Abstract
Legionella pneumophila is a rarely diagnosed microorganism in Serbia. It causes legionellosis, usually a mild respiratory infection. However, in some cases it can be severe and even life threatening. In June 2020, during the COVID-19 pandemic, a patient with symptoms of the aforesaid infection, namely severe pneumonia and acute respiratory distress syndrome, was admitted to the hospital. The multiplex polymerase chain reaction (PCR) test (The BioFire FilmArray Pneumonia Panel plus) detected the presence of L. pneumophila in the patient’s bronchial secretions. The specific culture for the detection of that organism, however, remained sterile. The patient’s paired sera had been sent for serology and the results in both of them came back positive for Legionella spp. 1–6, while the assays specific for each one of the 10 serogroups detected more than a fourfold increase of antibody titers in an uncommon serogroup 2 only. The patient was treated with moxifloxacin; he recovered well and was discharged after 26 days of hospitalization. Having being diagnosed with the L. pneumophila infection correctly through the multiplex PCR test, the patient was given the right therapy with moxifloxacin. The serologic assays corroborated this result and revealed the uncommon group 2, thus confirming the necessity of carrying out all the tests available to attain the exact diagnosis of legionellosis.
Introduction
The microorganism Legionella pneumophila lives freely in both fresh water and soil, but also in artificial water systems such as those for hot and cold water, cooling towers, fountains or whirlpool spas. It causes legionellosis in humans who contract the disease by inhaling the contaminated aerosols. 1
Legionellosis can present as either mild or severe respiratory infection. In the case of non-immunosupressed patients its occurrence is estimated to 5.4% of community acquired pneumonias 2 while among pneumonias which require hospitalization the numbers are 0.4 cases per 1,00, 000 adults per year. 3 The case fatality rate varies from 9.3% in Europe, 4 8.3% in United States 5 to 5.9% in Japan. 6 In Serbia, however, the occurrence of L. pneumophila has been reported in scarce cases,7,8 with successful outcome in spite of being described as a severe disease. Nevertheless, a systematic research of its occurrence has not yet been carried out. Currently, there are 48 species comprising 70 distinct serogroups in the genus Legionella. 9
A urinary antigen test is most widely used to detect legionellosis when suspected. According to the European Centre for Disease Prevention and Control’s report (ECDC), in 2018 91% of infections caused by Legionella spp. were diagnosed using this method while 10% of cases were confirmed by the culture. Serologic assays, either representing single high titer or fourfold titer rise, were reported in 1–2%. 10
Case presentation
In June 2020, a 47-year-old man, an economist by profession, was admitted to the Intensive Care Unit (ICU) at the Clinic for Infectious and Tropical Diseases as an emergency case. The disease began 3 days prior to his admission to the hospital with headaches and nausea, dry cough, difficulties with breathing and general weakness. On admission, he was somnolent, febrile 39.5°C, cyanotic, dehydrated, and with high respiratory rate (40/min). Oxygen saturation was 68%. Auscultation disclosed late inspiratory crackles and high-pitched wheezing. Radiography and a CT revealed dense confluent infiltrates in all parts of the right lung, and lower and middle parts of the left lung (Figure 1). Biochemical and blood gas analyses were considerably out of range (Table 1).
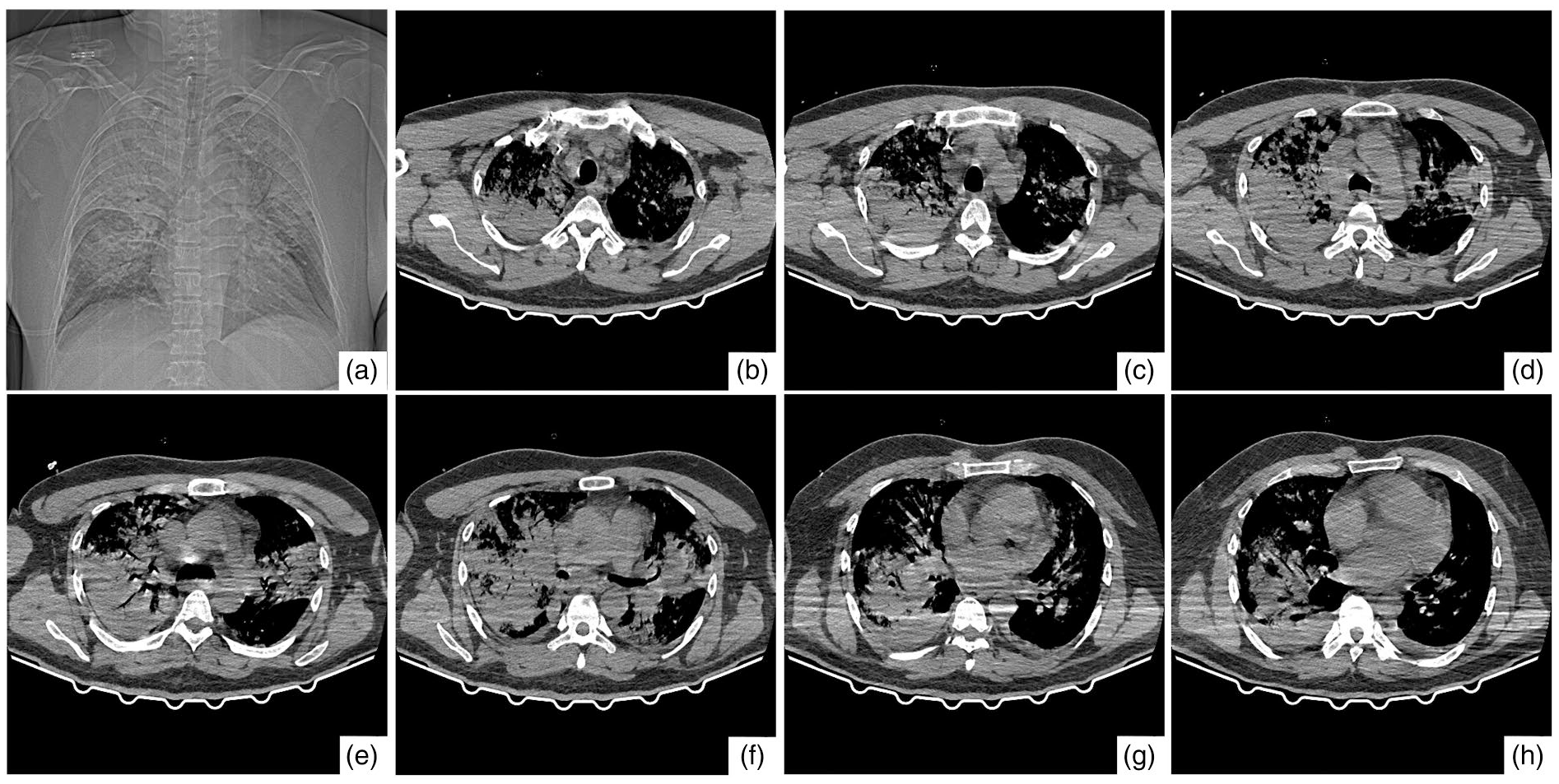
Chest computed tomography findings taken at the time of hospitalization. (a) Coronal plane image. (b)–(h) Axial plane images. Massive consolidation on both sides of lungs, predominantly on the right one, mostly in the upper parts and parachilary, as well as a ground glass opacity in the lower lobe of the right.
Results of the patient’s laboratory analysis.

Oxygenotherapy of high flow was administered immediately, but desaturation and tachypnea persisted. The relationship pO2/FIO2 was 127.8 indicating a moderate acute respiratory distress syndrome. 11 The patient was put on mechanical ventilation, which raised oxygen saturation to 98%. The initial antibiotic therapy in duration of the first 7 days, consisting of cefepime 3 g intravenously (IV) every 8 h, vancomycin 1 g IV every 12 h and fluconazole 400 mg IV once a day, was combined with the therapy for COVID-19 infection. Antibiotics were chosen to cover most of Gram-positive and Gram-negative pathogens, as well as fungi, since the cause of the life-threatening infection was unknown. The selection of antibiotics was based on knowledge about high resistance rates in invasive pathogens isolated in the same institution several years before. 12
They were administered for the first 7 days. Therapy for COVID-19 was consisted of a twice-daily dose of hydroxychloroquine 600 mg on the first day and 400 mg daily via nasogastric tube, and methylprednisolone 60 mg IV daily. This therapy was in accordance with the national treatment protocol at that moment. It was administered for 3 days, until this infection was ruled out, based on a PCR test to SARS-CoV-2 and antibody tests to this pathogen which were reported negative. After 5 days of incubation, standard blood cultures (BACTEC 9240, Becton, Dickinson and Company, Sparks, MD, USA) also came back negative, including the conventional sputum culture. On the third day of hospitalization, a newly introduced Multiplex PCR assay (The BioFire FilmArray Pneumonia Panel plus, Bio Mérieux) showed the presence of L. pneumophila in the tracheal aspiration fluid. During a run, the FilmArray system performs a nested multiplex PCR by two steps: the first step is a multiplex PCR. In that assay, different target DNA sequences belonging to different microorganisms can be simultaneously amplified by using multiple primer pairs in a reaction mixture under a single set of reaction conditions (annealing temperature, buffer concentration). 13 The product of the first amplification reaction is used as the template for the second PCR, which is primed by oligonucleotides that are placed internal to the first primer pair. The use of oligonucleotide pairs located on three spots of the test contributes to the higher sensitivity and specificity of the PCR. Finally, more sensitivity is attained through the analysis of the DNA melting curves, specific for target genes of each microorganism this test detects.
Upon receiving the test results, moxifloxacin 400 mg IV every 24 h was immediately introduced and was administered for the next 21 days. A urinary antigen test to L. pneumophila serotype 1 (BinaxNOW Legionella (Alere, Scarborough, ME, USA) still remained negative as well as the culture of tracheal aspiration, although a cultivation was attempted with selective media for Legionella spp: buffered charcoal yeast extract (BCYE) agar (Oxoid, Thermo Fisher Diagnostics Limited, Cheshire, UK), incubated at 35°C, under 5% of CO2 for 10 days.
After 13 days of mechanical ventilation, the patient’s state improved and he was successfully extubated. He continued to use the controlled oxygen therapy until the 20th day. In the third week of hospitalization the second serum sample was taken for serology testing after the first sample had been provided in the first week. The patient was discharged from hospital after 26 days. The inflammation parameters were normalized. Serology assays, performed at the French Reference Center for Legionella, Laboratoire de Biologie Medicale Multi Sites du Chu de Lyon, France, showed elevated antibody titers to L. pneumophila 1–6, through a screening method, in both serum samples: 4.68 in the first and 5.38 in the second sample (ELISA, Kit Captia Trinity Biotech). This means that they were positive for L. pneumophila serogroups 1–6. Specific antibody titers to each one of the 10 L. pneumophila serogroups (1–10) were determined by an Indirect immunofluorescence test (IIF, In House method). In IIF specific antigens bind antibodies targeted and a secondary antibody, which carries fluorophore, recognizes fixed antibodies (Supplementary Material). 14 A demonstration of a fourfold or a greater increase in antibody titers to at least 128 during convalescence provides serological evidence of a recent infection with the Legionella spp. Specific titers are the result of IgG, IgM, and IgA classes, individually or in various combinations. The sensitivity of the test is 79% and its specificity 99%. 15 In the two patient’s sera the IIF indicated an eightfold increase of antibody titers to serogroup 2: 512 in the first and 4096 in the second sample, while the antibody titers to other serogroups were negative.
Discussion
According to the current Case Definition for National Legionellosis Surveillance by the Centers for Disease Control and Prevention (CDC), 16 or the Public Health of England, 17 or the New Zealand’s Ministry of Health 18 the present case fulfilled all the criteria, clinical and laboratory, to be classified as a confirmed case. The clinical findings include pneumonia, diagnosed clinically and radiographically, while laboratory implies the detection of any Legionella species from lower respiratory secretions by a validated nucleic acid amplification test. A failure to obtain bacterial growth could be explained by an estimation that the sensitivities of the sputum cultures are usually less than 50% when serological findings are used as the standard. 19 In a study from Korea, for example, Legionella spp. was detected in 2 cases out of 59 endotracheal aspirations. However, the culture remained negative in both of them. 20 The BioFire FilmArray Pneumonia Panel plus, by contrast, has a much higher sensitivity compared to the culture, some 97.4%, and the specificity of 98.6%. 21 A good performance of the test is based on a two-step process: after the first step (multiplex PCR), follows the second step of the nested PCR, run in triplicate, to detect conserved regions of the targets of each microbial species in the test. 22
Although the serogroup 1 of L. pneumophila causes 79.3%–84% of infections, 23 the present disease triggered a fairly uncommon L. pneumophila serogroup 2. A fourfold or a greater rise in antibody titers to specific species or serogroups of Legionella other than L. pneumophila serogroup 1 is another criterium for diagnosing legionellosis. That serogroup was reported in 0.3% infections caused by L. pneumophila in Europe in 2018. 9 That is the reason why a urinary antigen test could not detect Legionella spp. in this case, since it detects only serogroup 1.
Conclusion
The present case is a confirmation of the reliability and utility of The BioFire FilmArray Pneumonia Panel plus assay, which played a major role in saving the life of a relatively young and healthy adult. Serology corroborated this result and even revealed the rare serogroup 2, thus confirming the necessity of applying all available tests to attain the exact and accurate diagnosis of legionellosis.
Learning points
Legionella pneumophila causes acute respiratory distress syndrome and symptoms similar to those of COVID-19
Multiplex PCR is a highly sensitive and accurate test to detect L. pneumophila
Serologic assay is a valuable tool in diagnosing legionellosis
The therapy with moxifloxacin proved to be effective in treating a L. pneumophila severe infection
Supplemental Material
sj-docx-1-eji-10.1177_1721727X221095035 – Supplemental material for Severe pneumonia caused by Legionella pneumophila detected by a multiplex polymerase chain reaction assay and confirmed by serology
Supplemental material, sj-docx-1-eji-10.1177_1721727X221095035 for Severe pneumonia caused by Legionella pneumophila detected by a multiplex polymerase chain reaction assay and confirmed by serology by Milica Jovanović, Nikola Mitrović, Letitia Beraud, Nikola Trboljevac, Branko Milošević, Aleksandra Radovanović Spurnić, Snežana Jovanović and Dragana Marić in European Journal of Inflammation
Footnotes
Acknowledgements
We express our gratitude to Lara Payne Halstrom, from the European Centre for Disease Prevention and Control (ECDC) and Søren Uldum, Statens Serum Institut, Copenhagen, for putting us in contact with Laetitia Beraud, France, who performed the serologic tests and critically reviewed the text.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from the Ethic board of Clinical Center of Serbia. (Approval number 251/3).
Informed consent
Written informed consent was obtained from the individual included in this study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
