Abstract
Detection of autoantibody against extractable nuclear antigens (ENAs) plays a critical role in the diagnosis and management of autoimmune diseases.In this study, we assessed the performance of LETIA in detecting anti-ENAs. Total 606 serum samples from the Second Hospital of Shanxi Medical University were collected. Anti-SSA, anti-SSB, anti-Sm, anti-U1-snRNP, and anti-Sm/RNP were parallelly detected by LETIA and line immunoblot (LIA). Besides, this study assessed LETIA for its repeatability in detecting anti-ENAs autoantibodies, and consistency with LIA. A receiver operating characteristic (ROC) curves was drawn to assess the accuracy of LETIA. The LETIA and LIA showed high coincidence rate in detecting anti-SSA, anti-SSB, anti-Sm, anti-U1-snRNP, and anti-Sm/RNP autoantibodies, with the results being 87.22%, 96.61%, 97.03%, 88.28%, and 92.06%, respectively. Almost perfect consistency (kappa > 0.8) were found in the detection of anti-SSB and anti-Sm by LETIA and LIA. While in the detection of anti-SSA, anti-U1-snRNP, and anti-Sm/RNP, moderate consistency (0.6 ⩽ kappa ⩽ 0.8) were shown. The AUCs of anti-SSA, anti-SSB, anti-Sm, anti-U1-snRNP, and anti-Sm/RNP detected by LETIA were 0.972 (95% confidence interval (CI): 0.941–1.000, p < 0.001), 0.986 (95% CI: 0.967–1.000, p < 0.001), 0.912 (95% CI: 0.763–1.000, p < 0.001), 0.829 (95% CI: 0.731–0.928, p < 0.001), and 0.828 (95% CI: 0.715–0.941, p < 0.001), respectively. LETIA and LIA showed considerable consistency in detecting anti-ENAs. Moreover, with the pronounced advantages of automatic and rapid detection, and high universality, LETIA can meet the requirements for quantitative detection of anti-ENAs.
Keywords
Introduction
Autoimmunity is a condition autoaggression of the immune system against constitutive antigens of an individual via production of autoantibodies, which may give rise to specific diseases. Serological detection is an important tool for diagnosing autoimmune diseases (AID) and related disorders.1–3 Internationally, antinuclear antibodies (ANAs) screening is recommended as the primary diagnosis standard of systemic autoimmune-rheumatic diseases. 4 However, the presence of positive ANAs does not necessarily indicate a disease state since low levels of ANAs can also be detected in healthy individuals. 5 Therefore, after the initial screen for ANAs, autoantibodies to extractable nuclear antigen (ENAs) are also frequently detected due to their diagnostic and prognostic significance. 6 A study suggested that a combined detection of ANA and ENA autoantibodies was more helpful for the diagnosis and prognosis of AID. 7
The identification of ENAs plays a critical role in the diagnosis and management of ANA-associated rheumatic disease (AARD), such as systemic sclerosis (SSc), systemic lupus erythematosus (SLE), Sjogren’s syndrome (SS), and mixed connective tissue disease (MCTD).8–10 In addition, study suggests anti-SSA/Ro can be detected in the majority of SS patients (70%–100%) and 35% to 70% SLE patients, and anti-La/SSB is detectable in 45% of patients with SLE and up to 40% to 90% of patients with SS. 11 Anti-Sm antibody is a highly specific marker of SLE. 12 In addition, anti-RNP reacts with proteins that are associated with U1RNA and form U1-snRNP. High titer of anti-U1-snRNP can be used to diagnose MCTD, with the clinical sensitivity up to 90%.13–15
Anti-ENAs have great significance and value in clinical diagnosis and classification, disease progression, and prognosis evaluation of AARD. 16 Testing for ENAs has historically relied on double immunodiffusion and enzyme immunoassay.17,18 In recent years, enzyme-linked immunoassays (ELISA) have been widely applied to detect the presence and specificity of anti-ENAs. 19 Many studies have shown that ELISA detection system has higher sensitivity than other detection methods, however, the increased sensitivity of ELISA may induce reduced diagnostic specificity.20,21 At present, the domestic clinical laboratories mainly rely on line immunoassay (LIA) for anti-ENA testing. 22 In addition, with an increased demand for anti-ENAs testing, several automated high throughput assays have been developed. Meanwhile, diagnostic accuracy and sensitivity of these new platforms has gained extensive attention of clinicians. To our knowledge, latex enhanced immunoturbidimetry (LETIA) has the advantages of automated quantitative detection, good stability, and and high repeatability. Herein, we assessed for the first time the clinical performance of LETIA assay in detecting anti-ENAs.
Material and methods
Sample collection
A total of 606 samples (460 females, 146 males, mean age 48.22 ± 14.72 years) in the Second Hospital of Shanxi Medical University were included in this study. Each autoantibody was detected as an independent assay in the LETIA analysis since the detection of each autoantibody by LETIA is an independent detection kit. Therefore, we selected randomly different samples for each autoantibody (anti-SSA (n = 133), anti-SSB (n = 118), anti-Sm (n = 101), anti-U1-snRNP (n = 128), and anti-Sm/RNP (n = 126)). These samples were parallelly tested by LETIA and LIA, and corresponding quality controls were performed. Furthermore, patients categorized as AARD fulfilled the classification criteria for the respective diseases, whereas inpatients that did not satisfy the classification criteria, were categorized as non-AARD (Table 1). All the blood samples were centrifuged to separate serum within 3 h after sampling and stored at –80°C. The test procedures were performed in accordance with the manufacturers’ instructions. Written informed consent was obtained from each patient in the study. The study was approved by the ethics committee of the Second Hospital of Shanxi Medical University (2016KY007).
A summary of baseline demographics and disease characteristics of all enrolled individuals.

LIA: line immunoassay; LETIA: latex enhanced turbidimetric immunoassay; F: female; M: male; AARD: ANA-associated rheumatic disease; SLE: Systemic lupus erythematosus; SjS: Sjogren’s syndrome; SSc: Systemic sclerosis; MCTD: Mixed connective tissue disease; PM/DM: Polymyositis/dermatomyositis; RA: Rheumatoid arthritis; USPA: Undifferentiated spondy loarthropathy.
LETIA for anti-ENAs
LETIA system for anti-ENAs detection, including automatic biochemical analyzer FAITH2000, and anti-SSA kit, anti-SSB kit, anti-Sm kit, anti-U1-snRNP kit, and anti-Sm/RNP kit provided by Shanghai Chuan-Zhi biotechnology co., LTD. In this study, each autoantibody was an independent kit. According to the instruction provided by the manufacturer, we added 150 μL reaction buffer (R1) and 15 μL serum samples incubated at 37°C for 3 min, and then 50 μL latex reagent (R2) coated with native antigen was added. Finally, the absorbance was measured at 500 nm. The cut-off values for anti-SSA, anti-SSB, anti-Sm, anti-U1-snRNP, and anti-Sm/RNP were 4.0, 16.0, 60.0, 10.0, and 6.0 RU/mL, respectively. All standards were re-dissolved with 0.5ml pure water and stood for 20 min at room temperature. Then we conducted multi-parameter curve fittings. Subsequently, a patient serum sample and a healthy control were randomly selected and repeated for 10 times to evaluate the repeatability of LETIA.
LIA for anti-ENAs
LIA for anti-ENAs research was performed by using a EUROBLOT ONE analyzer (Euroimmun, Germany) and EUROIMUN ANAs (IgG) diagnostic kit for the semi-quantitative determination of IgG autoantibodies against SSA, SSB, Sm, U1-snRNP, and Sm/RNP antigens in serum. These antigens were detected on a nylon membrane with a plastic backing. These strips were incubated sequentially with sera, alkaline phosphatase-labeled anti-human IgG antibodies, and substrate solution, according to the manufacturer’s instructions. All incubations were at room temperature. The sample buffer, wash solutions and substrate solutions were all provided in the test kit. All membrane strips were scanned by EUROBLOT ONE analyzer.
Statistical analysis
All data were analyzed by using the Statistical Package for the Social Sciences software (SPSS) version 22.0 or Graphpad Prism 7. Chi-square test was used to compare the test results. Kappa test was conducted to evaluate the consistency of results between LETIA and LIA. Concordance between two assays is the percentage of samples that give same results. At the same time, the predictive accuracy of the LETIA assay was expressed as the area under the curve (AUC) calculated by using receiver operator characteristic (ROC) curve analysis. P < 0.05 was considered statistically significant.
Results
Performance of LETIA in detecting anti-ENAs
The anti-ENAs standards of different concentration were tested by LETIA and standard curve was drawn through multi-parameter curve fitting (spline or Logit/log). The R squares were 0.9968, 0.9977, 0.9988, 0.9993, and 0.9982 for anti-SSA, anti-SSB, anti-Sm, anti-U1-snRNP, and anti-Sm/RNP, respectively. The results showed that, within a certain range, the absorbance of the standards have excellent correlation with the concentration of the autoantibodies, and all R squares were greater than 0.990 (Figure 1). Subsequently, a patient serum sample and a age- and sex- matched normal sample were repeatedly tested for 10 times, respectively. The excellent repeatability of LETIA in detecting anti-ENAs was shown. The variation coefficients of anti-SSA, anti-SSB, anti-Sm, anti-U1-snRNP, and anti-Sm/RNP autoantibodies were 2.50%, 3.71%, 1.36%, 2.16%, and 1.19% in disease status, respectively. In healthy individual, the variation coefficients of anti-SSA, anti-SSB, anti-Sm, anti-U1-snRNP, and anti-Sm/RNP autoantibodies were 9.40%, 9.82%, 5.84%, 5.56%, and 5.94%, respectively (Table 2).
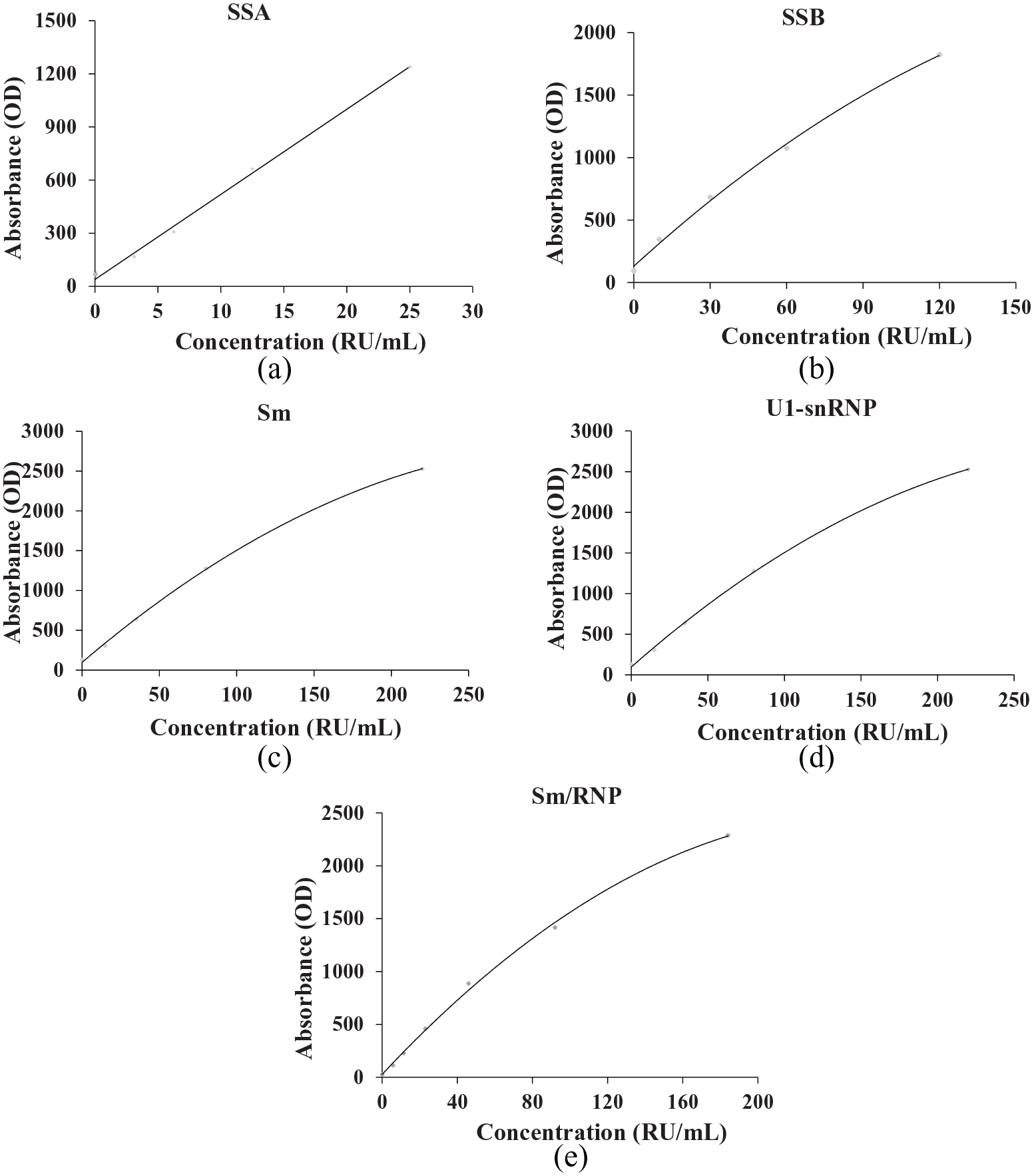
Standard curves of LETIA in detecting anti-ENA. They showed excellent relationship between absorbance and concentration for anti-SSA (a), SSB (b), Sm (c), U1-snRNP (d) and Sm/RNP (e), and the R2 ⩾ 0.990.
Repeatability of anti-ENAs was detected by LETIA assay in disease status and healthy control.

HC: healthy control; M: mean; SD: standard deviation; CV: coefficient of variation.
Comparison between LETIA and LIA in detecting anti-ENAs
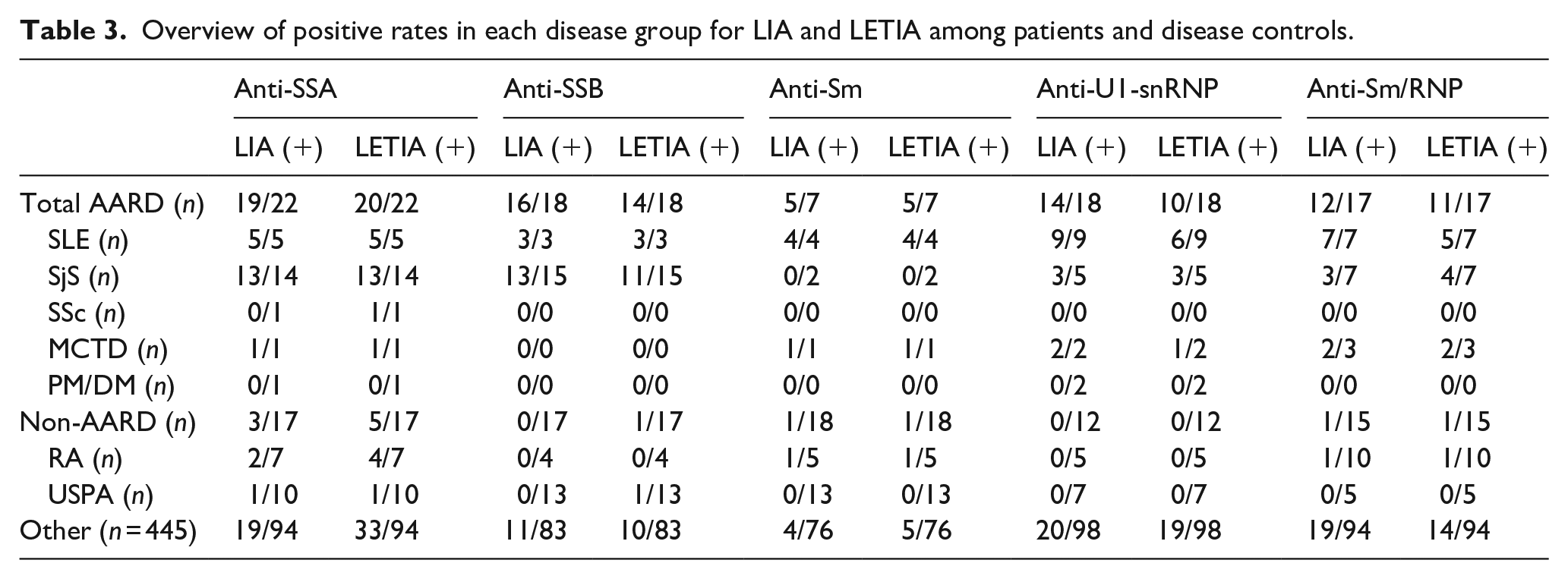
A total of 283 inpatients suspected of ARD and 323 outpatient were submitted for ENA testing by LIA and LETIA. In those inpatients, 82 patients (28.98%) were diagnosed with AARD. Of these 82 AARD patients, 28 were diagnosed with SLE, 43 with SjS, 1 with SSc, 7 with MCTD and 3 with PM/DM. The positive rates in each disease group for LIA and LETIA among patients and disease controls were shown in Table 3.
Overview of positive rates in each disease group for LIA and LETIA among patients and disease controls.
The positive coincidence rates of LETIA and LIA in detecting anti-SSA, anti-SSB, and anti-Sm were high, with each being100.00%, 88.89%, and 90.00%, respectively. However, when it comes to anti-U1-snRNP and Sm/RNP, a relatively high was shown, with each being 70.59% and 75.00%, respectively. As for the negative coincident rate, anti-SSA, anti-SSB, anti-Sm, anti-U1-snRNP, and anti-Sm/RNP were high, with each being 81.52%, 98.90%, 97.80%, 94.68%, and 97.87%, respectively. Under manufacturer’s cut-off values, the total coincidence rates of LETIA and LIA in detecting anti-SSA, SSB, Sm, U1-snRNP, and Sm/RNP were 87.22% (116/133), 96.61% (114/118), 97.03% (98/101), 88.28% (113/128), and 92.06% (116/126), respectively. Furthermore, the results showed that both LETIA and LIA had almost perfect consistencies (kappa > 0.8) in detecting anti-SSB and anti-Sm, with the kappa coefficients standing at 0.901 (p < 0.001) and 0.841 (p < 0.001), respectively. Anti-SSA, U1-snRNP, and Sm/RNP showed substantial consistencies (0.6 ⩽ kappa ⩽ 0.8), with the kappa coefficients standing at 0.731 (p < 0.001), 0.685 (p < 0.001), and 0.776 (p < 0.001) (Table 4).
The agreement of LETIA and LIA in detecting anti-ENAs.

LETIA: latex enhanced turbidimetric immunoassay; LIA: line immunoassay; P: positive; N: negative; C: confidence interval.
ROC-AUCs of LETIA in detecting anti-ENAs
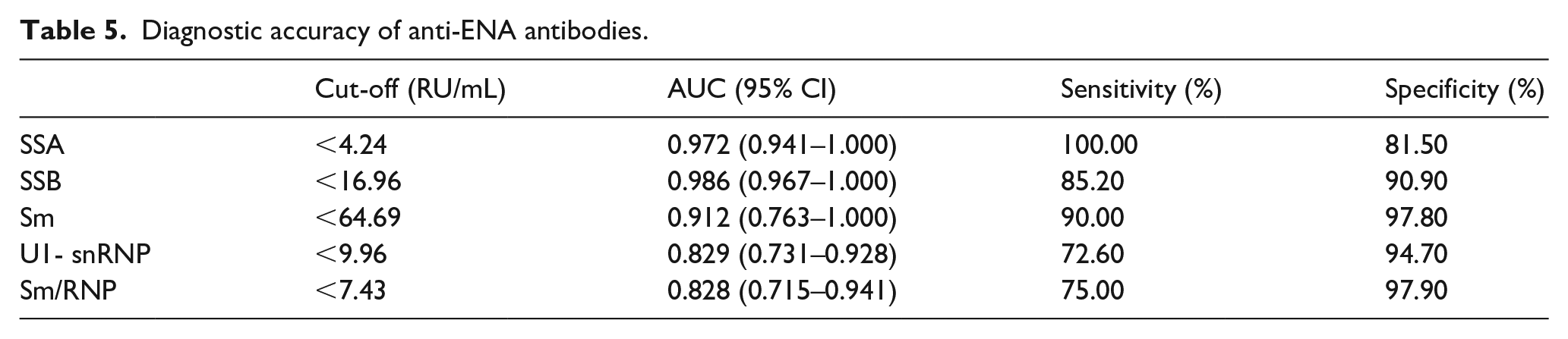
The ROC curves were performed with LIA as the reference method, and the accuracy of LETIA in detecting anti-ENAs was analyzed through AUC (Figure 2). The results showed that the LETIA method had good accuracy in detecting anti-SSA, anti-SSB, anti-Sm, anti-U1-snRNP and anti-Sm/RNP. When the cut-off value of anti-SSA, anti-SSB, anti-Sm, anti-U1-snRNP and anti-Sm/RNP was set at 4.24 RU/mL, 16.96 RU/mL, 64.69 RU/mL, 9.96 RU/mL and 7.43 RU/mL, the corresponding sensitivity and specificity stood at 100.00% and 81.50%, 85.20% and 90.90%, 90.00% and 97.80%, 72.60% and 94.70%, 75.00% and 97.90%, respectively (Table 5). Furthermore, the AUCs of anti-SSA, anti-SSB, anti-Sm, anti-U1-snRNP, and anti-Sm/RNP were 0.972 (95% confidence interval (CI): 0.941–1.000, p < 0.001), 0.986 (95% CI: 0.967–1.000, p < 0.001), 0.912 (95% CI: 0.763–1.000, p < 0.001), 0.829 (95% CI: 0.731–0.928, p < 0.001), and 0.828 (95% CI: 0.715–0.941, p < 0.001), respectively.

Receiver operating characteristics (ROC) curves of LETIA assay for anti-SSA, SSB, Sm, U1-snRNP and Sm/RNP. The orange full line indicated the AUC of anti-SSA; green full line indicated the AUC of anti-SSB; brown full line indicated the AUC of anti-Sm; gray full line indicated the AUC of anti-U1-snRNP; purple full line indicated the AUC of anti-Sm/RNP. The AUCs were 0.972, 0.986, 0.912, 0.829, and 0.828, respectively. The dotted line was the reference line.
Diagnostic accuracy of anti-ENA antibodies.
Discussion
ENAs are a class of ribonucleoproteins and nonhistone proteins, which can produce specific corresponding antibodies. The most common ENAs include RNP, Sm, SSA, SSB, Scl-70, and Jo-1, with high sensitivity and specificity, but others have been described. 23 The autoantibodies to ENAs are helpful for the diagnosis and classification of various AID, and are included in the classification criteria of various AID, therefore, the detection of autoantibodies to ENAs has attracted great attention.24,25 In recent years, with the development of coated antigens and technology, rapid and automated immunoassay methods were developed by numerous manufacturers. In the current study, we evaluated for the first time the clinical performance of LETIA in detecting anti-ENAs.
In our study, LETIA and LIA showed high agreements in detecting anti-SSA, anti-SSB, anti-Sm, anti-U1-snRNP, and anti-Sm/RNP, with the total agreement ranging from 87.22% to 97.03%. Our results also demonstrated that the kappa values in detecting anti-SSA, anti-SSB, anti-Sm, anti-U1-snRNP, and anti-Sm/RNP through LETIA and LIA assay were high (kappa > 0.6). For anti-SSB and anti-Sm, a almost perfect consistency were found (kappa > 0.8), while anti-SSA, anti-U1-snRNP, and anti-Sm/RNP showed the substantial consistent (0.6 ⩽ kappa ⩽ 0.8). 26 When analyzing the difference between two methods, it showed that a majority (31/49) of the inconsistent results came from specimens with a low ANA titers (1:100/-), which is in line with previous studies. 27 This may be due to the fact that the antibodies of patients with low titer of ANA has low binding activity. 6 On the other hand, because the international reference materials have been not developed, the values of calibrators for different assays were different and the detected results among different assays were diverse.
The ROC-AUC was used to evaluate the accuracy of the novel diagnostic method (LETIA) as compared to the already-established one (LIA). 25 The results demonstrated that LETIA had good predictive value in detecting anti-SSA, anti-SSB, anti-Sm, anti-U1-snRNP and anti-Sm/RNP. The AUCs of anti-SSA, anti-SSB and anti-U1-snRNP were over 0.900, and the anti-U1-snRNP and anti-Sm/RNP were greater than 0.8, indicating excellent detective performance. This may be resulted from the difference in the study populations. The LIA method coats dozens of antigens with completely different biochemical and physical properties onto a nitrocellulose membrane, and then share the same secondary antibodies, substrate, and the uniform reaction time and conditions, which makes it impossible that all the detection items attain the best reaction. While for the LETIA assay, it has independent antigen coating, independent calibration system and reaction conditions. Each autoantibody was detected as an independent assay. This may be another reason for the difference between two methods.21,28 However, LETIA has a limitation that samples with jaundice and hemolysis may interfere with the results.
It is widely accepted that clinical disease is critical to evaluate the performance of new method, therefore, we further analyzed the individual autoantibody in these AARD patients. Of the 14 patients with established SjS, 13 were anti-SSA positive (13/14) and 5/5 were anti-SSA positive in SLE patients. A total of 3/3 SLE patients and 11/15 SjS patients were positive for anti-SSB. Anti-Sm autoantibody was positive in 4/4 patients with SLE. The positive rates of U1-snRNP autoantibody detected through LETIA were SLE (6/9), SjS(3/5) and MCTD(1/2), respectively. Anti-Sm/RNP was positive in 5/7 SLE patients, 4/7 SjS patients and 2/3 MCTD patients (Table 3). In addition, LIA and ELISA are well established methods for the detection of anti-ENAs. Many studies have shown that, compared with other methods, ELISA detection system has higher sensitivity and is more convenient for a large number of samples.20,21,29 However, ELISA is tedious and time-consuming, therefore, it not well suited for daily clinical practice. 30 For example, samples need to be pre-diluted, and a lot of institutions still adopt manual operation, which increases the human error. 31 However, LETIA has the advantages of short detection time, wide detection range, and automatic quantitative testing.32,33 In addition, LETIA can detect anti-ENAs simply by using biochemical analyzer or protein analyzer, without requiring specific instruments and equipments, which will greatly enhance the universality of LETIA.
There are also some limitations in this study. First, the sample size was small, and sample size calculation was not done. Second, LETIA was compared only with commonly-used LIA. Therefore, we need further comparative analysis with other methodologies and reference methods in a larger sample cohort to confirm the performance of LETIA in detecting anti-ENAs.
Conclusion
This study gave an evaluation of the performances of LETIA in detecting anti-ENAs. The current results indicated that the performance of LETIA was satisfying compared with commonly-used LIA. From the perspective of clinical application, we believe that LETIA can provide clinicians with reliable and timely test results, which is conducive to the screening and monitoring of autoimmune diseases.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval for this study was obtained from the ethics committee of the Second Hospital of Shanxi Medical University (2016KY007).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Key Research and Development Project of Shanxi Province, China (201603D321091), the Key Research and Development Project of Shanxi Province, China (201803D421067), the Nature Fund Projects of Shanxi Science and Technology Department (201901D111377), the Scientific Research Project of Health commission of Shanxi Province (2019044) and Research Project Supported by Shanxi Scholarship Council of China (2020-191).
Informed consent
Written informed consent was obtained from all subjects before the study.
Trial registration
Not applicable
