Abstract
Induction of oxidative damage by the activation of histone deacetylase (HDAC) is an integral event that causes major membrane damage of ocular tissues and leads to the pathogenesis of cataract. It is elucidated that nuclear factor-κB is a mediator in the process of cataract development. However, studies on the role played by epigenetic proteins in regulating cataract pathogenesis are limited. Hence, in the current investigation, ARPE-19 human retinal epithelial cells were used as an experimental model to elucidate the role of HDAC inhibition and its mechanism behind the cataract pathogenesis. ARPE-19 cells were exposed to H2O2, with and without Trichostatin A (TSA), a pan-HDAC inhibitor, and maintained along with control cells without any treatment. On exposure to H2O2, cells were susceptible to oxidative stress as it is evident from the reduced expression levels of superoxide dismutase (SOD), catalase, and GSH levels. Simultaneously, H2O2-exposed cells showed the nuclear translocation of NF-κB with the activation of inflammatory cytokines such as CXCL1 and IL-6. In addition, the mRNA expression analysis revealed that the GADD45α, COX-2, MCP-1, and ICAM-1 expressions were increased in H2O2 group. Moreover, the activity of HDAC was increased to 2-fold with a significant reduction in the histone acetyltransferase (HAT) activity in cells that were maintained under oxidative conditions. However, TSA was able to inhibit the critical cytokines’ expression with attenuated HDAC activity and limited NF-κB translocation. Furthermore, pre-treatment of TSA significantly suppressed the transcript levels of up-regulated inflammatory markers in cells. Together, these findings offer new insight into the role of HDACs in regulating cellular processes involved in the pathogenesis of cataract as well as the potential use of HDAC inhibitors as therapeutics for controlling the disease progression.
Introduction
The clouding of eye lens that affects the vision in elderly people worldwide is the age-related cataract (ARC). The disease prolongs to the entire eye lens and can affect the vision completely if left untreated although it does not spread from one eye to the other. ARC continues to be most debilitating of all the disease of old age and treatment mostly involves surgical removal and it poses some potential risks apart from the high costs. 1 Dependent on the regions of the lens they originate—nuclear, cortex, or posterior capsule—cataracts are classified into nuclear, cortical, or posterior subcapsular cataracts, respectively.
Many factors such as genetics, environment, and diet can be attributed to the cataract formation. Epidemiological studies from worldwide populations show that oxidative damage is the most prominent hallmark of ARC. 2 Imbalance in the production of reactive oxygen species (ROS) and pro-oxidants (antioxidant defenses) can result in oxidative stress and is the initiating factor for the development of cataract in older individuals. Wide-ranging oxidation of lens protein has been related with elevated levels of H2O2 and is the major oxidant in affected individuals.
The underlying epigenetic mechanisms such as DNA methylation, histone modifications, and chromatin remodeling will be helpful in understanding the pathophysiology of the disease and help in treatment. The covalent modifications at the N-terminal tails of the histones, in particular deacetylations on Lysine residues by histone deacetylases (HDACs), 3 regulate the gene transcription by unwinding the chromatin and increase the availability of DNA-binding proteins and transcription factors to bind and transcribe genes available on them. 4 The role of epigenetic in the progression of age-related ocular diseases has already been established.5–8 Studies on streptozotocin-induced diabetic rat model indicate that hyperglycemia increases the interaction between the promoter regions of sod gene and the inflammatory transcription factor NF-kB, and covalent modifications on histones are dominant to induce downregulation of sod gene and promote oxidative stress and disease progression. 9 ROS, induced by H2O2, generated as a result of oxidation in mitochondria, accumulate in lens epithelial cells and damage the ocular tissues in human cataract.10,11 HDACs, known to be critical mediators of the normal gene silencing as precursors to the progression of optic nerve diseases in mice, 12 are frontrunners to be studied in the role of epigenetic proteins in the ROS-mediated pathogenesis of cataract. HDAC inhibitors have been already tested in animal models and cultured cells and that its safety in the nerve ocular disease treatment is satisfactorily addressed.1,13 Hence, we propose that HDAC inhibitor Trichostatin A (TSA) on H2O2-activated human retinal epithelial cells (ARPE-19) would be ameliorating the effects of ARC and its symptoms and it is a potent drug in cataract.
Methodology
Cell culture
The ARPE-19 human retinal epithelial cells, the approved cell line model for the study of ocular diseases, were acquired from the American Type Culture Collection (ATCC), USA. The cells were cultured in Dulbecco’s Modified Eagle Medium/Nutrient Mixture F-12 (1:1) culture medium supplemented with 10% heat-inactivated fetal calf serum with 2 mM/L glutamine, 100 U/mL penicillin, 50 µg/mL each streptomycin and neomycin. The cells were grown in culture plates and flasks according to the experiment until confluence. Along with untreated control, cells were developed in the presence of H2O2 (100 µM) for 1 h for oxidant induction group and TSA (50 nM in 0.1% dimethyl sulfoxide, Sigma Aldrich, USA) pretreatment for 2 h in another group of cells before H2O2 exposure was also employed.
Antioxidant enzymes
The cells after stimulations and treatments collected by scraping and centrifuged at 1500 g for 10 min at 4°C. The superoxide dismutase (SOD), catalase, GSH, and nitric oxide assays kits (Cayman Chemicals, USA) were used for the study, and hence the cells were lysed as per the protocol for the specific assay and the enzymes were analyzed as per manufacturer’s instruction.
Measurement of intracellular ROS
The ROS released in the cells was measured by the oxidant-sensitive fluorescent probe, 2,’ 7’-dichlorofluorescein diacetate (DCFHDA) (Sigma Aldrich, USA). About 1 × 105 cells grown in polylysine-coated coverslips were treated with/without H2O2 and TSA. The cells were washed with PBS and were incubated with DCFHDA (10 µM) at 37°C. The fluorescence released was detected in a fluorescence microscope, and the amount of fluorescence formed was observed using fluorimeter at an excitation and emission wavelength of 488 and 530 nm, and the images were taken in a confocal microscope (20× magnification).
Real-time reverse transcriptase-PCR
After the experimental period, RNA was extracted from the cells and the first-strand cDNA synthesized was done from 1 µg of RNA. Real-time RT-PCR was done using the Bio-Rad real-time PCR system and data obtained were compared with control. Sequence-specific oligonucleotides were obtained from Eurofins MWG (Operon). The forward (F) and reverse (R) primers used in the study were given in Table 1. The PCR amplifications were done in triplicates using SYBR Green PCR Master Mix (Qiagen, Valencia, CA, USA). The reaction settings: 95°C, 10 min 32 cycles at 95°C, 15 s 60°C, 1 min 1 cycle at 95°C, 1 min 55°C, 30 s 95°C 30 s. QPCR Software was used to calculate the CT values and the transcript level was determined by comparative Ct method (ΔΔCT) with expression values of GAPDH as an endogenous control.
Oligonucleotides used in this study.

Confocal microscope analysis
ARPE-19 cells (1 × 104) grown on coverslips for the experiment were fixed with 4% paraformaldehyde and permeabilized with 0.5 % Triton X-100 after stipulated treatment. Cells were then incubated with primary antibodies NF-κB (1:200) (Santa Cruz, CA, USA). The cells were incubated at 37°C for 1 h; then cells were washed with PBS and grown with FITC-labeled secondary antibody for 1 h. The cells were washed three times with PBS and counterstained using propidium iodide (PI) and imagined under Zeiss LSM 700 confocal microscope.
Enzyme-linked immunosorbent assay (ELISA)
ARPE-19 cells were grown and before stimulation, cells were serum-starved for 24 h in medium with 1% fetal bovine serum and the drug treatments were done. The cells were then scraped off, collected; centrifuged (1000 g × 5 min) to remove particulates and the supernatant was stored at −80°C until analysis. HAT and HDAC activities were evaluated using the ELISA assay kit as per the manufacturer’s protocol (HAT activity/inhibition kit or EpiQuik HDAC, Epigentek, Brooklyn, NY, USA). Absorbance was measure at 450 nm, and activities were measured using a standard curve for HAT (ng/h/mg protein) and HDAC (ng/min/mg protein). In addition, the level of cytokines such as vimentin (Abcam, USA), CXCL1, IL-6, ICAM-1, and IL-2 were also measured using commercial ELISA kits (R&D Systems, Minneapolis, MN, USA).
Western blotting analysis
ARPE-19 cells of control and experimental group were scrapped of culture flasks and sonicated to release the proteins and quantified using Brad ford reagent. The protein samples were mixed with 4× Laemmli SDS sample buffer, with reducing agents and loaded in SDS-polyacrylamide gels and the electrophoretic separation was done for 2 h at 100 V. The proteins were blotted into polyvinylidene difluoride membrane, blocked in 1% bovine serum albumin in PBS for 1 h, and probed with goat anti-human GADD45α, COX-2, MCP-1, ICAM-1 (1:500 dilution each, Santa Cruz, CA, USA), and β-actin antibody for endogenous control at 4°C. At the end of incubation, the membrane was washed and probed for the corresponding Horseradish Peroxidase (HRP) labeled secondary antibody for 1 h at room temperature, washed thrice with Phosphate buffered saline with Tween-20 (PBST) and visualized using Enhanced chemiluminescence (ECL) chemiluminescence system and the band intensities were determined densitometrically.
Data analysis
The data are expressed as mean ± standard error. One-way analysis of variance using GraphPad Prism Software (GraphPad Software, CA, USA). The value of P < 0.05 was considered significant.
Results
In order to test the hypothesis that HDACs are involved in the pathogenesis of cataract, we used ARPE-19 cells as culture model and tested it. Initially, the cells were divided into control, exposed to oxidant conditions and TSA pre-treatment, and the levels of anti-oxidant enzymes were elucidated. The results demonstrated that the levels of SOD, catalase, glutathione, and nitric oxide were diminished in H2O2-treated cells, while the levels of these cellular protective agents were restored in the cells grown with HDAC inhibitor TSA (Figure 1). It was documented that the elevation of oxidant status is the main occurrence in the cataract development; hence, the cells with and without H2O2 and TSA were analyzed for ROS release using the DCFHDA emitted fluorescence and the results confirmed the high ROS levels in H2O2 group, while TSA prevented the effect evidence the protective role of TSA against the oxidant mechanisms. On the other hand, the onset of cataract marker vimentin was analyzed in the experimental group, which evidenced that the H2O2-exposed cells elicited the 2-fold increase in vimentin expression while the TSA abrogated the cataract marker expression (Figure 2).
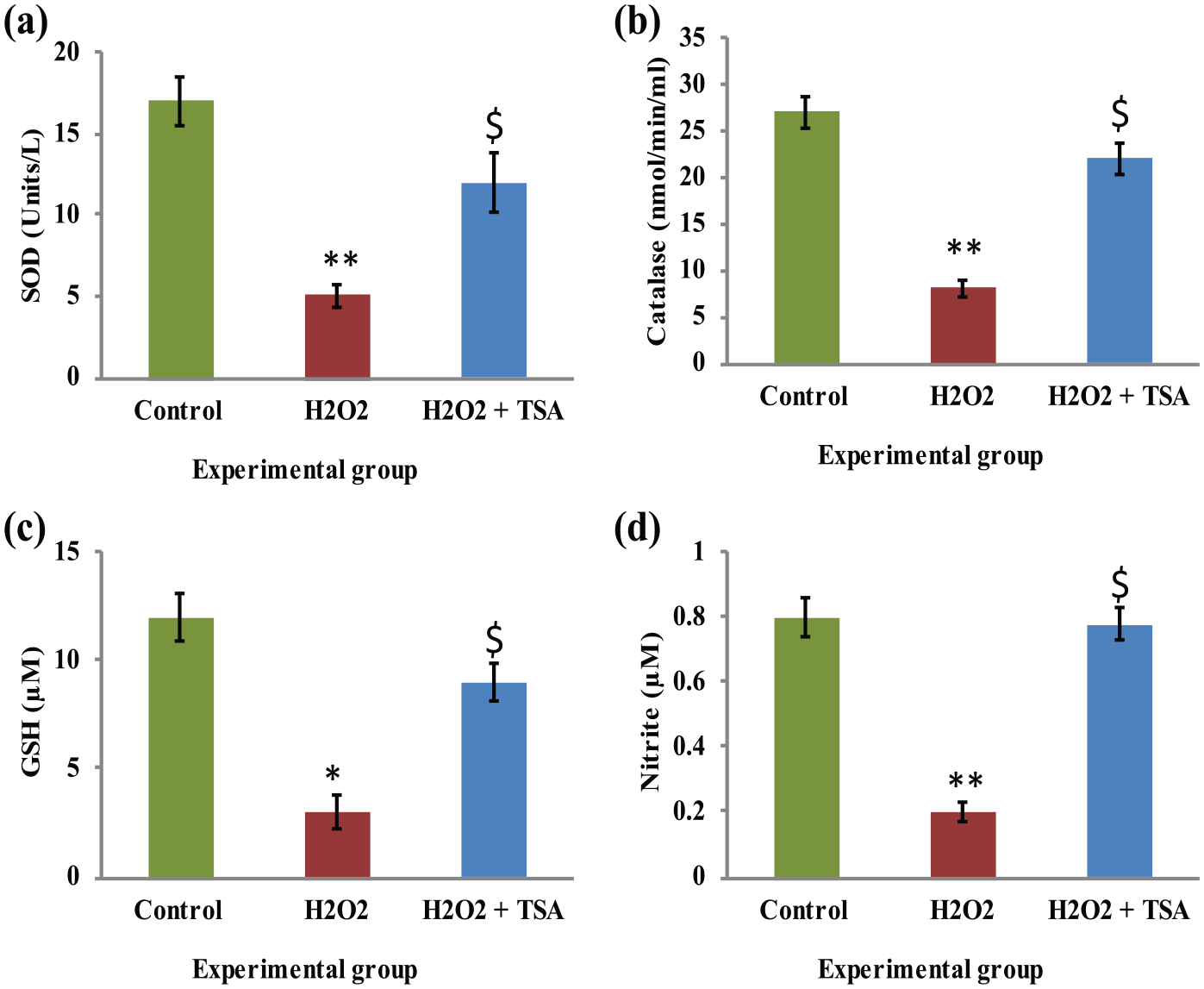
(a)–(d) Represents the levels of SOD, catalase, glutathione, and nitric oxide of control, H2O2, and TSA pre-treated H2O2 exposed cells, respectively. The enzyme activity unit for SOD and catalase is expressed as units/mg protein; GSH as µg/mg protein. Values are expressed as means ± SE (n = 6). Statistical significance expressed as *P < 0.05, **P < 0.01 compared with saline-treated controls, $P < 0.05 compared with H2O2 exposed cells.

Effect of TSA on the release of ROS in ARPE-19 cells. (a) Representative microscopic picture of cells emitting fluorescence in control, H2O2, and TSA pre-treated H2O2 exposed cells. (b) The amount of ROS release was measured as fluorescence with an excitation/emission wavelength of 488 and 530 nm. (c) The level of vimentin is represented. Values are expressed as means ± SE (n = 6). Statistical significance expressed as *P < 0.05, ***P < 0.001 compared with saline-treated controls, $P < 0.05, $$P < 0.01 compared with H2O2 exposed cells.
Furthermore, the role of HDAC inhibition in the nuclear transfer (translocation) of NF-κB was illuminated in the present study using confocal microscopy analysis. Cells exposed to H2O2 showed the nuclear transfer (translocation) of NF-κB, while cells with TSA pre-treatment demonstrated the prominent cytoplasmic localization suggesting that the nuclear translocation of NF-κB is one of the main events in the cataract development (Figure 3). It has been elucidated that the activation of HDAC is the major inducer of cataract growth. Figure 4 represents the activity of HDAC and histone acetyltransferase (HAT) activity in the experimental group of cells. In the present study, cells grown in oxidant conditions demonstrated 3.5-fold the activation of HDAC and with a 2.7-fold reduction in HAT activity, but the cells pre-treated with HDAC inhibitor abrogated the total HDAC activity with simultaneous improvement in HAT activity (Figure 4).

The role of HDAC inhibition in the nuclear translocation of NF-κB is represented. (a) Representative confocal microscopy immunofluorescence picture with colocalization. (b) The densitometry analysis of fluorescence is represented in cells stained with anti-rabbit NF-κB primary antibody and FITC-conjugated secondary antibody and counterstained with propidium iodide (red). The details of experiments were given in methodology section. Values are expressed as means ± SE (n = 6). Statistical significance expressed as *P < 0.05, **P < 0.01 compared with saline-treated controls, $P < 0.05 compared with H2O2 exposed cells. Magnification (63×).

Activity of histone deacetylase (HDAC) and histone acetyltransferase (HAT) activity in the control, H2O2, and TSA pre-treated H2O2 exposed cells. (a) and (b) Represents quantitative analysis of HDAC and HAT activity, respectively. Values are expressed as means ± SE (n = 6). Statistical significance expressed as *P < 0.05, **P < 0.01 compared with saline-treated controls, $P < 0.05 compared with H2O2 exposed cells.
Furthermore, the mRNA levels of GADD45α, COX-2, MCP-1, and ICAM-1 were studied in the present study. Simulated model of oxidant status in cells using H2O2 demonstrated the significant increase in expression of GADD45α (3-fold), COX-2 (3-fold), MCP-1 (2.5-fold), and ICAM-1 (2-fold); while the expression of markers related to cataract and cytokines are attenuated in the TSA-treated group of cells show that an obvious reduction in the markers demonstrated astonishing data that the HDAC inhibition preventing the aggressive cataract development (Figure 5). In addition, the expressed genes were tested for the level of protein expression using Western blotting analysis. The results demonstrated a significant increase in the protein expression of GADD45α (4.2-fold), COX-2 (5-fold), MCP-1 (3.8-fold), and ICAM-1 (3.2-fold) in H2O2-exposed cells, while these proteins found to be attenuated in the TSA-treated cells confirm the qPCR data supporting the protective role of HDAC inhibition.

(a)–(d) Represents qRT-PCR mRNA expression analysis of GADD45α, COX-2, MCP-1, and ICAM-1 normalized to GAPDH in the control, H2O2, and TSA pre-treated H2O2-exposed cells, respectively. Values are expressed as means ± SE (n = 6). Statistical significance expressed as *P < 0.05 compared with saline-treated controls, $P < 0.05 compared with H2O2-exposed cells.
Moreover, it was speculated and found from PCR analysis results that cytokines play a major role in cataract aggression; to further confirm the levels of cytokines, the ELISA was performed in the experimental cells. The results of the experiment demonstration that the amounts of cytokines were considerably (P < 0.01) enhanced—CXCL1 (2.5-fold), IL-6 (1.5-folds), ICAM-1 (2.5-folds) and IL-2 (3-folds)—in H2O2 group while and TSA pre-treated H2O2 exposed cells could endure the activation of pro-inflammatory cytokines (Figure 6 & 7). Thus, the results demonstrated the protective role of HDAC inhibition that can contribute to the development of novel treatment strategies.
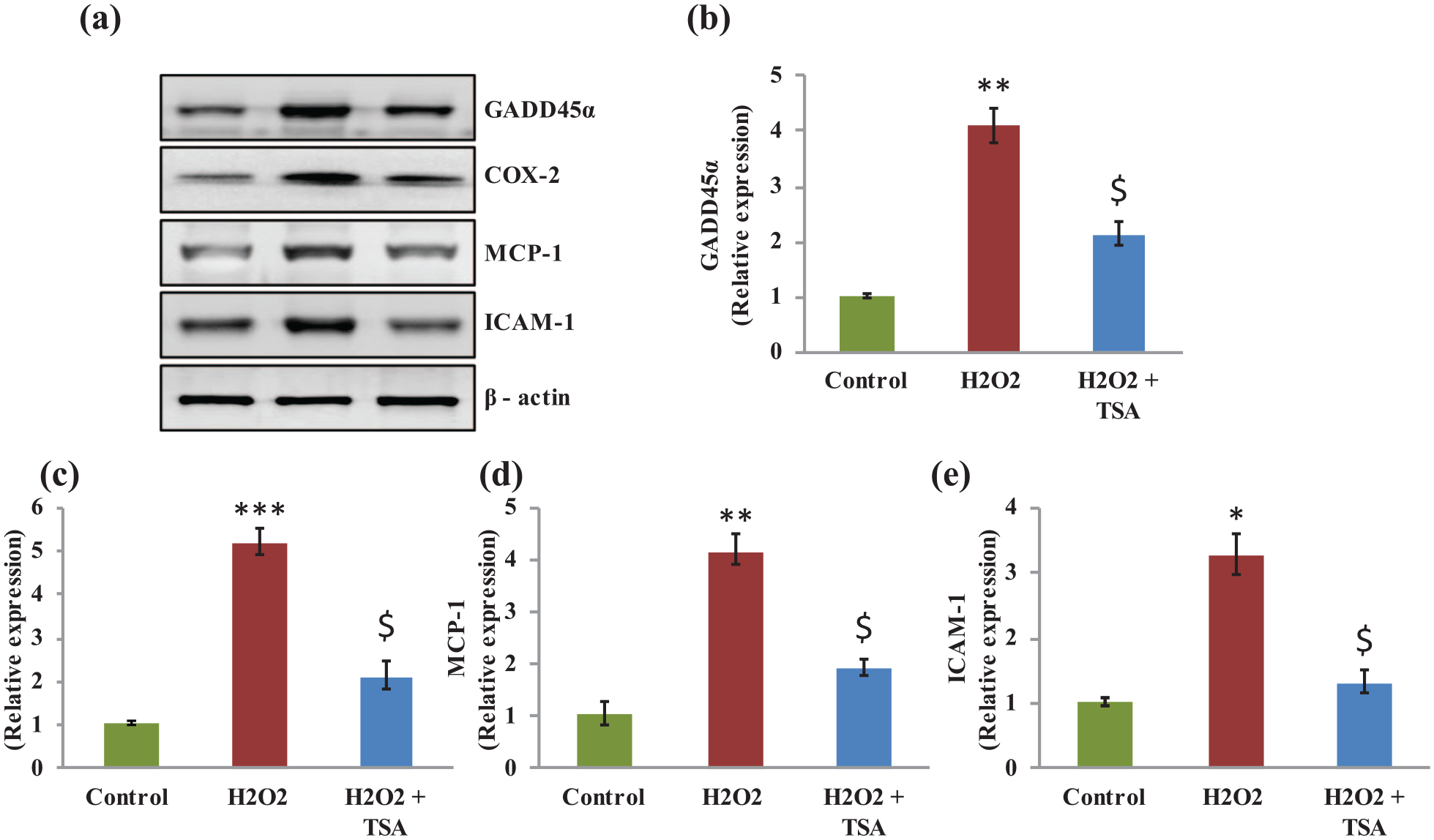
(a) Representative Western blotting protein expression analysis of GADD45α, COX-2, MCP-1, and ICAM-1 in the control, H2O2, and TSA pre-treated H2O2-exposed cells, respectively. (b)–(e) Densitometry analysis of proteins normalized to β-actin expression. Values are expressed as means ± SE (n = 6). Statistical significance expressed as *P < 0.05, **P < 0.01, ***P < 0.001 compared with saline-treated controls, $P < 0.05 compared with H2O2-exposed cells.

(a)–(d) Represents cytokine expression analysis of CXCL1, IL-6, ICAM-1, and IL-2 in control, H2O2, and TSA pre-treated H2O2 exposed cells. Values are expressed as means ± SE (n = 6). Statistical significance expressed as *P < 0.05, **P < 0.01 compared with saline-treated controls, $P < 0.05 compared with H2O2 exposed cells.
Discussion
Cataract is a neurodegenerative disease occurring in elderly people and is complicated by many other impending health complexities, for example, diabetes, hypertension, age-linked reduction of telomere length, hypertension, and accumulation of ROS. 14 Much of the complications include the symptoms such as blurring of vision to partial or total vision loss, aggravated by the associated risks with surgery. Surgical treatment is complicated due to increasing age and associated health conditions that include the posterior capsule rupture, postoperative infection, edema in cornea, and increased corneal pressure. 15 Among the various factors involved in mediating the pathogenesis of cataract, the role of HDAC has not been elucidated so far. Hence, in the current investigation, cultured lens epithelial cells were exposed to oxidative damage with or without HDAC inhibitors, and its mechanism behind the cataract pathogenesis was elucidated.
Oxygen free radicals are generated through various physiological and enzymatic pathways in the body and released from mitochondria. 16 This development of oxidative stress is a considerable factor in causing aging of the eye. On the other hand, an antioxidant defense that prevents oxidative damage to the body can be imbalanced and cause oxidative stress as a feedback mechanism. This includes accumulation of oxygen free radicals in the eye fluids and the tissues of the epithelial cells (Figure 2(a) and (b)). In addition to the increase in ROS in the cultured cells with H2O2, a corresponding upsurge in the expression of intermediate filament vimentin in the cells treated with H2O2 has been observed that shows the cataract onset. This observation goes well with the results obtained by Lai et al., 17 where an increase in vimentin expression was observed in the condition of hypoxic stress as obtained in our results with oxidative stress (Figure 2(c)).
We have demonstrated here the influence of H2O2 in the transcriptional regulation and transcript level of several enzymes and proteins that participate in the enzymatic and non-enzymatic defense mechanisms. These include SOD, glutathione peroxidase, catalase, and vitamins and glutathione. The action of H2O2 in the form of oxidative injury is manifested in the damage to the protective proteins 18 and H2O2-mediated ROS production was evidenced in DCF assay. Moreover, cells that are treated with only H2O2 where in the expression of SOD, catalase, GSH, and nitrite have been found to be decreased in all the experimental groups than control group. Upon treatment of HDAC inhibitor, TSA, the antioxidant defense system that included the amounts of these enzymes has been restored to levels on par with the untreated group (Figure 1) and that could delay the onset of cataract. 19
As stated earlier, directly or indirectly activation of superoxide radicals and reactive oxygen species is indicted for its involvement in the formation of cataract in the epithelial cells 20 and is mediated through the onset of inflammatory pathways in cultured cells. 21 In the current investigation, H2O2-treated ARPE-19 cells demonstrated an amplified expression of inflammatory cytokine IL-6 (Figure 6(b)) and it is achieved possibly by the activation of IL-6 promoter through the transcriptional factor NF-kB (Figure 3(a)). Similar results were obtained for rabbit lens epithelial cells when H2O2 was observed to activate NF-kB which in-turn switch-on the genes to modulate the oxidative stress. 20 It has been well documented that there is a significant role of HDACs in cellular functioning. The balance between the HDAC and HAT activity in H2O2-activated oxidative stress is maintained and is important for the epigenetic mechanism based activation of genes. 22 In the current investigation, the levels of HDAC and HAT in the H2O2-activated cells are inversely proportional to each other and it is conversely the same in cells treated with HDAC inhibitor TSA (Figure 4). 23
Finally, we looked for the up-regulation of mRNAs and proteins levels of some factors that are turned on by the transcriptional factor NF-kB in cells that are activated by H2O2 and those that inhibited by TSA. H2O2 can promote different types of cataracts at the onset of oxidative stress in eye epithelial cells. One of the hallmarks of epigenetic mechanisms is the epithelial-mesenchymal transition of cells that are under stress to induce apoptosis, migration of cells, and damage to proteins. ICAM-1 is a major marker found expressed in epithelial cells of cataract which might play an important role in the cataractogenesis. 24 A 2-fold increase in expression level of ICAM-1 is noted in cells treated with H2O2 and that would be decreased in cells treated with TSA, although a residual ICAM-1 mRNA expression is observed (Figure 5(d)). Another aspect of cataract onset observed through NF-kB activation is a 3-fold increase in mRNA expression of GADD45 and causing DNA damage. Inflammatory chemotactic proteins and cytokines that are expressed during NF-kB activation have been observed at the sites of oxidative damage, 25 and MCP-1 and IL-6 are observed to be increased several folds in ARPE cells activated with H2O2.
The results of our investigation specify that the onset and progression of cataracts in elder human subjects have been mainly through the actions of H2O2 and HDAC in modification of histones and turning on the expression of genes, by the transcriptional factor NF-kB, for the collective migration of epithelial cells and damage of the proteins on the retina. We have demonstrated in our in vitro studies that manipulating the systemic action of HDAC can be made possible by the use of HDAC inhibitor TSA and suggesting a potential usage of it as a drug to treat ARC to prevent the risks associated with surgery.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
