Abstract
Introduction
Lipopolysaccharide (LPS) tolerance is the downregulation of LPS signaling after pre-exposure to LPS, and it provides protection against hyperactive inflammation. Cytokine production decreases during LPS tolerance, and the phenotype of LPS-tolerant monocytes shifts toward M2 (anti-inflammatory) type. Magnesium sulfate (MgSO4) is a widely used anti-inflammatory agent. Although MgSO4 inhibits LPS signaling, the effect of MgSO4 on LPS tolerance is unknown. In the present study, we investigated the in vitro effects of MgSO4 on LPS tolerance.
Methods
To induce LPS tolerance, THP-1 cells were stimulated with LPS (200 ng/mL, 2 h) after pre-exposure to LPS (200 ng/mL, 24 h) with or without pre-treatment of MgSO4 (20 mM, 24 h).
Results
Our results revealed that MgSO4 enhanced LPS tolerance by downregulating nuclear factor-κB (NF-κB)-induced tumor necrosis factor-α or interleukin-6, and upregulating cluster of differentiation 163 (a M2-associated marker). Furthermore, the LPS-triggered upregulation of phosphoinositide 3-kinase (PI3K) was significantly increased during LPS tolerance. MgSO4 activated PI3K, but inhibited NF-κB in LPS-stimulated cells. Notably, MgSO4 mitigated the signaling of both PI3K and NF-κB in LPS-tolerant cells, suggesting the effect of MgSO4 on LPS tolerance relies on the modulation of the crosstalk between PI3K and NF-κB.
Conclusion
MgSO4 enhanced LSP tolerance, thus providing evidence for a novel underlying mechanism of the anti-inflammatory effects of MgSO4.
Keywords
Introduction
Binding of lipopolysaccharide (LPS) to toll-like receptor 4 (TLR4) triggers the activation of nuclear factor-κB (NF-κB) pathway and the production of pro-inflammatory cytokines, leading to various inflammatory pathologies. Internalization and trafficking of TLR4 (i.e., TLR4 endocytosis) modulate TLR4 signaling. 1 LPS tolerance, which is the downregulation of LPS-triggered inflammatory responses after pre-exposure to LPS, is observed in many inflammatory diseases including sepsis. 2 Downregulation of TLR4 signaling is critical for the development of LPS tolerance. 2 During LPS tolerance, the production of tumor necrosis factor-α (TNF-α) or other pro-inflammatory cytokines decreased, providing protection against hyperactive inflammation. 2 The phenotype of LPS-tolerant immune cells shifts toward the M2 (anti-inflammatory) type, as demonstrated by the increased expression of M2-associated surface markers including cluster of differentiation 163 (CD163). 3
Magnesium is an essential element for humans and is indispensable for many physiological functions. Low magnesium level is associated with increased inflammation. 4 In clinical practice, magnesium sulfate (MgSO4) is used not only for magnesium supplementation but also as an anti-inflammatory agent. For example, MgSO4 is effective in pain management5,6 because inflammation mediates the development of pain. 7 Although MgSO4 mitigates LPS-induced inflammation through inhibiting NF-κB pathway, 8 the effect of MgSO4 on LPS tolerance remains unstudied.
Materials and methods
Cell culture and stimulation
In this in vitro study, THP-1 cells (American Type Culture Collection, Manassas, VA, USA), a human monocyte cell line, were used to investigate the effect of MgSO4 on LPS tolerance. THP-1 cells were maintained in Roswell Park Memorial Institute 1640 medium (RPMI 1640; Sigma-Aldrich, MO, USA) containing 10% fetal bovine serum (Thermo Fisher Scientific, MA, USA) and 1% penicillin/streptomycin (Life Technologies, CA, USA) at 37°C in a mixture of 95% air and 5% CO2. To induce LPS tolerance, cells were stimulated with LPS (1st LPS, 200 ng/mL; from Escherichia coli O111: B4, Sigma-Aldrich, MO, USA) for 24 h and then washed with phosphate buffered saline (PBS), followed by re-stimulation of LPS (2nd LPS, 200 ng/mL) for 2 h. In the MgSO4-treated groups, MgSO4 (20 mM; Sigma-Aldrich, MO, USA) was added 24 h before the 1st LPS stimulation. The dose of MgSO4 was based on our previous study.
9
THP-1 cells were randomly allocated into PP, PL, LL, MPP, MPL and MLL groups (Figure 1(a)). The PP and MPP groups were used as control. LPS tolerance was induced in the LL and MLL groups. The PL and MPL groups received LPS stimulation without pre-exposure to LPS or the development of tolerance. Experimental protocol and levels of tumor necrosis factor (TNF-α), interleukin-6 (IL-6) and cluster of differentiation 163 (CD163). (a) To induce lipopolysaccharide (LPS) tolerance, THP-1 cells were stimulated with LPS (2nd LPS, 200 ng/mL) for 2 h after pre-exposure to LPS (1st LPS, 200 ng/mL) for 24 h. In the groups treated with magnesium sulfate (MgSO4), MgSO4 was administered 24 h before exposure to the 1st LPS. Cells were randomly allocated into PP, PL, LL, MPP, MPL and MLL groups. (b) The concentrations of TNF-α and IL-6 measured by enzyme-linked immunosorbent assay. Data are expressed as means ± standard deviations (n = 7). (c) Expression of CD163 was measured using flow cytometry. Data are expressed as means ± standard deviations (n = 4). *p < .05 versus PP group. #p < .05 versus PL group. & p < .05 versus LL group.
Enzyme-linked immunosorbent assay (ELISA)
The levels of TNF-α, interleukin-6 (IL-6), and CD163 were measured to determine the development of LPS tolerance. The concentrations of TNF-α and IL-6 in the supernatants were quantified using the commercial ELISA kit for human TNF-α (R&D System, MN, USA) and IL-6 (R&D System, MN, USA) respectively, in accordance with the manufacturer’s protocol.
Flow cytometry
Cell-surface TLR4 expression was measured using flow cytometry to evaluate TLR4 endocytosis triggered by the binding of LPS and TLR4. THP-1 cells (106 cells/ml) from each group were collected and resuspended in fresh medium. The cell suspensions were labeled with fluorescent-conjugated primary antibody against CD163 (Abcam, Cambridge, UK) or TLR4 (Thermo Fisher Scientific, MA, USA), as well as the isotype control (Alexa Fluor 488 Mouse IgG2b, BD), for 60 min. After washing with PBS, the immunolabeled-fixed cells were harvested and analyzed using the fluorescence-activated cell sorting flow cytometer (Cytomic FC50 Flow Cytometer; Beckman Coulter, Inc., Fullerton, CA, USA). For each group, 20,000 events accumulated were collected. The mean fluorescence intensity (MFI) was measured to determine the expressive levels of CD163 and TLR4.
Immunoblotting
We investigated the role of phosphoinositide 3-kinase (PI3K) and NF-κB in the effect of MgSO4 on LPS tolerance. After the cells (107 cells/10 mL) were treated as described earlier, the cell lysates were prepared using the lysis buffer (Cell Signaling Technology, USA), and the protein levels were quantified using the bicinchoninic acid assay kit (Thermo Scientific, USA). Protein samples from each group were separated using 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE), and transferred to polyvinylidene fluoride membranes (Bio-Rad, USA). The membranes were incubated with primary antibody against p65 (1:1000; Cell Signaling Technology, USA), phospho-p65 (p-p65; 1:1000; Cell Signaling Technology, USA), PI3K (1:200; Santa Cruz, USA), phospho-PI3K (p-PI3K; 1:500; Abcam, UK) and actin (1:1000; Sigma-Aldrich, MO, USA) followed by incubation with the secondary antibody (1:10000; horseradish peroxidase-conjugated anti-mouse IgG antibody; Amersham Pharmacia Biotech, Inc., NJ, USA). The protein bands were visualized using chemiluminescence (ECL plus kit; Amersham Pharmacia Biotech, Inc., NJ, USA), and the density of the protein bands was quantified using the ImageJ software.
Statistical analysis
Statistical analysis was performed using a commercial software package (SigmaStat for Windows; SPSS Science, Chicago, IL, USA). All data were presented as mean ± standard deviations, and analyzed by one-way analysis of variance in conjunction with Tukey’s post hoc test. A p-value <.05 was considered statistically significant.
Results
MgSO4 enhanced LPS tolerance
The levels of TNF-α and IL-6 (Figure 1(b), both n = 7 per group) in the PP group were low. LPS increased the concentrations of TNF-α and IL-6 (both p < .001, PL group vs PP group). Re-stimulation with LPS obviously decreased the LPS-triggered upregulation of TNF-α (p < .001, LL group vs PL group) and IL-6 (p = .002, LL group vs PL group), indicating successful induction of LPS tolerance. Moreover, the expression of TNF-α and IL-6 in the MLL group was significantly lower than that in the LL group (p = .019 and p = .021, respectively). In Figure 1(c), LPS stimulation did not altered the level of CD163 (n = 4 in each group). CD163 was significantly upregulated during LPS tolerance (p = .002, LL group vs PL group). Pre-treatment with MgSO4 further increased the expression of CD163 in LPS-tolerance cells (p = .003, MLL group vs LL group).
MgSO4 may modulate LPS tolerance through NF-κB pathway involving PI3K
The surface TLR4 expression was measured using flow cytometry (Figure 2(a), n = 4 per group). LPS stimulation induced TLR4 endocytosis and reduced surface TLR4 expression (p < .0001, PL group vs PP group). The development of LPS tolerance mitigated the LPS-triggered downregulation of the surface TLR4 level (p = .002, LL group vs PL group). MgSO4 did not significantly change the expression of surface TLR4, suggesting that the effect of MgSO4 on LPS tolerance is not dependent on the level of TLR4 endocytosis. Levels of nuclear factor-κB (NF-κB) p65, phospho-p65 (p-p65), phosphoinositide 3-kinase (PI3K), phospho-PI3K (p-PI3K), and toll-like receptor 4 (TLR4). (a) Expression of surface TLR4 was measured using flow cytometry. Data are expressed as means ± standard deviations (n = 4). (b) Expression of p65, p-p65, PI3K, and p-PI3K was measured using immunoblotting. The level of NF-κB or PI3K activation was defined as the ratio of p-p65/p65 or p-PI3K/PI3K to actin expression (the internal control), compared to that of PP group. Data are expressed as means ± standard deviations (n = 4). Cells were randomly allocated into six groups (PP, PL, LL, MPP, MPL and MLL groups as delineated in Figure 1(a)). *p < .05 versus PP group. #p < .05 versus PL group. & p < .05 versus LL group.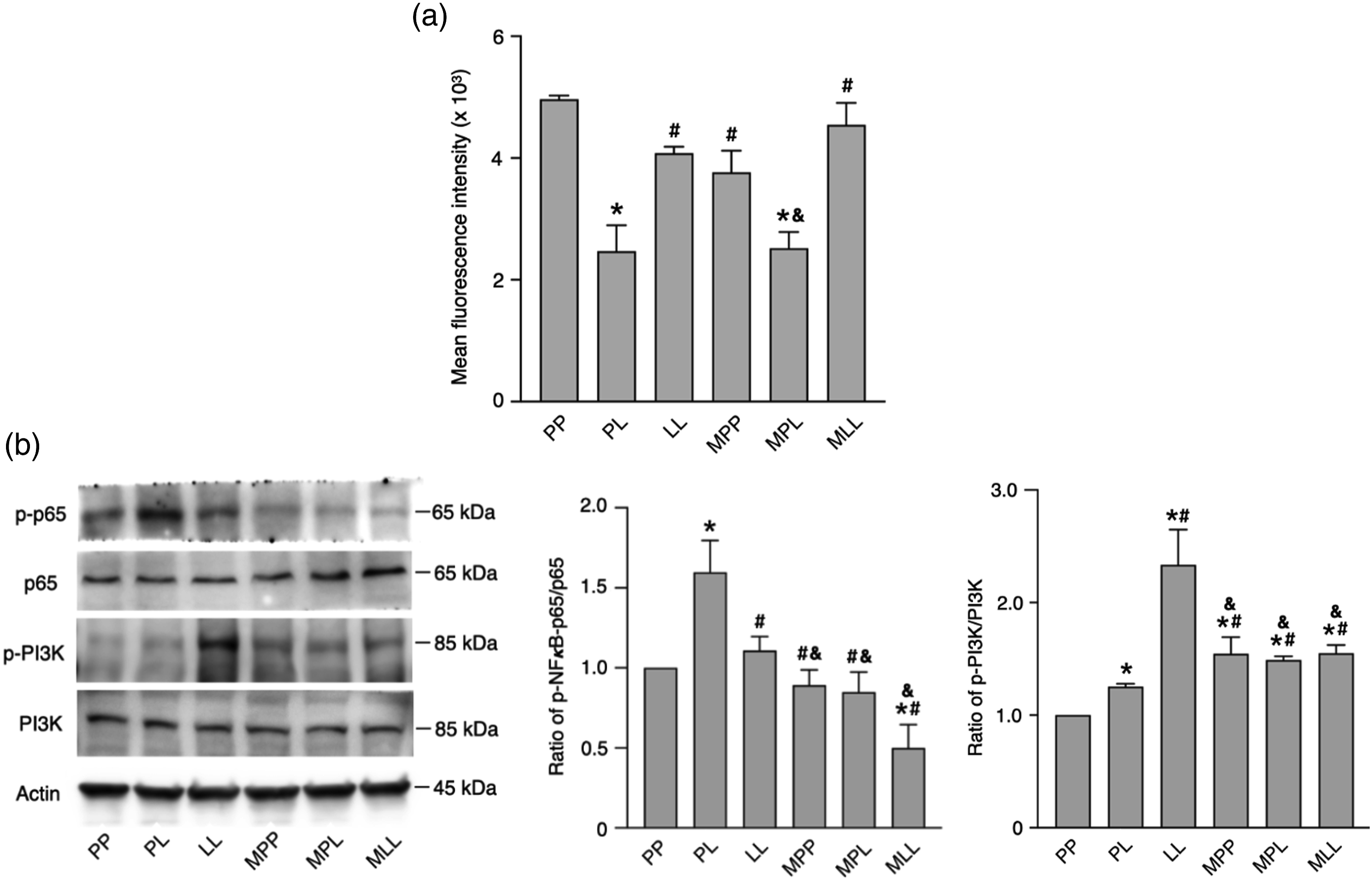
NF-κB regulates the expression of TNF-α, 2 IL-6, 2 and CD163 10 upstream. We measured the protein expression of NF-κB p65 and p-p65 (Figure 2(b), n = 4 per group). The expression of p65 were comparable in all groups. However, LSP stimulation increased the ratio of p-p65 to p65 (p < .001, PL group vs PP group), which was reduced by the development of LPS tolerance (p < .001, LL group vs PL group). MgSO4 significantly decreased the ratio of p-p65 to p65 in LPS-tolerant cells (p < .001, MLL group vs LL group), suggesting that MgSO4 enhanced LPS tolerance through inhibiting NF-κB pathway. We also measured the levels of PI3K and p-PI3K (Figure 2(b), n = 4 per group) because of the crosstalk between PI3K and NF-κB pathway. 11 PI3K expression in all group was comparable. LPS stimulation increased the ratio of p-PI3K to PI3K (p = .026, PL group vs PP group), which was further enhanced by MgSO4 treatment (p = .047, MPL group vs PL group). During LPS tolerance, the ratio of p-PI3K to PI3K was obviously increased (p < .001, LL group vs PL group), which was downregulated by MgSO4 (p < .001, MLL group vs LL group).
Discussion
In this study, we elucidated the effects of MgSO4 on LPS tolerance. MgSO4 significantly downregulated the NF-κB-triggered upregulation of TNF-α and IL-6, and upregulated CD163, suggesting that MgSO4 enhanced LPS tolerance. We also investigated the underlying mechanism of the effects of MgSO4 on LPS tolerance. Although the role of TLR4 endocytosis remains unclear, we confirmed the significant role of PI3K in the effect exhibited by MgSO4 on LPS tolerance. Based on our findings and previous studies, we draw the following inferences. First, PI3K expression was significantly upregulated during LPS tolerance. Though the role of PI3K in LPS tolerance remains unclear, PI3K may regulate the development of LPS tolerance by increasing IL-1 receptor-associated kinase-M (a negative regulator of TLR4 pathway) expression 12 and thus mitigates NF-κB pathway. Second, MgSO4 did exhibit an effect on LPS tolerance but not on LPS response repeatedly. We found that MgSO4 activated PI3K during LPS stimulation, but inhibited PI3K during LPS tolerance. Third, the effect of MgSO4 on LPS tolerance could be dependent on the modulation of the crosstalk between PI3K and NF-κB. During LPS stimulation, MgSO4 activated PI3K, but inhibited NF-κB pathway. However, during LPS tolerance, MgSO4 downregulated signaling of both PI3K and NF-κB, suggesting the modulation of the interaction between PI3K and NF-κB by MgSO4. Further investigation is needed to elucidate the underlying role of PI3K and NF-κB in effects of MgSO4 on LPS tolerance.
Our study had some limitations. For example, compared to the normal total magnesium level in human blood (0.73–1.06 mmol/L), the magnesium level in RPMI 1640 medium (0.4 mmol/L) of groups without MgSO4 treatment was relatively low. Although only free ionized magnesium, constituting 50% of total magnesium in the human blood, is biologically active, the clinical significance of inconsistent magnesium levels between the medium and human blood remains unknown. Besides, further studies are required to elucidate the in vivo effect of MgSO4 on LPS tolerance. Nevertheless, our findings confirm the immunomodulatory role of MgSO4. A previous clinical trial linked the detrimental effect of hypomagnesemia to poor outcomes of sepsis, 13 but the supplementation of MgSO4 for such cases has not reached a consensus. In clinical practice, MgSO4 supplementation could be considered for protection against inflammation in sepsis accompanied with LPS tolerance.
Conclusion
We demonstrated a novel underlying mechanism of the anti-inflammatory effect exhibited by MgSO4 via enhancing LPS tolerance. Our findings revealed the interplay between PI3K and NF-κB, which has been mostly reported in oncogenesis, but not in inflammation. Future application of MgSO4 in management of inflammatory pain or inflammatory pathologies accompanied with LPS tolerance is promising.
Footnotes
Author contributions
YYC and TYL contributed to the conception and design, data collection, analysis and interpretation, writing and critical revision of the article. WHJ and CWL contributed to data analysis and interpretation as well as writing of the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from Far-Eastern Memorial Hospital FEMH-2023-C-071 (awarded to TYL) and FEMH-2023-C-072 (awarded to YYC).
