Abstract
The aim of this study was to investigate the predictive value of peptidyl-prolyl cis-trans isomerase NIMA-interacting 1 (Pin1) with C-reactive protein (CRP) and white blood cell (WBC) count for community-acquired pneumonia (CAP) in infants. A total of 84 hospitalized infants with CAP and 69 healthy infants were included in this study. The clinical manifestations and laboratory assay results of infants were recorded. Serum Pin1 level was estimated by enzyme-linked immunosorbent assay. The median serum Pin1 concentration in infants with CAP was significantly higher than that in controls (1.44 vs. 0.21 ng/mL,
Introduction
Community-acquired pneumonia (CAP) is a major contributor to childhood morbidity and mortality, especially in developing countries. 1 There are approximately 21 million newly diagnosed cases of CAP every year in China. 2 At present, the diagnosis of CAP in infants is mainly based on clinical characteristics and chest radiography. Several studies have reported that white blood cell (WBC), C-reactive protein (CRP), erythrocyte sedimentation rate (ESR), and procalcitonin (PCT) values have contributed to the diagnosis of CAP. Owing to changing etiology, the diagnostic ability of clinical features for CAP in infants may vary. 3 Chest radiography is not recommended as a routine procedure for the diagnosis of CAP in infants because radiographic exposure may cause damage to the human body. 4 The utility of CRP, WBC, and ESR is usually hampered in clinical practice due to their low sensitivities and specificities. 3 Early diagnosis and timely treatment are very important for CAP in infants, especially in infants with hypoimmunity. Therefore, finding biomarkers with high predictive value for CAP in infants would benefit clinical practice.
Peptidyl-prolyl cis-trans isomerase NIMA-interacting 1 (Pin1) is frequently overexpressed and/or activated in human cancers. 5 It can regulate a large subset of key oncogenes and tumor suppressors by catalyzing cis/trans isomerization of specific pSer/Thr-Pro motifs. Therefore, Pin1 plays an important role in diverse cellular processes, including cell proliferation. 6 It has emerged as a common key regulator of oncogenic signaling pathways in multiple types of tumor. 7 Recently, the effect of Pin1 in inflammatory diseases has become a hot research topic. Pin1 can modulate prosurvival and proinflammatory cytokines expression in both eosinophils 8 and T-cells. 9 Moreover, several cytokines expressions, such as IL-4 and TNF-a, are considered Pin1-independent.10,11 It also has been reported that Pin1 plays a pivotal role in chronic inflammatory diseases such as rheumatoid arthritis, 12 asthma, 10 and liver fibrosis. 13 In terms of viral infection, Pin1 also potentially promotes the action of human immunodeficiency virus type-1. 14 Nonetheless, the role of Pin1 in CAP in infants is uncertain. Herein, we investigated the relationship between serum Pin1 levels and CAP in infants to determine whether Pin1 with CRP and WBC could serve as a biomarker for predicting CAP in infants.
Materials and methods
Study subjects
This study was conducted at a tertiary hospital, Fujian Maternity and Child Health Hospital, Affiliated Hospital of Fujian Medical University, from January 2017 to April 2020. The protocols for the study and informed consent were approved by the Biomedical Research Ethics Committee of Fujian Medical University (Approval number: 201624). Written informed consent was obtained from the parents or authorized representatives of the infants before the study. The sample size was calculated by using the equation15,16

where N is the sample size in each of the groups;
The inclusion criteria for infants with CAP were as follows: (1) infants ages from 1 month to 24 months old; (2) infants were hospitalized in the pediatric ward; (3) written informed consent was obtained from the parents or legal guardians of the infants; and (4) infants were diagnosed with CAP. CAP was diagnosed according to the Community-Acquired Pneumonia Guidelines (I, II) of the Chinese Medicine Association.17,18 In brief, the diagnosis criteria were: (1) new fever, cough, and/or breathlessness with sputum production; (2) auscultatory findings of rales; and (3) presentation with consolidation (a dense or fluffy opacity with or without air bronchograms), other infiltrate (linear and patchy alveolar or interstitial densities), or pleural effusion on chest radiography. 19
The inclusion criteria for healthy controls were as follows: (1) the age, weight, and gender of the healthy controls matched with CAP infants; (2) written informed consent was obtained from the parents or legal guardians of the infants; and (3) controls were confirmed by the doctor as healthy.
The exclusion criteria were as follows: (1) infants with congenital anomalies, chromosomal abnormalities, or inborn metabolic disturbance; (2) infants with severe immunodeficiency; (3) infants with a prior hospitalization within 2 weeks of the current diagnosis of pneumonia; (4) infants who were subsequently diagnosed with tuberculosis; and (5) infants in which a chest radiograph or blood sampling was not performed.
The clinical manifestations and results of laboratory examinations of infants were recorded including age, sex, weight, and laboratory data such as complete blood cell count, differential WBC count, and CRP. Blood samples were obtained from all study subjects. Sera were separated immediately and kept at −80°C before analysis. As for infants with CAP, blood was collected before the administration of antibiotics on the first day of hospitalization.
Measurement of serum Pin1
The serum concentration of Pin1 was detected according to the manufacturer’s instruction using the Human Pin1 enzyme-linked immunosorbent assay (ELISA) Kit ordered from Shanghai Lianshuo Biological Technology Co. Ltd. (China). The kit uses the quantitative sandwich enzyme immunoassay technique. Antibodies specific for Pin1 were precoated onto a microplate. This kit has high sensitivity and specificity for measurement of Pin1. No significant cross-reactivity or interference between Pin1 and analogs was observed. The detection range of the kit was between 0.156 and 10 ng/mL. The limit of detection of this kit was 0.054 ng/mL. The intra- and inter-assay coefficients of variance were less than 10% and 12%, respectively.
Measurement of CRP and WBC
The CRP was detected using the whole blood CRP test kit ordered from DiagVita Biological Technology Co. Ltd. (Suzhou, China), according to the manufacturer’s instruction. The direct measurement range of the kit was between1 mg/L and 200 mg/L. Both the intra- and inter-assay coefficients of variance were less than 10%. WBC count was determined using the Sysmex XN-3000 hematology analyzer (Sysmex, IL).
Statistical analysis
All data were analyzed with SPSS version 18.0 (SPSS Inc., Chicago, IL, USA). Continuous variables were expressed as median and interquartile range (IQR), and analyzed by the Mann–Whitney U test. A Chi-square test was used to compare the differences between groups for categorical variables. ROC curves were used to evaluate the diagnostic efficiency of the predictors. The differences in the areas under the ROC curve (AUCs) between the predictors were calculated using the nonparametric approach developed by DeLong et al.
20
The sensitivity and specificity of the predictors for discriminating infants with CAP were analyzed by McNemar test. A two-tailed
Results
Demographics and laboratory parameters of study infants
During the study period, 84 infants with CAP and 69 healthy controls were included. The median age and weight were not significantly different between infants with CAP and controls (4.0 vs. 5.0 months,
Serum Pin1 levels showed a positive correlation with CAP infection in infants
To determine the relationship between serum Pin1 levels and CAP, the serum Pin1 concentrations were measured. Our results showed that the median serum Pin1 concentrations in infants with CAP were significantly higher than those in controls (1.44 vs. 0.21 ng/mL,
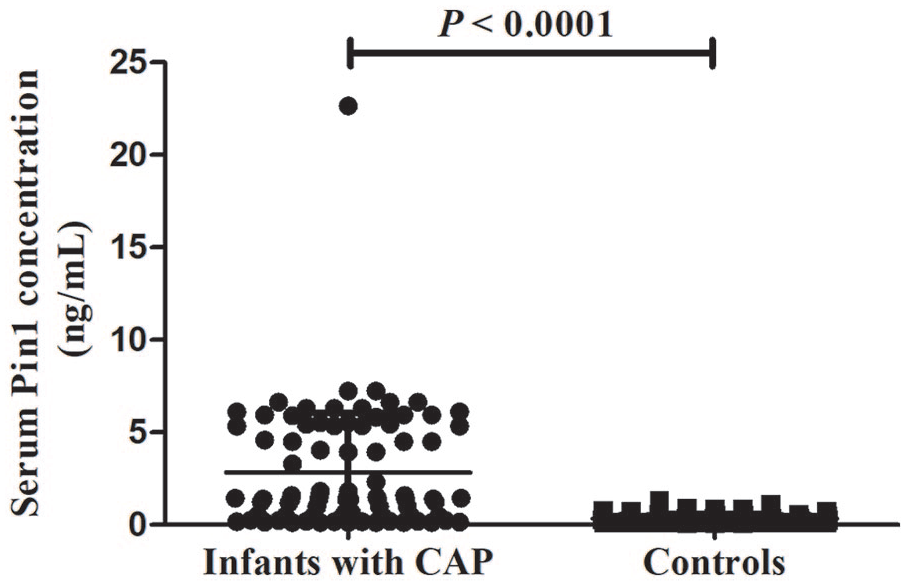
The relationship between serum Pin1 levels and CAP.
Diagnostic efficiency evaluation using ROC curve analysis for Pin1, CRP, and WBC to discriminate infants with CAP
Our results showed that the areas under the ROC curve (AUCs) of Pin1, CRP, or WBC alone were 0.820 (95% CI: 0.751–0.890), 0.763 (95% CI: 0.687–0.839) and 0.802 (95%CI: 0.732–0.873), respectively. The cut-off values of the serum Pin1, CRP, and WBC levels were 0.951 µg/mL, 0.530 mg/L, and 9.34 × 109/L, respectively (Figure 2(a)). The AUCs among Pin1, CRP, or WBC alone showed no significant difference (

Receiver operating characteristic (ROC) curve analysis for Pin1, CRP, and WBC to discriminate infants with CAP. (a) ROC analysis for Pin1, CRP, and WBC alone. (b) ROC analysis for combination of Pin1, CRP, and WBC. (c) Areas under the receiver operating characteristic curves (AUCs) of the predictors. The differences in the AUC among these predictors were calculated by the nonparametric approach developed by DeLong et al. 20
Although there was no significant difference in the AUC among Pin1 + CRP + WBC, Pin1 + WBC or CRP + WBC, the sensitivity of Pin1 + CRP + WBC was 94.0%, which was significantly higher than that of Pin1 + WBC (84.5%) or CRP + WBC (85.7%) together with other parameters (
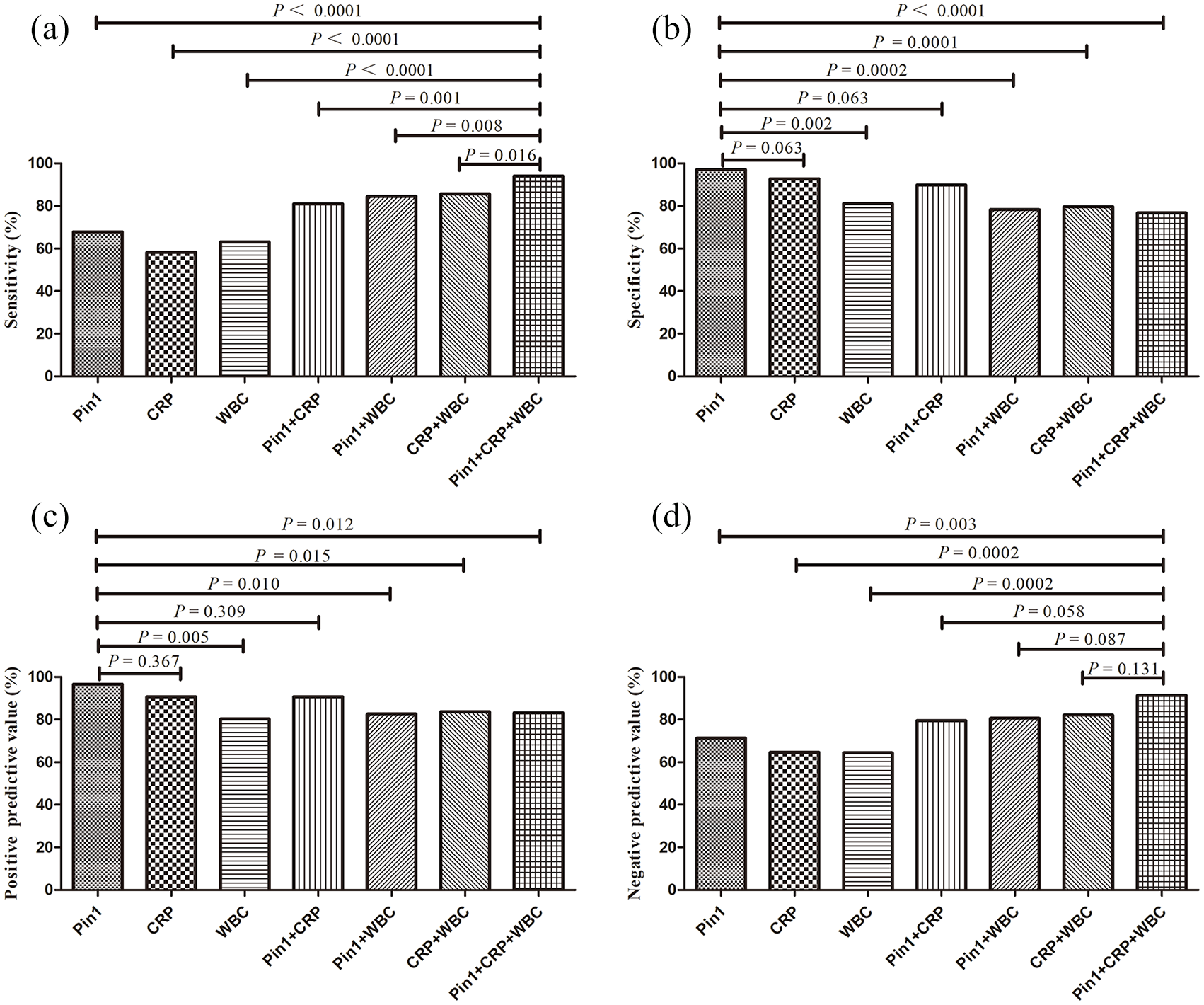
The sensitivities, specificities, and predictive values for Pin1, CRP, and WBC to discriminate infants with CAP. (a) The sensitivities of Pin1, CRP, and WBC alone or their combination. (b) The specificities of Pin1, CRP, and WBC alone or their combination. (c) The positive predictive values of Pin1, CRP, and WBC alone or their combination. (d) The negative predictive values of Pin1, CRP, and WBC alone or their combination.
Interestingly, the positive predictive value of Pin1 for infants with CAP was 96.6%, which was higher than WBC (80.3%), Pin1 + WBC (82.6%), CRP + WBC (83.7%) and Pin1 + CRP + WBC (83.2%), except CRP (90.7%) and Pin1 + CRP (90.7%) (Figure 3(c)). Moreover, the negative predictive value of Pin1 + CRP + WBC was 91.4%, which was higher than Pin1 (71.35), CRP (64.6%) and WBC (64.4%) alone, except Pin1 + CRP (79.5%), Pin1 + WBC (80.6%) and CRP + WBC (82.1%) (Figure 3(d)). Our results suggest that the combination of Pin1, CRP, and WBC had a high sensitivity and negative predictive value, while Pin1 alone had a high positive predictive value and specificity.
Discussion
In this study, we first evaluated the relationship between serum Peptidyl-prolyl cis-trans isomerase NIMA-interacting 1 (Pin1) levels and infants with CAP and found that serum Pin1 was highly expressed in infants with CAP. Our results also showed that the area under the receiver operating characteristic curve (AUC) of the combination of Pin1, CRP, and WBC count was higher than that of Pin1, CRP, WBC alone, or the combination of Pin1 and CRP. Moreover, the sensitivity of Pin1, CRP, and WBC in combination (94.0%) was also higher than that of either Pin1 and WBC or CRP and WBC. Interestingly, Pin1 alone had a high specificity (97.0%) and excellent positive predictive value (96.6%) for infants with CAP. Therefore, Pin1 alone or in combination with both CRP and WBC may be promising novel predictors for infants with CAP.
Pin1 is a cis-trans peptidyl-prolyl isomerase that modulates cytokine expression.10,11 It plays an important role in inflammation, an important pathophysiological process of CAP.21,22 Our study found that high serum Pin1 level was positively correlated with infants with CAP. Studies have found that interleukin-33 (IL-33) is abundantly expressed in inflammation, 23 and Pin1 is a crucial factor in IL-33-induced lung inflammation. 22 Therefore, the elevated serum Pin1 in infants with CAP in our study may also be due to the high expression of IL-33.
To our knowledge, some protein expression may relate to age or body weight, especially in neonates and infants. In agreement with this theory, our results showed that the median serum Pin1 concentration in healthy infants was significantly lower than the study 24 on healthy controls from 34 to 60 years old (0.21 vs. 25.76 ng/mL), which indicates that the expression of serum Pin1 might increase with age and weight.
CRP and WBC are important inflammatory markers usually used in the early detection of acute infections in infants. 25 CRP, an acute-phase protein, is rapidly increased in hepatocytes under the control of cytokines, in particular IL6. It is usually used as a useful predictor for infection, or even death, in infants with a probable serious bacterial infection.26,27 Our results showed that either CRP or WBC could discriminate infants with CAP, but the sensitivities of these two markers for the diagnosis of CAP were poor, although CRP showed higher specificity. The high specificity for CRP may be due to the presence of upregulated serum Pin1. 22 Similar to our findings, Berg et al. 3 also reported that a single elevated WBC or CRP concentration was not sensitive enough to diagnose CAP. Interestingly, the predictive potentials were good, with sensitivity markedly increased after adding Pin1 to CRP and WBC, which suggest that a combination of Pin1, CRP, and WBC may be a good predictor for infants with CAP. Our results were also concordant with the study of Berg et al., 3 who described that the performance of nonspecific inflammatory markers CRP and WBC in the diagnosis of pneumonia was increased when combining other related predictors.
This study had several limitations. First, all infants were from Fujian, Southeast China, which may not be representative of the general population. Second, Pin1 is ubiquitously expressed in human cells, and serum Pin1 level may be affected by the changes in the number of blood cells and tissue damage such as lung injury. Therefore, the expression of Pin1 in the lung tissue may be a more accurate predictor of pneumonia. Unfortunately, only the serum Pin1 could be detected in our study. Owing to a restricted serum volume, the dynamic measurement of Pin1 was also lacking in our study. In addition, the detecting efficiency, standard concentrations, and detecting ranges of Pin1 ELISA kits vary significantly, 28 which affects the comparability of results between different laboratories. Hence, further studies are needed to confirm this finding in a larger cohort of patients and in more extensive regions.
Conclusion
Serum Pin 1 was highly expressed in infants with CAP and can singly or in combination with CRP and WBC represent promising novel predictors for infants with CAP.
Footnotes
Acknowledgements
The authors thank Ms. Jennifer Kilbourn for language editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval for this study was obtained from the Biomedical Research Ethics Committee of Fujian Medical University (Approval number: 201624).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Natural Science Foundation of Fujian Province (no. 2018J01848) and Foundation of School of Medical Technology and Engineering, Fujian Medical University for Youth Teachers (no. 2017XY003).
Informed consent
Written informed consent was obtained from legally authorized representatives before the study.
