Abstract
Objective:
To study the role and underlying mechanisms of dihydromyricetin on the myocardial function in mice induced by CCl4.
Methods:
Eighteen C57BL/6 mice (6–8 W, female) were randomly divided into the following three groups: control group, CCl4-induced positive group (CCl4 group), dihydromyricetin group, six mice/group. NLRP3-deficient (NLRP3-/-) C57BL/6 mice used the same age, gender, and modeling method. The HL-1 cells were used for in vitro experiments. The HL-1 cells were treated with PBS, CCl4, and CCl4 + DMY respectively.
Results:
The RT-qPCR results showed that compared to the mice induced by CCl4, the dihydromyricetin increased the Arg-1 mRNA level in the mouse myocardial tissues. The mRNA expressions of the iNOS, IL-33, and ST2 were reduced by the dihydromyricetin. The results of immunohistochemistry showed that dihydromyricetin decreased IL-33 protein expressions in the myocardial tissues. Western blot results also showed that compared with the control group, the activation of NLRP3 inflammasomes in the myocardial tissues of mice injured by CCl4 was increased, and dihydromyricetin can reduce NLRP3 inflammasomes activation in the myocardial tissues induced by CCl4. The results of ELISA showed that dihydromyricetin could reduce the IL-1β level in the serum of the mice induced by CCl4. Consistent with the in vivo results, compared with the control group, the NLRP3 inflammasome activation and IL-33/ST2 expression were increased in the CCl4-treated HL-1 cells, while DMY significantly weakened this effect. Interestingly, NLRP3 deficiency enhanced the protective effect of DMY on myocardial function in mice.
Conclusions:
IL-33/ST2 signaling pathways and NLRP3 inflammasome activation may be involved in dihydromyricetin improving the myocardial function of the mice induced by CCl4.
Introduction
Currently, cardiovascular disease is a common disease in our country and even in the world. 1 Deaths from cardiovascular disease is twice as many as cancer, accounting for 31.5% of global deaths and 45% of non-infectious disease deaths. 1 Cardiac insufficiency is a common clinical symptom of cardiovascular disease. 2 Dihydromyricetin (DMY), a kind of flavonoids, has anti-inflammatory, antioxidant, and anti-tumor effects. 3 Whether dihydromyricetin can improve the myocardial function of the mice induced by CCl4 has not been reported.
NOD-like receptor protein 3 (NLRP3) inflammasomes belong to the innate immune receptors, mainly including NLRP3, apoptosis-associated speck-like protein containing a CARD (ASC) and pro-cysteinyl aspartate specific proteinase-1 (pro-caspase-1). 4 NLRP3 is activated by pattern recognition receptors (PRRs) on the surface of the cell membrane that recognize extracellular pathogen related molecular patterns (PAMPs) and intracellular damage related molecular patterns (DAMPs). 5 After the NLRP3 activation, ASC and pro-caspase-1 were recruited successively to form the NLRP3 inflammasome. After the activation of NLRP3 inflammasome, pro-caspase-1 was self-activated to form caspase-1. Caspase-1 is activated to catalyze pro-IL-33 to form IL-33, which is secreted to extracellular environment. 6
IL-33 belongs to the family of IL-1 and is usually expressed in the nuclei of a variety of cells. 7 The main cells involved include endothelial cells and cardiomyocytes in the vascular system. 8 In general, IL-33 in the nucleus binds to chromatin and plays a role in transcriptional inhibition. 9 When the cells are stimulated by exogenous substances, IL-33 will be released outside the cells. 9 IL-33 alerts neighboring cells as endogenous danger signal. 10 After IL-33 acts on suppression of tumorigenicity 2 (ST2) receptor on the surface of adjacent cell membrane, IL-1 receptor accessory protein (IL-1RAcP) is recruited to form heterodimer complex. 11 After heterodimer formation, nuclear factor-κ B (NF-κ B) and mitogen-activated protein kinase (MAPK) signal pathways are activated by myeloid differentiation primary response gene 88 (MyD88), which leads to the occurrence and development of the immune response. 12
In this experiment, myocardial function and cardiac index were detected at the mouse level. The microscopic state of the heart was observed by HE staining. The expression of IL-33 and ST2 in mouse myocardium was detected by RT-qPCR. The expression of IL-33 in mouse myocardium was detected by immunohistochemical. The activation of NLRP3 inflammasome in mouse myocardium was detected by Western blot. The secretion level of IL-1 β was detected by ELISA. The oxidative stress in the myocardial tissues were detected using biochemical kit. We observed whether dihydromyricetin can improve the myocardial function of CCl4-injured mice and whether IL-33/ST2 and NLRP3 inflammasome activation pathway are involved in this process, in order to find drugs to improve myocardial function caused by CCl4 poisoning and explore potential molecular targets.
Material and methods
The mice and main reagents
Female C57BL/6 mice aged 6–8 W and weighted 18–20 g were purchased from Hunan SJA Laboratory Animal Co., Ltd and maintained in the specefic pathogen free (SPF) environment. NLRP3-deficient C57BL/6 mice were provided by Xinxiang Medical University with support of 111 program (D20036). The age, weight, and feeding conditions of the mice were the same as those of C57BL/6 mice. The lack of NLRP3 expression in the NLRP3-deficient mice was verified by RT-qPCR and Western blot. This study was approved by the Experimental Animal Ethics Committee of Gannan Medical University (Approval number: 2023494). CCl4 and dihydromyricetin were purchased from sigma.
Establishment of the model
Eighteen female C57BL/6 mice and eighteen female NLRP3-deficient C57BL/6 mice were selected and randomly divided into the following three groups respectively: control group, CCl4-induced positive control group (CCl4 group), dihydromyricetin group, six mice in each group. Among them, CCl4 (10%) was administered intraperitoneally to mice from the day of modeling, two times/W for 8 W. The dihydromyricetin (10 mg/ml) was given to the mice intragastrically, seven times/W for 8 W, from 7 days before the modeling. The mice in the control group were given equal doses of dihydromyricetin and CCl4 solvent (Figure 1).

The schematic diagram of in vivo and in vitro experimental procedures.
Survival analysis and weight change
The survival of the mice was observed every day, and the body weight was measured and the weight change curve was drawn once a week.
Detection of cardiac function and blood pressure
The systolic and diastolic blood pressures of the mice were measured via the tail artery using a small animal non-invasive blood pressure measuring instrument. The mice were placed in a mouse bag and the sensor was put on the tail. After observing the pulse, inflate until the pulse disappears, and then deflate until the pulse appears. At this time, the reading is recorded as the systolic blood pressure; then continue to deflate until the pulse amplitude reaches its maximum, and at this time the reading is recorded as the diastolic blood pressure.
After anesthetizing the mice with pentobarbital sodium (1%, 0.1 ml), the mice were placed on the operating table on their back. Chest hair was shaved and the B-mode echocardiographic imaging detection is performed at the level of the cardiac papillary muscle (with a probe frequency of 16 MHz). The left ventricular end-systolic diameter (LVIDs), diastolic left ventricular diameter (LVIDd), left ventricular ejection fraction (LVEF) and left ventricular fractional shortening (LVFS) were measured by small animal echocardiography (Vivo 2100, VisualSonics). All data were measured three times and the average value was taken as the numerical value of the indicator.
Collection of the heart samples
At 8W after the modeling, the mice were sacrificed by CO2 anesthesia. Hearts were removed and cardiac index was calculated. Part of the heart was placed in paraformaldehyde (4%) for pathological examination, and the other part of the heart was frozen in a refrigerator (−80°C) for molecular biology detection.
HE staining
After myocardial tissue was fixed, it was routinely dehydrated and embedded in paraffin. The 4 μm slices were routinely dewaxed to water and stained with HE staining. The microscopic state of myocardial tissue was observed under a 100-fold microscope.
Immunochemistry
The sections were detected by IL-33 immunohistochemistry. The sections were routinely dewaxed to water, and the exposed antigens were used for microwave repair for 15 min. The antigens were incubated with endogenous hydrogen peroxide in the dark for 25 min, and were incubated with BSA for 30 min at room temperature. The sections were incubated with rabbit anti-mouse IL-33 antibody (1:100 dilution) overnight (4°C). The HRP-labeled secondary antibody (goat anti-rabbit) was incubated for 50 min. The sections were first developed with DAB for 2–4 min, and then counterstained with hematoxylin for 30 s. Taking the average optical density of brownish yellow in the visual field as positive, the percentage of positive area was analyzed by Image-ProPlus 6.0 software and semi-quantitative analysis was performed.
The culture and treatment of HL-1 cells in vitro
The HL-1 cell line was purchased from Procell Life Science&Technology Co., Ltd. The HL-1 cells were inoculated into six-well plates at 1 × 106/ml. After 24 h culture in the DMEM medium, the cells of logarithmic growth stage were selected for experiments. The HL-1 cells were treated with PBS, CCl4, and CCl4 + DMY respectively. The cells were given with CCl4 (40 mmol/l) for 1 h and DMY (50 μmol/l) for 24 h. After the treatment, cells were collected for the next experiments (Figure 1).
RT-qPCR
The myocardial tissue (100 mg) was extracted, and the Trizol solution (1 ml) was added to extract RNA. Reverse transcription and RT-qPCR amplification were completed according to the kit. The RT-qPCR result is represented by 2-△△CT. The sequences of the NLRP3, IL-1β, iNOS, Arg-1, IL-33, ST2, and GAPDH primers were synthesized by Sangon Biotech (Shanghai) Co. Ltd (Table 1).
Real time quantitative PCR primer sequence.

Western blot
The myocardial tissue (120 mg) was extracted, and the RIPA lysate (1.2 ml) was added to extract protein. Protein concentration was measured by BCA kit and protein denaturation was performed. The denatured proteins were subjected to SDS-PAGE gel electrophoresis and transferred to membrane. On the PVDF membrane transformed with denatured protein, we added primary antibody (NLRP3, pro-caspase-1, caspase-1, abcam, 1:1000 dilution), incubated overnight (4°C) and added secondary antibody (HRP labeled rabbit anti mouse IgG, Jackson, 1:5000 dilution) and incubated for 1 h. Protein bands were detected with enhanced chemiluminescence (ECL). Using GAPDH as an internal reference, the relative expression levels of the target bands were analyzed by ImageJ software.
Detection of oxidative stress-related indicators in the myocardial tissues
The oxidative stress-related indicators were detected using biochemical kit (provided by Nanjing Jiancheng Bioengineering Institute) according to the instructions. Myocardial tissues were extracted, homogenized and centrifuged to detect oxidative stress-related indicators such as hydrogen peroxide (H2O2), malondialdehyde (MDA), superoxide dismutase (SOD), peroxidase (POD), and creatine kinase (CK). Add the samples according to the reagent kit, and then conduct water—bath treatment at 95°C for 40 min. After centrifugation at 4000 rpm for 10 min, take the supernatant and measure the OD value. The OD value of each test tube was measured by a microplate reader, and the OD value of the relevant indicators in the myocardial tissues was measured according to the standard curve.
Statistical analysis
The experimental data were presented in the form of mean ± standard deviation, and all the data were analyzed by SPSS 17.0 or GraphPad Prism 5.0 software. Data of four groups were compared and one-way ANOVA was used. p < 0.05, the data were statistically significant.
Results
The survival of the mice with weight change curve
No mice died during the experiment. The mice in the control group were in good condition, exercising lively, eating and drinking normally, and their coat color was shiny. The mice in the CCl4 group and the dihydromyricetin group showed reduced food and water intake, poor exercise activity, psychasthenia, abnormal hair color, not smooth, and some mice appeared piloerection. There was no significant difference in the body weight between the control group and the CCl4 group, and the body weight increased steadily until the end of the experiment. The body weight of the mice in the dihydromyricetin group was generally lower than that in the control group and CCl4 group, especially at 14d, 21d, and 56d after CCl4 administration (Figure 2).
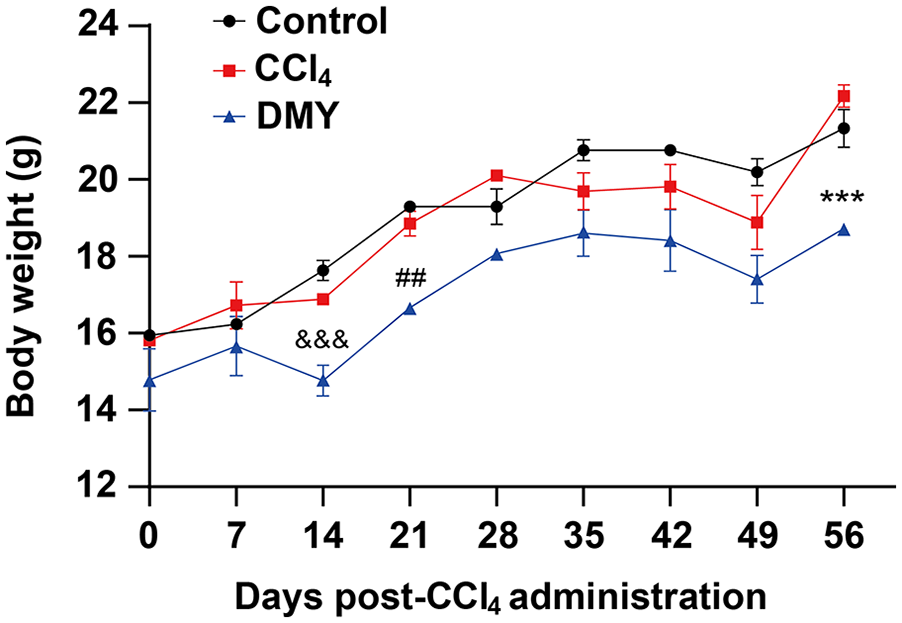
Body weight change curve of the mice in different groups. Eighteen female C57BL/6 mice were randomly divided into the three groups: the control group (given vehicle only), the CCl4 group (given CCl4 with vehicle) and the dihydromyricetin group (given dihydromyricetin 7 days prior to CCl4 administration), with six mice in each group. The body weights were monitored every day, and the weight change curve was drawn in weeks (n = 18).
The effect of dihydromyricetin on cardiac weight and cardiac index in mice
As shown by Figure 3(a), there was no significant difference in body weight and heart weight between the CCl4 group and the control group. Compared with CCl4 mice, DMY mice had lower body weight and heart weight (Figure 3(a and b)). There was no significant difference in cardiac index among the three groups (Figure 3(c)).

DMY reduced the increase of heart weight caused by CCl4. (a) The weight of the three group mice was weighed and compared before with the dissection (n = 18). (b) Take out the mouse hearts, weigh and compare the weight of the mouse hearts among the three groups (n = 18). (c) The heart indexes were calculated. The heart indexes = heart weight/body weight of mice before with the dissection (n = 18).
The effect of dihydromyricetin on cardiac function in mice
The left ventricular cardiac function of mice was detected by echocardiography. Compared with the control group, the LVIDs and LVIDd of left ventricle in CCl4 group increased, while LVEF and LVFS decreased. Compared with CCl4 group, DMY can reduce LVIDs and LVIDd, and improve the effect of LVEF and LVFS. In addition, the change trend of systolic blood pressure and systolic blood pressure is consistent with that of the LVID (Table 2).
The effect of dihydromyricetin on the cardiac function in mice (mean ± SD).

Compared with the blank control group, *p < 0.05, **p < 0.01; compared with the CCl4 group, #p < 0.05.
The effect of dihydromyricetin on cardiac hypertrophy in mice
The results of HE staining showed that the cardiomyocytes in the control group were neatly arranged, the structure was clear, and the number of apoptotic cells decreased. In CCl4 group, the nucleus of cardiac muscle shrank, the cell body enlarged and the intermuscular space widened. The morphology of cardiomyocytes in DMY group tended to be normal (Figure 4).

The effect of dihydromyricetin on histomorphology of the mouse heart. The myocardial tissues in the mice were extracted, embedded in the paraffin and stained with HE staining. The myocardial tissue morphology was observed under the microscope (100×). The arrow indicates the main site of the lesion.
The effect of dihydromyricetin on inflammatory indexes of myocardial tissues
Compared with the control group, the mRNA expression of iNOS in CCl4 group increased, while the mRNA expression of Arg-1 decreased. Compared with CCl4 group, DMY could reduce the mRNA expression of iNOS in the myocardial tissue (Figure 5(a)). Although there was no significant difference in the mRNA expression of Arg-1 in myocardial tissue between DMY group and CCl4 group, the mRNA expression of Arg-1 in myocardial tissue of DMY group was higher than that of CCl4 group (Figure 5(b)).

DMY alleviated the cardiac inflammation caused by CCl4. (a) The iNOS mRNA expressions in the myocardium tissues of the mice were detected by the RT-qPCR (n = 18). (b) The mRNA expressions of Arg-1 in the myocardium tissues were determined by the RT-qPCR (n = 18).
The effect of dihydromyricetin on the activation of NLRP3 inflammasome
In vivo experiments studied the effect of DMY on the activation of NLRP3 inflammasome in the mouse myocardial tissues. The results of Western-blot showed that compared with the control group, the activation of NLRP3, IL-1β, and Caspase-1 in myocardial tissue of mice in CCl4 group increased. Compared with CCl4 group, IL-1β and Caspase-1 activation in myocardial tissue of DMY group decreased (Figure 6(a–d)). The expression of IL-1β in myocardial tissue determinated by RT-qPCR and the content of IL-1β in serum detected by ELISA were also in line with the above trend (Figure 6(e and f)). In vitro experiments studied the effect of DMY on the activation of NLRP3 inflammasome in the HL-1 cardiomyocytes. Consistent with the in vivo results, compared with the control group, the levels of Caspase-1 and IL-1β increased in the CCl4-treated HL-1 cells, while DMY significantly weakened this effect (Figure 6(g–l)).

DMY inhibited the NLRP3 inflammasome activation induced by CCl4. Eighteen female C57BL/6 mice were randomly divided into the three groups: the control group (given CCl4 and dihydromyricetin solvent), the CCl4 group (given CCl4) and the dihydromyricetin group (given dihydromyricetin 7 days before CCl4 administration), with 6 mice in each group. (a–d) The protein levels of the NLRP3, IL-1β, pro-IL-1β, Caspase-1, pro-caspase-1, and GAPDH in the myocardial tissues were measured by Western Blot (n = 18). (e) The expression of IL-1β in myocardial tissue was determinated by the RT-qPCR. (f) The IL-1β levels in the serum were detected by the ELISA (n = 18). The HL-1 cells were treated with PBS (Control), CCl4, and CCl4 +DMY respectively. (g–j) The protein levels of the NLRP3, IL-1β, pro-IL-1β, Caspase-1, pro-caspase-1, and GAPDH in the HL-1 cells were measured by Western Blot (n = 3). (k) The expression of IL-1β in the HL-1 cells was determinated by the RT-qPCR (n = 3). (l) The IL-1β levels in the HL-1 cell supernatant were detected by the ELISA (n = 3).
The effect of dihydromyricetin on oxidative stress indexes in myocardial tissues
As shown in Table 3, compared with the control group, the contents of H2O2, MDA, and CK in serum of mice in CCl4 group increased, while the contents of SOD and POD decreased. Compared with CCl4 group, the content of H2O2 and MDA in serum of DMY group decreased, while the content of SOD and POD increased.
The effect of dihydromyricetin on oxidative stress indexes in the myocardial tissues.

Compared with the blank control group, *p < 0.05, ***p < 0.001; compared with the CCl4 group, #p < 0.05, ##p < 0.01.
The effect of dihydromyricetin on IL-33/ST2 signal pathway
In vivo experiments also studied the effect of dihydromyricetin on the IL-33/ST2 signaling pathway in the mouse myocardial tissue. The mRNA and protein expression of IL-33 and ST2 in the liver of CCl4 group was higher than that of control group. DMY could reduce the mRNA and protein expression of IL-33 and ST2 in the liver of mice in CCl4 group (Figure 7(a and b)). In addition, the results of immunohistochemistry showed that compared with the control group mice, the expression of IL-33 in the liver from the mice induced by CCl4 was increased; compared with the CCl4 group mice, the expression of IL-33 in the liver from the mice given by DMY was reduced (Figure 7(c)). In vitro experiments studied the effect of DMY on the IL-33/ST2 signaling pathway in the HL-1 cardiomyocytes. Consistent with the in vivo results, the RT-qPCR results showed that compared with the control group, the CCl4 increased the mRNA expression of IL-33 and ST2 in the HL-1 cells; however, compared with the CCl4 group, the DMY significantly decreased the mRNA expression of IL-33 and ST2 (Figure 7(d)). In addition, the Western blot results also showed a consistent trend (Figure 7(e)).

DMY decreased the expressions of IL-33 and ST2 induced by CCl4. (a and b) The mRNA and protein expressions of IL-33 and ST2 in the myocardium tissues of the mice were detected by the RT-qPCR and Western Blot, respectively (n = 18). (c) The immunohistochemistry showed the IL-33 expression (scale = 50 μm). (d) The expressions of IL-33 and ST2 in the HL-1 cells were detected by RT-qPCR (n = 3). (e) The expressions of IL-33 and ST2 in the HL-1 cells were also detected by Western Blot (n = 3).
NLRP3 deficiency enhanced the protective effect of DMY on myocardial function in mice
To understand whether NLRP3 is involved in the improvement of myocardial function by DMY, we used NLRP3-deficient mice to model. The changes of left ventricular function (systolic pressure, diastolic pressure, LVIDs, LVIDd, LVEF, and LVFS) were detected by echocardiography before dissecting the mice, as shown in Table 4, NLRP3 deficiency enhanced the protective effect of DMY on myocardial function and blood pressure in mice. The liver of mice was taken, and the NLRP3 expression was detected by RT-qPCR and Western-blot. The results suggested that the NLRP3 expression in the NLRP3-deficient mice is low (Figure 8(a and b)). We compared the myocardial function in the WT and NLRP3-deficient mouse models. The results showed that compared with the WT mice, LVIDs and LVIDd in the left ventricle of NLRP3-deficient mice decreased, while LVEF and LVFS increased. In addition, compared with the WT mice, the protective effect of DMY on myocardial function in the NLRP3-deficient mice was increased (Figure 8(c–f)).
The effect of dihydromyricetin on cardiac function in NLRP3-/- mice (mean ± SD).

Compared with the control group, *p < 0.05; compared with the CCl4 group, #p < 0.05.

NLRP3 deficiency enhanced the protective effect of DMY on myocardial function in mice. NLRP3-deficient (NLRP3-/-) C57BL/6 were selected and performed in the same grouping and modeling methods as WT mice. Taking the mouse tails, RT-qPCR (a) and western blot (b) detected the NLRP3 expression in the tails of NLRP3-deficient (NLRP3-/-) C57BL/6 (n = 18). (c–f) The changes of left ventricular function (LVIDs, LVIDd, LVEF, and LVFS) were detected by echocardiography (n = 18).
Discussion
The cardiovascular system plays a vital role in the body. Abnormal myocardial function is a common clinical symptom of cardiovascular disease. 13 The abnormal myocardial function is reflected by the hypertrophy of cardiomyocytes. 14 Therefore, it is urgent to improve the abnormal myocardial function. Dihydromyricetin belongs to flavonoids and is a common hepatoprotective drug. 3 The studies have shown that dihydromyricetin has anti-inflammatory, antioxidant, and anti-free radical effects. 3 Our study found that dihydromyricetin can effectively improve myocardial function in CCl4-induced mice, and NLRP3 inflammasome and IL-33/ST2 may be involved in this process.
NLRP3 inflammasome, as a member of pattern recognition receptors, is involved in the occurrence and development of various inflammatory diseases.15 –17 The process is that after the NLRP3 inflammasome is activated, it activates caspase-1, promotes the maturation and release of IL-1β and IL-18, and then triggers an inflammatory response. 5 Our results show that dihydromyricetin reduces NLRP3 inflammasome activation in the mouse myocardium tissues induced by CCl4. The studies have shown that dihydromyricetin attenuates azithromycin-induced cardiotoxicity by activating SIRT1 and inhibiting the activation of NLRP3 inflammasome. 18 Our findings are consistent with the report written by Zhenzhu Sun that dihydromyricetin can reduce cardiotoxicity and improve cardiac function. This process is mainly achieved by inhibiting the activation of caspase-1 and reducing the content of IL-1β. By comparing the effects of DMY on the myocardial function of WT and NLRP3-deficient mice, we found that NLRP3 deficiency enhanced the protective effect of DMY on the myocardial function in the mice. Our results were consistent with that DMY could reduce myocardial injury induced by azithromycin by inhibiting NLRP3 inflammasome. 18 The possible reason was that DMY played an anti-inflammatory role through NLRP3 to protect myocardial function. The latest research also reported that NLRP3 deficiency resisted Ang II-induced cardiac insufficiency by inhibiting mitochondrial function. 19 We speculated that the mitochondrial dysfunction may be the reason why DMY played an anti-inflammatory role through NLRP3. However, some studies reported that NLRP3 deficiency aggravated the infection of Coxsackie Virus B3 in mice, that is, NLRP3 deficiency could not protect the body. 20 We believe that NLRP3 responds differently to different antigens. Our findings provide experimental evidence for the treatment of NLRP3 inflammasome in cardiac diseases by dihydromyricetin.
Oxidative stress is involved in the occurrence and development of a variety of chronic diseases to clear senescent cells, but when it occurs excessively, the released oxidative factors will destroy cells and lead to tissue damage. 21 The studies have shown that dihydromyricetin can significantly increase the activity of glutathione reductase (GSH), while reducing the level of lipid oxidase (MDA) to play an antioxidant role. 22 In a mouse myocardial ischemia / reperfusion injury model, dihydromyricetin decreased the levels of ROS and SOD in the myocardial tissues to inhibit excessive oxidative stress. 22 We found that dihydromyricetin increased the contents of SOD and POD in the myocardium tissues injured by CCl4. Our report is inconsistent with the result presented by Prof. Xu, considering that mitochondrial function was lost in the myocardial ischemia / reperfusion injury model, and mitochondrial function is not fully recovered when the mice was treated with DMY.
IL-33 is a member of the IL-1 family. As a signaling “alarm,” it can promote immune cells to defend against cell/tissue damage through ST2 (IL-33R). 9 The studies have shown that a variety of inflammatory diseases are associated with the IL-33/ST2 signaling pathway. 23 It is reported that hepatic progenitor cells (HPC) can promote hepatocyte regeneration and inhibit the deposition of fibrotic collagen so as to reduce granuloma caused by Schistosoma japonicum infection. 24 During this process, the proliferation of HPCs was negatively correlated with IL-33 expression. 24 We found that CCl4 induced the expression of IL-33 and ST2 in mouse myocardial tissue. The results suggest that IL-33/ST2 signaling may be involved in CCl4-induced cardiac dysfunction. After the intervention of dihydromyricetin, the expression of IL-33 and ST2 in the myocardial tissues of CCl4-induced mice were decreased. The results suggest that dihydromyricetin may protect the cardiac function of the mice received CCl4 by inhibiting IL-33/ST2 signal. Whether the IL-33/ST2 signaling pathway is the way that dihydromyricetin protects the cardiac function of the mice injected by CCl4 needs further study. The natural compound resveratrol also has similar effects. 25 The combination of the DMY and resveratrol may be a potential therapeutic strategy for treating cardiovascular diseases.
The weight of mice in DMY group was generally lower than that in control group and CCl4 group, especially on the 14th, 21st, and 56th days, and the difference was statistically significant. This was consistent with the result reported by Xiong et al. that dihydromyricetin could decrease the weight of mice. 26 The reason may be that dihydromyricetin increased the number of mitochondria by promoting mitochondrial division, thereby enhancing the fat metabolism rate of mitochondria, and ultimately reducing the weight of mice.
Dihydromyricetin can reduce myocardial hypertrophy in the mice treated with CCl4 and contribute to the recovery of cardiac function in mice. It has been reported that dihydromyricetin has inhibitory effect on Ang-II-induced cardiomyocyte hypertrophy. 26 Dihydromyricetin inhibits excessive oxidative stress and alleviates cardiac hypertrophy induced by Ang-II by enhancing the production of cyclic guanosine monophosphate (cGMP) and phosphorylation of endothelial nitric oxide synthase (eNOS). 27 Therefore, we used echocardiography to detect the function of left ventricle in mice. Compared with CCl4 group, dihydromyricetin could reduce the levels of LVIDs and LVIDd and improve the levels of LVEF and LVFS12 in the heart from the mice damaged by CCl4. Therefore, dihydromyricetin may protect the cardiac function of mice.
Conclusion
To sum up, dihydromyricetin can improve the cardiac function of the mice given by CCl4, and there is the participation of IL-33/ST2 signal and the activation of NLRP3 inflammasome in this process. Our research provides new ideas for drugs to improve cardiac dysfunction caused by CCl4 poisoning. At the same time, our study provides a potential molecular target for dihydromyricetin to improve cardiac dysfunction in mice. However, whether NLRP3 inflammasome activation and IL-33/ST2 signaling are the pathways for dihydromyricetin to improve cardiac function in mice needs further investigation. In addition, the CCl4 induced myocardial injury is not a common model of the myocardial function, and its results may have limitations due to CCl4 toxicity.
Footnotes
Acknowledgements
The authors would like to thank Jing-Ru Qin, Qi Gao, and Ji-Sheng Hu for revision.
Abbreviations
DMY, Dihydromyricetin; NLRP3, NOD-like receptor protein 3; ASC, apoptosis-associated speck-like protein containing a CARD; pro-caspase-1, pro-cysteinyl aspartate specific proteinase-1; PRRs, pattern recognition receptors; PAMPs, pathogen related molecular patterns; DAMPs, damage related molecular patterns; ST2, suppression of tumorigenicity 2; IL-1RAcP, IL-1 receptor accessory protein; NF-κB, nuclear factor-κB; MAPK, mitogen-activated protein kinase; MyD88, myeloid differentiation primary response gene 88; SPF, specefic pathogen free; LVIDs, left ventricular end-systolic diameter; LVIDd, diastolic left ventricular diameter; LVEF, left ventricular ejection fraction; LVFS, left ventricular fractional shortening; ECL, enhanced chemiluminescence; GSH, glutathione reductase; MDA, lipid oxidase; HPC, hepatic progenitor cells; cGMP, cyclic guanosine monophosphate; eNOS, endothelial nitric oxide synthase.
Authors’ contributions
WJZ, SCZ, and BHH conceived, performed and designed the topics. WJZ wrote the first draft of the manuscript. WJZ and KYL conducted the experiments. LYL and YMS corrected and validated the manuscript. ZQX and JRZ analyzed the results and drew the figures. All contributing authors have participated in the revision of manuscript and concur with the submission.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Natural Science Foundation of China, Fund No. 82360404; Natural Science Foundation of Jiangxi Province, Fund No. 20232BAB206127; Open Project of the Key Laboratory of Cardiovascular and Cerebrovascular Disease Prevention and Control of the Ministry of Education, Fund No. XN202016; Jiangxi Provincial Department of Education Research and Technology Research Project, Fund No. GJJ211525 and No. GJJ2201410; The Doctoral Startup Fund of Gannan Medical University No. QD202111; Jiangxi Province College Students Innovation and Entrepreneurship Training Project, Fund No. S202410413027; Undergraduate of Gannan Medical College Biotechnology Innovation Project, Fund No. X202410413003.
Ethics approval
Ethical approval for this study was obtained from Scientific Research Ethics Committee, Gannan Medical University (APPROVAL NUMBER/ID: 2023494).
Animal welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
Consent for publication
Not applicable.
