Abstract
Monoclonal gammopathy is a premalignant condition associated with an abnormal circulating immunoglobulin indicative of an expanded B cell clone. Apart from monitoring, no other intensive management is prescribed in these cases. Periodic bone marrow biopsies when free light chain imbalances are detected are used to spot incidental cases of multiple myeloma. New reports have suggested the existence of a monoclonal gammopathy of renal significance where a circulating antibody and normal bone marrow biopsy results may be associated with proteinuric renal disease due to monoclonal immunoglobulin deposition. We report a case of a 65-year-old male with an immunoglobulin G kappa monoclonal gammopathy of undetermined significance, chronic kidney disease, proteinuria, and an initial inconclusive renal biopsy. His chronic kidney disease worsened with persistence of 0.5 g of proteinuria. Given the finding of ongoing Bence Jones proteinuria, a repeat renal biopsy was done revealing monoclonal immunoglobulin G kappa deposition disease. Bone marrow biopsy showed 20% plasma cells that could be consistent with smoldering myeloma. The patient’s renal disease has stabilized after starting treatment for the monoclonal gammopathy of renal significance. This case illustrates the importance of renal biopsy in making the diagnosis of monoclonal immune deposition disease and monoclonal gammopathy of renal significance.
Introduction
Monoclonal gammopathy of undetermined significance (MGUS) is a premalignant condition characterized by an abnormally high concentration of a circulating immunoglobulin detected in the blood and sometimes in the urine. Epidemiologically, 3% of patients older than 50 years old develop MGUS with a 1% per year risk of developing malignancy. 1 The presence of the abnormal immunoglobulin is indicative of an expansion of a clone of B-cells which produce the circulating immunoglobulin at the abnormal level. The finding of light chains in the urine is referred to as Bence Jones proteinuria. 2
The free light chain kappa to lambda ratio and the concentration of the M-spike on serum and urine electrophoresis are the usual noninvasive tests that can track the progression of MGUS and indicate incident multiple myeloma. 3 The finding of a rising specific monoclonal protein (M-protein) level and a skewed kappa to lambda ratio (>2 kappa:1 lambda or <1 kappa:2 lambda) indicates a higher likelihood of transition from a premalignant to a malignant disorder, and is an indication for bone marrow biopsy. 3 Hypercalcemia, worsening anemia, worsening chronic kidney disease (CKD), and proteinuria can often indicate renal injury from immunoglobulin deposition. Bone marrow biopsy often is indicated in patients with these findings. 4
An entity recently described might necessitate a renal biopsy rather than just a bone marrow biopsy. 5 Monoclonal gammopathy of renal significance (MGRS) is a newly coined pathological term to describe patients with MGUS who develop CKD and proteinuria due to immunoglobulin deposition similar to myeloma deposition disease syndromes. 5 These cases became evident after multiple incidences demonstrated renal impairment and pathological changes typical of myeloma kidney in patients who may not meet bone marrow biopsy criteria of multiple myeloma. These presentations are treated with chemotherapy like multiple myeloma to prevent worsening renal dysfunction and proteinuria. 6
We report a case of a 65-year-old male with a circulating kappa light chain monoclonal gammopathy who presented with worsening CKD and stable proteinuria. An initial renal biopsy was inconclusive, but with worsening of renal function, a repeat renal biopsy showed monoclonal immunoglobulin deposition disease (MIDD). A bone marrow biopsy confirmed incident multiple myeloma with 20% plasma cells leading to initiation of treatment with dexamethasone, bortezomib, and lenalidomide.
Case report
The patient is a 65-year-old Caucasian male with a history of CKD (stage IIIb down to stage IV) with an estimated glomerular filtration rate (eGFR) (non-African American) of 30 mL/min in 2015 decreased to 23 mL/min as of 11 May 2018. Initially, he was thought to have a renal disease of unknown etiology. The patient had a history of bipolar disorder, but no history of lithium treatment and hypertension diagnosed only 1 year prior. His original workup for proteinuria in 2015 revealed a negative serum protein electrophoresis and no M-protein.
The patient had originally been biopsied in another institution that revealed only one glomerulus on electron microscopy slide, without any glomeruli on the light microscopy core section, and five-sixths glomeruli on the immunofluorescence slide showing complete sclerosis. He presented to University of California, Los Angeles (UCLA) with worsening renal function without hypercalcemia and with mild to moderate proteinuria (0.9 g protein/gram creatinine). His urinalysis showed 2+ proteinuria on urinalysis, without any indication of lambda kappa mismatch. A 24-h proteinuria was noted to be at 552 mg/24 h. There was no evidence of anemia (hemoglobin of 17.2 mg/dL).
Serum protein electrophoresis and urine protein electrophoresis showed no evidence of an M-protein, and serum immunofixation showed no monoclonal immunoglobulin. Urine immunofixation, however, revealed the presence of a kappa monoclonal immunoglobulin. Despite only this mild abnormality on immunofixation, a severely imbalanced kappa to lambda ratio of 28.34 was noted on free light chain assay. Figure 1 summarizes the patient’s relevant laboratory data.
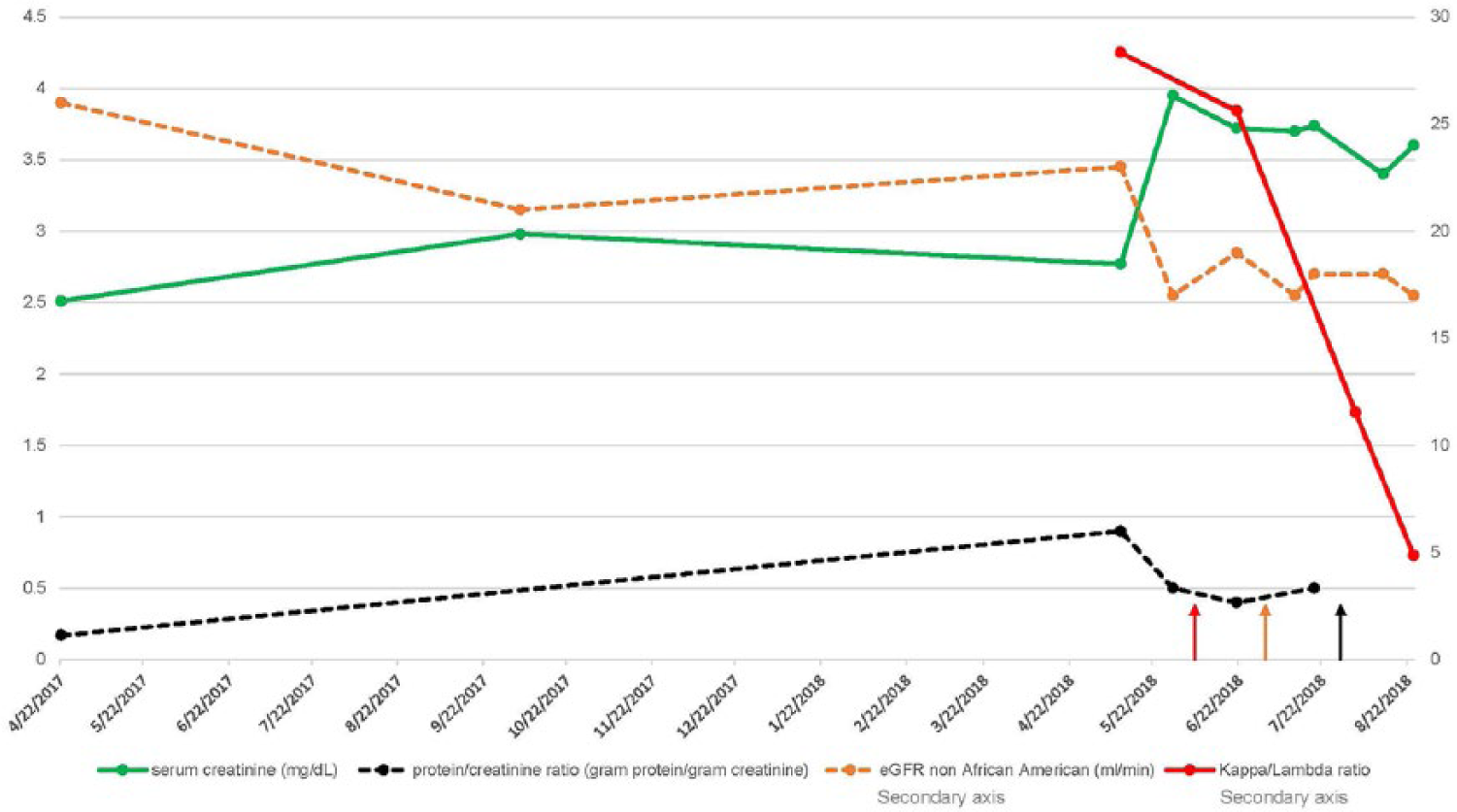
Graph of urine protein/creatinine ratio (gram protein/gram creatinine), serum creatinine (mg/dL), estimated glomerular filtration rate (mL/min), and kappa/lambda ratio (dimensionless) in a patient with LHCDD. Red line indicates bone marrow biopsy (11 June 2018), orange line indicates renal biopsy (28 June 2018), and black line indicates starting of cycle 1 of chemotherapy (4 July 2018).
A bone marrow biopsy was done showing 20% involvement with kappa light chain restricted plasma cells in the setting of hypo-cellular bone marrow that clinically could be consistent with smoldering myeloma. The patient’s fluorescent in situ hybridization (FISH) revealed low-level IgH gene rearrangement, but not (11;14), t(4;14), t(14;16), deletion 17p, +1q, del1p, or 13q abnormality. His positron emission tomography–computed tomography (PET-CT) scan on 22 June 2018 revealed no evidence of lytic lesions or hyper-metabolic activity. Figure 2 illustrates the bone marrow biopsy findings.
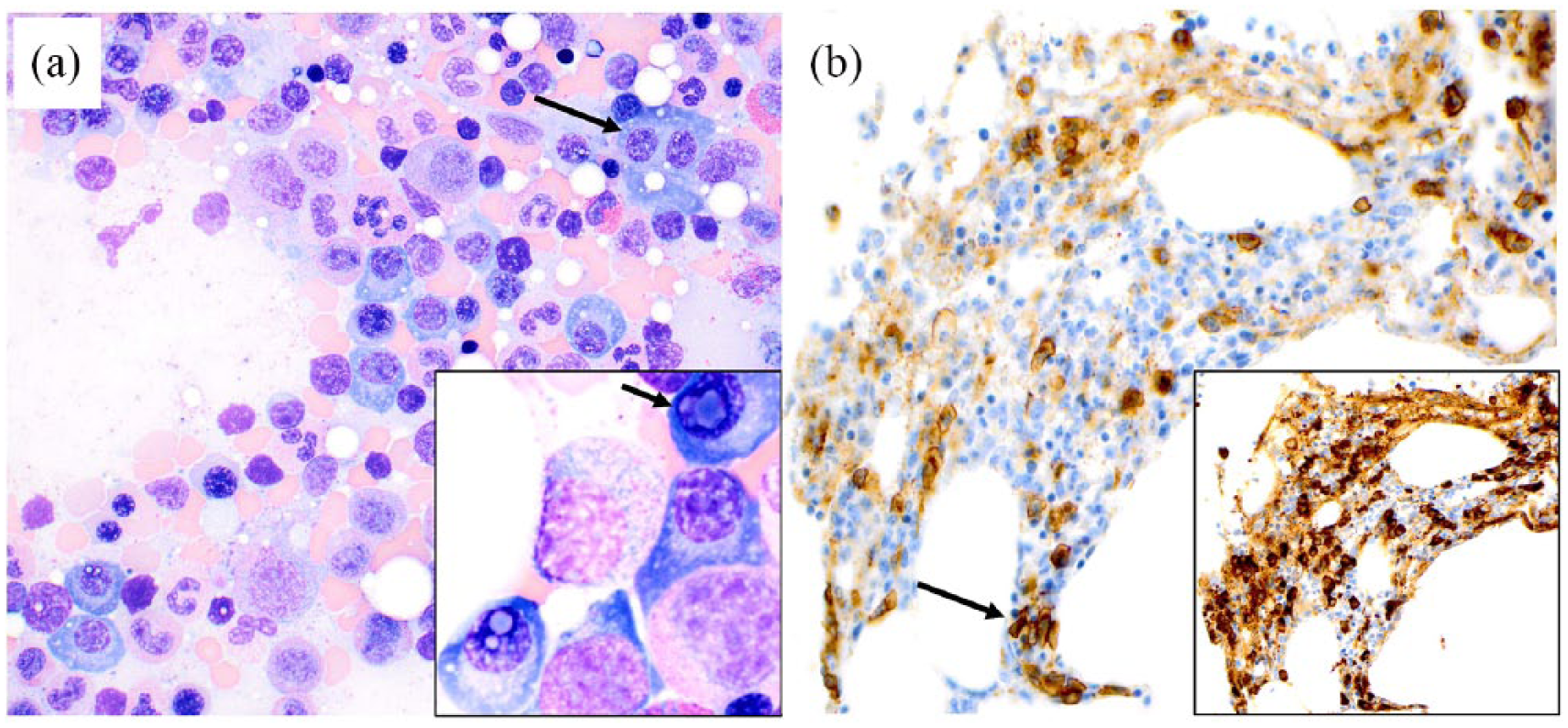
Bone marrow biopsy findings: (a) bone marrow aspirate smears demonstrated numerous plasma cell aggregates comprising 24% of the marrow cellularity. The plasma cells have abundant blue cytoplasm, prominent perinuclear hofs, and numerous waxy blue cytoplasmic inclusions (Dutcher bodies), which are often seen overlaying the nucleus and contain excess immunoglobulin (inset: short arrow). Scattered binucleated forms were also readily identified (long arrow); light microscopy, 400× and 1000× oil magnification. (b) Histologic sections of the marrow biopsy demonstrated clusters and aggregates (see arrow) of monoclonal plasma cells, which are highlighted by immunohistochemistry with CD138 and kappa (see inset); light microscopy, 400× magnification.
A renal biopsy (shown in Figure 3) was obtained that showed nine glomeruli, six of which are globally/near globally sclerotic. Eight glomeruli with extensive cortical scarring were in the immunofluorescence core. In the electron microscopy slide, three of four glomeruli were also sclerotic.
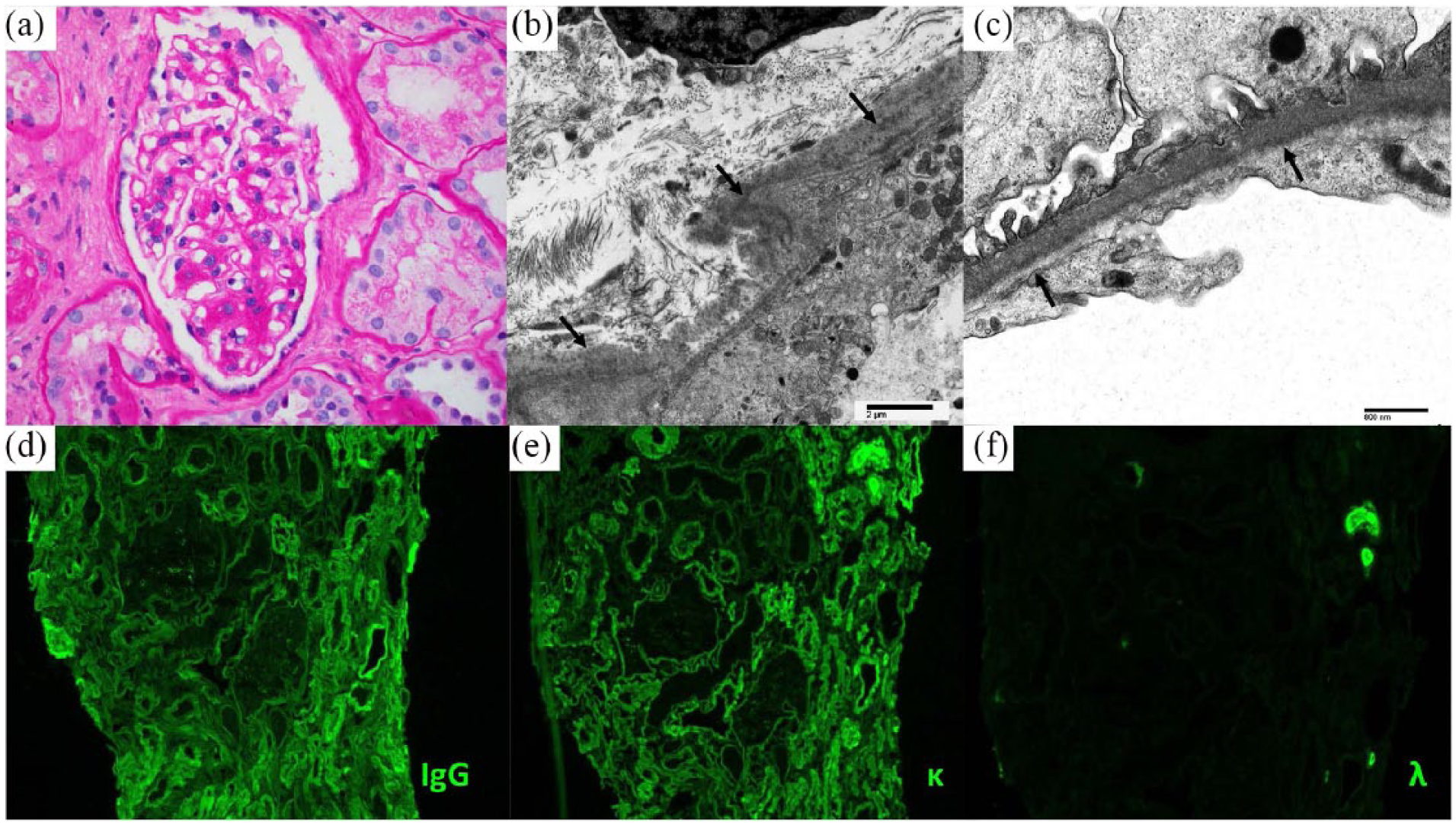
Renal biopsy findings. Renal biopsy demonstrated monoclonal immunoglobulin deposition disease (light chain/heavy chain subtype). (a) Light microscopy of a glomerulus showing segmental moderate mesangial sclerosis (periodic acid Schiff stained sections, 400× magnification). Electron micrographs of (b) tubular basement membranes (arrows) and (c) glomerular capillary loops showing deposition of “powdery” electron-dense material. d)–(f) Immunofluorescence studies demonstrating linear staining of tubular basement membranes with IgG heavy chain and kappa light chain without lambda light chain staining (200× magnification).
By light microscopy, the biopsy was most significant for segmental moderate mesangial sclerosis and very focal capillary loop double contour formation in one glomerulus. No atypical cast material was present. A Congo red stain was negative for amyloid deposition. There were extensive chronic changes including diffuse global glomerulosclerosis, moderate to severe cortical scarring, and severe arterial/arteriolar nephrosclerosis. Immunofluorescence studies were remarkable for diffuse bright linear staining of tubular basement membranes with immunoglobulin G (IgG) heavy chain and kappa light chain antibodies. Only globally sclerotic glomeruli were present and showed similar linear staining with IgG heavy chain and kappa light chain antibodies. Tubular casts showed equivalent staining with both light chains.
Ultrastructural studies were significant for powdery and focally granular electron-dense deposits found along glomerular capillary loops, within the mesangium, and along tubular basement membranes. The glomerular deposits closely followed the contour of the lamina rara interna and sub-endothelial spaces. The sub-endothelial spaces also showed mild accumulation of electron-lucent flocculent material with an early neomembrane formation and unusual double contour formation. Interestingly, very focal fibrillary deposits are present (12.5–17.3 nm) in mesangial and sub-endothelial locations. The latter finding has been reported in the setting of MIDD and was not favored to represent a separate coexisting process. 7
A diagnosis of MIDD of the light and heavy chain deposition disease (LHCDD) subtype was made. The patient was treated with dexamethasone, bortezomib, and lenalidomide treatment with stabilization of his renal function (lenalidomide dose was adjusted for renal function). Given that the patient’s biopsy and clinical course indicated likely advanced CKD and the sclerosis observed on the biopsy specimens, his treatment regimen was recently intensified. He recently has been switched to lenalidomide 15 mg every 48 h days 1–14, with dexamethasone 40 mg on days 1, 8, and 15 of each cycle. He also got bortezomib on days 1, 4, 8, and 11 of each cycle.
Discussion
We report a case of MIDD due to LHCDD in a 65-year-old male with a hitherto unidentified cause of CKD. The patient previously had a renal biopsy that failed to provide an adequate specimen to reach a pathological diagnosis. Importantly, screening tests for multiple myeloma including a serum and urine protein electrophoretic study were not diagnostic. Additional tests including immunofixation studies and the free light chain ratio were critical tests for determining the cause of the patient’s renal disease and the presence of a plasma cell dyscrasia. Our findings complement previous considerations in the literature. 8 This is because most MIDD disease has occurred in presence of MGRS; the need to distinguish MIDD from smoldering myeloma suggests an uncommon but important scenario. Treatment guidelines would then allow for treatment due to deleterious effects on renal function.
MIDD is a rare disease 9 with findings of both light and heavy chain deposits. Typically, the presence of the heavy chain fixes complement (C3 and C4 can be low) and can cause systemic disease. 10 Outcomes usually are poor with a 5-year survival of 67% and 5-year renal survival of only 57%. 11 The type of MIDD presented here is the Randall type of IgG LHCDD. In addition, a non-Randall type of granular IgA or IgM deposition has been previously described. 12 In our patient, a second renal biopsy was essential in making the diagnosis and initiating appropriate therapy. The patient was started after the biopsy on bortezomib, lenalidomide, and dexamethasone (RVD) and achieved a good response with a decreasing kappa to lambda free light chain ratio.
Conclusion
We review a case of a MGUS that leads to a bone marrow biopsy that could have suggested smoldering myeloma in the clinical context. This disorder is typically not treated but rather monitored. An initial renal biopsy failed to provide a conclusive diagnosis; hence, there was not enough pathological evidence to warrant chemotherapeutic treatment. A second renal biopsy, however, revealed clear MIDD that required chemotherapy treatment to be initiated. Table 1 reviews published case reports of LHCDD/MIDD and clinical characteristics of reported patients.7,13–21
Literature Review of Prior Reported Cases: Light and Heavy Chain Deposition Disease (LHCDD).
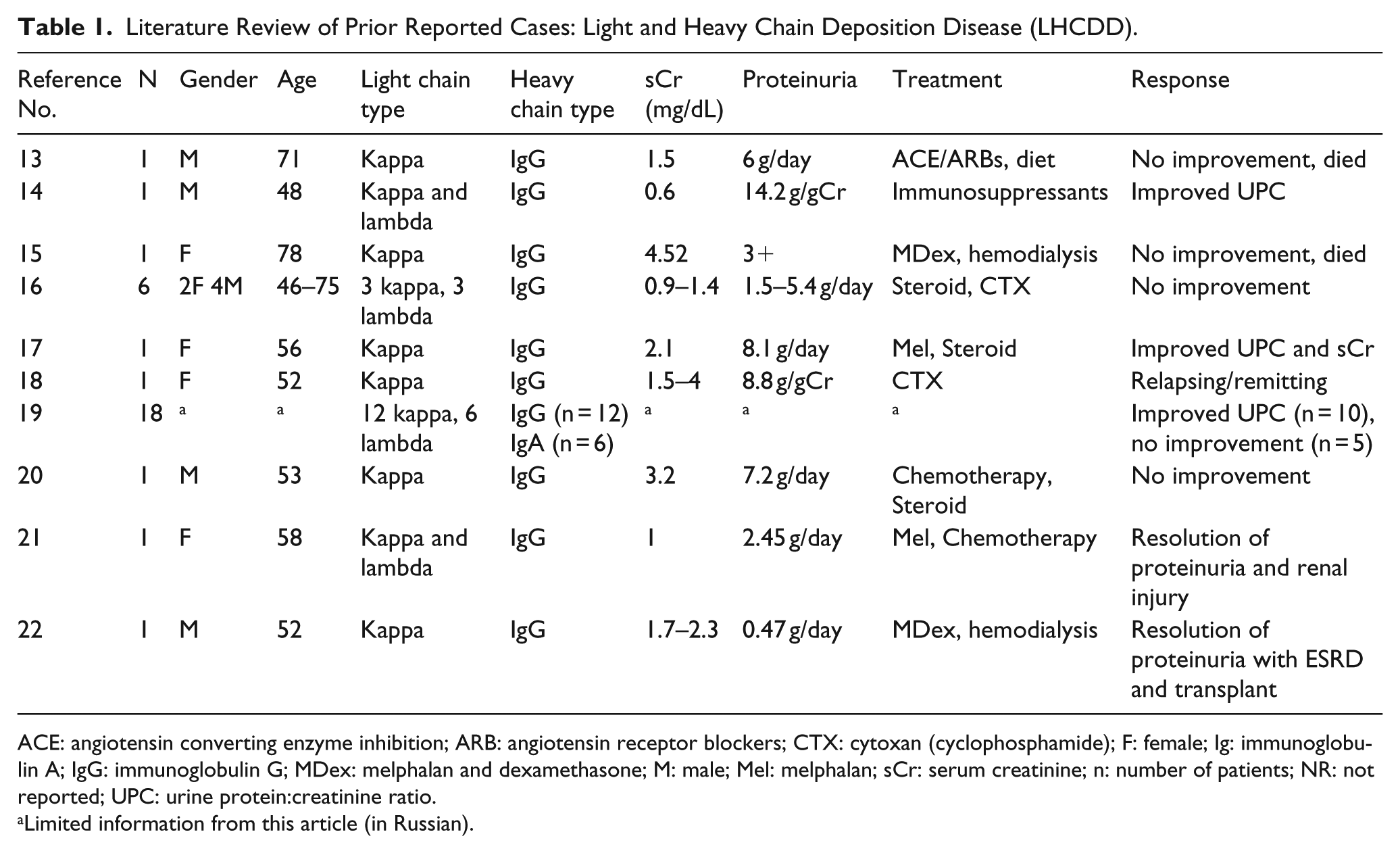
ACE: angiotensin converting enzyme inhibition; ARB: angiotensin receptor blockers; CTX: cytoxan (cyclophosphamide); F: female; Ig: immunoglobulin A; IgG: immunoglobulin G; MDex: melphalan and dexamethasone; M: male; Mel: melphalan; sCr: serum creatinine; n: number of patients; NR: not reported; UPC: urine protein:creatinine ratio.
Limited information from this article (in Russian).
Our case highlights that bone marrow biopsy is necessary but not sufficient in a patient with monoclonal gammopathy associated renal dysfunction. When proteinuria is present and free light chains are elevated, a diagnostic renal biopsy is indicated even if the bone marrow biopsy does not point to clearly active multiple myeloma. Hematological and renal remission were determined to have been induced as judged by the criteria published by the leukemia working group. 22 Furthermore, this case provides additional evidence that renal biopsies can be used in cases that could clinically be consistent with smoldering myeloma to discern active disease. Treatment then would be indicated if evidence of monoclonal antibody–related renal disease is present.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: I.K. is supported in part by funds from the NIH (R01-DK077162), the Allan Smidt Charitable Fund, the Factor Family Foundation, and the Ralph Block Family Foundation.
Informed consent
Written informed consent was obtained retrospectively from the patient(s) for their anonymized information to be published in this article.
