Abstract
This study investigated the effects of forsythiaside on the acute inflammatory response induced by
Introduction
Colibacillosis, a syndrome caused by
In the current study, the changes in the content of total protein and NO, and the levels of pro-inflammatory cytokines, including LITAF, IL-1β, IL-17, and IL-6, were examined to investigate the effects of the oral administration of different dosages of forsythiaside on acute liver inflammation in chicken induced by
Materials and methods
Chemicals and reagents
Forsythiaside, with a purity of 98.0%, was obtained from Herbpurify Co., Ltd. (Chengdu, China).
Ethical statements
All chicken experiments were performed according to the Northeast Agricultural University’s animal management approach and approved by the Institutional Animal Care and Use Committee of Northeast Agricultural University (SRM-10).
Animals and treatment
A total of 100 1-day-old commercial Arbor Acres broiler chickens were obtained from a local hatchery. The chickens were housed in electrically heated battery brooders and fed a commercial diet (220 g crude protein per kg and 12.55 MJ metabolizable energy per kg) ad libitum for 7 days. Then, about 80 7-day-old chickens were chosen on the basis of body weight to achieve body weight uniformity, and then were reared in stainless-steel wire cages in a temperature (25°C) and light (24 h/day) controlled room.
At the age of 15 days, 60 chickens with the similar weight were chosen and randomly assigned to three groups. Subsequently, forsythiaside (30 and 60 mg/kg of BW) was orally administered from day 15 to 21. The forsythiaside dose and the period of administration employed in the present investigation were chosen based on recent studies by Cheng et al.
7
The forsythiaside was mixed with water and was orally administered to each chicken through a syringe. The control group (0 mg/kg of BW) was orally administered the same volume of distilled water from day 15 to 21. At the age of 21 days, among the chickens in each group, 10 replicates were injected intravenously with either 200 μg/kg body weight of
Preparation of the liver tissue homogenate
A 0.5-g sample of liver was homogenized with 4.5 mL phosphate buffer saline (PBS, pH 7.4) using a hand-held electric tissue homogenizer in an ice bath. The resulting suspension was centrifuged at 12,000 × g, at 4°C for 30 min. Then, the supernatant was collected and stored at −80°C for further analyses.
Protein assay
The total protein concentration in the supernatant was determined using biuret method on an auto-analyzer (Technicon RA-100, USA) at wavelength of 540 nm. Pars Azmoon commercial kits (Tehran, Iran) were used according to the manufacturer’s instructions. The values are expressed in mg protein/mL of tissue homogenate.
Determination of NO levels
Production of NO in liver was measured by the quantification of its related end products, nitrite/nitrate. 8 Briefly, 100 μL of tissue homogenate was mixed with 100 μL of Griess reagent (0.1% N-(1-naphthyl)ethylenediamine dihydrochloride; 1% sulfanilamide in 5% phosphoric acid; Sigma) in a 96-well ELISA plate for 15 min at room temperature. The plate was read at 540 nm on an ELISA microplate reader (Thermo Scientific Multiskan FC, USA). The results are expressed as μmol/g protein.
Determination of LITAF, IL-1β, IL-17, and IL-6
The tissue samples were centrifuged at 3000 × g, at 4°C for 10 min. Then, the cytokine concentrations of LITAF, IL-1β, IL-17, and IL-6 were determined using ELISA assay. 7 The commercial sandwich-type ELISA kits (KMC0011 m IL-1β, KMC0061 m IL-6, KMC3011 m LITAF, and KMC0081 m IL-17; Biosource International, CA, USA) were used according to the manufacturer’s instructions.
Determination of LITAF, IL-1β, IL-17, IL-6, and iNOS mRNA expression
The detection of the mRNA expression of IL-1β, IL-6, LITAF, IL-17, and iNOS was performed by real-time polymerase chain reaction (RT-PCR). First, total RNA was isolated from the liver sample on ice using the TRIzol reagent according to the manufacturer’s instructions (Takara, Dalian, China). Second, 2 μg of total RNA was reverse-transcribed to complementary DNA (cDNA) using an oligo-dT primer in the presence of RNA guard and Superscript II reverse transcriptase (Thermo Scientific, MA, USA) according to the manufacturer’s instructions. Finally, quantitative polymerase chain reaction (qPCR) was conducted using a method based on the study of Cheng et al.
7
Briefly, 10 μL of LightCycler FastStart DNA Master SYBR Green I, 1.2 μL of cDNA, 0.6 μL of each primer, and 7.6 μL of ddH2O were mixed in 96-well plates (Roche Diagnostics). Then, standard cycling conditions were used, including a pre-amplification step of 95°C for 10 min, followed by amplification for 40 cycles of 95°C for 15 s, 60°C for 1 min, and 72°C for 20 s. All the samples were analyzed in triplicate. The mean cycle threshold (Ct) was calculated for the target and housekeeping (
Statistical analysis
A one-way analysis of variance (ANOVA; SPSS 18.0 Software) was used to test the effects of LPS and forsythiaside treatment on the dependent variables. The differences among each treatment group were tested by Duncan’s multiple-range test.
Results
Effect of forsythiaside on the level of the total protein
The effect of forsythiaside on the level of total liver protein after LPS injection or saline injection is shown in Figure 1(a). In the LPS group without forsythiaside treatment, the level of total protein in the liver was significantly increased to 1.59 ± 0.1 mg/mL compared to control group (
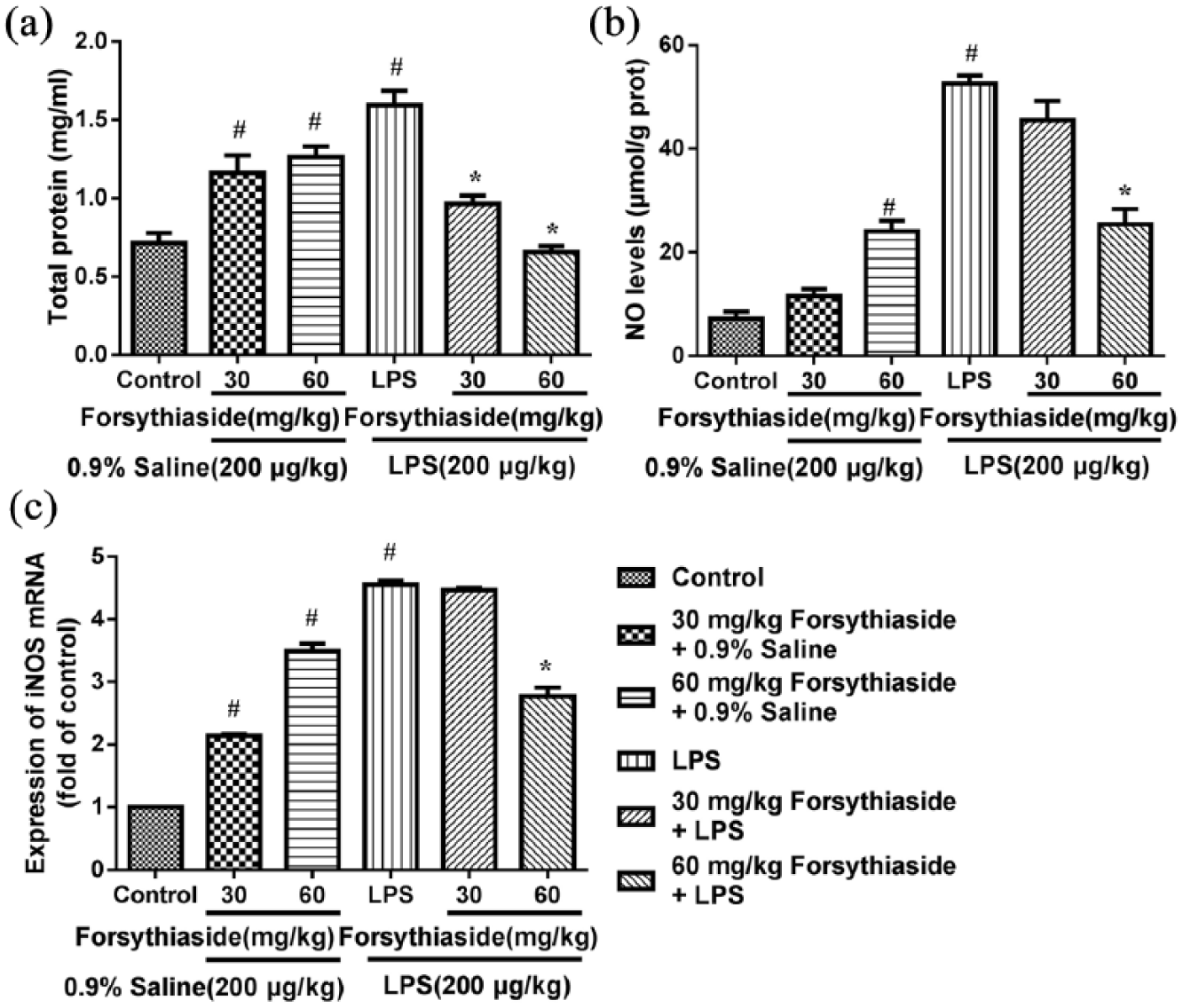
Effect of forsythiaside on the levels of total protein, NO, and iNOS mRNA expression in the liver: (a) total protein, (b) NO, and (c) iNOS.
Effect of forsythiaside on the levels of NO and iNOS
The effect of forsythiaside on the levels of the NO and iNOS in the liver after LPS or saline injection is shown in Figure 1(b) and (c). LPS treatment significantly increased NO production in the liver tissues compared to control group (Figure 1(b)), in which NO concentrations were 52.56 ± 1.57 and 18.08 ± 0.87 μmol/g protein, respectively. Then, the level of NO induced with LPS was significantly inhibited with 60 mg/kg forsythiaside treatment (
Effect of forsythiaside on the levels of LITAF, IL-1β, IL-17, and IL-6
The effect of forsythiaside on the concentration of pro-inflammatory cytokines, including LITAF, IL-1β, IL-17, and IL-6 in the liver after the LPS or saline injection is shown in Figure 2. LPS significantly induced the release of IL-1β, IL-6, and IL-17 in liver compared with the control group (
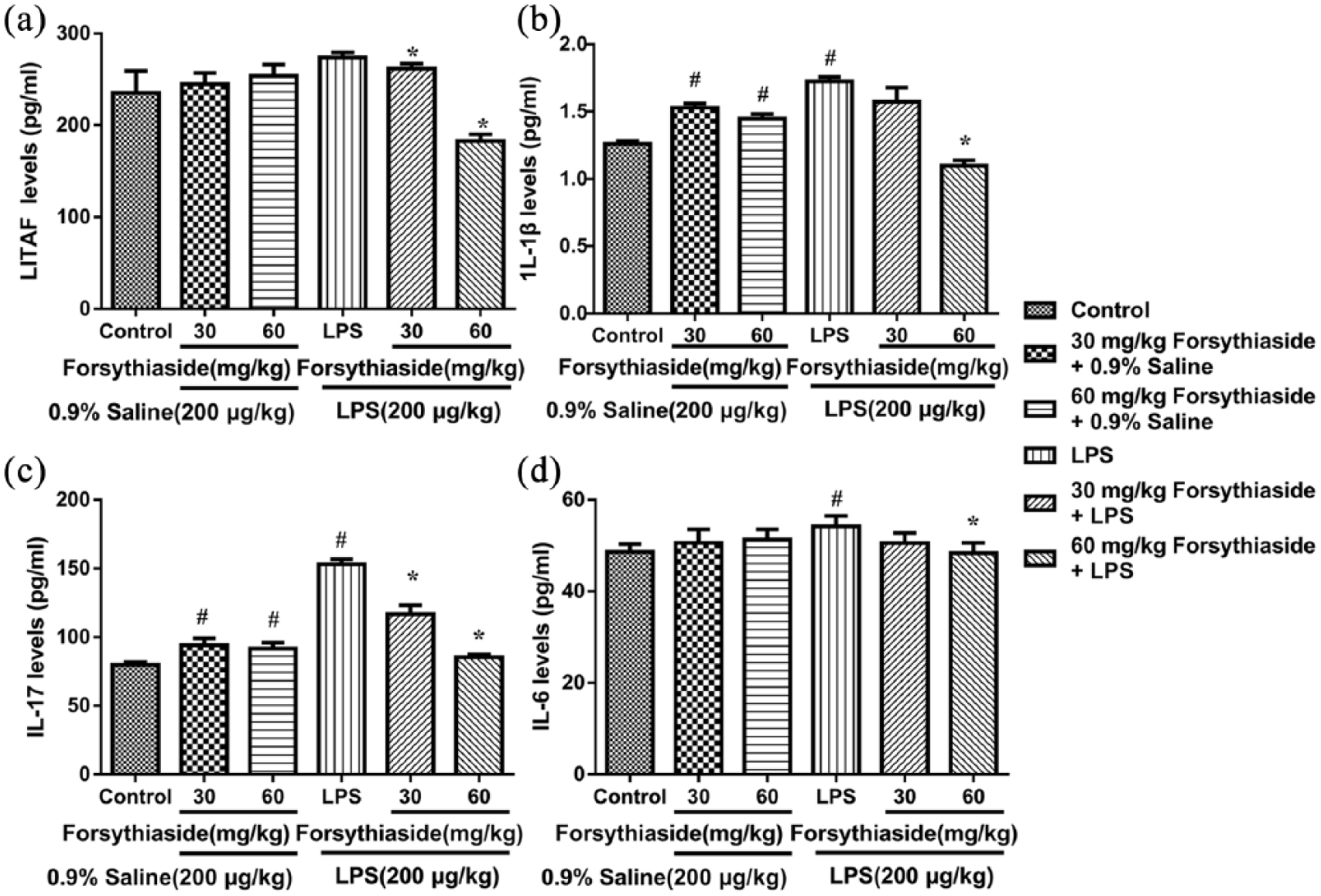
Effect of forsythiaside on the levels of LITAF, IL-1β, IL-17, and IL-6 in the liver: (a) LITAF, (b) IL-1β, (c) IL-17, and (d) IL-6.
Effect of forsythiaside on the mRNA expression of LITAF, IL-1β, IL-17, and IL-6
After the LPS injection, the mRNA expression of LITAF, IL-1β, IL-17, and IL-6 in the liver was significantly up-regulated to 1.13 ± 0.01-, 4.1 ± 0.34-, 2.43 ± 0.04-, and 2.38 ± 0.50-fold the expression levels of the control group, respectively (Figure 3). In addition, the mRNA expression levels of IL-1β and IL-17 in the liver were significantly increased in the saline groups that were pre-treated with forsythiaside (30 or 60 mg/kg). However, in the LPS groups, the pre-treatment with 60 mg/kg of forsythiaside significantly attenuated the mRNA expression of LITAF, IL-1β, IL-17, and IL-6 (
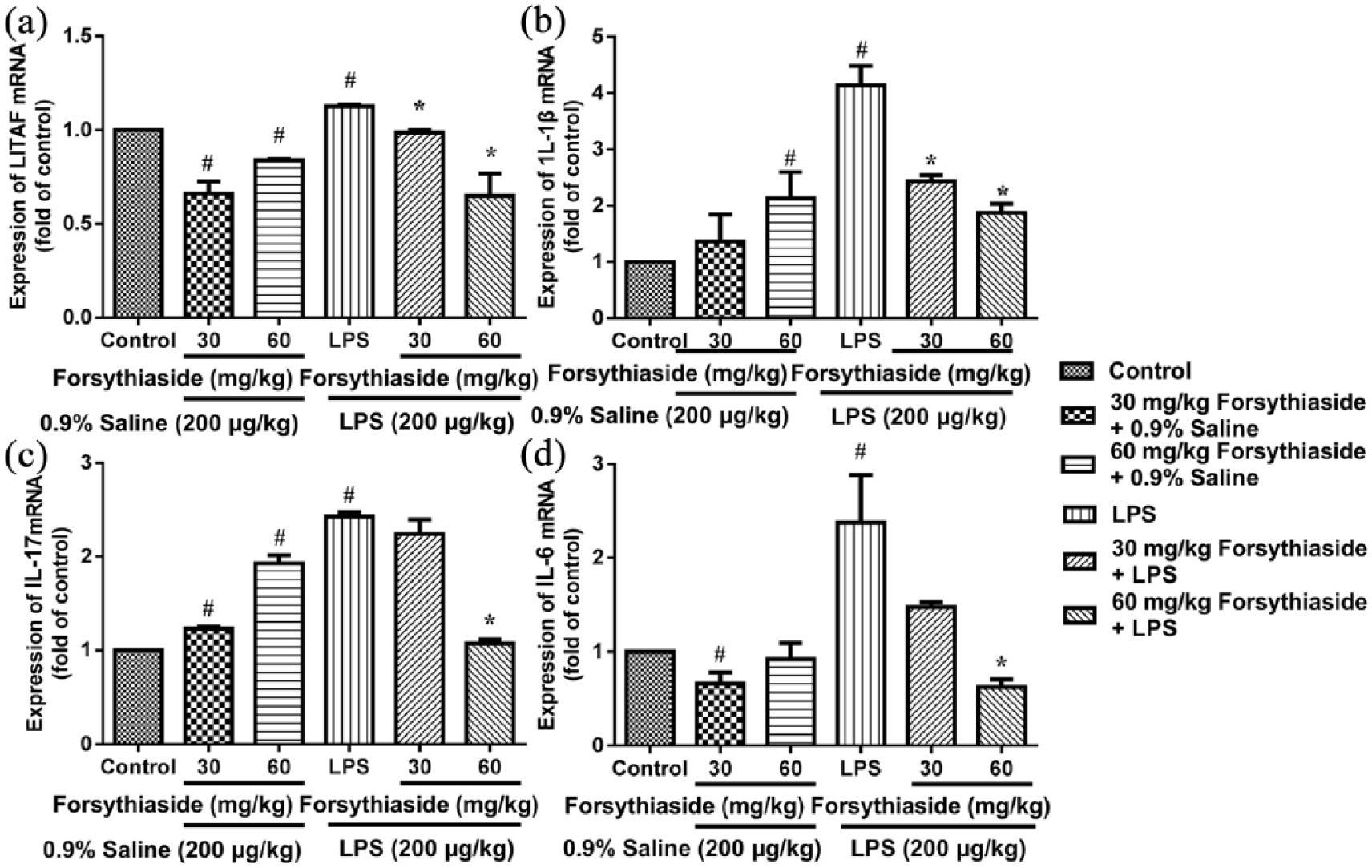
Effect of forsythiaside on the levels of mRNA expression of LITAF, IL-1β, IL-17, and IL-6 in the liver: (a) LITAF, (b) IL-1β, (c) IL-17, and (d) IL-6.
Discussion
In this study, the effects of forsythiaside on
Data from LPS-untreated groups provided some evidence that forsythiaside may regulate the host immune response. The results showed that forsythiaside alone seemed to significantly increase the total protein content, NO/iNOS, and the levels of IL-1β and IL-17 in the liver of chicken, while IL-6 and LITAF contents were not markedly affected by forsythiaside. This finding may be attributed to the immediate immune response by increasing the activation of macrophages.
In addition, the production of NO and the expression of iNOS in the LPS-treated group showed a significant enhancement compared with the control group. However, the higher dose of forsythiaside significantly reduced mRNA expression levels of NO and iNOS induced by LPS. These results suggested that forsythiaside reduced NO production likely by suppressing the expression of iNOS mRNA under LPS-induced liver inflammation. Thus, the inhibition of the iNOS enzyme may be one explanation of the anti-inflammatory mechanism of forsythiaside.
The pro-inflammatory cytokines such as IL-1β, LITAF, and IL-6 are considered pivotal mediators of the acute phase response.
10
The production of an appropriate amount of IL-1β and IL-6 protects against infection, but if are produced in a deregulated fashion, they might have a detrimental effect.
11
In addition, chick IL-1β induces the mRNA expression of several pro-inflammatory cytokines, including IL-1β, IL-6, LITAF, and INF-γ in chickens infected with
In conclusion, the current research shows that forsythiaside attenuated LPS-induced injury in the liver of chickens due to its anti-inflammatory properties. The protective effects of forsythiaside on
Footnotes
Acknowledgements
Yu Yang and Yanhua Li conceived the analysis; Yu Yang conducted the numerical calculations; and all authors discussed the results and reviewed the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Earmarked Fund for China Agriculture Research System (35), Heilongjiang Province Natural Science Foundation (C2018013), and Project Science and Technology Bureau of Harbin (2017RAQXJ033).
