Abstract
Objective:
Senile pruritus is a specific type of itching that occurs in elderly persons. Previously, we assessed antagonism of the nonselective ligand-gated cation channel transient receptor potential vanilloid 1 (TRPV1; capsaicin receptor or vanilloid receptor 1) and attenuation of atopic dermatitis by the non-steroidal TRPV1 antagonist PAC-14028 in clinical studies. The findings led us to postulate that PAC-14028 may also reduce itching in elderly people by antagonizing the TRPV1 pathway. In this study, we evaluated whether PAC-14028 modulates inflammatory markers present in senile pruritus.
Materials and Methods:
HaCaT, RAW264.7, and differentiated THP-1 cells under itching-inducing conditions were treated with zymosan or IL-17A and variety of experimental approaches such as molecular modeling simulations, site-directed mutagenesis, overexpression strategies, confocal microscopy, mRNA analyses, and immunoprecipitation/Western blotting analyses were assessed to check changes in inflammatory markers and explore the underlying mechanisms of PAC-14028 activity.
Results:
In the bioinformatic analyses, skin inflammation markers were found to be closely related to TRPV1, and the MAPK and NF-κB pathways were upregulated when TRPV1 was activated. In HaCaT cells, PAC-14028 was found to directly bind to TRPV1, inhibiting inflammatory cytokine gene expression and downstream MAPK and NF-κB signaling under various skin inflammatory conditions.
Conclusions:
By combining the results of multiple assays, we were able to elucidate the molecular mechanism of PAC-14028 to TRPV1. Taken together, the findings indicate that PAC-14028 as a potential therapeutic agent for elderly people with pruritus.
Introduction
Pruritus (itchy skin) is a common condition that can occur in anyone and may negatively impact physical or psychological well being.1,2 When symptoms of pruritus occur in elderly people, it is called senile pruritus. The causes of senile pruritus are not known, but attempts to alleviate the symptoms have involved treatment with emollients, corticosteroids, topical calcineurin inhibitors, and/or H1-antihistamines.3 –6 The history and presentation of senile pruritus may vary, but a common suspected cause is contact with allergens and/or infectious materials derived from bacteria, yeast, and fungi that elicit chronic inflammation by inducing pro-inflammatory cytokines.
The inflammatory cytokine IL-17A is associated with pruritus in elderly people and psoriasis.7,8 Previous studies reported that IL-17A increases endothelin-1 (ET-1) expression, which can induce pruritus.9,10 Zymosan, a β-glucan with repeating glucose units isolated from Saccharomyces cerevisiae, promotes the inflammatory signaling pathway mediated by toll-like receptor 2 (TLR2), a representative receptor recognizing Gram (+) bacteria and yeast. 11 Zymosan has been associated with various dermatological conditions and is known to exacerbate pruritic symptoms.12 –14 Additionally, skin infections caused by Saccharomyces cerevisiae can lead to psoriasis and pruritus in elderly people, including symptoms of itching.15,16 The non-selective cation channel transient receptor potential vanilloid 1 (TRPV1) is primarily expressed in sensory neurons of the dorsal root ganglion (DRG), but is also present in keratinocytes17,18 and is associated with inflammatory diseases of the skin.19 –21 The function of TRPV1 is detection and regulation of body temperature, as well as transduction of noxious stimuli such as exposure to heat and capsaicin, an active component in chili peppers.22 –24 Activation of TRPV1 leads to an influx of calcium and sodium ions, triggering neuronal excitation and the sensation of pain and itching. 25 Additionally, TRPV1 is involved in various inflammatory processes and has been implicated in numerous dermatological conditions characterized by chronic itching.26,27 Therefore, there have been many attempts to find a TRPV1 antagonist that may attenuate skin pruritus.19,28 The nonsteroidal compound PAC-14028 (Asivatrep, C21H22F5N3O3S) (Figure 1(a)) is a newly discovered potent and selective TRPV1 antagonist.29,30 Preclinical studies in mice and rats showed that PAC-14028 had noncarcinogenic activity and suppressed atopic dermatitis (AD)-like skin diseases. 31 Chan Woo Park et al. presented a paper on the relieving effects of PAC-14028 on atopic dermatitis patients.32,33

PAC-14028 inhibited TRPV1 activity by binding directly to TRPV1. (a) Scheme of molecular binding between TRPV1 and PAC-14028. (b−d) The predicted TRPV1 binding sites. (e) The protein sequence alignments of TRPV1 derived from different species were determined using Uniprot. (f) The expression of TRPV1 after mutagenesis of binding sites. (g, h) The CETSA assay after mutagenesis of TRPV1 for binding with PAC-14028.
In light of the above-noted finding, we aimed to elucidate the molecular mechanisms underlying the antipruritic effects of PAC-14028 and explore its therapeutic potential in the treatment of senile pruritus. We hypothesized that PAC-14028 would effectively reduce itch responses induced by zymosan and IL-17A, offering a novel approach to pruritus in the elderly. To clearly demonstrate this hypothesis and explore detained mechanism, molecular modeling simulations, site-directed mutagenesis, overexpression strategy, confocal microscopy, mRNA analysis, and immunoprecipitation/Western blotting analyses were employed in HaCaT, RAW264.7, and THP-1 cells under itching-like conditions.
Methods
Materials
PAC-14028 (Figure 1(a), Purity > 95%) was provided by AmorePacific Co. (Yongin, Korea). Hyclone (Logan, UT, USA) was the source of the penicillin-streptomycin (PC/SM), Dulbecco’s modified Eagle’s medium (DMEM), Roswell Park Memorial Institute 1640 Medium (RPMI), trypsin-EDTA, and phosphate buffered saline (PBS). Ethanol was purchased from Pure Chemical Company (Gyeonggi-do, Korea); fetal bovine serum (FBS) and Opti-mem™ reduced-serum minimal essential medium were obtained from Gibco (Grand Island, NY, USA). Molecular Research Center Inc. (Cincinnati, A, USA) was the source of the TRIzol® reagent. The HaCaT human keratinocytic, RAW264.7 mouse macrophagic, THP-1 human promonocytic, and HEK293 human embryonic cell lines were purchased from the American Type Culture Collection (ATCC, Rockville, MD, USA). Dimethyl sulfoxide, Phorbal-12-myristate-13-acetate (PMA), methanol, 2-propanol, 1-bromo-3-chloropropane, zymosan A from Saccharomyces cerevisiae and interleukin 17A recombinant protein were purchased from Sigma-Aldrich (St. Louis, MO, USA). Fluo-4 AM was purchased from BD Pharmigen™ (Franklin Lake, NJ, USA). Macrogen (Seoul, Korea) provided the primers used in the experiments. Anti-β-Actin, -p-p50, and -p-JNK antibodies were obtained from Santa Cruz Biotechnology (Dallas, TX, USA); anti-p50, -p65, -p-p65, -c-Fos, -p-c-Fos, -c-Jun, -p-c-Jun, -IκBα, -p-IκBα, -IKKα/β, -p-IKKα/β, -p-p38, p38, -p-MEK3/6, -MEK3/6, -p-TAK1, and -TAK1 antibodies were purchased from Cell Signaling Technology (Beverly, MA, USA). Polyclonal anti-TRPV1 antibody was obtained from Invitrogen (Waltham, MA, USA). The pCMV-TRPV1 plasmid was obtained from Sino Biological Inc. (Beijing, China).
Cell culture and cell viability
The HaCaT and HEK293T cells were cultured using DMEM with 10% FBS and 1% PC/SM in a 5% CO2 incubator, and RAW 264.7 cells and THP-1 cells were subcultured with RPMI supplemented with 10% FBS and 1% PC/SM. THP-1 cells were treated with 150 nM PMA for 24 h to induce their differentiation into macrophage-like state. The cells were cultured to 80% confluency, plated in 96-well plates at 5 × 104 cells/well, and incubated overnight. After incubation with 0−22.4 µM PAC-14028 for 24 h, MTT solution (5 mg/mL) was added to each well to generate formazan. 34 An MTT stop solution of 10% SDS solution with 0.1 N HCl was added after 4 h incubation to stop the reaction between the cells and MTT solution. 35 The absorbance was detected at 570 nm, and cytotoxicity was calculated by dividing the results of the non-treated and PAC-14028-treated groups.
mRNA isolation and real-time PCR
The HaCaT, RAW 264.7, and PMA-treated THP-1 cells were seeded into 6-well plates at 4 × 105 cells/well and incubated overnight in a 5% CO2 incubator. The cells were then incubated with 5.6−22.4 µM PAC-14028 for 1 h, followed by incubation with 20 nM IL-17A or 100 µg/mL zymosan for 2 h. Total mRNA was isolated using TRIzol® Reagent, 2-propanol, and 75% ethanol in DEPC water. Complementary DNA (cDNA) was synthesized using a kit containing Oligo dT primer, 5X Reaction Buffer, 10 nM dNTPs, reverse transcriptase, and RNase inhibitor from Thermo Fisher Scientific (Waltham, MA, USA). The cDNA mixture was combined with primers and SYBR™ Green for real-time PCR on a CFX 96 real-time PCR machine (Bio-Rad, Hercules, CA, USA). 36 Relative fluorescence units (RFU) were assigned to the SYBR™ Green fluorescence. The primer sequences used are listed in Table 1.
The primers used in the real-time PCR assays.

Calcium2+ influx assay
The HaCaT cells were plated and treated as described above. After removing the supernatant, the cells were fixed in a 4% formaldehyde solution for 10 min. The fixation solution was removed, and cells were washed three times with PBS and stained for 10 min with DAPI and Fluo-4 AM in PBS to identify the nuclei and calcium2+ ions, respectively. Images were acquired using a fluoresence microscope and analyzed using Opti-view software (Jacksonvile, FL, USA).
Transfection
The HaCaT and HEK293T cells were plated and cultured in DMEM with 5% FBS without antibiotics. Opti-MEM™ containing the transfection reagent and pCMV-TRPV1 plasmid was added to each well. After 24 h of incubation, the medium was replaced with DMEM containing 10% FBS and 1% antibiotics, and cells were incubated with PAC-14028 and IL-17A as previously described.
Luciferase reporter assay
The HEK293T cells were plated in 24 well-plates with the concentration of 1 × 105 cells per well. The NF-κB Luc-plasmid or AP-1 Luc plasmid was co-transfected with plasmids encoding TRIF or Myd88 with transfection reagent PEI in Opti-MEM™. β-galactosidase plasmid which could indicate the transfection efficiency was added to plasmid mixture.37,38 The mixture was treated to each well, the media was replaced with DMEM supplemented with 5% FBS and 1% antibiotics including 0–22.4 µM of PAC-14028 for 24 h. The cell lysis was examined with the lysis buffer in −70-degree deep freezer at least for 3 h. The absorbance was detected at 405 nm with β-galactosidase and cell lysis, and luminescence was detected with D-luciferin (Sigma Chemical Co.) and cell lysis. The luciferase mediated activity was normalized to absorbance of β-galactosidase activity. All the data was reorganized from GraphPad Prism 9.5.0 software.
Western blotting
The cells were collected in PBS after being treated with all compounds and were lysed using a lysis buffer containing 2% NP-40, 150 mM NaCl, 2 mM EDTA, 20 mM Tris-HCl pH 7.5, and 50 µM β-glycerol phosphate containing protease inhibitors (2 µg/mL of aprotinin, pepstatin, and leupeptin). 39 Intracellular protein was obtained from whole-cell lysates by centrifuging at 12,000 rpm for 5 min. Bio-Rad Bradford assay (Hercules, CA, USA) was used to measure protein concentration. Then, Western blotting was performed as described previously.40,41
STRING and GO analyses
The STRING network analysis was performed to identify potential protein-protein interactions (PPIs) between the NF-κB and MAPK signaling proteins and TRPV1. We added the target proteins into the STRING database to generate a network diagram, wherein nodes represented proteins and edges represented interactions. Gene ontology (GO) analysis was performed to categorize the proteins involved in the STRING network according to their associated biological processes, molecular functions, and cellular components. Each protein was annotated with relevant GO terms, and statistical enrichment analyses were conducted to determine which GO terms were significantly overrepresented when compared to a reference set.
Molecular modeling
Molecular modeling simulations were conducted to predict the binding structures of TRPV1 (Protein Data Bank [PDB] ID: 3j5p) and PAC-14028 using UCSF (University of California, San Francisco) Chimera X with the Autodock vina program. 42 All results were visualized and reorganized with Pymol (from Schrodinger.com).
Mutagenesis
The pCMV-TRPV1 plasmid was modified based on the molecular docking analysis. The TRPV1 sequence was downloaded from the National Center for Biotechnology Information (NCBI), and the protein sequence was aligned using the Protein Data Bank (PDB). Primer sequences used in the experiment are listed in Table 2.
The primers used in the TRPV1 mutagenesis procedures.

Cellular thermal shift assay (CETSA)
The HEK293T cells were transfected with wild-type and mutated pCMV-TRPV1 plasmids. Cells were incubated with PAC-14028 or DMSO for 24 h after the incubation. Cell pellets were harvested in 900 mL of PBS, divided into 1.5 × 105 cells/100 mL, heated from 45 to 75°C in 3 min, and incubated at 25°C for 3 min. 43 All samples were frozen in a −70°C deep freezer for 3 h and thawed at room temperature. Cell lysates were centrifuged at 13,000 rpm for 30 min, and supernatants were collected for Western blotting.
Statistical analyses
All data are presented as mean ± SD of three replicates of separate experiments. A one-way analysis of variance (ANOVA) was used to determine the statistical differences between the control and PAC-14028-treated groups. Significance was noted at p < 0.05, *, #; p < 0.01, **, ##; and p < 0.001, ***. All graphs were prepared using Prism GraphPad 9.5.
Results
PAC-14028 directly inhibits binding of TRPV1
PAC-14028 is known to inhibit TRPV1 agonist activity. Hence, the molecular docking of TRPV1 and PAC-14028 was examined to identify the binding sites. The predicted binding sites were visualized in Pymol (Figure 1(a–d)). The TRPV1 protein sequence alignment was analyzed using the PDB. Protein modification was performed based on the results of the alignment: Ser 404, Pro 407, Tyr 554, and Gly 700 were selected (Figure 1(e)) for modification. The mutagenesis of these binding sites was based on information from the PDB. We investigated whether TRPV1 expression could be changed by site-specific mutagenesis and found that expression was consistent even after mutagenesis (Figure 1(f)). Moreover, the pattern showed more stable expression during direct binding of PAC-14028 in the CETSA assay (Figure 1(g, h)). Taken together, these results suggest that PAC-14028 directly binds to TRPV1 sites by inhibiting TRPV1 activity without altering TRPV1 expression.
TRPV1 modulates the immune response via the AP-1 and NF-κB pathways
We performed STRING analyses to identify the relationship between TRPV1 and the AP-1 and NF-κB signaling proteins. The results indicated TRPV1 was associated with many proinflammatory cytokines, matrix metalloproteinases, and growth factors. The most common biological process was “regulation” (GO:0065007), the most enriched molecular function was “binding” (GO:0005488), and the most enriched cellular component was “anatomical entity” (GO:0110165; Figure 2(a–d)). In particular, the cluster that included TRPV1 showed “response to stimulus” (GO:0050896) as the most common biological process and “binding” (GO:0005488) as the most enriched molecular function. “Intracellular anatomical structure” (GO: 0005622) was the most enriched cellular component in this cluster. In addition, we employed the luciferase reporter assay to confirm that PAC-14028 attenuated NF-κB and AP-1 transcriptional activities. The luciferase activity, which meditated TRIF, Myd88, and AP-1, was reduced in a concentration-dependent manner, while p-p50 and p-p65 protein expression was increased by overexpression of TRPV1 and reduced by incubation with PAC-14028 (Figure 2(i–l)). Thus, TRPV1 activates NF-κB and MAPK signaling, and PAC-14028 attenuated the activation of this signaling.

TRPV1 modulates the inflammatory response, including NF-κB and MAPK pathway signaling. (a) The STRING analysis results indicated protein-protein interactions (PPIs) between AP-1, NF-κB, and TRPV1. (b−d) Gene ontology (GO) analysis. (e) PPI analysis of the cluster including TRPV1. (f−h) GO analysis of the cluster involving TRPV1. (i−k) The luciferase reporter assay was examined for transcription activities. (l) Protein expression was confirmed in a TRPV1-overexpression condition.
PAC-14028 reduced the inflammatory response by inhibiting TRPV1 activation
Capsaicin, a reported agonist of TRPV1 through promotion of Ca2+ ion flux, activates TRPV1.44 –46 We investigated the emolliating effects of PAC-14028 on skin inflammation by promoting TRPV1 activation. We used capsaicin to activate TRPV1 in HaCaT cells. We added PAC-14028 to the cell cultures in a range of concentrations that did not produce cytotoxicity (Figure 3(a)). The production of cytokines related to pruritus or itching was significantly upregulated by TRPV1 activation and downregulated after incubation with PAC-14028 (Figure 3(b–d)). We also assessed whether phosphorylation of NF-κB-related proteins was linked to upregulation of inflammation in cells incubated with PAC-14028. We found that TRPV1 activation increased phosphorylation of NF-κB-pathway proteins, and this pattern of NF-κB signaling factor phosphorylation was reduced in cells incubated with PAC-14028 (Figure 3(e)). Hence, we concluded that TRPV1 modulates the inflammatory response, and incubation with PAC-14028 reduced this activation by inhibiting TRPV1 activation. Moreover, in the Ca2+ influx assay using the Fluo-4 AM fluorescence reagent, the intercellular calcium concentration was restored by incubating the cells with PAC-14028 against Capsaicin treatment (Figure 3(f)). These findings indicate that PAC-14028 induces resolution of the inflammatory response by inhibiting cytokine expression, phosphorylation of NF-κB signaling, and cellular calcium ion influx.
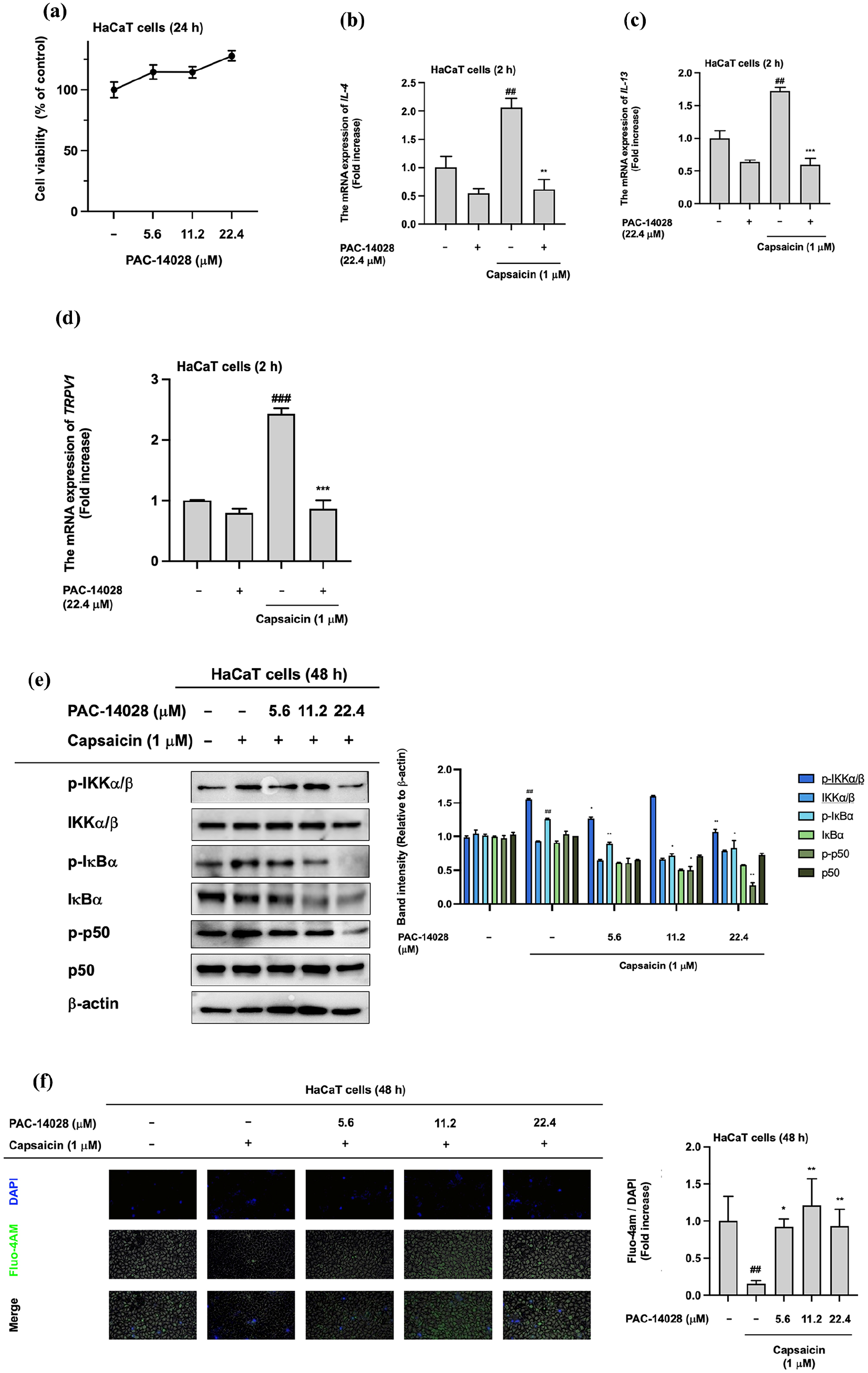
PAC-14028 attenuated TRPV1-mediated inflammation. (a) Cytotoxicity was assessed using the MTT assay. The experimental concentration range was 0−22.4 µM in HaCaT cells. (b−d) The mRNA expression of pruritus-mediated cytokine and TRPV1 in a capsaicin-treated condition. (e) The protein levels of NF-κB signaling in capsaicin- and PAC-14028-treated cells. (f) The Fluo-4 AM assay was used to detect Ca2+ ion flux induced by TRPV1 activation.
Attenuating the skin irradiation role of PAC-14028
We assessed the effects of IL-17A and zymosan, both inducers of senile pruritus, in RAW 264.7 cells, PMA-treated THP-1 cells, and HaCaT cells. While IL-17A has been reported to induce skin inflammation in conditions such as atopic dermatitis and psoriasis, zymosan has been found to activate macrophages by inducing reactive oxygen species (ROS) and proinflammatory cytokines. We found that zymosan-induced nitric oxide (NO) production was reduced by incubation with PAC-14028 at a concentration that does not affect cell viability (Figure 4(a, b)). Furthermore, cytokines involved in inflammatory responses to zymosan were downregulated by PAC-14028 at the mRNA levels (Figure 4(c, d)). In the case of THP-1 cells, the levels of IL-4, 6, TRPV1, and TNF-α were increased by zymosan treatment and PAC-14028 treatment alleviated these responses (Figure 4(e–h)). In HaCaT cells incubated with IL-17A, upregulation of TRPV1, IL-4, and IL-13 mRNA expression was reduced by incubation with PAC-14028 (Figure 4(i–l)). We examined the Western blotting results for NF-κB and MAPK signaling protein expression since p38 activation is related to TRPV1. 47 The phosphorylation of NF-κB (p-p65) and AP-1 (p-c-Jun)/MAPK (p-TAK1/p-MKK3/6/p-p38) pathway factors was increased by IL-17A and attenuated by PAC-14028 (Figure 4(m–o)). Consequently, we concluded that PAC-14028 inhibits the activation of NF-κB and AP-1/MAPK pathway proteins induced by TRPV1 activation.

PAC-14028 reduces the inflammatory molecule level in senile pruritus condition. (a) The experimental concentration was determined based on cell viability in RAW 264.7 and THP-1 cells. The MTT assay was performed to assess the toxicity of PAC-14028. (b) Measurement of NO production induced by the inflammatory response with zymosan treatment. (c, d) The mRNA expression of pro-inflammatory cytokines in zymosan-treated RAW 264.7 cells. (e–h) The mRNA levels of inflammatory cytokines in zymosan/PMA-treated THP-1 cells. (i–l) The mRNA expression of TRPV1 and inflammatory cytokines in IL-17A-treated HaCaT cells. (m–o) Levels of proteins related to NF-κB and AP-1 pathways in the presence of PAC-14028 and IL-17A.
Discussion
Inflammatory skin conditions such as AD, pruritus, and psoriasis have various etiologies, such as pathogen infection, dysbiosis of the skin microbiome, dysfunction of the skin barrier, and dysregulation of the immune system.48,49 Recent studies have suggested a relationship between TRPV1 and skin inflammation.50,51 Interestingly, activation of TRPV1 is known to be involved in the chronic inflammatory response that activates the NF-κB pathway. Thus, there are efforts to attenuate skin inflammation by deactivating TRPV1, implying that TRPV1 antagonists, such as PAC-14028 (Asivatrep, C21H22F5N3O3S), may be effective treatments for inflammatory skin conditions. Clinical studies have shown PAC-14028 to be the most effective drug to attenuate the symptoms of atopic dermatitis. 33 To further elucidate the mechanism of PAC-14028, we analyzed the effects on signaling cascades involved in skin inflammation.
Previous studies reported that many types of inflammatory cytokine signaling pathways are activated in inflammatory skin conditions.52,53 It has been suggested that the keratinocyte-intrinsic pathway has a vital role in immune balance in the skin. The AP-1 pathway proteins involving the Jun and Fos dimers are key regulators of inflammation. Patients with psoriasis showed upregulation of AP-1-mediated genes, 54 and the TNF pathway is a well-known inflammatory signaling pathway, with one study reporting that the epithelial-cell-specific IκB-knockout mouse model resulted in TNF-mediated overexpression of IL-24 in inflammatory skin lesions. 55 Based on those findings, we examined the expression of AP-1 and NF-κB after TRPV1 activation.
First, our results demonstrated that PAC-14028 effectively binds to TRPV1 at predicted sites such as Ser 404, Pro 407, Tyr 554, and Glu 700 without degradation of TRPV1 expression (Figure 1). Mutagenesis of these sites and CETSA assays demonstrated that PAC-14028 binding stabilized TRPV1, leading to its inhibition. The direct interaction with TRPV1 is important for its anti-pruritic effects, as TRPV1 is a key mediator in the transmission of itch signals. Our STRING analysis results demonstrated significant interactions between TRPV1 and various immune-response proteins, including components of the AP-1 and NF-κB pathways. Our luciferase reporter assay confirmed that PAC-14028 attenuates NF-κB and AP-1 transcriptional activities in a concentration-dependent manner. Overexpression of TRPV1 increased phosphorylation of NF-κB, which was subsequently reduced by PAC-14028 treatment. These results indicate that TRPV1 plays a substantial role in modulating inflammatory responses through the AP-1 and NF-κB pathways, and PAC-14028 effectively mitigates this activation (Figure 2). Meanwhile, capsaicin-induced TRPV1 activation, known to promote the Ca2+ ion flux and inflammation, was significantly reduced by PAC-14028 treatment. Our data also demonstrated that PAC-14028 can effectively reduce expression of cytokines related to pruritus and itching in keratinocyte cell lines, as well as phosphorylation levels of NF-κB pathway proteins. The Fluo-4 AM assays also indicated that PAC-14028 restores intracellular calcium concentrations disrupted by TRPV1 activation. These findings support the conclusion that PAC-14028 can reduce the inflammatory response by inhibiting TRPV1 activation, and thus downregulating cytokine production, NF-κB phosphorylation, and calcium ion influx (Figure 3).
In this study, we incubated the RAW264.7 mouse macrophage cell line and THP-1 human monocyte cell line with zymosan and IL-17A due to their known roles in macrophage activation and skin inflammation. Incubation with PAC-14028 significantly reduced both zymosan-induced NO production and the mRNA expression of pro-inflammatory cytokines (Figure 4(c, d)). Furthermore, PAC-14028 inhibited the upregulation of cytokine mRNA expression (Figure 4(i–l)) and reduced the phosphorylation of NF-κB and AP-1 pathway proteins (Figure 4(m–o)) at higher concentration (22.4 µM). Through this study, we could explore the anti-pruritic effects of PAC-14028 in RAW 264.7 cells, PMA-treated THP-1 cells or HaCaT cells incubated with zymosan and IL-17A.
Our study has some limitations that were not explored. First, we could not confirm the cytotoxicity of long-term PAC-14028 treatment in the skin. Further studies could investigate the long-term effects of PAC-14028 on skin tissue through in vivo experiments, potentially using animal models or human skin equivalents. Second, since we focused on specific pathways managed by NF-κB and AP-1/MAPK, other pathways such as calcium signaling or lipid metabolism pathways triggered by TRPV1 activation or inhibition should be considered in the following studies to provide a more comprehensive understanding of PAC-14028’s effects. Lastly, we demonstrated that the mutation of a major amino acid could affect the binding between TRPV1 and PAC-14028; however, the mechanism by which the interaction between TRPV1 and PAC-14028 can induce structural and functional changes remains unclear. To understand molecular dynamics between the receptor and the compound, advanced structural biology techniques, like cryo-electron microscopy or fine molecular dynamic modeling could be employed to elucidate precise structural changes in binding site of TRPV1.
Our findings provide significant insights into the potential mechanisms by which PAC-14028 alleviates itch and suggest its therapeutic value for treating senile pruritus as shown in Figure 5. Taken together, these findings suggest PAC-14028 as a promising therapeutic option for managing senile pruritus and other chronic itch conditions mediated by TRPV1 activation, such as atopic dermatitis, rosacea psoriasis, and prurigo nodularis.56 –59 Combining two or more drugs can mitigate toxicity and side effects associated with high doses of a single drug; for example, the combination of halobetasol propionate and tazarotene demonstrates treatment of dermatological conditions. 60 Therefore, combining a TRPV1 antagonist with PAC-14028 may enhance the therapeutic effects for conditions where TRPV1 is activated. What kind of TRPV1-targeting drugs can synergistically enhance the therapeutic activity of PAC-14028 will be further studied in the following study.

The scheme of the attenuating mechanism of PAC-14028 on senile pruritus.
Conclusions
In summary, PAC-14028 directly binds to TRPV1 and inhibits its activity. In particular, TRPV1 activation induces upregulation of the NF-κB and MAPK pathways, which are key activators of the inflammation response. Incubation of cell lines with PAC-14028 downregulated TRPV1 protein activation and expression of cytokines. We conclude that PAC-14028 may have the potential to attenuate senile pruritus symptoms by inhibiting TRPV1 activation and reducing the consequential upregulation of signaling pathways for NF-κB (p65) and AP-1 (c-Jun) activation. Since PAC-14028 displayed much stronger inhibitory activity on NF-κB pathway than on AP-1 pathway (Figure 5), anti-senile pruritus activity of PAC-14028 might be largely covered by its NF-κB inhibitory action. This seems to be due to a possibility that PAC-14028-binding sites in TRPV1 might be regions to associate with NF-κB activating proteins. To clarify this, hence, further experiments will be followed in the future. Interestingly, 5.6 µM of PAC-14028 treatment increased the phosphorylation levels of p85/PI3K, IKKα/β, and c-Fos without altering their total form levels. This phenomenon was also not seen in other proteins and functional assays, implying that the outcomes of this were marginal and non-specific, not affecting total pharmacological activity of this compound. Therefore, it is considered that a direct binding of this compound to the proteins may only increase the autophosphorylation of these enzymes. Since it is unexplainable yet for this phenomenon with current data, we will try to explain it in the future by using molecular modeling work.
Footnotes
Acknowledgements
We thank Mr. Yoonho James Cho (Michigan State University, East Lansing, USA) for English-editing a draft of this manuscript.
Abbreviations
TRPV1, Transient Receptor Potential Vanilloid 1;
PC/SM, Penicillin-Streptomycin;
DMEM, Dulbecco’s Modified Eagle’s Medium;
RPMI, Roswell Park Memorial Institute 1640 Medium;
PBS, Phosphate Buffered Saline;
FBS, Fetal Bovine Serum;
HaCaT, Human Keratinocyte Cell Line;
HEK293T, Human Embryo Kidney Cell Line;
THP-1, Human Monocyte Cell Line;
RFU, Relative Fluorescence Units;
PPI, Protein—Protein Interaction;
GO, Gene Ontology;
NCBI, National Center for Biotechnology Information;
PDB, Protein Data Bank;
ROS, Reactive Oxygen Species;
NO, Nitric Oxide;
AD, Atopic Dermatitis;
RAW 264.7 Cells, Mouse Macrophage Cell Line;
ANOVA, Analysis of Variance
Author contributions
Ji Hye Yoon: writing – original draft, visualization, methodology, investigation, formal analysis, and data curation. Byoung Young Woo: writing – review & editing, methodology, investigation, formal analyses, and data curation. Mi Yeon Kim: writing – review & editing, visualization, formal analyses, and conceptualization. Jae Youl Cho: writing – review & editing, visualization, supervision, funding acquisition, and conceptualization. All authors have read and approved the present manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by grants provided by the Basic Science Research Program (2017R1A6A1A03015642) through the National Research Foundation of Korea (NRF) funded by the Ministry of Education, and by the Korea Basic Science Institute (National Research Facilities and Equipment Center) funded by the Ministry of Education, Korea (grant # 2020R1A6C101A191).
Ethics approval
Not applicable.
Informed consent
Not applicable.
Trial registration
Not applicable.
Data availability
Data will be made available on request.
