Abstract
Introduction:
Proteinuria assessment is a key test in pregnancy to evaluate renal and systemic well-being. The finding of proteinuria may allow diagnosis and ready intervention for pre-eclampsia and glomerulonephritis, which may compromise a favorable delivery. Moreover, pathological-range isolated proteinuria is a risk factor by itself for adverse outcomes during pregnancy. An appropriate interpretation of pathologic values of proteinuria is therefore of crucial importance.
Patient presentation:
We present the case of a 33-year-old apparently healthy woman at her first pregnancy who developed a clinically significant proteinuria during the first trimester; we describe the clinical workup and the management of the patient up to delivery.
Conclusion:
Urine analysis with dipstick or protein-to-creatinine ratio is part of the routine prenatal clinical care during pregnancy. Detection of a pathological proteinuria (>300 mg/24 h or equivalent) should never be underestimated, and timed urine collection is required, as well as a thorough medical examination. The main goal is to exclude pre-eclampsia, whereas the suspect of a primary kidney disease can be managed together with the nephrologist, both for management and treatment.
Introduction
Periodic assessment of proteinuria during pregnancy is a key step to rule out or diagnose severe conditions associated with high morbidity and mortality, in particular, pre-eclampsia. In this article, we report an unusual case of glomerulonephritis first presented during pregnancy with proteinuria, focusing on how to use different tools to reach a diagnosis and hence act for an appropriate therapy.
Patient presentation
A 33-year-old woman was referred to our nephrology clinic during her 13th week of pregnancy due to the finding of proteinuria associated with microscopic hematuria and hyaline casts. The first urinalysis was performed at the eighth week of gestation by her general practitioner with a urinary dipstick that estimated 300 mg/dL of proteins. Based on these results, the patient was referred to a high-risk pregnancy clinic to be monitored throughout the whole pregnancy and where it was confirmed the pathological proteinuria, estimating 500 mg/dL with the same method. The patient was then referred by the obstetrician to the nephrology clinic for a diagnostic workup. The patient reported that no blood test nor urinalysis was performed before the eighth week. She did not have a family history of kidney pathologies, had not any past pregnancies, was not diabetic, was not a usual consumer of non-steroidal anti-inflammatory drugs (NSAIDs). She has been a regular smoker until pregnancy. At physical examination, the patient did not show limb edemas nor other pathological finding. Arterial blood pressure (BP) was in the normal range (110/60 mm Hg). During the nephrology consult, a monthly follow-up was suggested, monitoring renal function and proteinuria. In addition, diagnostic blood tests were required to be evaluated at the next nephrology consult. Heparin (enoxaparin sodium: 4000 U/day) was started empirically since the 20th week of gestation as prophylaxis for cardiovascular events prevention due to the onset of nephrotic-range proteinuria. This approach is not evidence-based as use of acetylsalicylic acid has been proposed by others. At the next nephrologic visit, 2 months later, the following laboratory tests were obtained: white blood cells (WBCs), red blood cells (RBCs), and platelets were in their reference intervals; serum creatinine 0.58 mg/dL; blood urea nitrogen (BUN) 24 mg/dL; uric acid 3.7 mg/dL; serum total proteins 5.2 g/dL; serum albumin 2.4 g/dL; urinary proteins 3.6 g/24 h; protein electrophoresis showed a peak in the α2-globulin region; immunologic exams (C3, C4, anti-nuclear antibodies, anti-neutrophil cytoplasmic antibodies, anti-cardiolipin antibodies, anti-beta2 glycoprotein I, lupus anticoagulant antibodies) were negative; anti-PLA2R antibodies were negative; serum electrolytes and liver tests were in their normal range. Based on these laboratory data, it was decided not to perform a renal biopsy considering the risk-to-benefit profile of an invasive procedure; particularly, despite the nephrotic syndrome, we delayed the procedure until after delivery since renal function was preserved, the fetus was in steady condition and there was no evidence of an evolving autoimmune disease. The patient was therefore asked to perform a second-level urinalysis with an examination of the urine sediment that showed hyaline casts, RBC casts, and 6–10 RBCs per field (59% of RBCs were dysmorphic, 38% isomorphic, and 3% were acanthocytes). It was then decided together with the patient and the obstetrician to monitor the proteinuria and renal function monthly. After 3 months, proteinuria reached 6.8 g/24 h with a urinary sediment positive for granular casts and a nephrotic syndrome (hypercholesterolemia, hypertriglyceridemia, hypoproteinemia); due to the evolving clinical picture, an empirical steroid treatment was started (intravenous methylprednisolone 300 mg for 3 days, followed by oral administration of prednisone 50 mg/day with progressive tapering). At her 33rd week of gestation, the patient was admitted in the gynecologic unit for the onset of oligohydramnios associated with nephrotic syndrome and fetal growth restriction (estimated birth weight below the 10th percentile). To minimize risks, a cesarean section was performed at 37th week of gestation as preventive measure due to fetal growth restriction, the oligohydramnios, and fetus in podalic position. The newborn was a female with a weight of 2220 g, APGAR score 10/10. After 2 days of the discharge, the proteinuria was 1 g/24 h, but it rebounded to nephrotic range (6.1 g/24 h) after 4 months with normal renal function (creatinine 0.65 mg/dL). It was then decided to perform a kidney biopsy that showed a membranous nephropathy, with sub-epithelial deposition of IgG and positivity for IgA and C1q. Henceforth, the patient was treated with ramipril 2.5 mg/day and, 1 month after the biopsy, with rituximab (two administrations of 1 g, 2 weeks apart). Despite this, the patient failed to respond, with 5.88 g/day of proteinuria after 5 months. The patient was therefore switched to cyclosporine 200 mg/day, with a significant reduction in proteinuria (up to 2.65 g/day).
Conclusion
Although not applicable in our case, as proteinuria was frankly in the pathologic range from the beginning, the first element to consider when evaluating a renal disease in pregnancy is the physiological changes occurring in the kidneys: renal blood flow increases with a proportional increase in the glomerular filtration rate (GFR); as a consequence, kidneys become larger (1–1.5 cm more) and serum creatinine decreases. 1 These changes also impact on urinary proteins, whose levels are higher than those in non-pregnant women, with average levels of 100 mg/24 h during the first trimester and 180–200 mg/24 h in the successive months. The cut-off to consider proteinuria as pathologic during pregnancy has been set at 300 mg/24 h. Due to progesterone effects on ureters and bladder muscular walls, there is a relaxation of the urinary tract, with increased vesicoureteral reflux and urinary stasis, favoring bacterial overgrowth. In these circumstances, microscopic hematuria or proteinuria in the pathologic may also be found. Microscopic hematuria, however, especially if isolated, is difficult to evaluate in pregnancy because up to 20% of healthy pregnant women show a positive dipstick test, and 15% are positive for hematuria at microscopy analysis. 2
We considered the different potential causes of proteinuria in pregnancy, where timing of onset and amount are critical factors. As an example, despite the high prevalence and the severity of complications, we could readily exclude pre-eclampsia as a possible diagnosis as, per definition, pre-eclampsia is characterized by the onset of hypertension (BP > 140/90 mm Hg) and proteinuria (>300 mg/24 h) or signs and symptoms of end organ dysfunction after the 20th week of gestation; 3 it affects 2%–8% of pregnancies and its complications can be organ specific (cerebral hemorrhage, hepatic failure, pulmonary edema, acute kidney injury) or diffuse (e.g. disseminated intravascular coagulation). This clinical picture can unfavorably evolve into eclampsia (development of seizures in a pre-eclamptic woman) or HELLP (Hemolysis, Elevated Liver enzymes, and a Low Platelet count) syndrome, which in 15%–20% of cases can develop independently from pre-eclampsia.
Despite this stringent definition, it should be considered that symptoms or clinical signs can have, although uncommonly, an unusual presentation, such as the onset of pre-eclampsia in normotensive women. 4 Moreover, isolated proteinuria (onset of pathological levels of proteinuria after the 20th week of gestation) is associated with a higher proportion of women that will develop pre-eclampsia later during pregnancy and seems to be also a negative predictive factor for pregnancy outcome in women with pre-eclampsia. 5
As proteinuria alone does not diagnose pre-eclampsia, but it is an important risk factor for its future development, especially in normotensive pregnant women, it is important using diagnostic tests with high sensitivity and to strictly follow up the patient, monitoring any pathologic changes, such as the onset of hypertension or signs of end organ dysfunction. Besides pre-eclampsia, a routine monitoring of proteinuria may be important to diagnose newly onset kidney diseases, which increase the risks of adverse events during pregnancy.
The 24-h urine protein excretion measurement is the gold standard for quantifying proteinuria, but it is cumbersome, inconvenient, time-consuming, and subject to errors leading to inaccuracies in nearly half of collections. 6 Urine dipstick analysis is widely used in obstetric practice because it is simple to perform and economic, but its usefulness is impaired by low sensitivity and specificity. Several factors, such as maternal hydration status or the presence of infections, may influence its accuracy. Sensitivity and specificity of the urine dipstick test vary greatly among different studies. Moreover, this test may also overestimate the risk of significant proteinuria. Automated dipstick methods appear to have greater sensibility than visual dipstick urinalysis for detection of proteinuria. Significant proteinuria cannot at the moment be diagnosed or excluded using only urine dipstick analysis. 7
The use of albumin-to-creatinine ratio (ACR) analysis on a random urine sample as a point-of-care test for chronic kidney damage has reached widespread use in clinical practices. Despite this, results of several studies concerning the use of this test in predicting significant proteinuria in pregnant women are quite variable, mainly depending on the studied population. Nisell et al. 8 reported 95% sensitivity and 100% specificity using a cut-off of 27 mg/mmol for estimating albuminuria >300 mg/24 h in a population of hypertensive women in order to pre-emptively diagnose pre-eclampsia, while Huang et al. 9 reported 82.4% sensitivity and 99.4% specificity (cut-off of 22.8 mg/mmol for proteinuria >300 mg/24 h) and 90.6% sensitivity and 99.6% specificity (cut-off of 155.6 mg/mmol for proteinuria >2 g/24 h), respectively, in patients with overt pre-eclampsia. Other studies documented lower accuracy with different cut-off values. Morris et al., 10 however, in a systematic review and meta-analysis, warned that there is no sufficient evidence regarding the accuracy of ACR and its use in clinical practice and underlined the need for further research. More data are available concerning the use of urine protein-to-creatinine ratio (uPCR) for detecting significant proteinuria in pregnant women. Protein-to-creatinine ratio (PCR) has been shown to correlate well with 24-h urine protein excretion measurement in non-pregnant women. Sensitivity and specificity obtained with PCR also vary depending on the cut-off and the studied population. In 2008, Papanna et al. 11 indicated that there is no consensus yet on which cut-off of uPCR or urine albumin-to-creatinine ratio (uACR) is most useful to estimate 300 mg/24 h proteinuria in acute diseases such as pre-eclampsia. A meta-analysis by Sanchez-Ramos et al. 12 reported a good sensitivity and specificity to detect significant proteinuria, while Morris et al. 10 reported less satisfactory results (Table 1). Both studies were performed on at-risk pregnant women for pre-eclampsia. These two meta-analyses suggested 0.30–0.35 mg/mmol as ideal uPCR cut-off values in order to detect a proteinuria >300 mg/24 h. Both meta-analyses considered uPCR as an effective test to rule out significant proteinuria with an acceptable cut-off of 0.30 mg/mmol. Maternal age, gestational age, or parity does not alter the accuracy of PCR. Guidelines are based on the findings of the latest studies. 13 All pregnant women should be screened for significant proteinuria, using an automated urinary dipstick or PCR: if a result of 1+ or more is obtained at dipstick, a timed urine collection should be performed. In Figure 1, a diagnostic algorithm for abnormal proteinuria is provided. BP measurement remains an essential diagnostic element and should be measured at each antenatal visit. The use of ACR for detection of significant proteinuria in pregnant women is not encouraged in the guidelines, probably because of the lack of studies, although it shows good correlation with PCR.
Results from different meta-analyses studying the effectiveness of uPCR in estimating significant proteinuria in pregnant women at risk for pre-eclampsia.
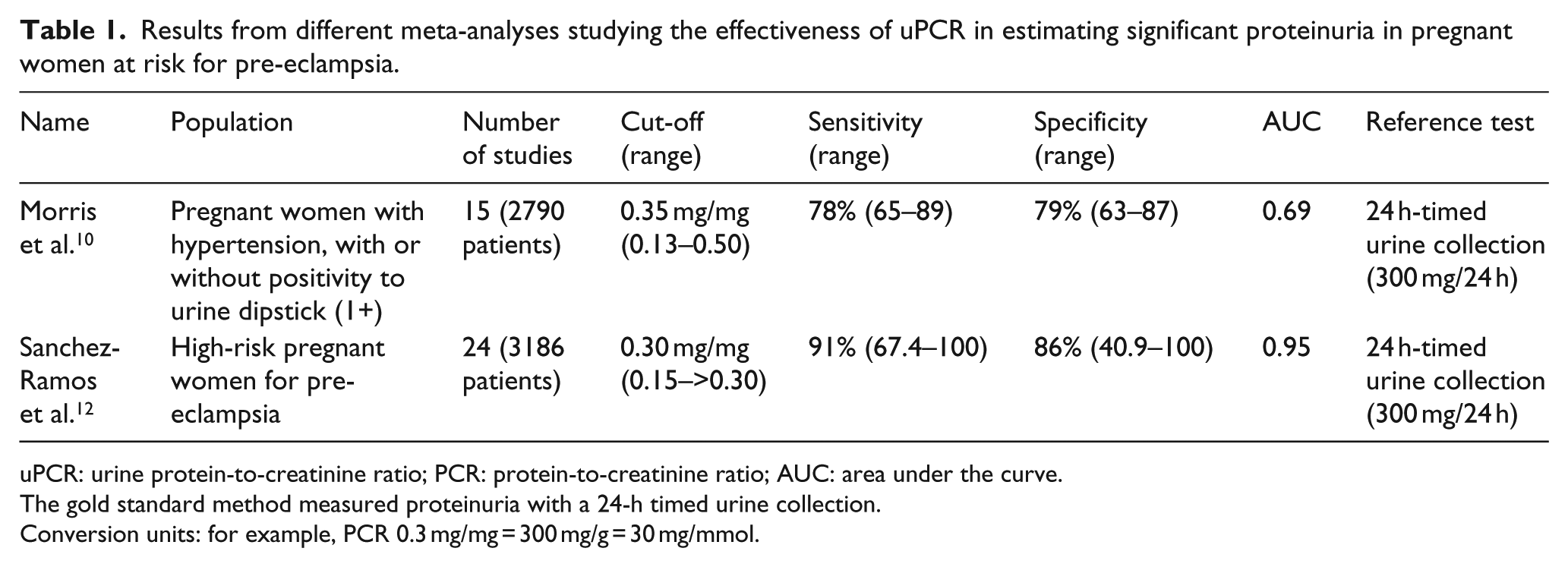
uPCR: urine protein-to-creatinine ratio; PCR: protein-to-creatinine ratio; AUC: area under the curve.
The gold standard method measured proteinuria with a 24-h timed urine collection.
Conversion units: for example, PCR 0.3 mg/mg = 300 mg/g = 30 mg/mmol.
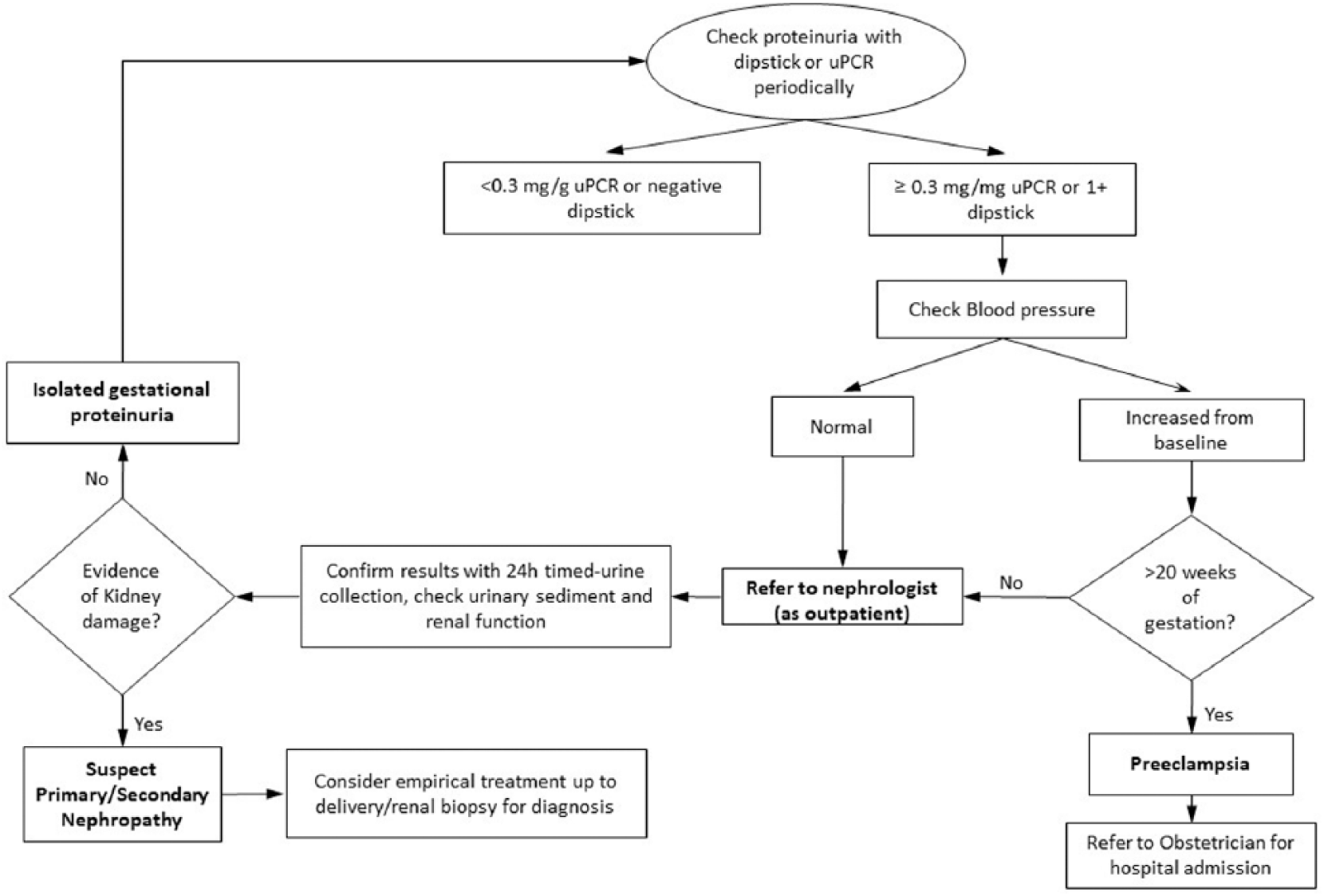
Summary of the diagnostic algorithm for abnormal proteinuria in pregnancy. Importantly, there is no documented upper decisional limit of proteinuria to diagnose isolated gestational proteinuria, pre-eclampsia, or primary kidney disease. Blood pressure, urinary sediment, renal function, and clinical signs and symptoms are needed to address a correct diagnosis.
In our patient, the chance that proteinuria could be related to pre-eclampsia was fairly low for several reasons: first, proteinuria was discovered early during pregnancy, well before the 20th week of gestation required for the diagnosis, which is typical of primary kidney diseases rather than pre-eclampsia. Second, the patient did not have any risk factor for the disease, except being at her first pregnancy. Moreover, during the follow-up, it was shown that proteinuria was steadily increasing up to nephrotic range, which, although possible, is unusual for pre-eclampsia. Arterial BP and renal function were both normal, whereas in pre-eclampsia, renal function could fall up to 40%, with a serum creatinine usually between 1 and 1.5 mg/dL. Other laboratory tests, such as a urinary sediment positive for granular or red cell casts or the presence of microhematuria, are not suggestive of pre-eclampsia.
Having ruled out pre-eclampsia, the differential diagnosis of isolated proteinuria requires evaluation of immune-mediated systemic pathologies and primary kidney diseases. Although rare, immune-related systemic diseases can have their first presentation during pregnancy and they represent dangerous risk factors for adverse outcomes. The most frequent are systemic lupus erythematosus (SLE), antiphospholipid syndrome, hemolytic-uremic syndrome and thrombotic thrombocytopenic purpura (HUS/TTP): each of these can be ruled out with serological tests that have reliable diagnostic efficacy. The diagnosis of primary kidney diseases instead can be done with renal biopsy. However, in this case, biopsy was not strictly indicated due to risk/benefit, considering the preserved renal function and a likely diagnosis, indicated by the active urine sediment. Findings relevant in the context of glomerulonephritis are RBC casts, hyaline-granulous casts, and dysmorphic RBC.
In conclusion, it is strongly recommended to carefully evaluate the finding of proteinuria during pregnancy using the appropriate tests, with the goal of ruling out pre-eclampsia. Moreover, a number of different primary kidney diseases may emerge during pregnancy, which need an appropriate diagnostic workup or, in the absence of biopsy-confirmed diagnosis, to establish a careful follow-up aimed at a safe delivery, minimizing the risks for the fetus and the mother.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
