Abstract
Music has been associated with alterations in autonomic function. Tempo, the speed of music, is one of many musical parameters that may drive autonomic modulation. However, direct measures of sympathetic nervous system activity and control groups and/or control stimuli do not feature in prior work. This article therefore reports an investigation into the autonomic effects of increases and decreases in tempo. Fifty-eight healthy participants (age range: 22–80 years) were randomly allocated to either an experimental (n = 29, tune) or control (rhythm of the same tune) group. All participants underwent five conditions: baseline, stable tempo (tune/rhythm repeatedly played at 120 bpm), tempo increase (tune/rhythm played at 60 bpm, 90 bpm, 120 bpm, 150 bpm, 180 bpm), tempo decrease (tune/rhythm played at 180 bpm, 150 bpm, 120 bpm, 90 bpm, 60 bpm) and recovery. Heart rate, blood pressure, respiration, and muscle sympathetic nerve activity were continuously recorded. The 60 bpm in the tempo decrease stimulus was associated with increases in measures of parasympathetic activity. The 180 bpm in the tempo increase stimulus was also associated with shifts towards parasympathetic predominance. Responses to the stimuli were predicted by baseline %LF. It is concluded that the individual tempi impacted upon autonomic function, despite the entire stimulus having little effect. The 60 bpm in an increasingly slower stimulus was associated with greater vagal modulations of heart rate than faster tempi. For the first time, this study shows that response direction and magnitude to tempo manipulations were predicted by resting values, suggesting that music responders may be autonomically distinct from non-responders.
Introduction
Music is a powerful stimulus with widespread effects on listener psychology, neurology, and physiology (Fancourt, Ockelford, & Belai, 2014). There is growing evidence that music can have beneficial effects on gut health (Krabs, Enk, Teich, & Koelsch, 2015), immune function (Charnetski, Brennan, & Harrison, 1998; Kuhn, 2002), depression (Cooke, Moyle, Shum, Harrison, & Murfield, 2010), pain and anxiety (Nilsson, 2008), and cardiovascular health (Möckel et al., 1994; Okada et al., 2009). One physiological system which influences all of these aspects is the autonomic nervous system (ANS). This therefore opens a question as to whether music can influence the ANS.
The ANS is a complex neural network, responsible for maintaining homeostasis (or balance) within the human body and is comprised of the sympathetic nervous system (SNS) and the parasympathetic nervous system (PNS). The SNS mobilises energy resources by increasing metabolic output to engage in “fight or flight” responses. In contrast, the PNS increases anabolic activity to promote restorative functions within the body, typically known as “rest and digest” activities. Activation of the SNS is associated with increases in heart rate (HR) and blood pressure (BP), whereas activation of the PNS is associated with decreases in HR and BP. In healthy individuals, the PNS is usually predominant over the SNS. This balance shifts towards sympathetic predominance in a variety of conditions, including depression (Kemp et al., 2010), heart failure (Triposkiadis et al., 2009), and hypertension (see Thayer, Yamamoto, & Brosschot, 2010 for a review).
There is a range of stimuli that are considered to promote parasympathetic (vagal) predominance. Music is one such stimulus. For instance, elderly patients with cerebrovascular disease and dementia showed enhanced measures of parasympathetic activity and a shift away from sympathetic predominance following a music therapy intervention (Okada et al., 2009). Furthermore, in a more acute setting, listening to Mozart K.545 boosted parasympathetic tone, thereby promoting a shift away from sympathetic predominance in children with epilepsy (Lin et al., 2013). In addition, in non-patient samples, music was more effective at limiting the effects of a stressor compared to a no-music control (Knight & Rickard, 2001). Also, measures of parasympathetic activity were significantly greater during sedative music than during excitative music (Iwanaga, Kobayashi, & Kawasaki, 2005).
Music is the amalgamation of many parameters, often characterised as dynamics, pitch, timbre, rhythm, articulation, mode, and tempo. Many studies have demonstrated that alterations in specific musical parameters can have precise effects on sympathetic and parasympathetic activity. For example, crescendos (increases in the intensity of music) were accompanied by decreases in skin vasomotion and increases in HR and systolic and diastolic blood pressures, indicative of shifts towards sympathetic predominance (Bernardi et al., 2009). Interestingly, the influence of changes in sound intensity on autonomic function is apparent in early life: foetuses between 33 weeks and 36 weeks gestational age and term foetuses showed accelerations in HR to a lullaby played at increasingly louder intensities (Kisilevsky, Hains, Jacquet, Granier-Deferre, & Lecanuet, 2004). In addition, happy music (characterised by a major mode) has been associated with significantly greater BP and skin conductance responses than sad music (characterised by a minor mode, Khalfa, Roy, Rainville, Bella, & Peretz, 2008). Musical genre also plays a large role in determining sympathovagal activity (see, e.g., Pérez-Lloret et al., 2014 and Kume et al., 2017).
Tempo is another musical parameter considered to have an influential role on autonomic function. Tempo is the speed (or rate) at which music is played, usually indicated at the beginning of a piece of music and measured in beats per minute (bpm, e.g., 90 bpm). Increases in musical tempo have been associated with shifts towards sympathetic predominance, signified by increases in HR (Gomez & Danuser, 2007; Watanabe, Ooishi, & Kashino, 2017), skin conductance level (Chuen, Sears, & McAdams, 2016; Dillman Carpentier & Potter, 2007; Gomez & Danuser, 2007), and decreases in measures of vagal tone (Iwanaga et al., 2005; van der Zwaag, Westerink, & van den Broek, 2011). Tempo is considered to have a particularly large impact on autonomic function due to its intimate connection with HR. Indeed, preferred tempo (measured by adjusting a control dial) was 1, 1.5, and 2 times as fast as resting HR, with this being dependent on the direction of change in tempo (Iwanaga, 1995a, 1995b). Furthermore, direction of change in HR can be influenced by the direction of change in tempo: HR increased with an increase in musical tempo, with the degree of change being related to resting HR (Watanabe et al., 2017).
The strength of the tempo-autonomic relationship also appears to be influenced by the presence/absence of other musical parameters. For example, Khalfa, Roy, Rainville, Bella, and Peretz (2008) showed that original happy excerpts evoked more skin conductance responses and higher diastolic blood pressure (DBP) than the same excerpts following the removal of pitch and temporal variations. Additionally, original sad excerpts and those which had pitch variations removed elicited significantly greater respiration rates, compared to sad excerpts which had only tempo preserved.
Collectively, these findings demonstrate that tempo is a highly influential musical parameter. Studies illustrate that the direction of tempo change can determine the orientation of autonomic shift: towards sympathetic predominance for faster tempi and towards parasympathetic predominance for slower tempi. Furthermore, degree of change seems to be related to resting autonomic activity and interacts with the presence/absence of other musical parameters (such as pitch and rhythmic variations). However, it is important to note that most previous studies have employed indirect measures of autonomic activity, primarily HR, particularly for those exploring the relationship between autonomic function and tempo. Consequently, this renders it challenging to determine the extent to which tempo and its changes impact the SNS, given that most indirect autonomic measures do not reflect purely sympathetic activity. Furthermore, control conditions in studies investigating the effects of tempo on autonomic function have tended to comprise periods of silence, rather than control auditory stimuli. Therefore, by employing a control group similar to Khalfa et al. (2008) as well as a control stimulus (constant/stable tempo auditory stimulus), this study used tightly controlled auditory stimuli that manipulated tempo only to investigate the effects of tempo changes on autonomic function. Autonomic function was assessed by measuring heart rate variability (HRV), BP, and respiration. Microneurography was also used to directly record muscle sympathetic nerve activity (MSNA) during the auditory stimuli. It was hypothesised that: A stimulus which increased in tempo will be associated with significantly greater shifts towards sympathetic predominance compared to a baseline (resting) period or a stimulus whose tempo remained constant. A stimulus which decreased in tempo will be associated with significantly greater shifts towards parasympathetic predominance compared to a baseline (resting) period and a stimulus whose tempo remained constant. Direction of sympathovagal shift will be related to the direction of individual tempi within a stimulus which increases or decreases in tempo. Direction of change in autonomic function will be influenced by the presence/absence of a melody.
Methods
Participants
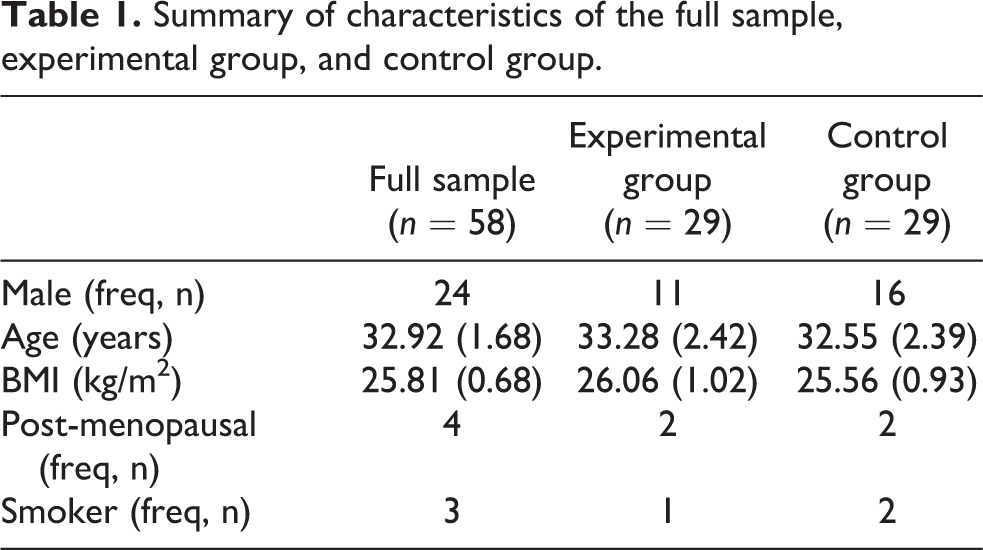
Fifty-eight healthy participants (27 males) free from cardiovascular disease and obstructive sleep apnoea were recruited for the study. Age ranged from 22 years to 80 years. None of the post-menopausal females (n = 4) were receiving hormone replacement therapy. Table 1 summarises the initial characteristics of the sample.
Summary of characteristics of the full sample, experimental group, and control group.
On each testing occasion, the study was conducted between 14:00 and 18:00 and participants were asked to abstain from caffeine, nicotine, alcohol, and strenuous exercise for the preceding 12 hours and to empty their bladder directly before study commencement. Upon study completion, all participants received £20 reimbursement (as funded by SEMPRE’s Arnold Bentley New Initiatives Fund). The study was approved by the local (Faculty of Biological Sciences) University Ethics Committee (ethics reference: BIOSCI 15-014). Written informed consent was obtained from all participants prior to testing.
Twenty-nine participants were randomly allocated to an experimental group (11 males) and 29 were randomly allocated to a control group (16 males, see Table 1 for a summary). Allocation to the experimental group entailed hearing a melody, whereas participants in the control heard the rhythm of the melody (pitch variations were removed).
Materials
Auditory stimuli
Seven auditory stimuli with piano instrumentation were created. The first included a one-octave C major scale starting on middle C. Two comprised five discreet tempi (60 bpm, 90 bpm, 120 bpm, 150 bpm, and 180 bpm) organised in ascending order (tempo increase stimulus). One version consisted of a nursery rhyme (“baa baa blacksheep”) administered to those in the experimental group. The second version consisted of the rhythm of the same nursery rhyme, given to those in the control group. Two additional stimuli comprised the same five tempi but organised in descending order (tempo decrease stimulus) with the original version presented to the experimental group and the rhythm-only version presented to the control group. The duration of each tempo was at least 2 minutes, creating 10-minute stimuli. The final two stimuli consisted of the original tune (experimental group) or rhythm (control group) repeatedly played at 120 bpm for 10 minutes (stable tempo stimulus). All auditory stimuli were created in Sibelius Version 7, exported as a MIDI file, recorded in Audacity at 44.1 kHz at 76dB, exported as a .wav file and stored on a Sanza Fuse mp3 player. All stimuli were presented to participants via wired over-the-head headphones.
Procedure
Prior to physiological monitoring, participant height and weight were measured. Physiological equipment was then attached: three-lead electrocardiogram (ECG) to record HR (sampled at 2000 Hz with the PowerLab system); BP recorded via finger plethysmography (sampled at 400 Hz); and a pneumograph to record respiration rate (sampled at 400 Hz). A sub-set of the sample agreed to undergo microneurography (n = 24; n = 10 in the control group).
Muscle sympathetic nerve activity (MSNA) was recorded as previously described (Greenwood, Stoker, & Mary, 1999; Macefield, Wallin, & Vallbo, 1994). The peroneal nerve was identified by palpation and confirmed with the use of a PowerLab stimulator unit (ADInstruments). Upon confirmation of the peroneal nerve, two tungsten microelectrodes (FHC Inc., USA) were inserted percutaneously. The microelectrodes were 35 mm in length with a diameter of 200 μm tapering to a tip. The recording microelectrode was epoxy insulated with an impedance of 0.3 ± 0.6 MΩ. The recording microelectrode was inserted into the peroneal nerve, and the second (reference) microelectrode was inserted into subcutaneous tissue 1–2 cm away from the recording electrode.
To identify MSNA units, participants undertook two cardiovascular tests: cold pressor test, submerging the left hand into ice water for 1 minute; isometric hand grip test, squeezing a handgrip at 50% maximum voluntary contraction for 2 minutes. To confirm single MSNA units, the following criteria were met: (a) the unit occurred in diastole only; (b) the unit increased in firing with increases in blood pressure; (c) the unit firing frequency increased during the second half of the cold pressor and hand grip tests; (d) the unit shape and firing rate did not change during the stroking of the upper foot; and (e) the unit shape was consistent when superimposed (Corel Draw, Version 6).
Participants then underwent an adaptation period of approximately 3 minutes. The one-octave C major scale was presented to ensure the audio was played at a comfortable volume. Physiological measures were continuously recorded during five, 10-minute conditions: baseline (silence with headphones in place), stable tempo (rhythm or tune repeatedly played at 120 bpm), tempo increase (rhythm or tune repeatedly played at five increasingly faster tempi), tempo decrease (same as tempo increase but tempo order reversed), and recovery (same as baseline). The presentation order of the stimuli was randomised between participants. At the end of each condition, brachial HR and BP were measured three times. Figure 1 provides an overview of the study procedure.
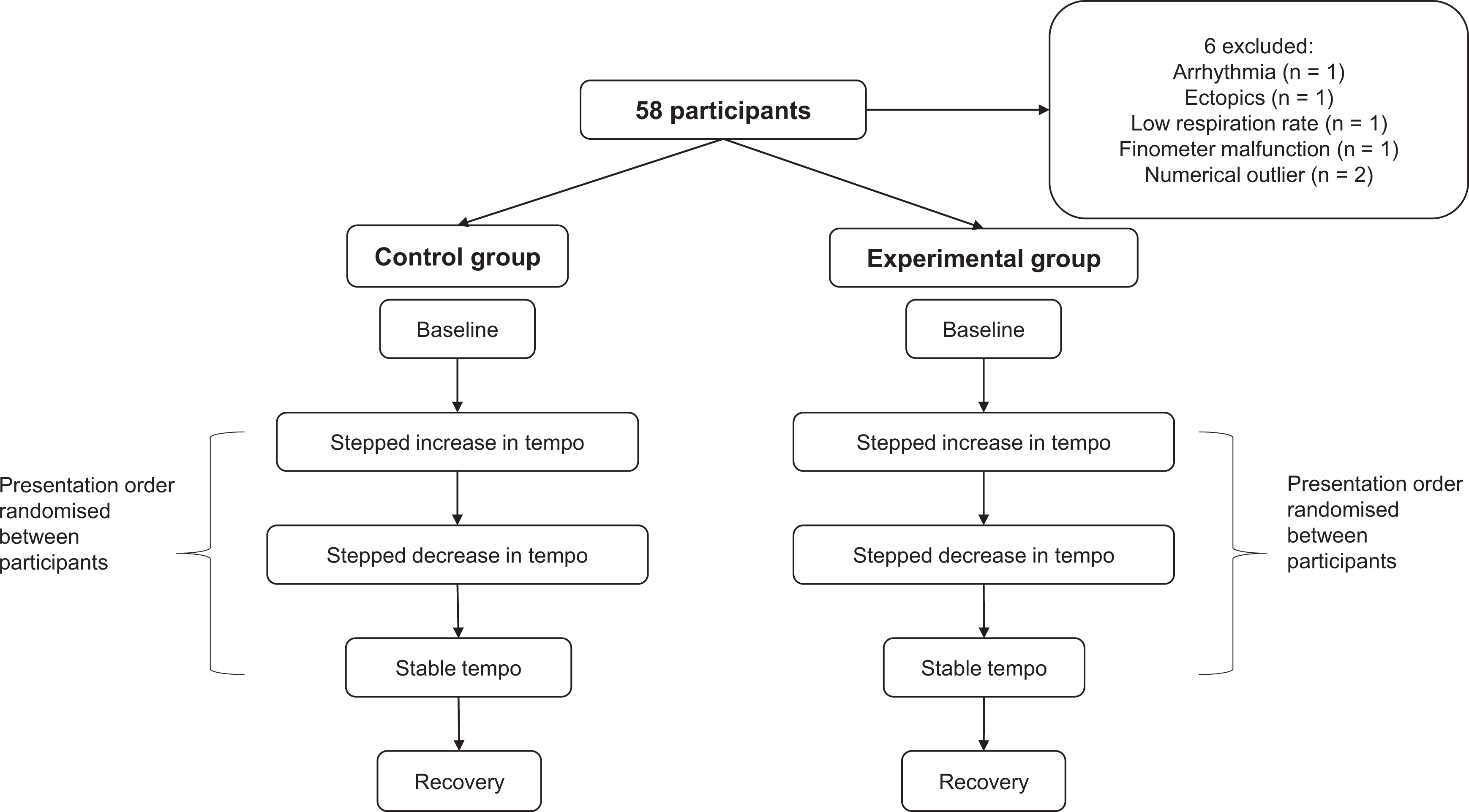
Study procedure.
Participants were asked to relax, remain still throughout, and to refrain from talking or falling asleep. The study room was kept at a constant temperature of 21± 2°C.
Data analysis
All data analysis was performed offline. The following time periods were analysed: final 5 minutes of the baseline and recovery conditions; entire duration of the stable tempo, stepped increase and tempo decrease stimuli; and five individual tempi of the stepped increase and decrease in tempo stimuli. The following measures were derived (see Table 2 for a summary).
Summary of measures derived in the study.

Time-domain HRV
Sequences of RR intervals were analysed statistically to determine: the difference between the maximum and minimum RR intervals (Δ RR), representing overall variability; standard deviation of successive RR intervals (SDRR), also reflecting overall variability; square root of the mean of the squared differences between adjacent RR intervals (RMSSD), reflecting parasympathetic activity; and the percentage of the number of RR intervals that are larger than 50 ms (pRR50), also representing parasympathetic activity (Camm et al., 1996). All of these parameters, along with mean RR, were derived in LabChart 8 (ADInstruments).
Frequency-domain HRV
Frequency-domain HRV applies spectral density analysis algorithms (e.g., Fast Fourier Transform) to a tachogram to identify frequency components that reflect specific autonomic functions. The in-built HRV module in LabChart 8 was used to derive: VLF power, 0.00–0.04 Hz, reflecting thermoregulatory function; LF power, 0.04–0.15 Hz, reflecting combined sympathetic and parasympathetic activity; HF power, 0.15–0.4 Hz, representing vagal (parasympathetic) modulation of HR; and total power, representing global contributions of sympathovagal activity (Camm et al., 1996). From this, the percentage of low and high frequency power relative to total power (%LF and %HF respectively) and normalised LF and HF power (nuLF and nuHF respectively) were derived. Autonomic balance was obtained by deriving the ratio of LF to HF power (LF/HF).
Non-linear HRV
Non-linear analysis entails generating a scatterplot which plots current RR interval against the previous RR interval. Also known as a Poincaré plot, each point represents two successive heartbeats (Guzik et al., 2007). It generates a visual representation of the temporal correlations between the RR intervals and is analysed by fitting an ellipse with its centre point of the markings (Tulppo, Mäkikallio, Seppänen, Laukkanen, & Huikuri, 1998). In healthy volunteers, the Poincaré plot typically resembles a comet shape, whereby a long RR interval is followed by another long RR interval (Woo, Stevenson, Moser, Trelease, & Harper, 1992).
From the Poincaré plot quantitative measures were derived. SD2, the standard deviation of the projection of the Poincaré plot on the line of identify (i.e., the length of the ellipse), is thought to represent both long- and short-term variations in HR (Guzik et al., 2007). SD1, the standard deviation of projection of the Poincaré plot on the line perpendicular to the line of identify (width of the ellipse), is interpreted as a short-term measure of HRV, correlating with HF power (Guzik et al., 2007; Kamen, Krum, & Tonkin, 1996). Both measures were obtained via LabChart 8.
From SD1 and SD2, additional variables were derived, including: nSD1 and nSD2, which are SD1 and SD2 corrected for mean RR interval respectively (formula: (SD1 or SD2/RR interval)*1000); S, the area of the ellipse, representing total variability (formula: π*SD1*SD2); and SD2/SD1, analogous to LF/HF.
Spontaneous sequence BRS
Spontaneous BRS was non-invasively derived via the sequence method. This involved detecting a sequence of three or more beats during which there was either: a progressive increase of ≥1 mmHg in systolic blood pressure (SBP) accompanied by a lengthening of ≥2 ms in RR intervals (“up” sequences); or a progressive decrease in SBP of ≥1 mmHg accompanied by a shortening ≥2 ms in RR intervals (“down” sequences; Parati et al., 1988). BRS values were calculated by plotting RR interval against SBP and calculating the slope of the line fitted through the data points in Excel 2013. The slope of the line was used as an index of BRS: the steeper the slope, the greater the BRS (Parati et al., 1988).
Respiration rate
Respiration rate (per minute) was derived by counting the number of complete inhalation-expiration cycles in a condition and dividing by the condition’s duration.
Muscle sympathetic nerve activity (MSNA)
MSNA frequency (per minute) was calculated by counting all single units that occurred in each condition and dividing by the condition’s duration. MSNA incidence (number of units per 100 heartbeats) was also derived to limit the effect of any changes in HR. This involved dividing MSNA frequency by mean HR and multiplying by 100.
Statistical analysis
Normality was ascertained by the Shapiro-Wilk test. If data were not normally distributed non-parametric equivalents were implemented.
To explore differences in demographic and baseline measures between the experimental and control groups, independent sample t-tests (or Mann-Whitney U tests for not normally distributed data) were performed.
Mixed mode ANOVAs were performed to explore the effects of condition and tempo (within-participants variable) and group (between-subjects variable) on autonomic function. Bonferroni pairwise comparisons were performed on statistically significant main effects and interactions. The Greenhouse-Geisser correction was used when data did not meet sphericity.
Linear regressions were performed to determine whether stimuli responses could be predicted by baseline measures. To examine differences between response types, chi-square tests of independence were performed.
All statistical tests were two-tailed and the alpha level was set to 0.05 except for non-parametric pairwise comparisons. In these cases, the alpha level was divided by the number of pairwise comparisons undertaken.
All data are presented as the mean ± 1 standard error of the mean (SEM) unless otherwise stated.
Results
Data from 6 participants were excluded. This included: 2 males (n = 1 control group) who showed extreme responses (i.e., extreme outliers); 1 female (control group) who had an undiagnosed arrhythmia; 1 female (experimental group) who had >2 ectopic heartbeats in any given 5-minute section; 1 female (control group) whose BP measurement malfunctioned; and 1 male (control group) who had a respiration <10 breaths per minute.
No statistically significant differences in demographic or baseline measures emerged between the control and experimental groups (p > 0.05, see Table 3).
Summary of characteristics of the final sample, experimental group and control group.

The entire block of tempi significantly altered a measure of parasympathetic activity
Comparison of the entire blocks of tempi (whole of the tempo increase and tempo decrease stimuli) to baseline, stable tempo, and recovery revealed that one measure of parasympathetic activity changed, while those of sympathetic or mixed sympathetic/parasympathetic did not.
Reflecting increases in parasympathetic activity, overall variability differed between the conditions (Δ RR main effect of condition: F(4, 200) = 6.12, p < 0.001, η2 = 0.11). Δ RR was significantly higher in tempo increase compared to baseline (p = 0.034), stable tempo (p = 0.038) and recovery (p = 0.005), and during tempo decrease compared to recovery (p = 0.030). Further analysis demonstrated that nSD1, considered a measure of parasympathetic function, significantly changed (main effect of condition: F(2.49, 124.60) = 2.99, p = 0.042, η2 = 0.06): nSD1 was significantly higher in baseline than during the stable tempo stimulus (p = 0.017, see Table 4). Comparing the experimental with the control group revealed no significant differences (main effect of group: p > 0.05), nor were there any interaction effects (p > 0.05).
Summary of results for the five conditions (baseline, stable tempo, tempo increase, tempo decrease, recovery). * = significantly different to baseline; † = significantly different to tempo increase; # = significantly different to recovery. Data presented as the mean (± 1 SEM).

Single MSNA units were confirmed in 10 participants. An example unit with traces from the corresponding cardiovascular tests are provided in Figure 2. Sympathetic nerve activity (MSNA frequency and incidence) did not significantly differ between the conditions (main effect of condition: p > 0.05, see Table 4), indicating that the above change in Δ RR could be primarily due to parasympathetic activity fluctuations.

Example traces from cold pressor (a) and hand grip (b) tests. (c) shows putative individual action potentials that may originate from one MSNA. Overlaying the action potentials (d) confirms one MSNA unit.
Respiration rate significantly differed between the conditions (main effect of condition: F(3.03, 151.30) = 16.97, p < 0.001, η2 = 0.25). Breathing was significantly lower during baseline compared to the three auditory stimuli (all p < 0.001); during stable tempo than tempo increase (p = 0.012); and during recovery compared to tempo increase and tempo decrease (both p = 0.001, see Table 4).
Since there were increases in measures of parasympathetic function but no apparent change in sympathetic function when the whole period of a particular series of tempi changes were compared to baseline as above, further analysis was undertaken of distinct tempi within each increase or decrease series.
The tempo increase manipulations significantly increased parasympathetic measures
Analysis during the 2-minute periods of presentation of relevant tempi revealed that measures reflecting parasympathetic activity increased, while those of sympathetic or mixed sympathetic/parasympathetic did not change.
Consistent with increases in parasympathetic activity, overall variability significantly differed between the tempi of the tempo increase stimulus (main effect of tempo, SDRR: F(3.33, 166.60) = 5.10, p = 0.001, η2 = 0.09; Δ RR: F(4, 200) = 3.47, p = 0.009, η2 = 0.07). Further analysis revealed that measures of parasympathetic function significantly increased during the tempo increase stimulus (RMSSD: F(3.01, 150.60) = 4.77, p = 0.003, η2 = 0.09, SD1: F(3.01, 150.70) = 4.77, p = 0.003, η2 = 0.09 and nSD1: F(3.05, 152.50) = 4.35, p = 0.005, η2 = 0.08, Table 5). SDRR was significantly higher during 180 bpm than during 90 bpm (p = 0.025), 120 bpm (p = 0.003) and 150 bpm (p = 0.049). Also, Δ RR was significantly greater during 180 bpm compared to 120 bpm (p = 0.048) and 150 bpm (p = 0.039). RMSSD, SD1 and nSD1 were all significantly higher during 180 bpm compared to 60 bpm (p = 0.003, p = 0.003 and p = 0.002 respectively).
Summary of results for the five tempi of the tempo increase stimulus. ▪ = significantly different to 60 bpm; • = significantly different to 180 bpm; ♦ = significantly different to 90 bpm. Data presented as the mean (± 1 SEM).

Also consistent with changes in parasympathetic measures, main effects of tempo reached statistical significance for measures of sympathovagal activity: SD2 (F(4, 200) = 4.84, p = 0.001, η2 = 0.09), nSD2 (F(4, 200) = 4.73, p = 0.001, η2 = 0.09) and SD2/SD1 (F(3.20, 159.70) = 2.90, p = 0.034, η2 = 0.06). SD2 and nSD2 were significantly higher during 180 bpm than during 90 bpm (p = 0.041 and 0.029 respectively) and 120 bpm (p = 0.004 and 0.010 respectively). Also, SD2/SD1 was significantly higher in 60 bpm compared to 120 bpm (p = 0.016), suggesting there was a shift towards parasympathetic predominance during 120 bpm (see Table 5).
Sympathetic nerve activity did not significantly differ between the tempi (main effect of tempo: p > 0.05), suggesting that the above changes could be primarily due to parasympathetic activity. In addition, parasympathetic, sympathetic and mixed sympathovagal measures did not significantly differ between the groups (main effect of group: p > 0.05) or interact with tempo and group (interaction effect: p > 0.05).
The tempo decrease manipulations also significantly increased parasympathetic measures
Measures reflecting parasympathetic activity increased for the analysis of the 2-minute periods of presentation of decreasing tempi. In contrast, measures of sympathetic and mixed sympathetic/parasympathetic activity did not change.
Representing increases in parasympathetic activity, overall variability significantly differed between the tempi of the tempo decrease stimulus (main effect of tempo: Δ RR, F(3.46, 173.00) = 3.05, p = 0.018, η2 = 0.06, see Table 6). Measures of parasympathetic activity significantly increased during the tempo decrease stimulus (main effect of tempo, HF power: F(1.96, 98.18) = 7.32, p = 0.001, η2 = 0.13; RMSSD: F(2.09, 98.18) = 7.32, p < 0.001, η2 = 0.18; pRR50: F(2.40, 119.50) = 7.82, p < 0.001, η2 = 0.14; SD1: F(2.09, 104.70) = 11.08, p < 0.001, η2 = 0.18; nSD1: F(2.24, 112.00) = 9.66, p < 0.001, η2 = 0.16, see Table 6). Δ RR was significantly shorter during 150 bpm than 60 bpm; (p = 0.039) and HF power was significantly higher in 60 bpm and 90 bpm compared to 180 bpm (p = 0.018 and p = 0.030 respectively). RMSSD, SD1 and nSD1 were significantly higher during 60 bpm than 180 bpm (p = 0.003, p = 0.003 and p = 0.005 respectively), 150 bpm (p = 0.004, p = 0.004 and p = 0.015 respectively) and 120 bpm (p = 0.019, p = 0.019 and p = 0.041 respectively). Also, RMSSD, SD1 and nSD1 were significantly higher during 90 bpm than 180 bpm (p = 0.006, p = 0.006 and p = 0.008 respectively) and 150 bpm (p = 0.007, p = 0.007 and p = 0.017 respectively). pRR50 was significantly lower during 180 bpm compared to 90 bpm (p = 0.010) and 60 bpm (p = 0.007).
Summary of results for the five tempi of the tempo decrease stimulus. • = significantly different to 180 bpm; ♦ = significantly different to 90 bpm; ▪ = significantly different to 60 bpm. Data presented as the mean (± 1 SEM).

Consistent with the increases in parasympathetic measures, BRS significantly varied between the five tempi (main effect of tempo: F(2.70, 135.20) = 4.17, p = 0.009, η2 = 0.08): BRS was greater during 60 bpm than 180 bpm (p = 0.022) and 120 bpm (p < 0.001, see Table 6).
Sympathetic nerve activity did not significantly differ between the tempi (main effect of tempo: p > 0.05), implying that the above changes could be primarily due to parasympathetic activity. Similarly, autonomic measures did not significantly differ between the groups (main effect of group: p > 0.05) or interact with tempo and group (interaction effect: p > 0.05).
Baseline %LF predicted responses to stimuli
Linear regressions were performed to assess the extent to which baseline measures of autonomic function significantly predicted change during the three stimuli. Baseline %LF power significantly predicted responses to all three stimuli: stable tempo (R 2 = 0.150, p = 0.005), tempo increase (R 2 = 0.149, p = 0.005), and tempo decrease (R 2 = 0.169, p = 0.002). The higher the baseline %LF the greater the decrease in %LF between baseline and stimulus (see Figure 3). Other HRV measures, as well as BRS, MSNA frequency, and MSNA incidence were entered into linear regressions, but none were significant predictors for all three stimuli (p > 0.05).

Baseline %LF significantly predicted responses to stable tempo (a), tempo increase (b), and tempo decrease (c).
Participants were classified into one of three groups: non-responders, if they showed <20% change in %LF between baseline and stimulus; type 1 responders, for ≥20% decrease in %LF between baseline and stimulus; type 2 responder, ≥20% increase in %LF between baseline and stimulus.
Responder types for the three stimuli were significantly associated with each other: response type to tempo decrease was significantly associated with response type to stable tempo (p = 0.002) and tempo increase (p < 0.001). This was also the case for the tempo increase and stable tempo stimuli (p = 0.011). Type 1 responders showed consistent responses to all three stimuli (chi-square test of independence).
As baseline %LF is a significant predictor of response to all three stimuli and ageing is associated with deteriorations in measures of autonomic function, we considered whether age was associated with response type (non-responder, type 1 responder, type 2 responder) for each stimulus (stable tempo, tempo increase, tempo decrease). Mean age of the final sample was 31.98 years. However, since the age range of the sample was large (22–80 years) and not normally distributed (Shapiro-Wilk test: p < 0.001), the median age (26 years) was used as the criterion for determining participant allocation to age group. Therefore, participants aged <26 years were allocated to one group (n = 23, mean age = 23.91 years, SEM = 0.21 years), and those aged ≥26 years were allocated to the other group (n = 29, mean age = 38.38 years, SEM = 2.57 years). Chi-square tests of independence revealed that age group was not significantly associated with response type for any of the three stimuli (p > 0.05).
Discussion
This study showed that when the entire block of tempi was analysed, one measure of parasympathetic activity changed (nSD1). Additionally, parasympathetic activity parameters increased for the final tempo of the tempo increase stimulus (180 bpm) and the final tempo of the tempo decrease stimulus (60 bpm). Measures of sympathetic and mixed sympathetic/parasympathetic did not change at any point, suggesting that the observed increases in overall variability in HR were primarily due to increases in parasympathetic activity. In addition, for the first time, this study shows that it is possible to predict changes in measures of autonomic function to tempo manipulations based on resting levels of some of these variables. Figure 4 summarises these main findings.

Take-home message; 180 bpm in the tempo increase stimulus and 60 bpm in the tempo decrease stimulus were associated with increases in measures of parasympathetic activity. When the entire block of tempi was analysed, tempo increase had significantly greater overall variability than stable tempo and tempo decrease.
Tempo manipulations impact measures of parasympathetic activity
Music played at slow speeds is associated with increases in measures of parasympathetic activity. For instance, van der Zwaag, Westerink, and van den Broek (2011) showed that RMSSD was significantly higher during slow compared to fast music. Iwanaga, Kobayashi, and Kawasaki (2005) demonstrated that HF power was significantly higher during sedative compared to excitative music. Also, Iwanaga (1995b) and van Dyck et al. (2017) showed that stimuli decreasing in tempo were potentially more effective at boosting autonomic tone, reflected by decreased HR, than those that increased in tempo. Similarly, in this study, when the 2-minute distinct tempi of the tempo decrease stimuli were analysed, measures reflecting parasympathetic activity increased. For instance, 60 bpm was associated with the increases in HF power, RMSSD, pRR50, SD1, nSD1, and BRS. This pattern was accompanied by increases in measures of overall variability in HR (Δ RR) in the absence of any change in sympathetic nerve activity (ascertained from MSNA frequency and incidence) or mixed sympathetic/parasympathetic activity. This suggests that the observed increases in measures of overall variability in HR could have been due to the detected increases in measures reflecting parasympathetic activity.
Similar changes in measures of autonomic function occurred for the final tempo of the tempo increase stimulus (180 bpm): parasympathetic activity measures increased, as did those for overall variability in HR, but there was no change in sympathetic or mixed sympathetic/parasympathetic activity. Initially, these findings parallel those of Iwanaga (1995b), Peretz, Gagnon, and Bouchard (1998), and van Dyck et al. (2017), who showed that the direction of stepped change (increase or decrease) impacts autonomic responses to the tempi. This is further supported by the results of the analysis for the entire block of tempi reported here: parasympathetic activity parameters only differed between stable tempo and baseline, compared to between tempo increase/decrease and baseline. But, the findings are also rather surprising: one may have expected the 180 bpm tempo to be associated with low measures of parasympathetic activity and high values of sympathetic nerve activity. As 180 bpm of the tempo increase stimulus and 60 bpm of the tempo decrease stimulus were the final tempi, it is feasible that listener expectations may have influenced these autonomic measures (Meyer, 1956). From the outset of the study, participants were aware that they would hear stimuli that altered tempo, lasting 10 minutes in duration. Therefore, shifts towards parasympathetic predominance and increases in overall variability in HR that occurred for both tempi may have been driven by the anticipation of reaching the end of the stimulus. An alternative interpretation could pertain to the metrical interpretation of the tempi listeners used when listening to the stimuli. As this factor was not ascertained in the study, it is unclear how participants interpreted the 180 bpm tempo of the tempo decrease stimulus. Consequently, a future study could explore the effects of random changes in musical tempo on measures of autonomic function while obtaining a measure of metrical interpretation that has little cardiovascular impact – for example, finger tapping.
Attenuated parasympathetic predominance is associated with poor health outcomes. For instance, vagal tone is negatively correlated with all-cause mortality (see Thayer & Lane, 2007 for a review) and age (Stein, Klieger, & Rottman, 1997), and is lower in a number of conditions, including heart failure (Binkley, Nunziata, Haas, Nelson, & Cody, 1991). As the tempo manipulations appeared to be effective at modulating measures of parasympathetic activity, it would also be interesting to investigate the effects of presenting stimuli similar to those employed here to specific groups in which vagal tone is impaired – for example, heart failure patients and individuals aged over 60 years. Furthermore, it would be interesting to combine stimuli such as these with other stimuli that have different autonomic effects. For instance, a non-invasive form of neuromodulation which targets small electrical impulses to parts of the outer ear innervated by the auricular branch of the vagus nerve has been shown to reduce sympathetic nerve activity (assessed by microneurography, Clancy et al., 2014). This stimulation is known as transcutaneous vagal nerve stimulation (tVNS) and is being incorporated into “music therapy” devices that administer tVNS and music (e.g., the Net-1000 device and Nervana headphones). As tVNS reduces sympathetic nerve activity (Clancy et al., 2014) and the 60 bpm tempo of the tempo decrease stimulus and 180 bpm of the tempo increase stimulus augmented measures of parasympathetic outflow, combining similar tempo manipulations with tVNS would be an interesting research avenue to pursue.
Interestingly, measures of autonomic function did not significantly differ between the group who received the original melody (experimental) and the group who received the rhythm-only stimuli (control). This is surprising, as we initially expected the control group to show lower values for measures of parasympathetic activity (e.g., HF power, RMSSD, pRR50, SD1, and nSD1) and overall variability in HR (e.g., SDRR and Δ RR), and larger values indicating heightened sympathetic nerve activity (e.g., MSNA frequency and incidence). These expectations were based on findings from Khalfa et al. (2008), who showed that DBP was significantly higher during an original melody compared to a rhythm control stimulus (pitch variations removed) and a tempo control stimulus (rhythmic variations removed). It is possible that these discrepancies are due to differences in the duration and familiarity of the stimuli employed, as well as the types of control stimuli used. Nevertheless, these two studies highlight the importance of including a control group in studies investigating the autonomic effects of music-related stimuli.
Unlike previous work which has controlled respiratory rate (e.g., Watanabe et al., 2017), participants in the current study were allowed to breathe spontaneously. This was to ensure participants remained focused on the stimuli and not on their breathing (which could be interpreted as a form of meditation). As respiration rate significantly increased during the three auditory stimuli, it needs to be carefully considered whether the changes in HRV can be accepted as real or as due to respiratory fluctuations. As there were few differences between baseline, stable tempo, tempo increase, tempo decrease, and recovery in the HRV measures, it could be argued that the changes in respiration did not significantly alter the measures of autonomic tone. This reinforces the point that studies measuring HRV should always record respiratory rate in order to ascertain the extent to which changes in autonomic tone resulting from music are real or a result of fluctuations in respiration. Since the spontaneous breathing used here could be a limitation with the current study, comparing the autonomic impacts of controlled versus free breathing while presenting tempo manipulations would be a worthwhile future research project.
Baseline %LF power predicts responses to tempo manipulations
Intra-individual variation in response patterns to music have previously been reported (e.g., Ellis & Brighouse, 1952; Siddle & Heron, 1976; Haas, Distenfeld, & Axen, 1986; Khalfa, Isabelle, Jean-Pierre, & Manon, 2002). Although it is not fully known whether these responses can be ascertained from resting values, for musical tempo there appears to be a close relationship between resting HR and the speed of music. For instance, Iwanaga (1995a) identified a preference (measured by adjusting a control dial) for tempi that were 1, 1.5, and 2 times as fast as resting HR. In a different study, Watanabe et al. (2017) revealed a negative correlation between baseline HR and change in HR between baseline and stimulus, suggesting that HR during the stimulus shifted towards the speed of the music. However, there is little work exploring whether other (perhaps more sensitive) indicators of autonomic tone correlate with responses to tempo manipulations.
Despite employing a different stimulus (tVNS), baseline LF/HF (taken to reflect sympathovagal balance) significantly predicted change in LF/HF between baseline and stimulation (Clancy et al., 2014). Although tVNS is different to music, we aimed to ascertain whether autonomic responses to music may also be predicted by baseline autonomic balance.
For the first time, this study showed that the percentage of LF power during baseline (%LF) was a significant predictor of change in %LF between baseline and the three stimuli (stable tempo, tempo increase, tempo decrease). Higher resting %LF was associated with larger decreases in %LF between baseline and the stable tempo, tempo increase, and tempo decrease stimuli. Both parasympathetic and sympathetic activity are thought to contribute to LF power (Akselrod et al., 1981). Some participants showed similar changes in %LF to all three stimuli, meaning that changes in autonomic function to one stimulus (e.g., tempo increase) were similar to those for the other stimuli (tempo decrease, stable tempo). Although a (somewhat arbitrary) 20% threshold was used to ascertain response type (non-responder, type 1 responder, type 2 responder), it was a conservative criterion for differentiating between response directions and magnitudes. Due to the novelty of this finding, replication studies that test the predictive power of this and other measures of autonomic function would be worthwhile.
Age group was not significantly associated with response type for any of the three stimuli in the study. However, since ageing is associated with shifts towards sympathetic predominance and attenuations in parasympathetic outflow, and baseline %LF predicted direction and magnitude of change during the three stimuli, further exploration of the link between response type and age would be worthwhile. Indeed, it is plausible that when tasked with lowering the amount of LF power (or sympathetic predominance), the effectiveness of simple tempo manipulations (and indeed, more sophisticated musical stimuli) may be higher in specific age groups.
By combining indirect measures of autonomic function with a direct measure of sympathetic outflow (MSNA), we have been able to provide a more mechanistic account of the relationship between tempo manipulations and changes in autonomic tone. This therefore has methodological impacts for future work exploring the effects of music on autonomic function, together with wider research exploring how alternative stimuli may confer autonomic benefits. Exploration of intra-individual differences in autonomic responses to tempo manipulations is another important aspect of this article. This is particularly noteworthy given the increasing use of music as a therapeutic tool. Indeed, these results highlight the possibility of using baseline readings to identify potential individuals who are likely to gain greater benefits from interventions such as music and tVNS, reducing costs and improving outcomes. With further work exploring the reproducibility of baseline measures in predicting the magnitude and direction of response to stimuli, it will be possible to further understand why individuals respond differently to music (along with other stimuli) and if it is possible to determine the nature of this response from the outset.
Conclusion
The tempo manipulations employed here significantly modulated measures of parasympathetic activity, and in turn overall variability in HR. In contrast, the tempo manipulations used here had little impact on measures of sympathetic or mixed sympathetic/parasympathetic activity. The 60 bpm in an increasingly slower stimulus and 180 bpm in an increasingly faster stimulus were associated with the greatest shifts towards parasympathetic predominance, suggesting that direction of tempo change and listener expectations may have played an important role. Autonomic responses to the stimuli were predicted by resting %LF, a novel finding that may or may not generalise to more complex musical stimuli. This more detailed finding is particularly important given the demand for “alternative” therapies in treating both physical and mental conditions, and more or less structured attempts to meet this demand by general and specialist practitioners. It should not be a surprise to find that volunteers might respond differentially to musical treatments. Identifying responders and non-responders on the basis of baseline data should be an aspiration for research on music’s application to health and wellbeing if one wishes to maximise its effectiveness.
Footnotes
Author contribution
BB, JD, and LW researched the literature and conceived the study. BB was involved in the study design, gaining ethical approval, participant recruitment, data collection, and data analysis. BB wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by SEMPRE’s Arnold Bentley New Initiatives Fund.
Peer review
Loïc Damm, University of Montpellier, EuroMov laboratory. One anonymous reviewer.
