Abstract
To explore the effects of two different administration routes of Endostar on the survival of patients with medium and advanced hepatocellular carcinoma (HCC) and underwent trans-arterial chemoembolization (TACE). Seventy-two patients with medium and advanced HCC were enrolled. Among them, 42 patients underwent the hepatic arterial infusion of Endostar combined with TACE (infusion therapy group); and the remaining 30 patients underwent the hepatic treatment of TACE combined with the intravenous application of Endostar (intravenous therapy group). All patients underwent regular examinations of CT (or MRI) and DSA to observe the conditions of tumor recurrence or metastasis, and to determine the existence of tumor angiogenesis. The response rate of treatment in the Endostar hepatic arterial infusion group was higher than that in the control group, and the difference was statistically significant (31/42:14/30, X2 = 5.501, p < 0.05). In addition, median progression free-survival time of the two groups were 8.67 months and 6.67 months, respectively (p = 0.046); and the difference was statistically significant. The hepatic arterial infusion of Endostar combined with TACE can significantly improve recent clinical efficacy and mPFS in the treatment of medium and advanced HCC. However, improvement on the overall survival of long-term efficacy is not significant.
Keywords
Introduction
Primary hepatocellular carcinoma (HCC) is one of the common malignant tumors in clinic. Its mortality is high, is lower than that in lung cancer, and ranks second in grade. However, the rate of surgical resection is only 20% to 30%. 1 Trans-arterial chemoembolization (TACE) is currently the first choice for the treatment of unresectable or postoperative recurrence of medium and advanced HCC. However, its long-term efficacy is not ideal, and its three-year survival rate is only 14% to 30%. 2 After TACE treatment, the hypoxia ischemia of the tumor focus can promote the production of vascular endothelial growth factor (VEGF), transform growth factor-beta 1 (TGF-b1), and induce tumor vascular regeneration; which are the main causes of tumor recurrence and metastasis, and are also important factors that affect the efficacy of TACE. 3 Recombinant human endostatin (Endostar) can specifically inhibit the proliferation and migration of vascular endothelial cells and inhibit the growth and metastasis of tumors. Thus, it has been considered to be an important drug in the treatment of cancer.4–6 However, which administration routes of Endostar can result in better treatment effects is unknown. Thus, to explore the effects of two different administration routes of Endostar on the survival of patients with medium and advanced hepatocellular carcinoma (HCC) and underwent trans-arterial chemoembolization (TACE), from March 2009 to March 2015, a total of 72 cases of patients with medium and advanced HCC were admitted in our hospital and underwent TACE treatment combined with the application of Endostar via the routes of hepatic arterial infusion and intravenous drips. The comparative survival analysis is as follows.
Materials and methods
Clinical data
A total of 72 cases of patients with medium and advanced HCC, who were admitted in our hospital from March 2009 to October 2014, were selected for a case-control trial. All patients were primary therapeutic patients, who were all diagnosed with primary HCC by imaging examination and AFP or liver puncture biopsy and had measurable focuses. All patients were diagnosed in accordance with the diagnosis and treatment norms of primary HCC. Furthermore, all patients provided a signed informed consent prior to enrollment into the study. This protocol was approved by the Ethics Committee of the hospital. Among these patients, 50 patients were male, and 22 patients were female; and the age of these patients ranged from 46 to 68 years old, with an average age of 54.3 ± 6.8 years old. All the patients were randomly divided into two groups: group A, hepatic arterial infusion of Endostar combined with TACE treatment (42 cases); group B, intravenous drips of Endostar combined with TACE treatment (30 cases). There were no significant differences in age, gender, tumor size and other aspects between these two groups (p > 0.05) (Table 1). This study was conducted in accordance with the Declaration of Helsinki and approved by the ethics committee of our hospital. All participants signed informed consent.
The stage of Barcelona clinic liver cancer.
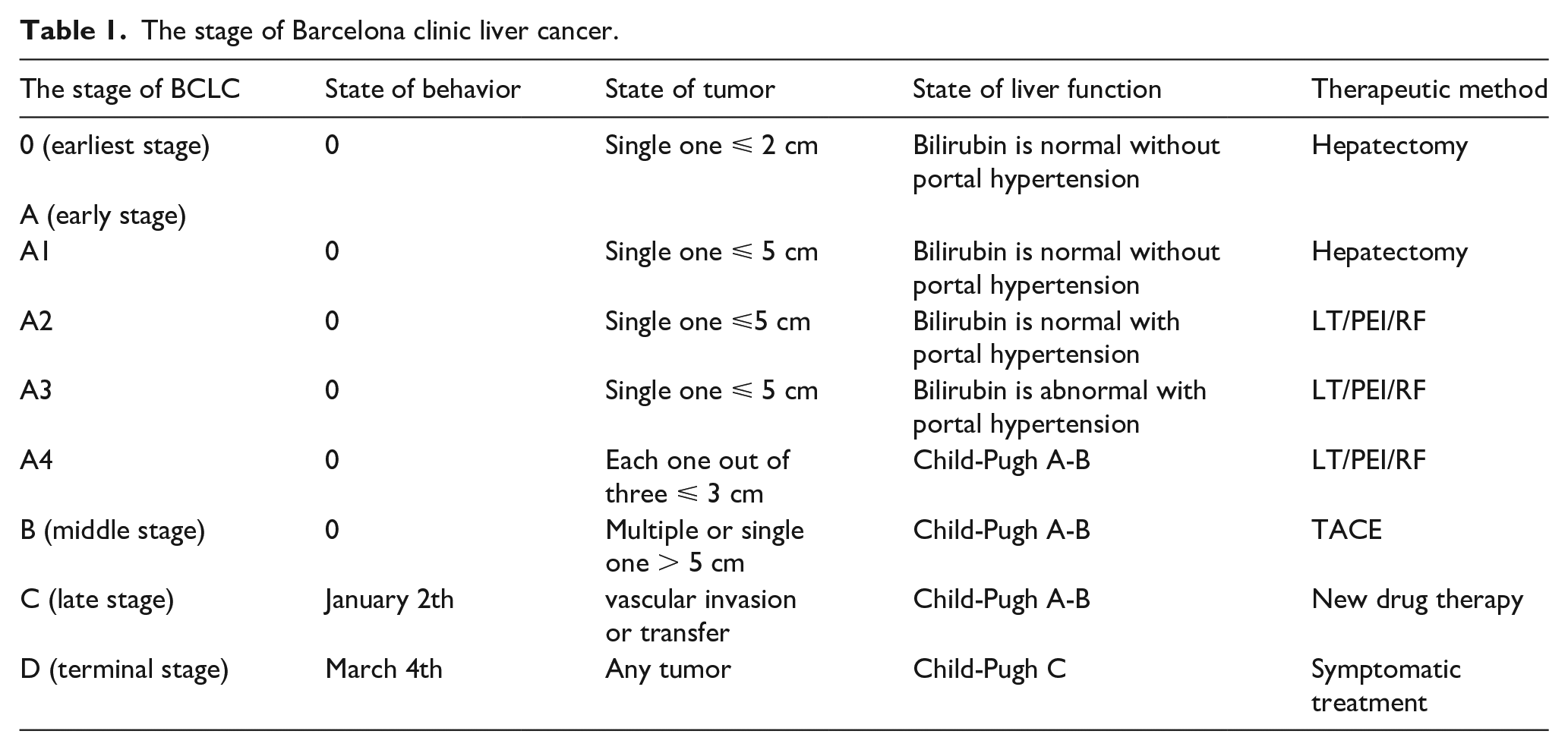
Exclusion criteria: (1) liver function was grade C in Child grading, and there was a tumorous thrombosis in the portal stem vein; (2) the expected survival time was less than 6 months according to the staging and metastasis of HCC and patients’ status, and there was a wide range of intra-liver or distant metastases; (3) patients with other severe organic diseases such as cardiac and cerebral disease or renal disease; (4) Hepatic artery angiography revealed that the tumor had a poor blood supply; (5) patients combined with mental disease or language expression was not clear. A total of 72 cases were treated for at least two cycles.
Treatment methods
All cases in group A underwent TACE treatment twice, and the interval of each treatment was 30 days. Arteriography was performed after the catheter was punctured into the hepatic proper artery or its branches via the femoral artery using the Seldinger technique. Tumor staining was observed. Further super-selective catheter was inserted into the arteries that supply blood to the tumor. Sixty mg of epirubicin and 10 to 20 mL of super liquefaction iodipin were fully mixed into the emulsion and slowly infused into the arteries. Then, the blood supply arteries were embolized with PVA particles. For patients in group A, after two times of embolization, the hepatic arterial chemotherapy pump tube was placed in the arteries that supply blood to the tumor (right or left liver artery), and the arterial pump was placed in the right abdominal subcutaneous. At 1 week post-operation, a mixture of 7.5 mg/m2 of Endostar and 500 mL of normal saline in the chemotherapy pump was infused into the artery, and maintained for 4 h. The patients were continuously administered from the first to the fourteenth day, and the administration was repeated with an interval of 16 days. The average application time was 6.45 cycles. For patients in group B, after two times of TACE treatment, Endostar was administrated 2 days before the third TACE treatment; and was continuously administered for 14 days. The usage was: 7.5 mg/m2 of Endostar was mixed with 500 mL of normal saline, intravenously dripped at a constant speed, and maintained for 4 h. Then, TACE treatment was performed 2 days after Endostar administration, and the TACE technique was the same with group A. These procedures were repeated during the second TACE treatment (Endostar was administrated with an interval of 16 days), and average application time was 6.24 cycles.
Efficacy evaluation
All patients underwent enhanced CT (MRI) examination. This was repeated each time before interventional treatment, and the efficacy was compared. The efficacy was evaluated by the Response Evaluation Criteria in Solid Tumors v1.1 (RECIST v1.1): the complete disappearance of all focuses was assigned as complete remission (CR), a reduction of 30% in the long diameter of the baseline focus was assigned as partial remission (PR), a 20% increase in the long diameter of the baseline focus or new focus appeared was assigned as progressive disease (PD), and the decrease in the level of the long diameter did not reach PR or the level of increase in long diameter did not reach PD was assigned as stable disease (SD). Response rate (RR) = (CR + PR)/total number of cases × 100%; the disease control rate (DCR) = (CR + PR + SD)/total number of cases × 100%. Regular outpatient follow-up or telephone follow-up were carried out for all patients, imaging contrast examination was performed each time before the application of Endostar, and comparison were performed on the same instruments for CT or MRI examination used during the first visit, in which the interval of each time was 1 month. The last follow-up time was December 15, 2015. Progression-free survival (PFS) time was defined as the time from the beginning of intervention treatment to the time when PD was identified, and the overall survival (OS) time was defined as from the first intervention treatment beginning to death or last follow-up time.
Statistical methods
SPSS 17 software was used for statistical analysis. Count data were analyzed by X2-test. The survival curve was plotted by the Kaplan-Meier method, values of PFS and OS were calculated, and the survival difference was analyzed by the log-rank method.
Results
Clinical data
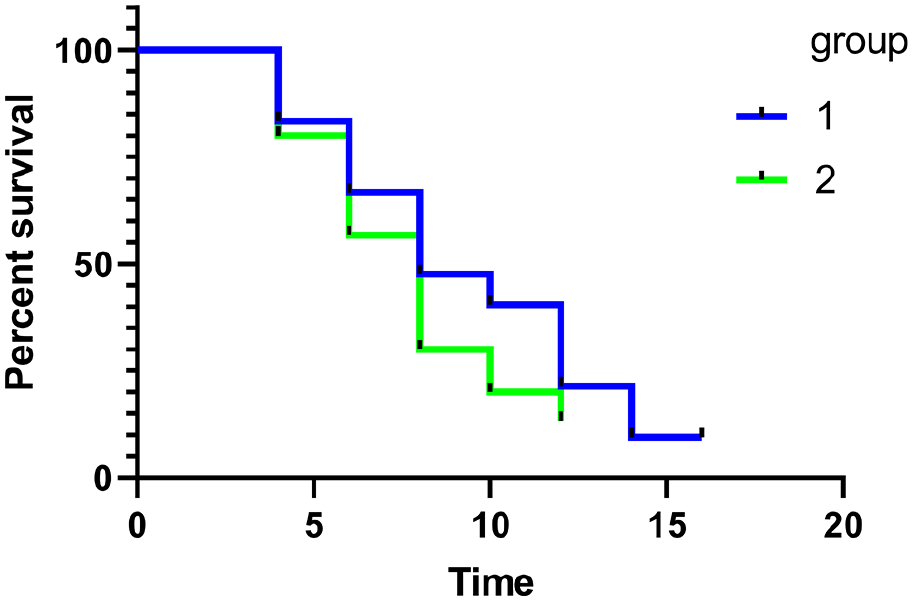
In the Endostar hepatic arterial infusion group, after treatment, two cases of patients were CR, 29 cases were PR, six cases were SD, and five cases were PD (Tables 2 and 4). In the Endostar intravenous therapy group, after the treatment, no case was CR, 14 cases were PR, 12 cases were SD, and four cases were PD. In comparing between the two groups, there was a significant difference in response rate (RR; 31/42:14/30, X2 = 5.501, p < 0.05). Furthermore, there was a significant difference in disease control rate (DCR; 37/42:26/30, X2 = 0.03, p > 0.05), and the differences were statistically significant (Figure 1).
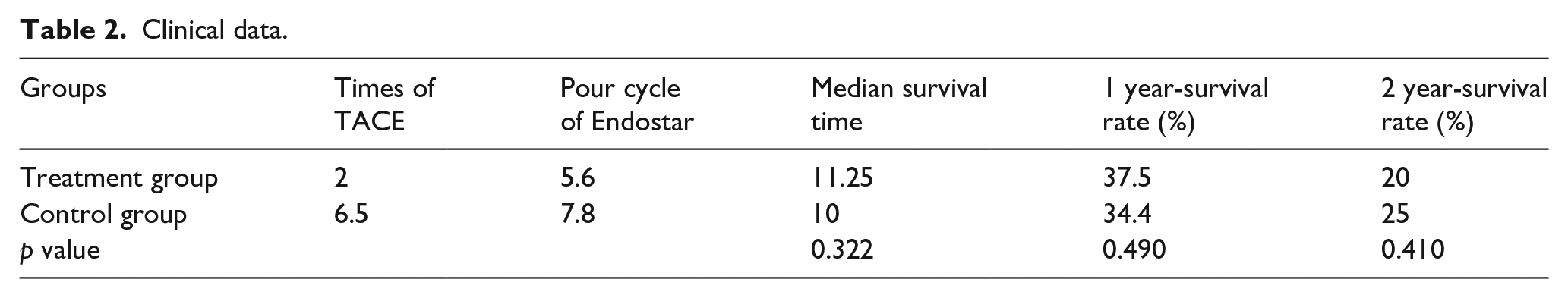
Clinical data.
The comparison of toxic reaction of the two groups.

Basic data before treatment.

Clinical data.
Survival results
Median progression-free survival (mPFS) time in the Endostar hepatic arterial infusion treatment group and intravenous therapy group was 8.67 months and 6 months (p = 0.046), respectively, and median overall survival (mOS) time was 11.25 months and 10.00 months, respectively (p = 0.322). The difference was not statistically significant (Figure 2).

Survival results.
Toxicity reaction
Cardiac toxicity, bilirubin, serum aminotransferase and diarrhea were observed in the two groups of patients, and no III–IV degree of toxicity reactions appeared. For cardiac toxicity: in the treatment group, four cases were in degree I and two cases were in degree II; in the control group, two cases were in degree I and one case was in degree II. There were no significant differences in bilirubin, serum aminotransferase, and diarrhea between the two groups (p > 0.05). Results are shown in Table 3.
Discussion
Approximately 110,000 people die of HCC in China each year. Among the current clinical visiting patients with HCC, patients who were completely suitable for surgical resection account for approximately 20%; and TACE treatment is preferred for non-surgical patients.7,8 However, after simple TACE treatment, tumor necrosis rate was only 20% to 50%; hence, the treatment effect is not ideal 9 if tumor cells cannot be completely necrotic, that is, no ideal operational effects were achieved. 10 Tumor angiogenesis is a complex process that involves in a variety of factors, including a series of steps of enhancement of angiogenesis promoting factor activity, endothelial basement membrane degradation and the upregulation of adhesion molecule expression, the formation of the lumen wall of blood vessels, and the formation of mature angiogenesis. 11 VEGF is the strongest angiogenesis promoting factor and endothelial cellular specific potent mitogen. It can specifically act on vascular endothelial cells to promote the proliferation and migration of endothelial cells, in order to promote tumor angiogenesis. As a result, a lot of new vessels with abnormal structures and functions appeared, and the growth and metastasis of tumor is consequently accelerated. In addition, after the TACE operation, ischemia and hypoxia in part of the tumor cells would lead to increased VEGF expression levels, and further lead to increased microvascular growth around the tumor, as well as the invasion and metastasis of the tumor. 12
Through specific actions on the endothelial cell of new vessels and the inhibition on migration of endothelial cells, as well as the induction of cellular apoptosis, Endostar can play an important role in anti-angiogenesis effect, and indirectly lead to tumor dormancy or retreat.13,14 Recombinant human endostatin combined with other treatment methods can improve the curative effect on multiple malignant tumors. This treatment model breaks through the clinical research dilemma that the effective rate of simple drug treatment of recombinant human endostatin is not high. 15 Many domestic and foreign research results revealed that Endostar has a significant effect on anti–angiogenesis, and the application of Endostar can reduce the extrahepatic metastases of HCC after TACE treatment, which would significantly prolong the survival of patients.16–18 Furthermore, TACE treatment can make chemotherapy drugs act on local tumors, and embolize the blood supply of tumor artery, as a result to directly kill the tumor cells and destroy the formed blood vessels of tumors. TACE and anti-angiogenesis drugs are complementary. The combination of the two treatments can achieve better therapeutic effect, especially via the direct hepatic artery perfusion.19,20
Through the research of application, we found that there were significant differences in the comparison between the therapy group of Endostar combined with TACE and the control group of simple TACE treatment. 21 The response rate was satisfactory in the treatment of HCC with the hepatic arterial infusion of Endostar combined with TACE, the progression-free survival time had been prolonged, and the difference has statistical significance. 21 However, there was no significant difference in overall survival time.
When the group of TACE combined with the hepatic arterial infusion of Endostar was compared with the group of TACE combined with intravenous drips of Endostar, there was significant difference in the response rate, and the progression-free survival time had been prolonged. The difference was statistically significant, but there was no significant difference in overall survival time between these two groups. The reason mainly comprised of two aspects: one was that in the arterial Endostar application group, although the acting time is short, the concentration of anti-angiogenic drugs in the liver is high, the ability of angiogenesis inhibition is stronger than that in the venous group, and progression-free survival time was prolonged in certain times. The second was: in the arterial treatment group, as the arterial pump was placed after two cycles of treatment, TACE could not be continued; while TACE treatment was not be interrupted in the intravenous application of Endostar, which made its role of anti -angiogenesis generate the maximum effect on the proliferation of tumor cells, and resulted in the slowdown of the tumor progression rate, so as to achieve the effect of survival time prolonging. Compared with the control group, there was no significant increase of side effects in the heart, liver function, diarrhea and others in the experimental group, which indicate that this study is safe and reliable. In patients in the two groups, 7.5 mg/m2 of Endostar were applied during the treatment. Whether the 15 mg/m2 of Endostar should be applied in patients to prolong the mPFS and mOS, higher requirements are proposed for the next step of study. The sample size in this study was small, and the results were not satisfactory. We hope to expand the sample size in the future and make the study more extensive.
Conclusion
The hepatic arterial infusion of Endostar combined with TACE can significantly improve recent clinical efficacy and mPFS in the treatment of medium and advanced HCC. However, improvement on the overall survival of long-term efficacy is not significant.
Footnotes
Acknowledgements
We are particularly grateful to all the people who have given us help on our article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
I confirm that I have read the Editorial Policy pages. This study was conducted with approval from the Ethics Committee of Petroleum Hospital Affiliated to HeBei Medical University. This study was conducted in accordance with the declaration of Helsinki.
Informed consent
Written informed consent was obtained from all participants.
Trial registration
This randomized clinical trial was not registered because retrospective study.
