Abstract
This study sought to investigate the role of miR-206 in polymyositis/dermatomyositis (PM/DM) development. Transwell and 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium inner salt (MTS) assay were performed to investigate cell migration and proliferation. Real-time polymerase chain reaction (PCR) analysis was used to determine the expression of miR-206, interleukin-17A (IL-17A), IL-17 receptor A (IL-17RA), and regenerating islet-derived protein 3-alpha (REG3A). Significantly, miR-206 mimics decreased macrophage migration and proliferation, while miR-206 inhibitors exhibited the opposite effects. Indeed, elevating IL-17RA levels resulted in increased REG3A expression, which was inhibited by IL-17RA siRNA. Besides, miR-206 mimics decreased IL-17A and REG3A expressions, but miR-206 inhibitors showed opposite effects. Moreover, miR-206 expression in PM/DM patients was significantly reduced compared with the healthy controls, while IL-17A and REG3A expressions substantially increased among PM/DM patients. These findings suggested that downregulation of miR-206 increased the migration and proliferation of macrophages via IL-17A/REG3A signaling pathway, which could promote the inflammatory infiltration in PM/DM.
Introduction
Polymyositis/dermatomyositis (PM/DM), a systemic inflammatory disorder, can affect organs such as skeletal muscles and skin, which is characterized by symmetric proximal muscle weakness and elevated serum muscle enzymes. 1 In PM, the perivascular infiltration in muscle biopsies is composed predominantly of CD8+ T cells and macrophages, while inflammatory infiltration in DM mainly consists of CD4+ T cells, B cells, and macrophages. 2 Interleukin-17 (IL-17), a cytokine produced by T-helper cells, is involved in PM/DM pathogenesis via IL-17/intercellular adhesion molecular 1 pathway, and can culminate in PM/DM deterioration. Regenerating islet-derived protein 3-alpha (REG3A) is an antimicrobial protein which controls bacteria proliferation.3,4 Furthermore, existing literature supports that the induction of REG3A by interleukin-17A (IL-17A) via activation of keratinocyte-encoded IL-17 receptor A (IL-17RA) could facilitate proliferation of skin keratinocyte. 5 However, the role of REG3A in PM/DM pathogenesis is unclear. In this regard, this study aims to investigate the role of IL-17A and REG3A in PM/DM. Indeed, microRNAs (miRNAs) are increasingly becoming popular among researchers due to their usefulness as indicators for the diagnosis and prognosis of diseases.
Notably, miR-206 (specific to muscles) might participate in PM/DM pathogenesis owing to its involvement in cell proliferation and differentiation.6,7 Indeed, miR-206 is vital for generation and growth of muscle tissues with its expression restricted mainly to skeletal muscle and osteoblasts. Speculatively, the expression of miR-206 could be correlated with IL-17A secretion due to the close proximity of the former to the Il17a/f gene locus. Besides, available data suggest that miR-206 is expressed in diabetics with Krüppel-like factor 4 (KLF4) as one of its multiple targets, albeit negative correlation with Th17 cell percentages. Nevertheless, the known targets and potential interactions of miR-206 with IL-17A and REG3A in PM/DM have not been completely clarified. The present study proposed that miR-206 could regulate the migration and proliferation ability of macrophages via IL-17/REG3A pathway.
Materials and methods
Patients and samples
Twenty patients were diagnosed with PM or DM according to the criteria of Bohan and Peter at the Third People’s Hospital of Wuxi from March to October 2016. 8 The inclusion body myositis and non-autoimmune myopathies (such as muscular dystrophies) were excluded. All the participants were enrolled in this study based on the protocols approved by the Ethics Committee of the Third People’s Hospital of Wuxi, while written informed consent was provided for the use of their tissues and data in this study. The clinical characteristics of the participants are presented in Table 1. Macrophages are commonly observed to infiltrate into the muscle fibers of PM/DM patients. Therefore, the influence of miR-206 on the migration of macrophages was investigated through the isolation of the macrophages from the peripheral blood mononuclear cells (PBMCs) of healthy donors prior to Transwell assaying.
Clinical data of patients and HCs.

HCs: health controls; M: male; DM: dermatomyositis; ANA: antinuclear antibody; Ro52, Sjoegren syndrome type A antigen; Ro60, Sjoegren syndrome antigen A2; F: female; PM: polymyositis.
Cell isolation and culture conditions
Human macrophages were prepared from PBMCs obtained from healthy donors as described elsewhere.
9
Briefly, the cells were plated at 4 × 106/well in 24-well plates (Costar Corning, Cambridge, MA, USA) for 1 h in Dulbecco’s modified Eagle’s medium (DME) (Gibco, Gaithersburg, MD, USA), and non-adherent cells were rinsed off with Hanks’ balanced salt solution (HBSS) (Gibco). The macrophages were then cultured for 3 days in
Cell transfection
The miR-206 mimics, miR-206 inhibitors, NC siRNA, IL-17A siRNA, IL-17RA siRNA, and REG3A siRNA were bought from GenePharma (Shanghai, China) and transfected into cells using Lipofectamine™ 2000 (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instruction. In brief, cells in
In vitro migration assay
An in vitro migration assay was performed using a 24-well Transwell culture plate (Costar Corning) with a polycarbonate filter (pore size: 8 µm) based on the instructions of the manufacturer. Briefly, macrophages were first treated with miR-206 mimics, miR-206 inhibitors, or siRNA for 24 h, and then 2 × 105 cells were suspended in
Cell proliferation assay
In accordance with standard protocol, cell proliferation was assessed using the Cell Titer 96® Aqueous One Solution Reagent (Promega, Madison, WI, USA) comprising a tetrazolium compound (3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium inner salt) (MTS) and a reagent of electron coupling (phenazine ethosulfate). Macrophages were seeded into 96-well plates at a density of 1 × 104cells/well prior to treatment with miR-206 mimics, miR-206 inhibitors, or siRNA for 12, 24, and 48 h. After incubation, 20 µL of Cell Titer 96® Aqueous One Solution Reagent was pipetted into each well that contained samples in 100 μL of culture medium. Then, the sample was incubated at 37°C in a humidified atmosphere containing 5% CO2. After 1 to 3 h incubation period, the absorbance was recorded at 490 nm using a universal microplate reader (ELx 800TM, BioTek, Winooski, VT, USA).
Real-time polymerase chain reaction
Total RNA from cultured macrophages and human tissues (muscle biopsies) was prepared via Trizol (Invitrogen) concordant with the specifications of the manufacturer. Next, RNA (1 µg) was converted to cDNA using SuperScript™ II Reverse Transcriptase (Invitrogen). The resultant cDNA was subjected to quantitative polymerase chain reaction (PCR) with SYBR® Green Real-Time PCR Master Mixes (Invitrogen) using StepOnePlus™ Real-Time PCR System (Applied Biosystems, Foster City, CA, USA). Each measurement was performed in triplicate, and the relative gene expression was normalized with the expression of the glyceraldehyde-3-phosphate dehydrogenase (GAPDH) mRNA levels. Data were analyzed via the 2-ΔCtΔCt method. The primers of GAPDH, IL-17A, IL-17 RA, and REG3A were as follows: GAPDH forward primer: GGAGCGAGATCCCTCCAAAAT, reverse primer: GGCTGTTGTCATACTTCTCATGG; IL-17A forward primer: TCCCACGAAATCCAGGATGC, reverse primer: GGATGTTCAGGTTGACCATCAC; IL-17 RA forward primer: GCTTCACCCTGTGGAACGAAT, reverse primer: TATGTGGTGCATGTGCTCAAA; REG3A forward primer: AGCTACTCATACGTCTGGATTGG, reverse primer: CACCTCAGAAATGCTGTGCTT.
To analyze miR-206, the real-time PCR was performed with a TaqMan PCR kit (Invitrogen) on the account of the manufacturer’s instructions. Then, relative expression of miR-206 was normalized to U6. Data were analyzed with the 2-ΔCtΔCt method. The primers of U6 and miR-206 were as indicated below: U6 forward primer: CTCGCTTCGGCAGCACA, reverse primer: AACGCTTCACGAATTTGCGT; miR-206 forward primer: GAGTGCTGGAATGTAAGGAAG, reverse primer: GCAGGGTCCGAGGTATTC.
Statistical analysis
Data are presented as mean ± standard deviation (SD). Two-tailed
Results
Increased IL-17A promotes regenerating family member 3 alpha (REG3A) expression in macrophages depending on IL-17RA
Macrophages were treated with different concentrations of IL-17A (R&D Systems, Minneapolis, MN, USA), and the result showed that REG3A expression was elevated at increased IL-17A concentration with its strongest expression achieved at 1.5 µg/mL of IL-17A (Figure 1(a)). Besides, when macrophages were treated with siRNA targeting IL-17RA, the expression of REG3A induced by IL-17A was inhibited (Figure 1(b)). Altogether, these findings suggest that increased expression of IL-17A promoted REG3A expression in human macrophages through IL-17RA.
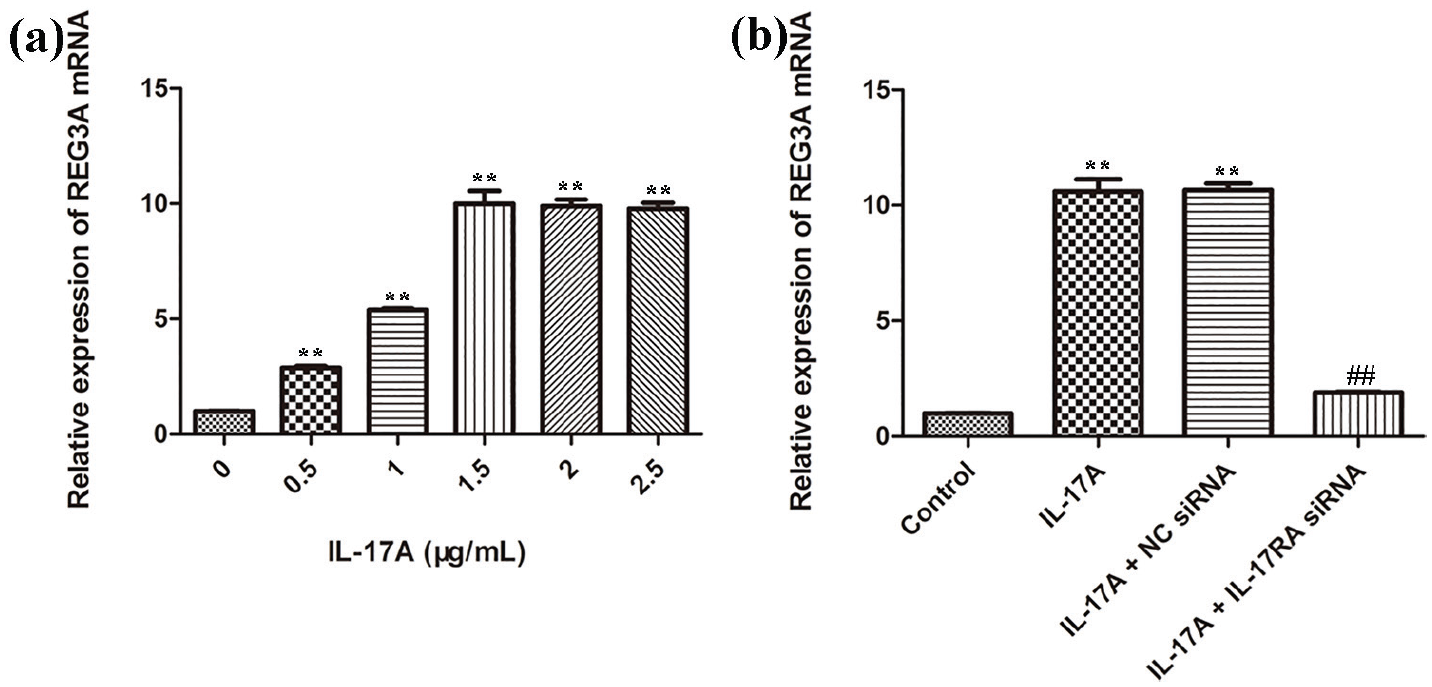
IL-17 promotes REG3A expression in macrophages depending on IL-17RA. (a) The mRNA expression levels of REG3A in macrophages treated with different concentrations of IL-17A. (b) The mRNA expression levels of REG3A in macrophages treated with siRNAs. **
MiR-206-induced inhibition of macrophage migration and proliferation ability is involved in the IL-17A/REG3A signaling pathway
Prior to the study, it was postulated whether miR-206 could regulate IL-17A/REG3A pathway, and thus inhibited the migration and proliferation ability of macrophages. To test this hypothesis, some experiments such as separate treatment of cells with miR-206 mimics and inhibitors as well as performance of real-time PCR analysis were performed. Imperatively, Figure 2(a)–(c) showed that the cells treated with miR-206 mimics demonstrated decreased expression levels of IL-17A and REG3A compared with the control group. Moreover, the cells treated with miR-206 inhibitors displayed increased IL-17A and REG3A expression levels compared with that of the control group. However, neither miR-206 mimics nor inhibitors had effect on the IL-17 RA expression (

MiR-206 regulates macrophage migration and proliferation through the IL-17/REG3A signaling pathway. (a–c) Cells were pretreated with mimics, inhibitors, and siRNAs for 24 h, and the mRNA expression levels of IL-17A, IL-17RA, and REG3A were analyzed. (d, e) Cells were pretreated with mimics, inhibitors, and siRNAs for 24 h, and migration assay was performed. Absolute number of migrated cells was the average value of cells in three fields under 200× view. (f–h) Cells were pretreated with mimics, inhibitors, and siRNAs for 12, 24, and 48 h, and proliferation ability was analyzed with MTS in the indicated time point. **
In addition, the MTS results confirmed that inhibition of miR-206 by miR-206 inhibitors caused an increase in cell proliferation, which was counteracted by IL-17A siRNA, IL-17RA siRNA, or REG3A siRNA transfection (Figure 2(e)–(h)). Overall, these findings indicated that IL-17A/ REG3A signaling pathway might act as a functional mediator in miR-206-induced inhibition of macrophage migration and proliferation.
Decreased expression of miR-206 and increased expression of IL-17A and REG3A in PM/DM patients
In order to affirm our results in PM/DM patient, the expressions of miR-206, IL-17A, IL-17RA, and REG3A in 20 healthy controls and 20 PM/DM patients were assessed separately. Real-time PCR analysis was performed, and the result showed that miR-206 expression in PM/DM patients was significantly decreased to a lower level comparable to the healthy controls (

Decreased expression of miR-206 and increased expression of IL-17 and REG3A in PM/DM patients: (a–d) The expression levels of miR-206, IL-17, IL-17RA, and REG3A in the healthy controls and PM/DM patients. **
Altogether, these results indicated that downregulation of miR-206 might increase the migration and proliferation ability of macrophages via IL-17A/REG3A signaling pathways, thereby promoting the inflammatory infiltration in PM/DM patients.
Discussion
The two major and distinct subsets of idiopathic inflammatory myopathies (IIMs) are PM and DM. 10 Immunohistochemical studies of muscle biopsies obtained from PM/DM patients showed infiltration of muscle fibers by macrophages. Clinical manifestations of PM/DM vary from individuals; thus, treatment should be personalized amid based on depth understanding of the pathogenesis. Although various reports have established the crucial role of inflammatory reactions in the pathogenesis of PM/DM, little is known about the underlying mechanisms. Therefore, further study of the exact mechanisms of PM/DM is imperative for the individualized therapy. In Figures 1 and 3, the migrated cell number and cell proliferation were significantly decreased after adding miR-206 mimic. This could probably support the fact that the expression of miR-206 might suppress cell migration and proliferation in PM/DM as reported in other disease models. 11 Therefore, inhibition of miR-206 by its inhibitor might have resulted in significant increased migrated cell numbers and proliferation in the inhibitor group compared with the controls. Likewise, targeting the inhibition of miR-206 via its inhibitor might have not only reversed the effect of miR-206 but also induced increased responses. Notwithstanding, further works are required for detailed understanding of the role of miR-206 in the cell migration and proliferation in PM/DM. Besides, the mRNA expressions of IL-17 mRNA and REG3A were significantly increased in miR-206 inhibitor group compared with the control group (Figure 3). This finding could be attributable to a possible negative correlation between miR-206 inhibition and mRNA expressions of IL-17 and REG3A. 12 Nevertheless, the actual role of miR-206 inhibition in mRNA expressions of IL-17 and REG3A will be comprehensively investigated in future. Herein, it was found that downregulation of miR-206 might increase the migration and proliferation ability of macrophages via IL-17/REG3A signaling pathway. Notably, different subsets of T cells may also be involved in the pathogenesis of PM and DM. Therefore, the role of miR-206 and IL-17/REG3A signaling pathway in T cells in PM/DM should be investigated further.
Footnotes
Author contributions
X.H. helped in the proposal of the investigation and implementation plan; Z.L. and F.G. carried out the acquisition, analysis, and interpretation of data; and J.Z. and Q.Y. contributed to the drafting or revision of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
