Abstract
Do premenopausal female systemic lupus erythematosus (SLE) patients have a low incidence of hyperuricaemia (HU) as healthy premenopausal females? As of yet, there have been few studies. This study aims to investigate the serum uric acid (UA) levels of premenopausal female SLE patients and the associated clinical risk factors. 107 premenopausal female SLE patients were divided into two groups: the high UA SLE group (n = 45) and the normal UA SLE group (n = 62). In total, 50 age-matched healthy premenopausal females served as the control group. Serum UA concentration, kidney damage index, lupus index, disease activity score of lupus and serum lipid index were collected and compared between the SLE subgroups. Binary logistic regression and multiple linear regression analyses were used to analyse the association of high UA levels with clinical features. The mean UA level of the SLE group was significantly higher than that of the control group (509.73 ± 150.28 μmol/L vs 296.78 ± 69.87 μmol/L,
Introduction
Systemic lupus erythematosus (SLE) is a complex autoimmune disease of unknown aetiology that is characterized by multiple end-organ damage and predominantly affects premenopausal females aged 15–45 years. 1
Uric acid (UA) levels in premenopausal females are significantly lower than those in males, whereas in postmenopausal females, UA levels are similar to those in males. Therefore, the World Health Organization (WHO) defines hyperuricaemia (HU) in females as a UA level greater than 360 μmol/L before menopause or greater than 420 μmol/L after menopause. 2 This phenomenon may be related to the role of oestrogen in promoting urinary excretion of UA. High levels of oestrogen in premenopausal females trigger SLE. 3 There are many factors affecting the UA level in SLE patients, including oestrogen, inflammation, nephritic damage and medications. 4 In recent years, several studies have focused on the UA level in SLE patients.
Since Frocht et al. 5 found that 29% of SLE patients exhibited HU, there have been many reports regarding the UA level in SLE patients. Yang et al. 6 reported that the UA level in SLE patients may be independently associated with the development of lupus nephritis (LN). Reategui-Sokolova et al. 7 studied 186 SLE patients (102 with LN at baseline) in a 3-year follow-up and found that high UA levels appeared to predict new renal damage. However, the cohort was not an inception cohort, and the author could not be sure of the exact relationship between high UA levels and renal damage. Furthermore, none of the previous studies included a healthy control group. Calich et al. 4 investigated the UA level in 46 female SLE patients with normal renal function (18 had the presence of LN and 28 had the absence of LN) and 28 age-matched healthy females, and the results showed that an elevated UA level was an independent variable related to LN. This study confirmed the relationship between HU and LN.
However, it is still unknown whether premenopausal female SLE patients have relatively low serum UA levels because Yang et al.’s 6 study included female and male SLE patients and did not limit age. In addition, in Calich et al.’s 4 study, although its focus was on female patients, it did not limit premenopausal subjects. Therefore, this study aimed to evaluate the UA level of premenopausal female SLE patients and their characteristics and discuss the clinical significance of HU.
Subjects and methods
Subjects
In this cross-sectional study, 107 premenopausal female SLE patients aged between 15 and 45 years were recruited as the SLE group. In total, 50 age-matched healthy premenopausal females were selected as the control group. All of the SLE patients fulfilled the 1997 revised criteria of the American College of Rheumatology, 8 and the main inclusion criteria were female gender and premenopausal status. 9 Exclusion criteria were (1) pregnancy, (2) patients with severe renal damage and those under haemodialysis, (3) no serum UA measurements and (4) diabetes mellitus or severe infection. The SLE patients were divided into a high UA SLE group and a normal UA SLE group based on the UA level (HU being UA level > 360 μmol/L in premenopausal females). 2 The ethical committee of our hospital approved the study, and written informed consent was obtained from all participants in this study.
Assay of serum UA
Before the blood samples were taken, the subjects maintained a normal diet that contained few purines. Blood samples from the fasting individuals were centrifuged to obtain the serum samples using classic approaches at 4°C. Serological levels of UA were then measured using a Beckman Synchron LX20 Chemistry Analyzer (Beckman Coulter Inc., Beckman Coulter Diagnostics, Brea, CA, USA) and quantified against a standard curve that was derived from a Beckman standard UA detection reagent.
Analysis of clinical biomarkers
The following parameters were measured in SLE patients: renal indices (serum creatinine (CRE), urinary protein and blood), SLE disease indicators (white blood cells (WBCs), haemoglobin, platelet count, erythrocyte sedimentation rate (ESR), C-reactive protein, alexin C3, alexin C4 and anti-double stranded DNA (dsDNA)) and blood lipid levels (cholesterol, triglyceride (TG), low-density lipoprotein (LDL) and high-density lipoprotein (HDL) levels).
The measured clinical variables of the SLE patients were as follows: age, weight, menstrual status, current and previous renal manifestations (oedema, foamy urine, haematuria, decreasing urine output and nocturia), the Systemic Lupus Erythematosus Disease Activity Index (SLEDAI) score and current therapy (including daily doses).
The clinical variables of the control group were as follows: age, weight, serum UA, serum CRE, blood lipid levels and menstrual status.
Statistical analyses
Statistical analyses were performed using SPSS statistical software for Windows 19.0 (SPSS Inc., Chicago, IL, USA). The mean and standard deviation were used for continuous data that were normally distributed. The median and interquartile range (IQR) were used for data presented as frequencies and percentages. Student’s t-test was used to identify levels of statistical significance between groups of data. The chi-square test was used for comparison of the categorical variables. Binary linear regression analysis and multiple linear regression analysis were used to analyse the independent factors, such as the UA level in SLE patients. The level of significance was set at 0.05.
Results
General conditions
The mean ages of the SLE group and the control group were comparable (t = −0.290,
Comparative analysis of clinical biomarkers in high and normal UA groups of female SLE patients and normal controls.

UA: uric acid; SLE: systemic lupus erythematosus; CRE: creatinine; CH: cholesterol; TG: triglyceride; LDL: low-density lipoprotein; HDL: high-density lipoprotein; WBC: white blood cell; Hb: haemoglobin; PLT: platelet count; ESR: erythrocyte sedimentation rate; CRP: C-reactive protein; C3: alexin C3; C4: alexin C4; SLEDAI: Systemic Lupus Erythematosus Disease Activity Index; dsDNA: double stranded DNA; UPRO: urinary protein; UBLD: urinary blood.
Renal manifestation: current and previous renal manifestation.
In the SLE group, there were 39 cases with current or previous renal manifestations (oedema, foamy urine, haematuria, decreasing urine output and nocturia), with 29 cases in the high UA SLE group and 10 cases in the normal UA SLE group. The frequency of patients with renal manifestations in the high UA SLE group was significantly higher than that in the normal SLE group (χ2 = 26.278, P < 0.001) (Table 1). Seven SLE patients were diagnosed with renal failure, with serum CRE levels higher than 129 μmol/L. The average serum CRE level of all SLE patients was 88.93 ± 76.54 μmol/L.
In the SLE group, the mean disease duration was similar between the two subgroups (t = 0.251,
UA concentrations
The mean serum UA level of the SLE group was significantly higher than that of the control group (367.43 ± 159.86 μmol/L vs 296.78 ± 69.87 μmol/L, respectively,
Among the SLE group, the high UA SLE group had a higher mean UA level than that of the normal UA SLE group (509.73 ± 150.28 μmol/L vs 264.15 ± 48.33 μmol/L, respectively,
Effect of medication on UA levels
Sixteen SLE patients received medications, including azathioprine and cyclosporine. In the two subgroups, the frequency of use of these medications was comparable (χ2 = 0.160,
Clinical variables independently associated with serum UA levels.

UA: uric acid.
Medications that affect UA level include azathioprine and cyclosporine.
Binary logistic regression analysis of the impact of medication on UA levels; corrected R2 was 57.9%.
All of the SLE patients were being treated with prednisone, and 48 patients were taking doses greater than 20 mg/day, with 24 patients in each subgroup. The frequencies of prednisone use >20 mg/day between the two SLE subgroups were similar (χ2 = 2.254,
Laboratory parameters
In SLE group, serum CRE, cholesterol, TG, LDL levels significantly higher than those in the control group (t = −2.654, −2.012, −2.304, −2.130, respectively;
In the high UA SLE group, serum CRE, TG, LDL and ESR levels were significantly higher than those in the normal UA SLE group (t = 2.582, 3.538, 2.280 and −2.607, respectively;
The rates of positive urinary blood and anti-dsDNA were higher in the high UA SLE group than those in the normal UA SLE group (χ2 = 7.489 and 7.010, respectively;
Association between laboratory values, clinical characteristics and UA levels
In the SLE patients, the variables that measured
Independently associated clinical biomarkers with serological UA levels in SLE.
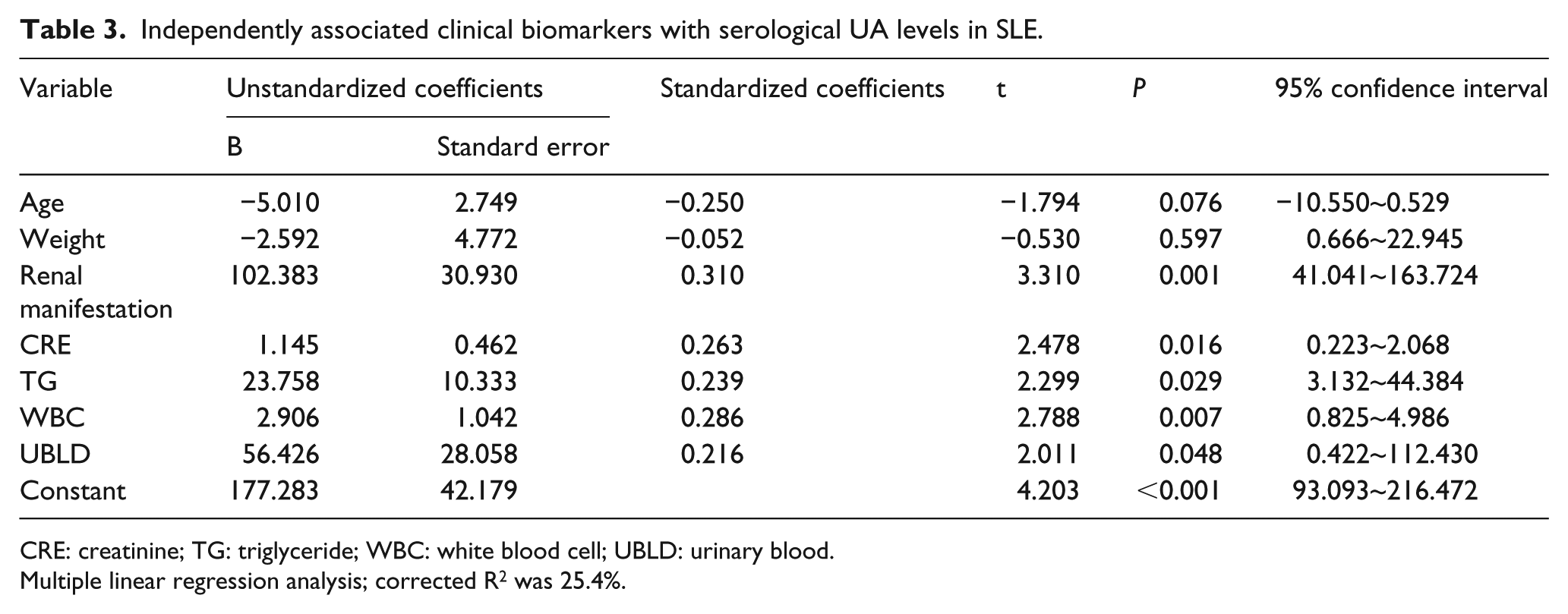
CRE: creatinine; TG: triglyceride; WBC: white blood cell; UBLD: urinary blood.
Multiple linear regression analysis; corrected R2 was 25.4%.
Discussion
It is widely known that serum UA levels may be influenced by several factors. Both Calich et al. 4 and Yang et al. 6 showed an association between elevated UA levels and LN. Particularly, Calich et al. 4 excluded the patients with renal failure, and the results still indicated that the high UA levels reflected nephritic damage. In addition to nephritic damage, there are some other factors, especially oestrogen, that can interfere with serum UA levels. It is still unknown whether premenopausal female SLE patients maintain relatively low serum UA levels. Therefore, we assessed UA levels in premenopausal female SLE patients.
In this study, HU was found in 42.06% of premenopausal female SLE patients, compared with only 14.00% in the control group. In the high UA SLE group, the UA level was 509.73 ± 150.28 μmol/L, compared with 264.15 ± 48.33 μmol/L in the normal UA SLE group. Further analysis showed that there were some factors influencing the UA level, including current or previous renal manifestations, urinary blood and CRE and serum WBC count. It would be expected that UA levels have a positive association with age. However, to our surprise, age showed no association with serum UA level. This result may be because we have restricted the age range of patients. Interestingly, although a high incidence of HU was observed in these premenopausal female SLE patients, none of them had current or previous attacks of gout, which may be because the use of glucocorticoids inhibits attacks of gout.
In recent studies on the UA levels in SLE patients, almost all the results implied that HU was related to LN. In our study, multiple linear regression analysis showed that the current or previous renal manifestations and urinary blood and CRE all showed positive associations with serum UA levels. This result demonstrated that high serum UA levels may indicate potential or existing renal damage, similar to other studies.4,6,7 Therefore, UA levels may be a useful predictor for renal damage. However, as HU could be part of a metabolic syndrome and is currently considered as a cardiovascular risk factor, it is important to monitor UA levels in SLE patients, including premenopausal females without renal failure.
The relationship between UA and renal damage may be due to two reasons. On one hand, a high UA level is known to be caused by renal failure, which is marked by elevated serum CRE. However, serum CRE rises only after losing one-third of renal function, 10 and UA rises when renal damage is latent (before CRE increases). 11 The elevation in UA levels may be caused by tubular damage resulting from proteinuria. On the other hand, UA is supposed to be a potential mediator of kidney damage. There are at least two different roles in the mechanism of renal injury induced by UA. One is that elevated UA could be contributing to renal damage through endothelial dysfunction, anti-angiogenesis, oxidative stress and disturbances in renal autoregulation. 12 The second is that UA has proinflammatory activity, including inducing the expression of MPC-1, NFκB, P38 MAPK and C-reactive protein. The two mechanisms just mentioned could trigger or accelerate the progression of renal damage occurring as a result of LN.
In conclusion, this study demonstrates that premenopausal female SLE patients have a higher incidence of HU than that of healthy premenopausal females. The elevated UA level has a positive association with potential or existing renal injury. Further study should be carried out to investigate the exact mechanisms.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This study was supported by grants from the Guangzhou Planned Project of Science and Technology (201607010145) and the Guangzhou Medical Science and Technology Project (20151A011105).
