Abstract
Saturated fatty acids (SFAs) can directly stimulate innate immune responses, thereby exacerbating inflammatory aspects of metabolic syndrome. Dietary SFAs act as ligands of Toll-like receptor 4 (TLR4), triggering associated signaling pathways. In this study, we investigated the role of TLR4 in palm oil SFA-associated inflammatory cytokine gene expression in monocytes/macrophages and adipose tissue using TLR4-overexpressing genetically modified sheep. SFA stimulation resulted in upregulation of interleukin-6 (IL-6), tumor necrosis factor-α (TNF-α), interleukin-8 (IL-8), interferon-γ (IFN-γ), and interleukin-10 (IL-10), and TLR4 overexpression enhanced such SFA-induced inflammatory cytokine expression. Moreover, SFAs markedly activated MyD88-dependent signaling, including IL-1 receptor–associated kinase 4 (IRAK4), TNF receptor–associated factor 6 (TRAF6), and nuclear factor-κB (NF-κB). Taken together, our results indicate that TLR4 overexpression enhances the SFA-induced inflammatory response through MyD88-dependent signaling in monocytes/macrophages and adipose tissue.
Introduction
Dietary saturated fatty acids (SFAs) have been implicated in metabolic syndrome and atherosclerotic cardiovascular disease, and there appears to be a connection between metabolic syndrome and the innate immune system.1,2 SFAs can activate innate immune responses, exacerbating inflammatory aspects of metabolic syndrome.3,4 This inflammatory activity is characterized by increased production of pro-inflammatory cytokines, further contributing to the pathogenesis of this condition.
Toll-like receptor 4 (TLR4) is critical for the recognition of gram-negative bacterial lipopolysaccharide (LPS)/endotoxin by various host cells. Emerging evidence suggests that TLR4 can also be activated by dietary components and metabolic intermediates, which induce sterile inflammation. 3 As with microbe-induced inflammation, sterile inflammation is marked by neutrophil and macrophage recruitment and the production of pro-inflammatory cytokines and chemokines, notably, tumor necrosis factor (TNF) and interleukin-1 (IL-1). 5 TLR4 may be involved in the crosstalk between inflammatory and metabolic signals. Some studies have shown that SFA may be natural ligands for TLR4 and can activate TLR4-mediated pro-inflammatory signaling pathways, 6 consequently increasing the risk of metabolic syndrome. 1
In our previous study, the effects of TLR4 overexpression on the LPS-stimulated inflammatory response were examined in vivo and in vitro. 7 The main purpose of this study was to investigate the role of TLR4 in palm oil SFA-associated inflammatory cytokine gene expression in monocytes/macrophages and adipose tissue.
Materials and methods
Ethics statement
Sheep husbandry was performed using standard practices. All experimental protocols involving animals were approved by and performed in accordance with the requirements of the Animal Care and Use Committee of Northeast Agricultural University (approval ID: 2013-028).
Animals
The TLR4-overexpressing genetically modified (GM) sheep were produced by microinjection during our previous study. The expression vector is shown in Figure 1(a).These sheep exhibit normal growth performance and reproductive traits, 8 and their meat has no adverse effects on Sprague–Dawley rats in comparison with that from wild-type (WT) sheep. 9 In this study, male TLR4-overexpressing GM sheep (n = 10) and WT sheep (n = 10) of the same age were selected and housed in Daqing Yinlang Sheep Farm in Heilongjiang Province, China. The animal diet was 2000 g per animal per day. All animals had unlimited access to water.
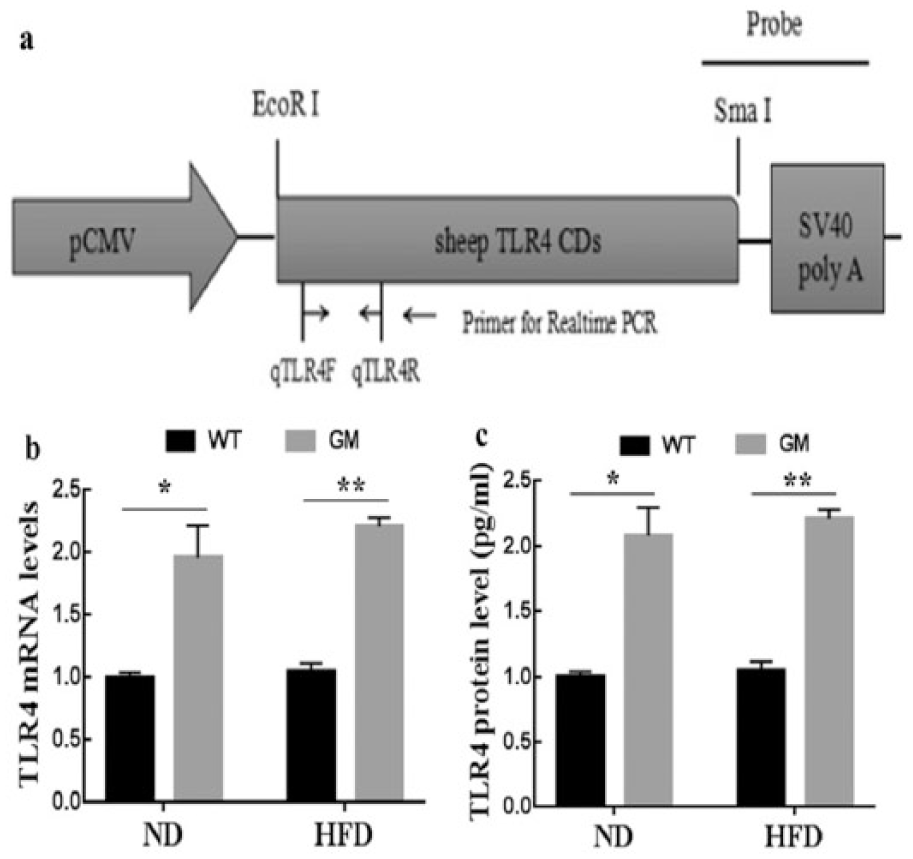
Production and identification of genetically modified (GM) sheep and the expression of TLR4 in monocytes/macrophages. (a) The expression vector, (b) TLR4 mRNA level in monocytes/macrophages, and (c) TLR4 protein level in monocytes/macrophages. Results are presented as means ± SEM. *P < 0.05 and **P < 0.01 compared with WT sheep. ND: normal diet; HFD: high-fat (saturated fatty acid) diet.
Experimental design and sample collection
Sheep of each genotype (GM and WT) were randomly assigned to one of two experimental diet groups (n = 10/genotype/diet): (1) regular chow (normal diet, ND) or (2) regular chow with 5% palm oil (high-fat diet, HFD). The composition of each diet is shown in Table 1. The palm oil was obtained from Fortbon, Harbin, China. All sheep were fed for 8 weeks under the same conditions. Peripheral blood was collected at weeks 3, 6, and 8, and monocytes/macrophages were isolated using Lymphocyte Separation Medium (TBD, Tianjin, China), as described previously.16 The cell population acquired was monocytes/macrophages, which has been proven in our previous study. 8 Perirenal adipose tissue was collected at week 8, immediately frozen in liquid nitrogen, and stored at −80°C prior to RNA extraction and analysis.
Diet composition.

SFA: saturated fatty acid.
Quantitative real-time polymerase chain reaction
Total RNA was extracted using TRIzol (TaKaRa, Dalian, China). Complementary DNA (cDNA) was then synthesized from total RNA with a PrimeScript™ RT Reagent Kit with gDNA Eraser (#RR047A; TaKaRa) according to the manufacturer’s protocol. The relative expression of all target genes in monocytes/macrophages and adipose tissue was quantified using an ABI 7500 Fast Real-Time PCR System (Applied Biosystems, Foster City, CA, USA) and FastStart Universal SYBR Green Master (ROX) (Roche Diagnostics GmbH, Mannheim, Germany). Relative expression was determined using the 2−ΔΔCT method, with β-actin expression serving as an internal control.
Enzyme-linked immunosorbent assay for inflammatory factors
Protein concentration of TLR4 in monocytes/macrophages was examined using Elisa (Shanghai, Xinle, China). Protein concentrations of IL-6, TNF-α, IL-8, IFN-γ, and IL-10 in the adipose tissue were measured using the enzyme-linked immunosorbent assay (ELISA) kit (Cusabio Biotech. Co., Ltd, Wuhan, China) following the manufacturer’s instructions. The concentrations were calculated according to standard curves with CurveExpert software.
Statistical analysis
Statistical analysis was performed using the general linear model in SPSS 18.0 (SPSS Inc., Chicago, IL, USA). The Shapiro–Wilk test showed that all data were distributed normally. Significant differences among means were identified with Duncan’s multiple range test. Differences were considered statistically significant when associated with P < 0.05. Results are presented as means ± SEM.
Results
The expression vector and baseline TLR4 expression
The linear vector containing sheep TLR4 gene was microinjected into the pronuclei (Figure 1(a)). The GM sheep were found to be positive by southern blot, carrying exogenous TLR4. 8 Then, TLR4 expressions in peripheral blood monocytes/macrophages of GM and WT sheep were analyzed by real-time PCR and ELISA at the beginning of the experiment. It showed that expression of TLR4 was significantly higher in GM sheep than in WT sheep (P < 0.05, Figure 1(b) and (c)).TLR4 and inflammatory cytokine gene expression in monocytes/macrophages during SFA stimulation
The expression of TLR4 and inflammatory cytokine genes in monocytes/macrophages was analyzed at weeks 3, 6, and 8 of SFA stimulation (Figure 2). The GM sheep exhibited significantly higher TLR4 expression than the WT sheep at all three time points (P < 0.05). Furthermore, administration of SFAs had no significant influence on TLR4 expression throughout the experiment (P > 0.05, Figure 2(a)). Expression of IL-6 was increased after 6 weeks of SFA stimulation (P < 0.01). Moreover, at weeks 6 and 8, IL-6 expression in GM sheep fed a high-SFA diet was significantly higher than that in WT sheep given the same diet (P < 0.05, Figure 2(b)). The TNF-α gene was expressed at a similar level in all groups at the early stage of the experiment. However, 8 weeks of SFA stimulation resulted in heightened TNF-α expression (P < 0.01), and of the high-SFA diet groups, GM sheep demonstrated significantly higher TNF-α messenger RNA (mRNA) levels than WT sheep (P < 0.01, Figure 2(c)). Expression of IL-8 mRNA was significantly upregulated after SFA stimulation, and this effect was still evident at week 6 (P < 0.05). In addition, IL-8 expression in GM sheep fed the diet high in SFAs was significantly higher than that in WT sheep of the same diet group at all time points (P < 0.05, Figure 2(d)). The interferon-γ (IFN-γ) gene was found to be expressed at high levels on week 6 and at the end of the experimental period (week 8) under SFA stimulation (P < 0.05, Figure 2(e)). About 6 weeks of SFA administration led to significantly increased level of IL-10 mRNA (P < 0.01), and expression of this gene was higher at all three time points in GM sheep fed high-SFA diet than WT sheep given the same feed (P < 0.01, Figure 2(f)).

Pattern of TLR4 and inflammatory cytokine gene expression in monocytes/macrophages after 3, 6, and 8 weeks of saturated fatty acid (SFA) stimulation. (a) Toll-like receptor 4 (TLR4), (b) interleukin-6 (IL-6), (c) tumor necrosis factor-α (TNF-α), (d) interleukin-8 (IL-8), (e) interferon-γ (IFN-γ), (f) interleukin-10 (IL-10). Results are presented as means ± SEM. #P < 0.05, ##P < 0.01, and ###P < 0.001 compared with the normal diet (ND) group. *P < 0.05, **P < 0.01, and ***P < 0.001 compared with wild-type (WT) sheep. HFD: high-fat (SFA) diet, GM: genetically modified.
Inflammatory cytokine expression in adipose tissue during SFA stimulation
In adipose tissue, the IL-6 and TNF-α genes were upregulated on week 8 in sheep fed a diet high in SFAs compared to their counterparts given the ND (P < 0.01, Figure 3(a) and (b)). However, IL-8, IFN-γ, and IL-10 expression was not affected by the high-SFA diet (P > 0.05, Figure 3(c)–(e)). IL-6 mRNA levels in GM sheep fed the diet high in SFAs were significantly higher than those in WT sheep given the same diet (P < 0.01, Figure 3(a)).
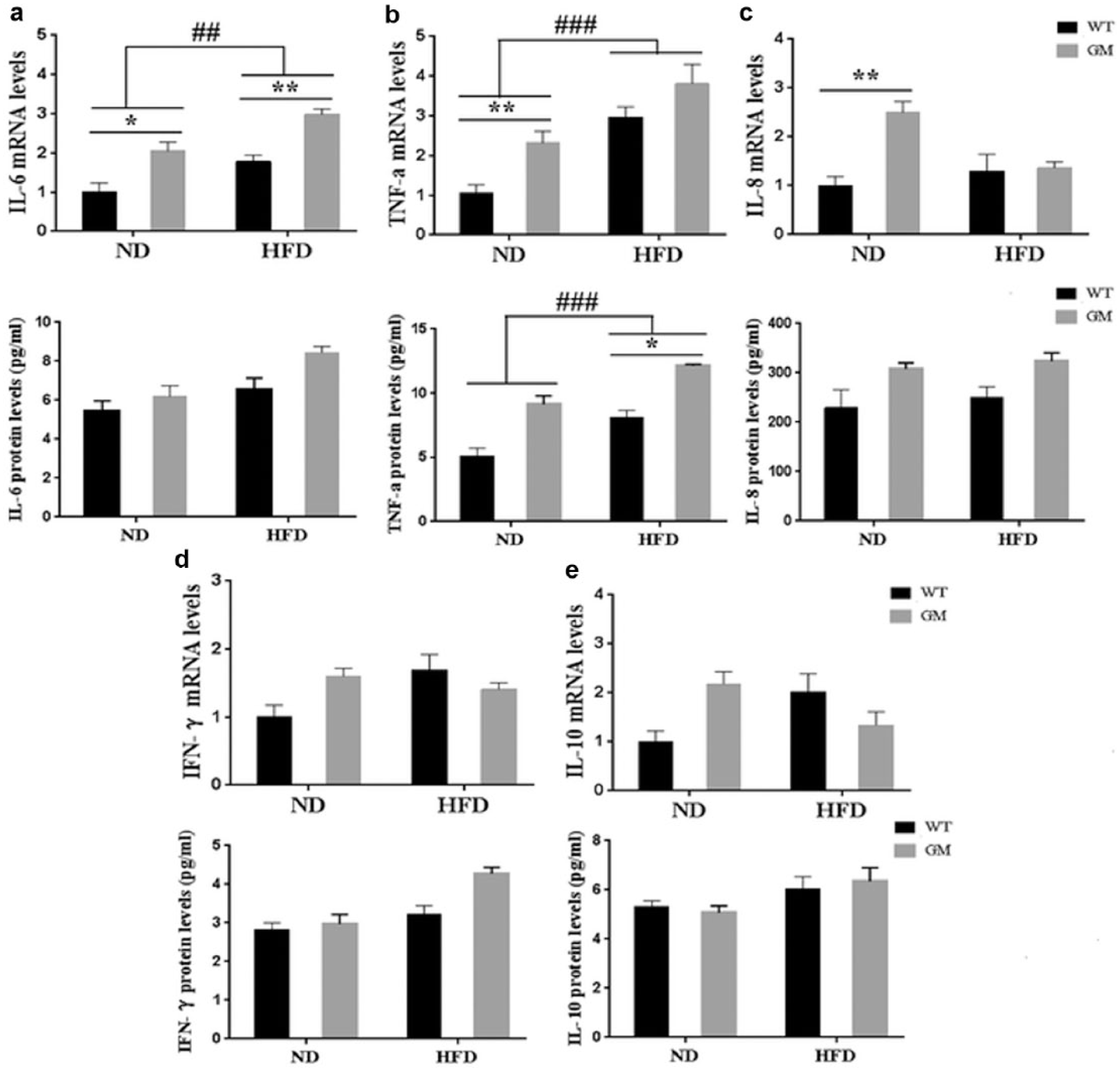
Expression of inflammatory cytokine gene mRNA levels and protein levels of inflammatory cytokine in perirenal adipose tissue under saturated fatty acid (SFA) treatment at 8 weeks. (a) interleukin-6 (IL-6), (b) tumor necrosis factor-α (TNF-α), (c) interleukin-8 (IL-8), (d) interferon-γ (IFN-γ), (e) interleukin-10 (IL-10). Results are presented as means ± SEM. #P < 0.05, ##P < 0.01, and ###P < 0.001 compared with the normal diet (ND) group. *P < 0.05, **P < 0.01, and ***P < 0.001 compared with wild-type (WT) sheep. HFD: high-fat (SFA) diet; GM: genetically modified.
Similarly, TNF-α protein concentration was significantly upregulated after SFA stimulation (P < 0.01, Figure 3(b)). IL-6 protein expression level was also upregulated, but there was no significant difference (P > 0.05, Figure 3(a)). At the same time, IL-8, IFN-γ, and IL-10 protein concentrations were not affected by the high-SFA diet (P > 0.05, Figure 3(c)–(e)). After fed with 8 weeks of high-SFA diet, GM sheep demonstrated significantly higher TNF-α protein concentration than WT sheep (P < 0.05, Figure 3(b)).
Expression of TLR4 signaling pathway components in monocytes/macrophages and adipose tissue
TLR4 signaling activity was tested by measuring the expression of several key components of the myeloid differentiation factor 88 (MyD88)-dependent (including IL-1 receptor–associated kinase 4 (IRAK4), TNF receptor-associated factor 6 (TRAF6), and nuclear factor-κB (NF-κB)) and MyD88-independent (including TRAF3, TRAF family member–associated NF-κB activator (TANK)-binding kinase-1 (TBK1), and interferon regulatory factor 3 (IRF3)) TLR-signaling cascades. At week 3, the IRAK4 and NF-κB genes were upregulated in SFA-stimulated monocytes/macrophages (P < 0.05, Figure 4(A) and (C)). In adipose tissue, expression of the TRAF6 gene was elevated due to SFA stimulation (P < 0.05, Figure 4(b)). However, the high-SFA diet did not influence TRAF3, TBK1, or IRF3 expression in monocytes/macrophages or adipose tissue (P > 0.05, Figure 4(d)–(f)).
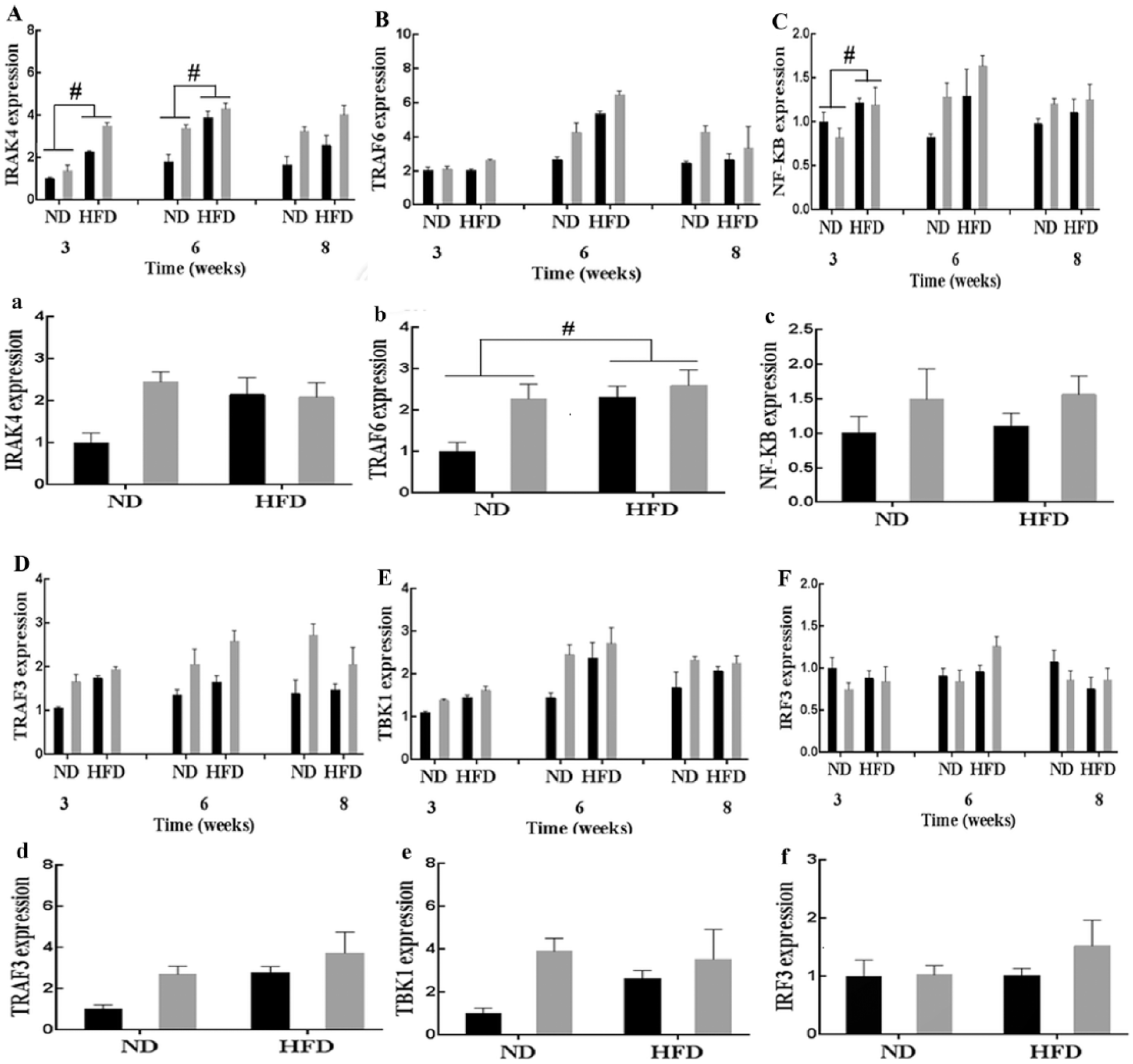
Expression of MyD88-dependent signal pathway factors IRAK4, TRAF6, NF-κB, and MyD88-independent signal pathway factors TRAF3, TBK1, and IRF3 in the monocytes/macrophages and adipose tissue under saturated fatty acid (SFA) treatment at 8 weeks. (a) IL-1 receptor–associated kinase 4 (IRAK4), (b) TNF receptor-associated factor 6 (TRAF6), (c) nuclear factor-kappa B (NF-κB), (d) TNF receptor-associated factor 3 (TRAF3), (e) TRAF family member–associated NF-κB activator (TANK)-binding kinase-1 (TBK1), and (f) interferon regulatory factor 3 (IRF3). Capital letters represent the expression of signaling pathway factors in the monocytes/macrophages and small letters represent the expression of signaling pathway factors in the adipose tissue. Results are presented as means ± SEM. #P < 0.05 compared with the normal diet (ND) group. HFD: high-fat (SFA) diet; GM: genetically modified; WT: wild type.
Discussion
SFAs are pro-inflammatory lipids that can directly stimulate pro-inflammatory cytokine expression. 4 For instance, mice fed a diet high in SFAs exhibit increased levels of such cytokines in adipose tissue. 10 TLR4 appears to be a critical link between SFAs and low-grade inflammation. SFAs can act as TLR4 activators and trigger pro-inflammatory responses involved in the development and progression of many chronic diseases and metabolic disorders. 11 In this study, TLR4 overexpression enhanced IL-6, TNF-α, and IL-8 expression in SFA-stimulated monocytes/macrophages and raised IL-6 expression and TNF-α protein concentration in adipose tissue. In contrast, C3H/HeJ mice, which carry a spontaneous TLR4-inactivating mutation, exhibit decreased TNF-α mRNA levels and associated attenuated adipose tissue inflammation compared with control mice when fed a diet high in fat. 12 Taken together, TLR4 overexpression enhanced both the pro- and anti-inflammatory response during SFA stimulation.
In this study, treatment with SFAs resulted in upregulation of downstream genes of MyD88-dependent signaling, including IRAK4, TRAF6, and NF-κB. However, SFA administration had no significant impact on the expression of MyD88-independent signaling components, including the TRAF3, TBK1, and IRF3 genes. As discussed above, our results demonstrate that activation of the MyD88-dependent signaling, but not the MyD88-independent, constitutes a principal mechanism in the response of sheep monocytes/macrophages and adipose tissue to SFAs.
Footnotes
Acknowledgements
X.X. and M.-Y.Q. have contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by grants from the National Transgenic Creature Breeding Grand Project (2014ZX08008-005, 2016ZX08008-003), Harbin Municipal Natural Science Foundation (2013RFQXJ031), and Open Projects of Key Laboratory of Animal Genetics, Breeding and Reproduction, College of Heilongjiang Province (GXZDSYS-2012-06).
