Abstract
T cell immunoglobulin and mucin domain-containing molecule-3 (Tim-3) has been found to play important roles in systemic lupus erythematosus (SLE), however, whether Tim-3 is involved in apoptosis of NK cells in SLE remains unknown. The proportion of CD3−CD56+ NK cells and the percentage of AnnexinV+ NK cells were analyzed by flow cytometry in SLE patients and healthy controls. Tim-3 expression on NK cells was also evaluated by flow cytometry. We firstly observed a decreased proportion of NK cells and an increased proportion of apoptotic NK cells in SLE patients. The proportion of apoptotic NK cells was positively correlated with anti-dsDNA and SLEDAI. Tim-3 expression on NK cells was up-regulated in SLE patients. Further analysis showed that Tim-3 expression on NK cells was negatively correlated with the proportion of apoptotic NK cells, anti-dsDNA and SLEDAI, while positively correlated with the proportion of NK cells. The present results suggest that Tim-3 might play roles in SLE by regulating the apoptosis of NK cells and Tim-3 might serve as a potential target for the treatment of SLE.
Introduction
Systemic lupus erythematosus (SLE) is a systemic autoimmune disease. The characterization of SLE is persistent inflammation which leads to severe tissue damage in multiple organs, such as skin, kidney, the central nervous system (CNS) and so on. Alterations in immune system have been discovered in SLE, like autoantibodies produced by B cells, formation of immune complexes, and complement deficiencies.1,2 Given the genotype and clinical presentation of SLE is broadly heterogeneous, there is no single drug which is capable of improving all manifestations. A better understanding of pathogenetic mechanisms in immune system of SLE could result in a better investigation of biological therapy targets. Therefore, to explore the crucial immune molecules and cells in the pathogenesis of SLE would illuminate novel targets for immunotherapy.
NK cells play important roles in immune system. Either the reduced NK cell number or impaired NK cell function might contribute to autoimmune disease, immune deficiency, or malignancy.3–9 Previous research suggests that NK cells may participate in the pathogenesis of SLE through several mechanisms, such as increased capacity of producing IFN-γ, decreased ADCC, and altered natural cytotoxicity.10–12 Published evidences have shown that the proportion and absolute number of peripheral blood NK cells are greatly decreased in SLE.12,13 However, it remains unknown whether NK cells have a protective or a disease-promoting effect in SLE.5,14
Numerous evidence indicates that NK cell function is tightly regulated by receptor-ligand interactions. 15 The molecular alterations responsible for NK cell defects or NK cell deficiencies are largely unknown in SLE. Tim-3 (T cell immunoglobulin and mucin domain containing molecule-3), which is expressed on various immune cells including NK cells, T cells and macrophages, has been proved an important immune checkpoint molecule.16–18 It was demonstrated that the aberrant Tim-3 expression plays key roles in several types of human diseases (e.g. autoimmune diseases, infection and tumor) by affecting innate and/or adaptive responses.19–20 And all these findings suggested that Tim-3 might be an important target in immune checkpoint therapy. 21 More recently, we disclosed a dysfunctional effect of Tim-3 on CD14+ monocytes in mediating the engulfment of apoptotic cells in SLE, which strongly suggests the involvement of Tim-3 in the pathogenesis of SLE. 22 Our previous results from HBV-infected patients indicated that abundant Tim-3 expression on NK cells may contribute to the inhibition of NK cells function and the persistence of HBV infection. 17 In addition, we also detected that Tim-3 plays an important role in NK deficency by promoting TNF-α induced NK apoptosis during atherogenesis. 23 However, it is still unclear about the effects of Tim-3 on NK-cell status of SLE patients.
In the present study, we first detected a decreased proportion of NK cells and an increased proportion of AnnexinV+NK cells (apoptotic NK cells) in periphery blood of SLE patients, which suggested the defects of NK cells, might be the result of apoptosis. To confirm whether Tim-3 played roles in NK apoptosis, we examined Tim-3 expression on NK cells and analyzed their correlation with clinical indices of SLE patients. Our results showed that Tim-3 expression on NK cells in SLE patients were significantly higher than those in control subjects. Tim-3 expression on peripheral NK cells in SLE negatively correlated with the disease activity and the proportion of AnnexinV+NK cells, while positively correlated with the proportion of NK cells. Our present results give new insights into the innate immune system and might provide new evidences for clinical practice of Tim-3-based immunotherapy in SLE.
Materials and methods
Subjects
This was a prospective study. All 44 incipient SLE patients enrolled for this study were from Qilu Hospital of Shandong University fulfilling the criteria of the American College of Rheumatology 24 between May and September 2018. Sex and age-matched 30 control subjects were recruited from medical examination center of Qilu Hospital of Shandong University during the study period. Individuals who were positive for hepatitis B virus, hepatitis C virus, HIV, or attacked with other autoimmune diseases, active infection, and blood system diseases were excluded before sampling. None of the enrolled subjects have taken glucocorticoids or immunosuppressive drugs before sampling. Fresh peripheral blood cells from all subjects were collected for flow cytometry assay. Cells from 30 SLE patients and 30 control subjects were collected for apoptosis detection, and cells from 44 SLE patients and 30 control subjects were analyzed for Tim-3 expression. Disease activity of SLE patients was assessed by anti-dsDNA (IU/ml), complements (C3 (g/l), C4 (g/l)) and the SLE disease activity index (SLEDAI) score. We obtained 38 SLE patients with anti-dsDNA, 34 SLE patients with C3 and C4, and 44 SLE patients with the SLEDAI scores. Informed consent was obtained from all participants and the study received approval from the Ethics Committee on Scientific Research of Shandong University Qilu Hospital. The characteristics of both SLE patients and control subjects were summarized in Table 1.
Clinical characteristics of SLE patients and control subjects.
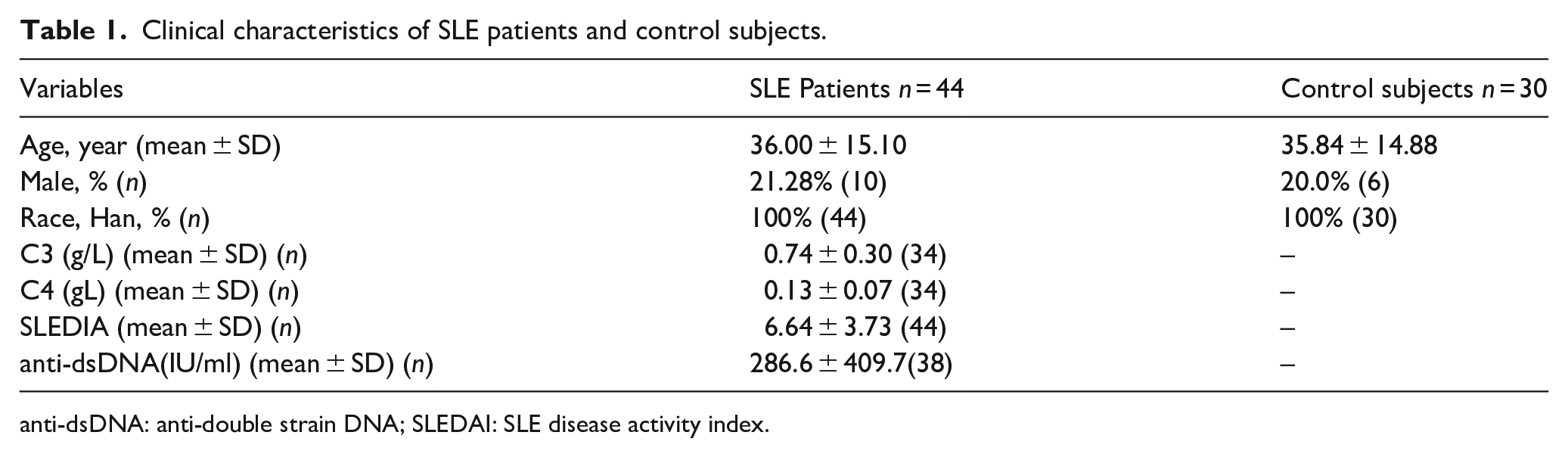
anti-dsDNA: anti-double strain DNA; SLEDAI: SLE disease activity index.
Flow cytometry
100 μl blood sample was incubated for 30 min at 4°C in a dark room with PEcy7-conjugated anti-human CD56 (Biolegend), Percpcy5.5-conjugated anti-human CD3 (Biolegend) and PE-conjugated anti-human Tim-3 (Biolegend) and then submitted to RBC lysis buffer (Beyotime, China) to get rid of red blood cells (RBC) according to the manufacturer’s instructions. CD3-CD56+cells are defined as NK cells. For apoptotic cell examination, Cells were resuspended in 100 μl AnnexinV Binding buffer, and 5 μl of FITC AnnexinV reagent (Dakewe, China). Each tube was gently vortexed and incubated for 15 min at RT (25°C) in the dark room. 400 μl AnnexinV Binding buffer was added to each tube. AnnexinV+ CD3−CD56+cells (AnnexinV+ NK cells) were defined as apoptotic NK cells. At least 10,000 cells were acquired on BD FACS Canto II Flow Cytometer. All the data was analyzed by using BD FACS Diva software (BD Biosciences, San Jose, CA, USA).
Statistical analysis
GraphPad Prism 5 (GraphPad Software Inc., San Diego, CA) was used to analyze all the data. Student’s
Results
Decreased frequency and increased proportion of circulating apoptotic NK cells in SLE patients
The immune state of NK cells in SLE is poorly understood, so we detected the proportion of NK cells in the peripheral blood lymphocytes by flow cytometry. As shown in Figure 1(a) to (c), the proportion of NK cells was significantly lower in SLE patients than control subjects. Previous reports25–27 showed that there is a significantly higher level of AnnexinV+ cells in peripheral blood from SLE patients compared to control subjects. We postulated that the decreased frequency of circulating NK cells might be the result of apoptosis. To confirm this hypothesis, the proportions of AnnexinV+NK cells in the peripheral blood were determined by flow cytometry. The results showed an increased proportion of apoptotic NK cells in SLE patients compared with control subjects (Figure 1(d) and (e)). All these data indicated that the increased apoptosis might be the cause of decreased proportion of NK cells.

Decreased NK cells frequency and increased proportion of circulating AnnexinV+NK cells in SLE patients. Summary data of percentage of CD3-CD56+NK cells from both SLE patients and control subjects (a). The gating strategy for identification of lymphocytes, CD3-cells (b) and CD3-CD56+NK cells (c). Summary data of percentage of Annexin V+ NK cells from both SLE patients and control subjects (d). Representative dot plots of Annexin V expression on NK cells from both a SLE patient and a control subject (e).
The percentage of apoptotic NK cell in SLE patients is associated with disease activity
It is well known that high levels of anti-dsDNA, SLEDAI score and lower levels of C3, C4 are in accordance with of SLE disease activity. We then further analyzed the correlation of apoptotic NK cells with disease activity in SLE patients. The precentage of apoptotic NK cells in peripheral blood was positively correlated with SLEDAI scores and anti-dsDNA (Figure 2(a) and (b)). However, it showed no obvious correlation with C3 and C4 (Figure 2(c) and (d)).

AnnexinV+NK cell populations are associated with disease activity in SLE patients. Positive correlation is shown between AnnexinV expression on NK cells and anti-dsDNAs (
Augmented Tim-3 on NK cells negatively correlates with AnnexinV expression and disease activity in SLE patients
Our previous results showed that Tim-3 on macrophages played roles in the pathogenesis of SLE, 22 however, whether Tim-3 expression on NK cells involved in SLE is still unclear. To our interest, the proportion of Tim-3+NK cells in SLE patients was significantly higher than that of control subjects (Figure 3(a) and (b)). Consistently, MFI (mean fluorescence intensity) of Tim-3 on NK cells was also up-regulated in SLE patients compared to control subjects (Figure 3(c)).

Tim-3 expression on NK cells is increased in SLE. Summary data of percentage (a) and MFI (c) of Tim-3 expression on NK cells from SLE patients and control subjects, and (b) representative dot plots of Tim-3 expression on NK cells from both a SLE patient and a control subject.
We further analyzed the correlation between Tim-3 expression and the proportion of apoptotic NK cells in SLE patients. Correlation analysis showed that the proportion of Tim-3+NK cells was positively correlated with the proportion of NK cells, while, negatively correlated with apoptotic NK cells in SLE patients (Figure 4(a) and (b)).To explore whether Tim-3 on NK cells was correlated to disease activity in SLE patients, We then analyzed the correlation of Tim-3+NK cells with SLEDAI scores, anti-dsDNA, C3 and C4. It is showed that the percentage of Tim-3+NK cells was negatively correlated with SLEDAI scores and anti-dsDNA, while positively correlated with C3 and C4. All these results indicated that Tim-3 might involve in the pathogenesis of SLE by regulating the apoptosis of NK cells.

Augmented Tim-3 on NK cells negatively correlates with AnnexinV expression and disease activity in SLE patients. Tim-3 on NK cells was positively correlated with the proportion of NK cells (
Discussion
This study represents our attempt to explore the numerical deficiencies of NK cells in SLE patients. Our results showed that NK cell deficiency is related to apoptosis (supplemental Figure 1). In particular, the apoptosis of NK cells was found to be associated with SLE disease activity. We also detected that NK cell apoptosis was associated with elevated Tim-3 expression in SLE.
NK cells played important roles in the regulation of autoimmunity. 28 Previous studies have described an NK cell deficiency in SLE.29,30 Consistently, in this study, we also found reduced circulating NK cells in SLE patients. The mechanisms underlying these numerical deficiencies of NK cells in SLE is still unknown. It is reported that circulating haemopoietic stem cells (HSC) numbers are decreased in SLE patients 31 and HSCs have a defective differentiation into mature NK cells than did those from control subjects. 32 However, the defective differentiation of HSCs into NK cells was not the only explain. There is also evidence that HSCs in the bone marrow of active SLE patients have an increased propensity of apoptosis. 33 NK cells are more susceptible to apoptosis in the complex serum factor. As showed that combinations of IL-18 and IL-15 or IL-18 and IL-12 could induce NK cell death in patients with systemic autoimmune diseases. 34 It has previously been reported that NK cells were susceptible to apoptosis with the involvement of CD16 after stimulated with IL-2 or IL-12, which suggests that immune complexes might be responsible for NK cell depletion. 35 All these observations indicate that NK cell apoptosis induced by circulating immune complexes or serum cytokines might contribute to NK cell deficiency in SLE. Consistently, in the present study, our data shows that apoptotic NK cells are greatly increased in SLE patients. Moreover, the percentage of peripheral apoptotic NK cells is positively correlated with disease activity (e.g. anti-dsDNA and SLEDAI). Interestingly, no significant correlations were found between the percentage of peripheral apoptotic NK cells and complement levels. These findings suggest that it might be the chronic inflammatory activity rather than circulating immune complexes leads to NK cells apoptosis during SLE process. However, the question of why NK cells exhibit increased susceptibility toward apoptosis in SLE patient needs further be speculated.
Tim-3 is a well-known co-inhibitory molecule that is highly expressed on NK cells in series of diseases (e.g. Chronic hepatitis B and atherosclerosis).17,23 To our interest, in this study, we found that Tim-3 expression was increased on NK cells from SLE patients compared to control subjects. The increased expression of Tim-3 may be due to activation of peripheral NK cells by some serum factors in SLE like we have confirmed in our previous studies.22,36 Furthermore, the factors that are responsible for the induction of Tim-3 expression on NK cells in SLE need to be further explored.
Tim-3 plays critical roles in regulating immune activities during infection, cancer, and organ transplantation 37 by interacting with its ligand to induce T cell apoptosis. Consistently, our previous results demonstrated augmented Tim-3 expression on peripheral NK cells in atherosclerosis and blockade of Tim-3 reduced the TNF-α-induced death of NK cells. 23 Although Tim-3 has been regarded as an inhibitory molecule, Tim-3 might play various, or even opposing roles. It was reported that Tim-3 may synergize with the TLR (Toll-like receptor) system to influence inflammatory conditions by promoting Th1 immune activity. 18 Tim-3 was higher expressed in DSCs (decidual stromal cells) from normal pregnancy. Tim-3, in turn, protected DSCs from TLR-mediated apoptosis in an ERK1/2 pathway-dependent manner. 38 More interestingly, in the present data, correlation analysis showed the following observations. Tim-3 expression on NK cells was positively correlated with the proportion of NK cells in periphery blood lymphocytes and serum complement levels from SLE patients. However, augmented Tim-3 expression on NK cells in SLE patients showed a negative correlation with the percentage of AnnexinV+ cells, SLEDAI and anti-dsDNA. All these suggest that Tim-3 might play protective roles in the apoptosis of NK cells and SLE progression. Phenotypic analyses of NK cells in SLE patients showed an increase of the activating receptor NKG2C and CD69. 39 The activation of NK cells in SLE might result in up-regulation of Tim-3 expression. Thus, we hypothesized that Tim-3 represents a negative feedback mechanism employed by NK cells to avoid activated induced apoptosis, which might be helpful in preventing NK depletion in SLE. Nonetheless, further experiments are still needed to identify the effect of Tim-3 on NK apoptosis in SLE progression.
In conclusion, the expression of Tim-3 is up-regulated on NK cells, which is more susceptible to apoptosis in SLE patients. The correlation analysis showed the higher Tim-3 expression on NK cells, the lower apoptosis on NK cells and the lower disease activity. Our data suggest that Tim-3 might be induced by activated NK cells to inhibit apoptosis of NK cells in SLE progression. However, there are some liminations in the study. First, we just included the subjects from May to September 2018, according to the inclusion and exclusion criteria; however, we did not calculate and justify the sample size. Second, the status of NK cells was not explored precisely and the mechanism that Tim-3 was up-regulated and the signal pathways that regulate the apoptosis of NK cells were still not explained. Further elucidation of the exact regulative mechanism of Tim-3 expression on NK cells apoptosis might be helpful in novel approaches to SLE immunotherapy.
Supplemental Material
sj-tif-1-eji-10.1177_20587392211000570 – Supplemental material for Tim-3 associated with apoptotic NK cells and disease activity in SLE
Supplemental material, sj-tif-1-eji-10.1177_20587392211000570 for Tim-3 associated with apoptotic NK cells and disease activity in SLE by Di Zhao, Xiao Yang, Jie Zhang and Yi Zhang in European Journal of Inflammation
Supplemental Material
sj-tif-2-eji-10.1177_20587392211000570 – Supplemental material for Tim-3 associated with apoptotic NK cells and disease activity in SLE
Supplemental material, sj-tif-2-eji-10.1177_20587392211000570 for Tim-3 associated with apoptotic NK cells and disease activity in SLE by Di Zhao, Xiao Yang, Jie Zhang and Yi Zhang in European Journal of Inflammation
Footnotes
Author contributions
Di Zhao performed the experiments. Xiao Yang helped in patients’ recruitment and obtaining clinical history. Jie Zhang helped in data analysis and in developing experiment methods. Di Zhao and Yi Zhang authored and drafted the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Nature Science Foundation of China (No. 81601419) and the Key Research and Development Plan of Shandong Province (No. 2019GSF108242, No. 2019GSF108013).
Ethics approval
Ethical approval for this study was obtained from the Ethics Committee on Scientific Research of Shandong University Qilu Hospital (KYLL-2016(KS)-117).
Informed consent
Written informed consent was obtained from all subjects before the study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
