolar and blood vessel walls.30,31 This causes fluid to leave the vascular space and fill alveolar gaseous exchange spaces with fluid and debris. Lastly, the alveoli collapse due to loss of surfactant.
Researchers have examined the clearance of bacteria in mice.26,27 These studies found that removal of bacteria is chiefly achieved by the phagocytic activity of macrophages and only a small amount of clearance is due to mechanical removal of bacterium. Green et al. go on to describe how pulmonary clearance is impaired by immune suppression. 26
In the case of severe thermal injury, both the direct inhalation burn injury and resultant ARDS, as well as the indirect effect of burn injury on immunity and thus bacteria clearance, could increase the risk of both primary and reactivation PTB. 27
The development of biofilms in PTB
The development of biofilms in the airways where the PTB can grow without being dislodged or detected could play a role as well.32,36
It has recently been established that like multiple other microbial species, Mycobacterium tuberculosis too forms biofilms.28,32 Biofilms are an organized assembly of microbes which are encapsulated by a matrix with an ability to attach to surfaces. These biofilms incur tolerance to antimicrobials and other environmental stresses in addition to complicating diagnosis.
The role of phosphatidylinositol mannoside in PTB
Phosphatidylinositol mannoside (PIM) is integral in the composition of the mycobacterial envelope. PIM2, the mycobacterial envelope antigen, has many biological functions, including fostering active macrophages which in turn activate protein kinases, activator proteins and nuclear factor-κB (NF-κB). PIM also has granuloma-forming properties and leads to the recruitment of NKT cells into those granulomas. Finally, PIM induces adherence of Tuberculosis bacilli to nonphagocytic cells and initiates the inflammatory response. 37
In summary, through the literature review, six mechanisms which could contribute to reactivation of PTB in severe burn patients were identified. Further research, by means of a detailed case-cohort analysis of the clinical observations and laboratory results looking at the benefit of empiric PTB treatment for selected patients will add value to the current body of knowledge.
Limitations
The retrospective chart review looked only at survivors to determine if the protocol had improved outcomes in patients who previously had significant, and in the case of HIV, up to 100% mortality. A review of patients who did not survive is still needed to determine the percent mortality in this patient group with consideration for the impact of TBSA burn on outcome. Additionally, other factors other than PTB treatment may have increased survival. There are also other bacterial infections that may have responded to the empiric TB treatment and led to the improved outcomes observed.
Finally, a global survey in collaboration with the WHO with an accompanying robust systematic review of global policies and outcomes in these patients is needed.
Conclusion
In our clinical experience a common pattern in patients with inhalation burn and negative PTB laboratory tests was observed. There was an improved outcome for these patients with the clinical diagnosis of reactivation of PTB when they were started on empirical PTB treatment. The literature shows that there are several potential mechanisms that can contribute to reactivation of PTB and negative PTB screening tests in susceptible severely burned patients. The GeneXpert test should not be relied upon in these patients for a diagnosis, but rather all other clinical evidence such as X-ray changes and the clinical course of the patient should be used to inform treatment choice.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article
How to cite this article
Kulenkampff C, Kleintjes WG, Kotzee EP and Kankam H. Protocol for clinical diagnosis and empiric treatment of pulmonary tuberculosis in severely burned patients: observations and literature review. Scars, Burns & Healing, Volume 9, 2023. DOI: 10.1177/20595131231175794

