Abstract
Background
The coronavirus disease 2019 (COVID-19) is a respiratory virus, the spread of which has caused a global pandemic with catastrophic consequences. The current study aimed to investigate the association between vitamin D deficiency and the clinical presentation of COVID-19.
Patients and methods
The current study included 166 COVID-19 patients recruited from Prince Mohammad Bin Abdulaziz Hospital in Riyadh, Saudi Arabia. The study was conducted from October 2020 to January 2021. Patients were diagnosed by positive polymerase chain reaction (PCR) results. History and clinical data were collected for all subjects. In addition, laboratory analysis was done to estimate blood levels of 25 hydroxyvitamin D (25(OH)D), C-reactive protein (CRP), ferritin, parathyroid hormone (PTH), alanine aminotransferase (ALT), D-dimer, calcium, and relative lymphocytic count. COVID-19 patients were divided into three subgroups according to their vitamin D status. Patients were considered sufficient when their vitamin D level was above 30 ng/mL. Patients with vitamin D levels below 20 ng/mL were considered deficient. Patients with vitamin D levels ranging from 20 ng/mL to 30 ng/mL were considered insufficient.
Results
Our results showed that 81 patients (49%) were deficient in vitamin D, and 48 patients (29%) were insufficient in vitamin D. Only 37 patients (22%) had normal vitamin D levels. Moreover, a significant difference was found regarding the inflammatory markers of COVID-19 severity. Also, vitamin D levels were inversely correlated with the markers used for monitoring the condition of COVID-19 patients: ferritin, CRP, and D-dimer.
Conclusion
Our results showed that vitamin D deficiency was associated with increased levels of inflammatory markers of COVID-19 infection.
Introduction
The coronavirus infection has spread all over the world. The outbreak started in Wuhan, Hubei, China, late in 2019 and was officially named COVID-19 by the World Health Organization (WHO) on 11 February 2020.1,2 This infection is associated with severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) and threatens the world. 3 There is variation in the clinical features of COVID-19, as 17.9% of COVID-19 infections are mild, while 15.7% of the patients developed severe illness after being admitted to the hospital. On initial presentation, no radiologic anomalies were found in 2.9% of patients with serious disease. 4
COVID-19 shows its effect as initial immune suppression, followed by exaggerated immune system response, resulting in a cytokine storm. COVID-19 can have severe consequences, such as the development of acute respiratory distress syndrome (ARDS) and systemic inflammatory response syndrome (SIRS).5,6
Vitamin D deficiency represents an important health problem; more than one billion people are estimated to have vitamin D deficiency worldwide.7,8 In Saudi Arabia, the prevalence of vitamin D deficiency is around 60%. Its occurrence has been reported in different ages and both genders.9,10
Vitamin D exerts significant antiviral and anti-inflammatory effects via its immunoregulatory actions as vitamin D receptors have been recognized in many immunological cells, and certain cells of the immune system can synthesize the active form of vitamin D. 11
Vitamin D can reduce the risk of infection through several effects: first, it plays an antiviral role by enhancing antimicrobial peptides cathelicidin and B-defensin that diminish the viral replication; second, enhancement of anti-inflammatory and diminishment of pro-inflammatory cytokines (IL-6, TNF-α, and IFN-γ) that cause pneumonia and lung insult. 2 The pro-inflammatory cytokines are recognized as predictors for bad outcomes in COVID-19 infection. 12
Previous research has reported that vitamin D deficiency may enhance the possibility of respiratory infections, including respiratory syncytial virus, tuberculosis, and flu. In addition, it is considered a risk factor for ARDS. 13
The COVID-19 virus principally involves the pulmonary type-II alveolar pneumocytes by binding to the increased angiotensin‐converting enzyme 2 receptors (ACE2) of the infected person, 14 decreasing surfactant production, and increasing surface tension, which results from alveolar type-II pneumocytes dysfunction. 15 Vitamin D metabolites can stimulate surfactant synthesis by modulating the renin-angiotensin system in alveolar type‐II cells protecting against acute lung injury. 16 Thus, vitamin D deficiency can be a pathogenic element in COVID-19.
Before the emergence of COVID-19 vaccines, vitamin D supplementation and exposure to sunlight were included in treatment protocols. In the current study, we aimed to define the prevalence and clinical significance of vitamin D deficiency in hospitalized patients diagnosed with COVID-19.
Subjects and methods
Study design and participants
We planned a retrospective case control study including 166 patients with COVID-19, aged 23–88 years, who were admitted to the Prince Mohammad Bin Abdulaziz Hospital in Riyadh, Saudi Arabia, between October 2020 and January 2021. They were diagnosed by RT-PCR and further assessment was done by computed tomography (CT) scans of the chest.
Patients were further classified into three subgroups according to their vitamin D status: a vitamin D sufficient group (vitamin D > 30 ng/mL), a vitamin D–deficient group (vitamin D < 20 ng/mL) and a vitamin D–insufficient group (vitamin D from 20–30 ng/mL). 17 Specific criteria were used to determine the number of cases to be recruited in the study according Miaoulis and Michener. 18 For calculation of the minimum sample size, the formula of Cochran 19 was used, with confidence value 95% at a significant concentration of 5%.
We have excluded patients with malabsorption diseases, liver cirrhosis, and serum creatinine levels of > 2 mg/dL. Patients who obtained oral vitamin D supplements or previous anticonvulsant treatment were also excluded from the study.
Data collection
Demographic and clinical data were collected from hospital records of COVID-19 patients, stored in an electronic database, and independently examined by two researchers. Procedures were performed after prior approval by the research ethics committee of Taif University (42-0010).
The outcome variable for COVID-19 severity was defined as the combination of intensive care unit (ICU) admission, mechanical ventilation prerequisite, or in-hospital mortality. Generally, the ICU admittance criteria were set by following the rules by the American Thoracic Society and the Infectious Diseases Society of America. 20
Laboratory measurements
Fasting venous blood samples were collected from COVID-19 patients and allocated into two portions. The sera were separated from the plain tubes for the estimation of vitamin D, calcium, and the inflammatory markers. The whole blood from ethylenediaminetetraacetic acid (EDTA)–containing tubes were used for complete blood count (CBC) tests. Serum 25(OH)D levels were determined by using an Abcam human vitamin D enzyme-linked immunosorbent assay (ELISA) kit, USA (Cat No. ab213966), following the manufacturer’s protocol. The range of detection was 0.5–1010 ng/mL, and the sensitivity of the assay was 1.98 ng/mL. Serum vitamin D levels of less than 20 ng/mL were considered deficient. The PTH serum levels were estimated by the Abcam Human PTH ELISA kit, USA (Cat No. ab230931), based on the company guidelines, with a detection range of 4.69–300 pg/mL and a sensitivity of 0.761 pg/mL.
CRP was evaluated by using the immunoturbidimetric method (CRP II Latex X2, Denka Seiken Co. Ltd., Tokyo, Japan), utilizing an autoanalyzer (Toshiba, Tokyo, Japan). The measurement range of this assay was 0.01–32 mg/dL. Serum ferritin levels were evaluated using an ELISA kit (RCD012R, BioVendor) with an intra-assay CV of 7.3% and an inter-assay CV of 4.5%. Serum D-dimer was measured by a human Abcam ELISA kit (Cat No. ab260076), with a sensitivity of 2.36 ng/mL, intra-assay of 4.4%, and inter-assay of 4.3%.
Serum calcium was estimated by a calcium colorimetric assay kit, Abcam, USA (Cat No. ab102505), with a detection range 0.4–100 mg/dL, according to the manufacturer’s protocol.
Statistical analysis
Statistical Package for Social Sciences (SPSS) for Windows version 20.0 (IBM SPSS Statistics, IBM Corporation, Armonk, NY, USA) was used for data analysis. Data were presented as mean ± standard deviation (SD) and one-way analysis of variance (ANOVA), followed by Tukey’s honestly significant difference (HSD). Post-hoc analyses were used for multiple comparisons between groups. The chi-square (x2) test of significance was applied to compare proportions, and the Pearson correlation coefficient was used to assess the association between vitamin D and the studied parameters. p values were considered statistically significant at < 0.05.
Results
Demographic and laboratory characteristics of all patients included in the study.
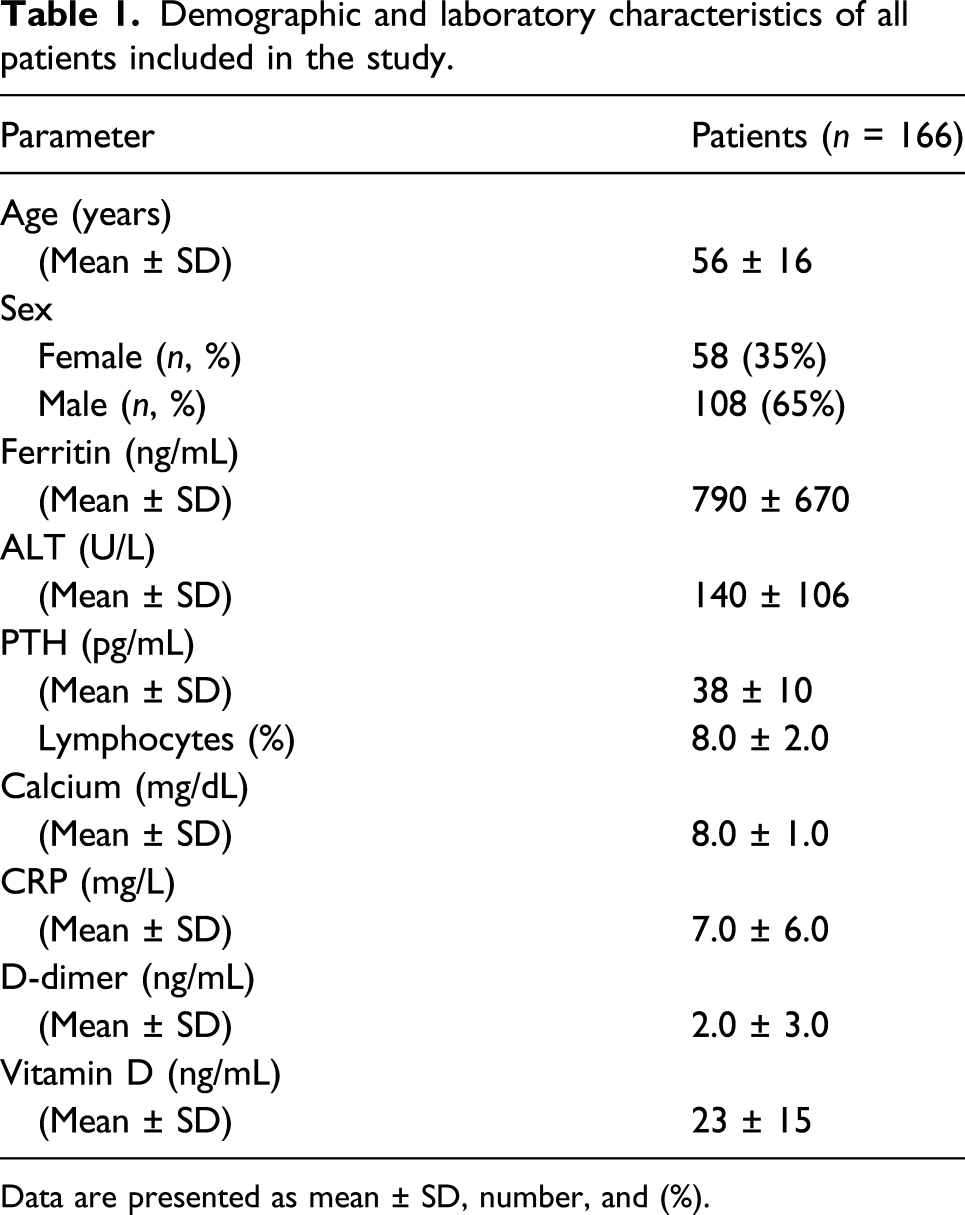
Data are presented as mean ± SD, number, and (%).
Comparison between subgroups of COVID-19 patients according to demographic data.

Data are presented as mean using F-one-way analysis of variance.
Data are presented as number and (%) using the chi-square test.
**p-value < 0.001 HS; *p-value < 0.05 S.
Comparison between COVID-19 patients subgroups according to laboratory data.

Using: F-one-way analysis of variance.
Post-hoc test, LSD: a: statistically significant difference with the deficiency group; b: statistically significant difference with insufficiency group.
p-value > 0.05 NS; *p-value < 0.05 S; **p-value < 0.001 HS.
Clinical characteristics of COVID-19 patients in the three subgroups.

Data are presented as number using the chi-square test.
Abbreviation: CPAP: continuous positive airway pressure.
*p-value < 0.05 S; p-value > 0.05 NS.
Correlation between vitamin D with all parameters in COVID-19 patients group.

Using r-Pearson correlation coefficient.
p-value > 0.05 NS; *p-value < 0.05 S; **p-value < 0.001 HS.

Scatter plot between Vitamin D with ferritin in patients group. r = −0.44; p = 0.01.

Scatter plot between Vitamin D with PTH in patients group. r = −0.60; p < 0.001.

Scatter plot between Vitamin D with D-dimer in patients group. r = −0.36, p = 0.03.
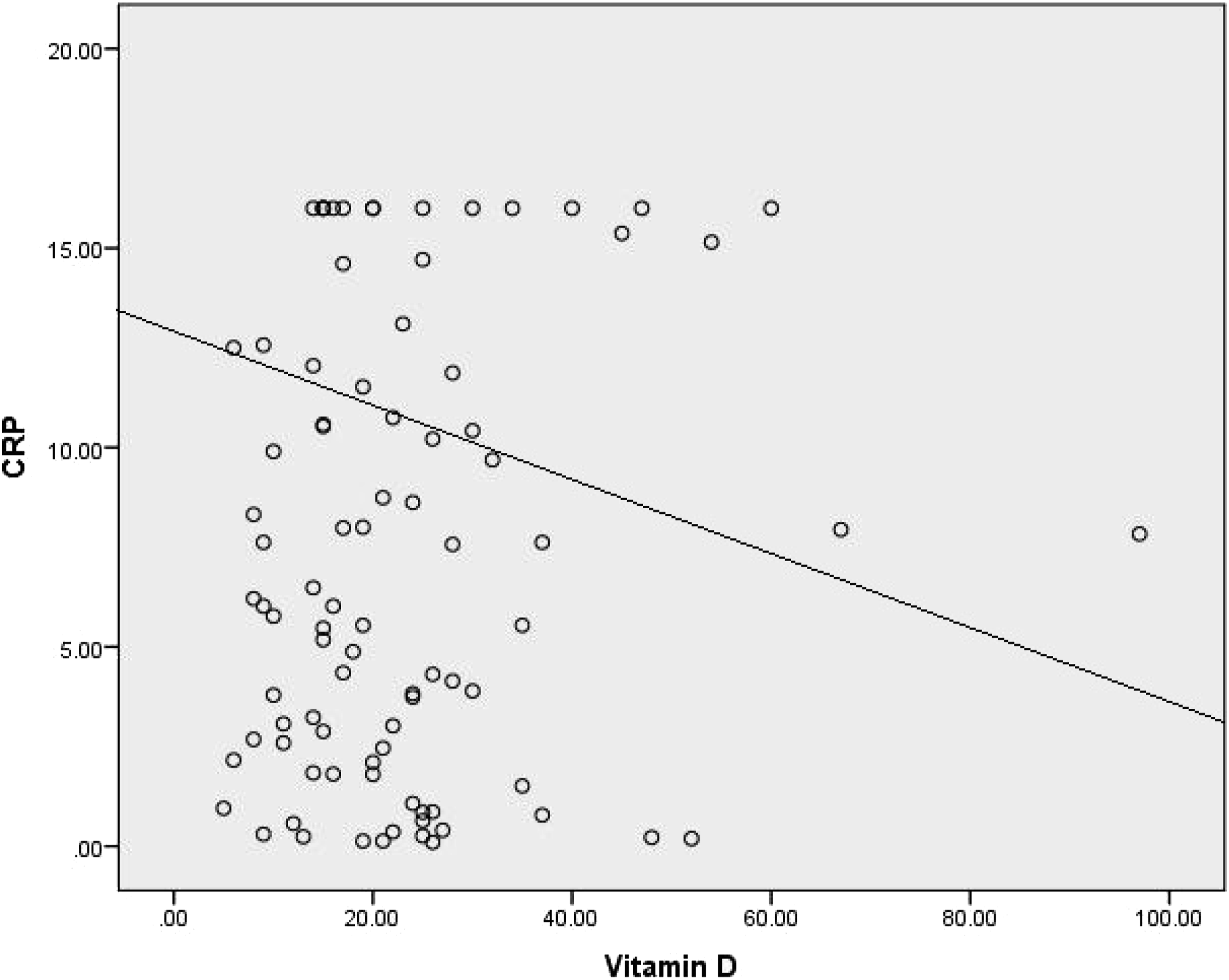
Scatter plot between Vitamin D with CRP in patients group. r = −0.6, p < 0.001.

Scatter plot between Vitamin D with calcium in patients group. r = 0.5, p = 0.007.
Discussion
The current study was conducted to evaluate the vitamin D status among COVID-19 patients and to study the association of vitamin D deficiency with clinical data and inflammatory biomarkers in COVID-19 patients.
Although little is known about the effects of vitamin D deficiency on the clinical presentation and the outcome of COVID-19 infection, 21 several studies have demonstrated the relationship between other respiratory infections and vitamin D. Vitamin D has been reported to protect against respiratory infections.22–24
In our current research, we found that 49% of COVID-19 patients enrolled in the study were deficient in vitamin D, while 29% were insufficient in vitamin D, resulting in a total of 78% with deficiency or insufficiency in vitamin D. Also, we found that the vitamin D deficiency group included elderly patients.
In accordance with our findings, Zhou et al. 25 revealed that patients with a serum vitamin D level of less than 20 ng/mL had a 64% higher chance of getting community-acquired pneumonia. Ricci et al. 26 reported that 80% of their COVID-19 patients had vitamin D deficiency and 6.5% had vitamin D insufficiency. Paiz et al. 27 reported that vitamin D has recently become more prominent in studies due to its possible effect on the incidence of COVID-19 infection. Moreover, Ilie et al. 22 found that the oldest portion of the population, which is the most susceptible to COVID-19 infection, is also the group with the lowest vitamin D levels. On the other hand, Ali, 28 revealed that there is insufficient evidence to link vitamin D levels with COVID-19 severity and mortality.
The current study evaluated the effect of vitamin D deficiency on the inflammatory markers that are used to assess the clinical conditions of patients, such as serum ferritin, CRP, ALT, and D-dimer. We observed higher concentrations of inflammatory markers in the vitamin D deficiency group when compared to the other two groups.
Our findings agree with a previous study by Yilmaz and Şen, 29 which found that vitamin D may reduce the incidence of inflammatory markers, which are effective predictors of worse outcomes in children with COVID‐19 infection. The pathology of COVID‐19 includes a complex interaction between the virus and the host immune system. COVID‐19 triggers the release of pro-inflammatory cytokines. Vitamin D has been reported as a modulator of the immune response of macrophages, preventing them from releasing several inflammatory cytokines and chemokines.12,30
In our current research, we studied the correlation between vitamin D and inflammatory markers used in clinical practice to assess the severity of COVID-19. Significant negative correlations were reported between lower vitamin D levels and higher inflammatory markers CRP, ferritin, and D-dimer. In accordance with our findings, Daneshkhah et al. 31 observed that high CRP was inversely correlated with vitamin D levels. Our results suggest a possible role for vitamin D in the reduction of complications caused by the cytokine storm, considering C-reactive protein as an important marker for the severity of COVID-19 inflammation and the cytokine storm. Moreover, Ricci et al. 26 found a significant relation between high levels of D-dimer and low levels of vitamin D.
The relationship between vitamin D and inflammatory diseases has triggered a lot of debate because the mechanism of the link between low vitamin D concentrations and increased inflammatory cytokines levels has not been completely explored. A recent meta-analysis conducted by Kazemi et al. 32 discovered that in COVID-19, the association between vitamin D deficiency and lung inflammation was contradictory and the relationship between vitamin D deficiency and mortality was ambiguous. Vitamin D deficiency is prevalent in hospitalized patients with severe illnesses, especially in those who are critically ill.33–35
There are several conflicting results at this point. Yousefzadeh et al. 36 reported that vitamin D levels measured during an acute disease may be an inaccurate biomarker of vitamin D status, and the changes in vitamin D–binding protein (DBP) levels should be noted as confounding factors when interpreting 25(OH)D concentrations in blood. Another explanation by Quraishi and Camargo 37 is that lower 25(OH)D levels can be caused by lower levels of DBP due to interstitial leakage caused by increased vascular permeability during inflammatory diseases. Amrein et al. 38 revealed that vitamin D levels assessed at the beginning of an acute inflammatory injury can represent the acute phase of the illness rather than true vitamin D levels. Moreover, Waldron et al. 39 suggested that serum 25(OH)D is a negative acute phase reactant, and after an acute inflammatory injury, serum 25(OH)D is an ineffective biomarker of vitamin D status.
On the other hand, vitamin D regulates immune reactions induced by macrophages and dendritic cells, which are the first line of host defense, preventing them from the production of excessive inflammatory cytokine and chemokine. 40 The value of vitamin D sufficiency in serious disease is strongly supported by the study of Braun et al., 41 in which 25(OH)D was measured at the start of critical treatment. Mata-Granados et al. 42 observed that large doses of oral 25(OH)D can correct vitamin D deficiency and could lead to improvement in general health and most likely, a decrease in the overall mortality rate of critically ill patients.
Regarding the clinical presentations of the COVID-19 patients we studied, the clinical features of COVID-19 infection were more prominent among the vitamin D–deficient group in comparison to other groups. Patients who were deficient in vitamin D had a higher frequency of fever and fatigue in comparison to the other groups, and the difference was statistically significant. In consistency with our results, Mardani et al. 43 observed that adequate vitamin D may have a controlling effect on the symptoms of COVID-19 infection by interfering with the rennin-angiotensin-aldosterone system (RAAS) and immune system functions through the vitamin D receptor (VDR), which is a ligand-activated transcription factor. Pereira et al. 44 observed a positive association between vitamin D deficiency and the severity of the disease. Moreover, Ilie et al. 22 found a significant relation between vitamin D levels and the number of COVID-19 cases in different European countries, with increased mortality.
Considering the causes of clinical diversity in the course and mortality rates of COVID-19 cases, it is necessary to point out that vitamin D deficiency may also underlie the comorbidity of patients. 45
This study has limitations. The retrospective design of the study may possibly involve incomplete or incorrect data, and the number of patients in the study may be small, but it may provide guidance which helps researchers perform larger studies to explore the effects of vitamin D deficiency on COVID‐19 infected patients. The study was underpowered to examine severity due to the small numbers of mortality and ICU-admitted patients. In addition, vitamin D levels before the start of COVID-19 infection were not reported for included patients. So, vitamin D levels should be measured before and at the onset of acute diseases in future research, with adjustment for all possible affecting factors such as age, sex, BMI, diet, medication, vitamin D supplementation, and socioeconomic factors.
Conclusion
We evaluated vitamin D status and its relationship with clinical features in hospitalized patients with COVID‐19. Our results suggested that vitamin D deficiency was associated with increased levels of inflammatory markers of COVID‐19 infection.
Footnotes
Acknowledgments
The authors would like to thank Taif University, Taif, Saudi Arabia, for their support (Taif University Researchers Supporting Project number: TURSP-2020/80), Taif University, Taif, Saudi Arabia.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was funded by Taif University, Taif, Saudi Arabia. (Taif University Researchers Supporting Project number: TURSP-2020/80).
