Abstract
T-helper (Th) cells play a critical role in inflammatory bowel disease (IBD), especially the two new types: Th17 and Th22. But whether they are protective or pathogenic in the gut is still controversial. Unlike them, regulatory T (Treg) cells have undoubtedly suppressive function and can maintain immune homeostasis. Our current aims were to examine the change of Treg/Th17 balance, Th22 proportion, and the expression of Th17-related and/or Th22-related cytokines in inflammatory bowel disease. In this study, 3% dextran sulphate sodium (DSS) was administered orally to C57BL/6 mice to induce colitis. The morbidity was evaluated by daily body weight, colon length, disease activity index, and histology score of colonic lesions. Th17, Th22, and Treg cells were measured using flow cytometry. IL-17A, IL-17F, and IL-22 were quantified using enzyme linked immunosorbent assay (ELISA) kits. DSS administration induced severe clinical manifestations, such as body weight loss, colon shortening, relatively high disease activity index, and histological damage in colitis mice. Both CD4+IL-17A+IL-22− cells and CD4+IL-22+IL-17A− cells were significantly increased, which associated with the increasing expression of IL-17A, IL-17F, and IL-22. On the contrary, CD4+CD25+Foxp3+ cells reduced. In brief, our findings clearly show that: the drift balance of Th17/Treg is apparently in DSS-induced colitis. The increasing concentration of IL-17A, IL-17F, and IL-22, as well as the increasing Th17 and Th22 cells count, suggesting Th17 and Th22 are involved in the pathological process. Further studies are warranted to develop drugs, which are effective for colitis by targeting the Th17/Treg and Th22 pathways.
Introduction
Inflammatory bowel disease (IBD) is chronic remittent or progressive inflammatory conditions related to the mucosal immune system response to antigenic stimulation from the human intestinal microbiome on a background of genetic susceptibility.1,2 The two most common forms are ulcerative colitis (UC) and Crohn’s disease (CD). Despite hundreds of studies, the etiology of IBD still remains ambiguous.
Animal models of IBD have been central to enhance the understanding of human disease and have made significant contributions to the development of powerful new therapies and drugs.3,4 There are more than 50 different models of IBD used for studies Dextran sodium sulfate (DSS) induced colitis is one of the most widely used models of IBD historically, because it has similarities to human UC in etiology, pathology, pathogenesis, and therapeutic response.5,6 DSS administration triggers intestinal inflammation by binding to medium-chain-length fatty acids present in the mouse colon, inducing disruption of colonic epithelial barrier. 7 The ensuing tissue damage allows exposure of innate immune cells to commensal bacteria accompanied by a robust immune response to eliminate infiltrating pathogens and promote tissue healing. 7
IBD was regarded as a kind of disease due to Th1/Th2 abnormal immunoregulation for a long time. T-helper cell (Th) 17, a new CD4+ T cell subset, has transformed our understanding of the IBD pathogenetic basis of an increasing number of chronic immune-mediated diseases.8,9. The Th17 cells express cytokines interleukin (IL)-17A (commonly denoted IL-17), IL-17F, IL-21, IL-22, and IL-26; however, not all Th17 cells secrete all these cytokines, probably reflecting the heterogeneity of this Th cell subset. 10 On one hand, Th17 cells have major effects on host protection against a wide spectrum of pathogens, especially at mucosal surfaces,11,12 on the other hand, sustained activation of Th17 cells can also be potent mediators of colitis as well. Interestingly, regulatory T cells (Treg) have been found a fine balance with Th17 cells regulating by the concentration of transforming growth factor beta (TGFβ). 12
Th22 cells, as the major source of IL-22, represent a T cell subset uniquely competent to regulate epidermal responses in inflammatory diseases. 13 Unlike Th17, the cell is the second major source of IL-22. Th22 cells do not secrete IL-17. 14 IL-22 belongs to the IL-10 family of cytokines and binds to a heterodimer composed of IL10 receptor (IL-10R) β chain and IL-22R. Despite a similar molecular structure and shared receptors, the bioavailability of IL-22 is also regulated by a soluble IL-22 binding protein (IL-22BP) that acts as a natural antagonist, and IL-22 signaling acts independently of IL-10, also triggers STAT3 and MAPK pathways.13–15 Lauren A. Zenewicz et al. reported that colon epithelial cells are a target for IL-22 activity. 16 They concluded that IL-10Rβ was ubiquitously expressed in the liver, spleen, skin, kidney, lung, colon, and small intestine; however, IL-22R was highly expressed in the colon and small intestine and to a lesser extent in the other tissues. 16
Being interested in the balance of Th17/Treg in IBD, and the curious relationship between Th-22 and pathogenesis of IBD, we determined the change of Treg/Th17 balance, Th22 proportion, and the expression of Th17-related and Th22-related cytokines in DSS-induced colitis model.
Materials and methods
Mice and DSS-induced colitis model
C57BL/6 female mice (weight range, 20–22 g) were obtained from the Vital River Laboratories. All of the animal protocols were reviewed and agreed upon the Ethics Committee of Animal Experiments of Lanzhou University. Mice received one cycle (5 days) of drinking-water containing 3% dextran sulfate salt (DSS) (3600–5000 KDa, MP Biomedicals), beginning on day 1 of the experiment. 17 The normal control mouse received tap water only. Body weights, stool consistency, and occult or gross bleeding were measured every 24 h. The colon and spleen were separated immediately after the last weight check.
Clinical assessments
The disease activity index scoring system involved a combination of various features: changes in weight, stool consistency, and occult or gross bleeding. The disease activity index was between 0 and 12 for each mouse. The disease activity index component was scored according to the following criteria: 18 weight loss: 0, none; 1, 1–5%; 2, 5–10%; 3, 10–20%; 4, >20%. Stool consistency: 0, normal; 2, loose stools; 4, diarrhea. Occult or gross bleeding: 0, normal; 2, hemoccult; 4, gross bleeding.
Histopathological analysis and histological scoring
A specimen from the distal third of the colon was fixed overnight with 4% phosphate-buffered (pH 7.2) paraformaldehyde and embedded in paraffin. Sections were cut at 5 μm thickness in a longitudinal fashion and stained with hematoxylin and eosin (H&E). 19 Three sections per slide were scored on a blind basis and histological analysis was performed on a blind basis as standard protocols. Specifically, colonic histology was scored as follows: 20 Inflammation: 0, none; 1, slight; 2, moderate; 3, severe. Extent: 0, none; 1, mucosa; 2, mucosa and submucosa; 3, transmural. Regeneration: 4, no tissue repair; 3, surface epithelium not intact; 2, regeneration with crypt depletion; 1, almost complete regeneration; 0, complete regeneration or normal tissue. Crypt damage: 0, none; 1, basal one-third damaged; 2, basal two-thirds damaged; 3, only surface epithelium intact; 4, entire crypt and epithelium lost. Percent involvement: 1, 1–25%; 2, 26–50%; 3, 51–75%; 4, 76–100%.
Isolation of cells and flow cytometry
Briefly, spleen was removed and carefully cleaned. Splenic lymphocyte cells were prepared from the spleen by forcing the organs through a 100 μm nylon mesh into Lymphocyte Separation Medium (Mouse) according to the manufacturer’s direction (dakewe). The following antibodies were used in flow cytometry analysis: PE anti-mouse CD4 (clone GK1.5), PE/Cy5 anti-mouse CD4 (clone RM4-5), FITC anti-mouse CD25 (clone 3C7), FITC anti-mouse IL-17A (clone TC11-18H10.1), PE anti-mouse FOXP3 (clone MF-14), all from Biolegend; and anti-human/mouse IL-22 APC (clone IL22JOP) from eBioscience. Cells were stimulated for 4 h with PMA (50 ng/mL, Sigma), ionomycin (750 ng/mL, Calbiochem), and BFA (10 ng/mL, Biolegend). Cells were fixed with fixation buffer (Biolegend) and permeabilized with permeabilization buffer (eBioscience) and incubated with antibodies (13). Cells were analyzed using FACSCalibur (BD Biosciences), and data were illustrated by CellQuest-Pro software (BD).
Cytokines quantification in serum
Mouse IL-22 (Dakewe), IL-17A (Dakewe), and IL-17F (Shyuanye) enzyme linked immunosorbent assay (ELISA) kits were used according to the manufacturers’ instructions.
Statistical analysis
Data values were expressed as mean values with their standard errors for each group. Statistical comparisons were performed using one-way ANOVA, as appropriate, with
Results
Weight loss in DSS-induced colitis
After receiving one cycle of drinking-water containing 3% DSS, mice tended to lose weight compared to control animals (study 2–5), as showed in Figure 1. We found that DSS-induced mice lost significantly body weight compared with drinking-water mice at day 5 (14.91 vs. 1.15%,

Changes in body weight after dextran sulphate sodium (DSS) administration in C67BL/6 mice. Body weight change was measured by dividing body weight on a specified day by body weight at day 0 (starting body weight) and expressed in percentage. N = 9, ***
Colon shortening in DSS-induced colitis
In this study, the total colon length becomes shortened in mice treated with DSS. As showed in Figure 2, the DSS administration significantly caused colon shortening compared to control mice at day 5 (10.50 vs. 11.45 cm,
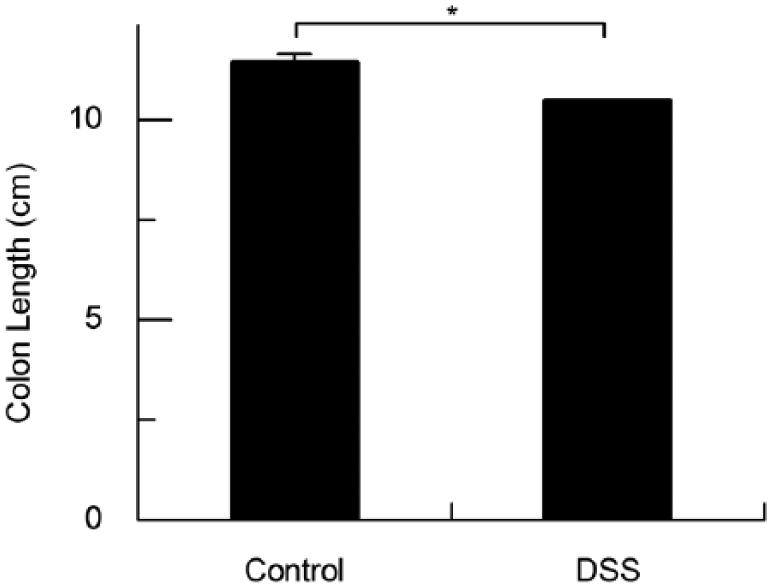
Colon length decreased after dextran sulphate sodium (DSS) administration in C67BL/6 mice. The colon length was measured on day 5 after administration of DSS. N = 9, mean values were significantly different compared control group at day 5: *
Clinical colitis in DSS-induced colitis
Clinical symptoms such as loss of body weight, loose stools, and fecal blood appeared in DSS group, which are consistent with other studies.
21
The clinical symptoms first occurred on day 3. Compared to control mice, DSS-induced mice increased significantly in the disease activity index at day 5 (7.40 vs. 1.20,
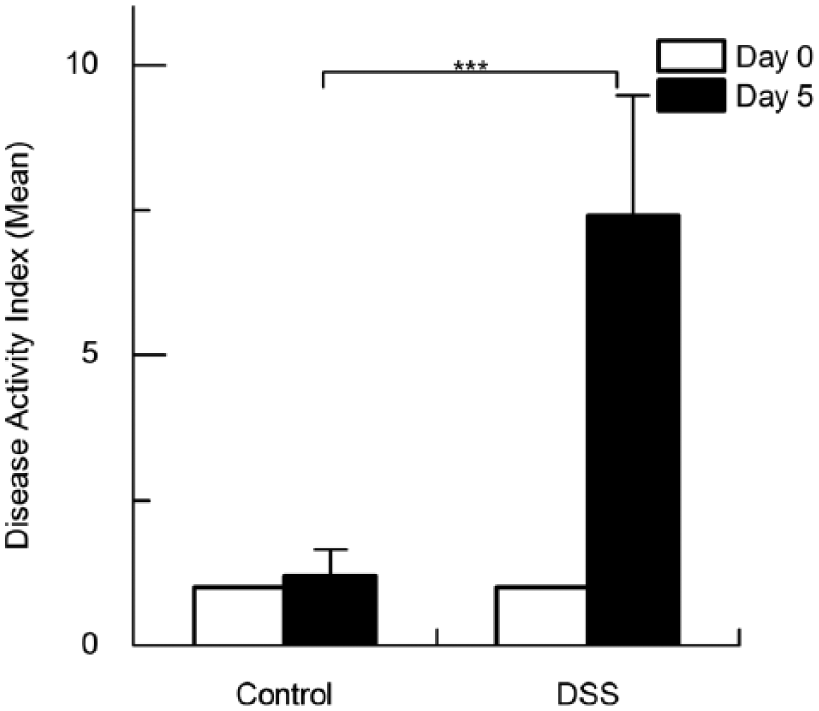
Disease activity index (DAI) increased after dextran sulphate sodium (DSS) administration in C67BL/6 mice. DAI score was assessed on day 5 after administration of DSS. N = 9, ***
The histological damage of colonic mucosa in DSS-induced colitis
Control group showed normal architecture with intact epithelium in colonic mucosa, while the DSS group showed severe damage to the surface epithelium, intense transmural inflammation, and submucosal neutrophils infiltration (Figure 4a and b). Quantification of these colonic histology results was shown in Fig 4e. DSS group had significantly (14.67 vs. 2.00,

Histological pathology increased after dextran sulphate sodium (DSS) administration. (a) Control group, (b) DSS group (hematoxylin and eosin staining; magnification 200×). (c) Histological score. N = 9, ***
Th17 and Treg cell populations in DSS-induced colitis
Plots are gated on CD4+ T cells were shown in Figure 5a. In DSS-induced colitis, CD4+IL-17A+IL-22− cells (1.47 vs. 0.47%,

CD4+IL-22+IL-17A− cells, CD4+IL-17A+IL-22− cells and CD4+CD25+Foxp3+ cells production changes in dextran sulphate sodium (DSS)-induced colitis. (a) Shown are intracellular stainings for splenic lymphocyte cells conditions and after stimulation with PMA/ionomycin. (b-d) The data of flow cytometry analysis of CD4+IL-22+IL-17A− cells (b), CD4+IL-17A+IL-22− cells (c), and CD4+CD25+Foxp3+ cells (d). N = 9, mean values were significantly different compared control group at day 5: *
Cytokine levels in serum
To analyze the cytokine production change in serum during colitis, we investigated representative Th17 (IL-17A, IL-17F, and IL-22) cytokine levels by ELISA after 5 days of DSS administration. The level of IL-17A (235.33 vs. 142.05 pg/mL,

IL-17A, IL-17F, and IL-22 production increased in dextran sulphate sodium (DSS)-induced colitis serum. (a) IL-17A concentration, (b) IL-17F concentration, (c) IL-22 concentration. N = 9, mean values were significantly different compared control group at day 5: *
Discussion
The present study detected the change of Treg/Th17 balance, Th22 proportion, and the expression of Th17- and Th22-related cytokines on the DSS-administrated mice. The experimental data showed that there are significantly difference between DSS-administrated mice and the control group in colitis clinical symptoms, Treg/Th17 balance, Th22 proportion, and IL-17A, IL-17F, L-22 concentration.
Animal models of IBD have contributed significantly to the present understanding of IBD, as they provided a platform through which some of these complex mechanisms could be systematically investigated, sometimes with manipulations and interventions, that would not have been possible with human studies.
5
We chose the DSS-induced colitis model, which caused hematochezia, body weight loss, shortening of the intestine, mucosal ulcers, and infiltration of neutrophils.22,23 In this study, DSS-induced colitis, made mice weight drops (14.91%, Figure 1), caused colon shrinkage (
Mice with acute DSS-induced colitis exhibit pathogenetic similarities to human IBD, particularly UC.5,24 Insights into IBD are advancing rapidly owing to the discovery of new CD4+ T cell subsets. Dieleman reported in 1998 that the cytokine response in the DSS-induced colitis appears to be a Th1 and Th2 type with increased levels IFN-γ and IL-4 in inflamed colonic tissue compared to controls in SW mice.
20
In 2009, data indicated that DSS colitis switches from a Th1-Th17-mediated acute inflammation to a predominant Th2-mediated inflammatory response in the chronic state.
25
Whereafter, increasing the relative balance of Tregs to Th17 is a novel strategy for IBD, such as histone-deacetylase inhibitors that ameliorate DSS colitis through expansion of Tregs.11,26 In 2009, Eyerich et al. have identified a subset of human Th cells that infiltrates the epidermis in individuals with inflammatory skin disorders and is characterized by the secretion of IL-22 and TNF-α, but not IFN-γ, IL-4, or IL-17.
13
Recent genome-wide analysis carried out in the initial phase of DSS-induced colitis revealed that the IL-22/STAT3 axis enforces a gene profile in IEC that especially connects to the vital task of intestinal wound healing.
27
In the present study CD4+IL-17A+IL-22− cells and CD4+IL-22+IL-17A− cells increased (
IL-17A and IL-17F belong to IL-17 family, as well as IL-17B-E. IL-22 is IL-10 family cytokine. IL-17F possesses the highest degree of homology with IL-17A, sharing similar functions through binding to the common cell surface receptor complex that is consisted of IL-17RA and IL-17RC.24–28). IL-17A and IL-17F essentially contribute to the development of bowel inflammation, in contrast, biological roles of other IL-17 family members have not been fully characterized.
24
Both IL-17A and IL-17F have a redundant but highly pathogenic role in gut inflammation, and had to be deficient or neutralized to block disease after transfer of wild type CD4+CD45RBhi T cells.9,29 However, studies also have shown that IL-17A and IL-22 are produced at early stages in the cecum of mice with
Collectively, the drift balance of Th17/Treg is apparently in DSS-induced colitis. The increasing concentration of IL-17A, IL-17F, and IL-22, as well as the increasing Th17 and Th22 cells count, suggests that Th17 and Th22 are involved in the pathological process. Further studies are warranted to develop drugs targeting the Th17/Treg and Th22 pathways, by which these agents are able to protect against ulcerative colitis.
Footnotes
Acknowledgements
We thank Professors Hongyu Li and Jinbo Yang for help with flow cytometry and are grateful to Professor Qiang Chen (Lanzhou University) for animal husbandry.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to research, authorship, and/or publication of this article.
Funding
This work was supported by the foundation of the Key Laboratory for Gastrointestinal Diseases of Gansu Province (no. gswcky-2012-007).
