Abstract
Reactive airway dysfunction syndrome (RADS) has a clinical manifestation similar to asthma, but some features are different between both the diseases. To probe the effect of CD19+ cells in RADS pathogenesis by inhalation of sulfur dioxide (SO2), rats were exposed to SO2 at 600 ppm for 2 h per day for 7 days and the CD19 expression in lung tissue was detected both at mRNA and protein levels by RT-PCR and western blot. The percentages of CD19+ and CD19+ CD23+ cells were measured by flow cytometry. IgG, IgA, and IgE in serum and bronchoalveolar lavage fluid (BALF) were detected by enzyme-linked immunosorbent assay (ELISA). Histological analysis was performed. The results showed that expression of CD19 in SO2 exposure group was lower than that in the control both at mRNA and protein levels (P < 0.05). Flow cytometry analysis showed that the percentages of CD19+ and CD19+ CD23+ were significantly lower in the SO2 exposed group than that in the control (P < 0.05). There was no difference between the control and SO2 exposed groups in both serum and BALF levels of IgG, IgA, and IgE. Pathological changes, such as chronic bronchitis, local alveolar hemorrhage, and lymphocytes infiltration were observed in SO2 exposed. RADS is a non-immunogenicity, chronic airway inflammatory disease caused by irritation of harmful factor and manifests as airway hyperresposiveness.
Reactive airway dysfunction syndrome (RADS), which is also called irritant-induced asthma, is included in occupational asthma by many scholars because its onset is often related to occupational exposure. 1 But with further study on the pathogenesis of asthma in recent years, RADS was found to be different from past classic asthma and its onset is rapid and it can be caused by inhalation of high-concentration irritation gas without features of immunological response and allergy. Asthma is thought to be an immune associated disease, which occurs in atopic individuals. 2 At first, it is necessary that the immune system of a body is sensitized by exposure to an allergen and asthma is triggered when the immune system is exposed to the same allergen. The patient with asthma has a series of symptoms such as wheeze. However, both diseases share many similar features. The clinical manifestations of both diseases are much similar and both are common characteristic of asthma, that is, airway hyperresponsiveness. Therefore, RADS can be thought as a special type of occupational asthma, or an earlier subset developed to asthma. 3 Most factors for inducing RADS are chemical gases and more definite gases are chlorine, acetic acid, hydrochloric acid, sulfuric acid, hydrogen sulfate, sulfur dioxide, combustion product of oil paint, welding fume, lachrymator gas, and so on. 1
Because of the similarity of clinical manifestations between RADS and asthma, it is necessary to clarify whether the basis of inflammation in RADS is similar to that in asthma, in order to provide a basis for treatment of RADS. Owing to the non-immune feature of RADS, the inflammation cells and cytokines in RADS may be significantly different from those in asthma and further experiments will be needed to make certain the exact mechanism.
We attempt to probe the effect of CD19+ cells in RADS pathogenesis by inhalation of SO2 in high concentrations in rat models.
Materials and methods
Chemicals and reagents
SO2 gas: the product of Beijing Ya-nan Gas Scientific and Technology Corporation Ltd with a purity of 99.9%. Collagenase Ⅳ (Sigma, USA). Anti-CD19-FITC monoclonal antibody, anti-CD16 polyclonal antibody, rabbit anti-rat CD3 monoclonal antibody, goat anti-rat IgG antibody, goat anti-rat IgA antibody, rat IgE enzyme-linked immunosorbent assay (ELISA) detection kit were purchased from Abcam Corporation Ltd. Anti-CD32 antibody, Goat ABC staining system, purchased from Santa Cruze Corporation Ltd. Mouse anti-rat CD19 monoclonal antibody, R&D Corporation Ltd. Rat IgG ELISA and IgA ELISA kit, purchased from eBioscience Corporation Ltd. Rabbit anti-CD23-PE antibody, purchased from Bioss Corporation Ltd. Ttizol reagent, the product of Takara Corporation Ltd.
Experimental animals
Twenty SD male rats with a body weight of 180–200 g were purchased from Liaoning Changsheng Biotechnology Corporation, Ltd, China. The experiment protocol complied with Chinese regulations of Administration of Affair about experimental animals and was approved by the Experimental Animal Ethics Committee of General Hospital of Shenyang Military Command.
Exposure to SO2
Exposure chamber
We designed an exposure chamber of SO2 based on a model published previously with a slight modification.4,5 The devise consists of SO2 source, air pump, intake port, SO2 chamber and SO2 detector, and some connective tubes and valves. The SO2 was diluted with fresh air at the intake port of the chamber to yield the desired concentrations. The desired SO2 was delivered to animals via a tube positioned at the upper level of the chamber and distributed homogeneously via a fan in each chamber. The concentration of SO2 was determined in a real-time manner by an SO2 sensor (JSA5- SO2 sensor, Shenzhen Ji-shun-an Technology Co., Ltd., China). The concentration of SO2 in the chamber was adjusted by opening and closing a valve between intake port and SO2 chamber according to the quantitative value of the SO2 sensor.
Exposure protocol
The SD rats were divided randomly into two groups with 10 rats in each group. The rats of the SO2 exposure group were placed in the exposure chamber described above and exposed to 600 ppm SO2 for 2 h/day for 7 days consecutively. The rats of the control group were exposed to filtered air in another identical chamber for the same period of time. When not being treated, all of the rats had free access to food and water ad libitum.
Microarray analysis
The total RNA was prepared from the lungs of the rats in both the groups and, after quality control, subjected to Agilent rat whole genome 4 × 44K Array GeneChip microarray test according to the manufacturer’s instructions (service provided by Kangchen Biotech, Shanghai, China). The scanning procedure was performed using an Agilent Microarray Scanner (Agilent G2565BA). Agilent Feature Extraction Software (version11.0.1.1) was used for data analysis.
Real-time quantitative RT-PCR
After the experiments were finished, the rats of both the groups were sacrificed and the lungs were removed. The total RNA was isolated from less than 100 mg of lung tissue using TRIzol reagent (Invitrogen Life Technologies) according to the manufacturer’s instructions. QRT-PCR was performed using a sequence detection system (ABI PRISM 7000; Applied Biosystems, Life Technologies, Grand Island, NY), and all reaction components were purchased from the same source (SYBR PrimeScript RT-PCR Kit obtained from TaKaRa Biotechnology). Standard experiments were carried out based on the instructions of the provider. All reactions were performed in triplicate. The threshold for the cycle threshold analysis of all samples was set automatically by the ABI PRISM 7000 software. The mRNA expressions of the indicated genes were normalized relative to β-actin mRNA. The primers were designed as follows:
β-actin primers,
F:5’CCTCTATGCCAACACAGTGC3,’ R: 5’GTACTCCTGCTTGCTGATCC3.’
Annealing temperature: 60°C, 211 bp
CD19 primers,
F:5’ATGTGGGTTTGGGGGTCTC3,,’ R: 5’AGGGTCGGTCATTCGCTTC3..’
Annealing temperature: 60°C, 273 bp
Flow cytometry
After completion of the experiments, the rats were sacrificed and bronchoalveolar lavage was performed with 3 mL PBS and BALF was collected. The lung tissues were minced and digested with 0.04% collagenase °C in DMEM for 1 h at 37°C. The digested cells were washed with PBS to remove collagenase. BALF was centrifuged and the cells were pooled into digested cells and suspended in PBS with 1% BSA with a cell density of 1 × 107 cells/mL. The anti-rat CD16 and CD32 antibodies were added for 1 μg/106 cells to block Fc receptors by incubation for 10 min on ice. The cells were washed with PBS and stained with 1 μg of anti-rat CD19-FITC and CD23-PE antibodies at 4°C for 30 min in dark. The cells were washed again and analyzed by flow cytometry.
Western blot analysis
Western blot (WB) analysis was performed as described previously. 6 Briefly, the proteins were separated by SDS–polyacrylamidegel Electrophoresis (PAGE; 10% separating, 5% stacking) and transferred to PVDF membranes (Millipore). After the membranes were blocked in 5% nonfat dry milk in PBS buffer containing 0.1% Tween 20, immunostaining was performed with monoclonal antibody specific for rat CD19 at a concentration of 1:400 (for CD19) overnight at 4°C. Anti-mouse secondary antibody at a concentration of 1:5000 was added to membranes and incubated for 45 min at 37°C. The protein signal was amplified and visualized via chemiluminescence using the ECL western blotting detection system and Hyperfilm ECL autoradiography film (AmershamPharmaciaBiotech, Inc.) To verify equal protein loading and transfer, membranes were stripped with stripping buffer and reprobed with anti-β-actin antibody. Images were quantified using the Labworks v3.0.2 image scanning and analysis software (Gel-Pro-Analyzer).
Immunohistochemical and histological analysis
The partial left lung was fixed in 4% formaldehyde. The fixed tissue samples were dehydrated in graded ethanol, embedded in paraffin. Each paraffin block was cut into 5 μm thick slices. Then, these slices were dewaxed in xylene, rehydrated in gradient alcohols, and rinsed with distilled water. For immunohistochemical analysis, nonspecific binding was blocked by incubating samples in 10% BSA prepared in PBS for 1 h, and the samples were incubated with 3% H2O2 for 15 min at room temperature. Then, the specimens were incubated with an anti-rat CD19 or CD3 antibodies at 4°C overnight. The samples were incubated with biotinized second antibodies for 30 min at 37°C. Specimens were incubated with HRP-labeled avidin for 30 min at 37°C and subsequently incubated with 3,3_-diaminobenzidine (DAB). Nuclei were stained with hematoxylin. For histological analysis, each section placed on a glass slide was stained with hematoxylin and eosin (H&E) and double-blind evaluated under microscope for histological changes by a professional pathologist.
ELISA
The levels of IgG, IgA, and IgE in serum and BALF were measured by commercially available ELISA kit according to the manufacturer’s instruction (Wanleibio, China). The concentrations of IgG, IgA, and IgE were calculated by generating a standard curve using standard proteins and analyzed using curve expert 1.3 software.
Statistical analysis
All values were expressed as mean ± standard deviation (SD). Significance testing was performed using the unpaired student’s t test. P < 0.05 was considered statistically significant.
Results
The expression of CD19 in microarray analysis
The result of microarray analysis showed that the fluorescent intensity of CD19 in the lungs of SO2 exposed group and the control were 11.172 and 12.218, respectively. The fold change between the two groups was 2.066 and the P value was 0.007. The results are shown in Table 1.
The expression of CD19 in microarray analysis.

SO2 reduced the expression of CD19 mRNA in lung
In order to verify the results of the microarray analysis, the total RNA was isolated from the lungs of both SO2 exposed and control rats and QRT-PCR was performed. The results showed that the expression of CD19 mRNA in SO2 exposed and the control group was 0.592 ± 0.096 and 1.027 ± 0.332, respectively. The expression of CD19 mRNA in the SO2 exposed group was significantly lower than that in the control group (P < 0.05; see Figure 1).

The expression of CD19 mRNA in the SO2 exposed group and the control. The SD rats were exposed to 600 ppm SO2 for 2 h/day for 7 days consecutively and the rats in the control were exposed to filtered air for the same period of time. The rats were killed and the lungs were removed. Total RNA was isolated from less than 100 mg of lung tissue and QRT-PCR was performed. The mRNA expressions of the indicated genes were normalized relative to β-actin mRNA.
The protein level of CD19 was decreased in the lungs of SO2 exposed rats
To further study if CD19 in protein level was influenced by SO2 exposure, the protein expression of CD19 in both the SO2 exposed and control groups was detected by WB analysis. The results showed that the gray value of CD19 in the control and SO2 exposed groups was 1.027 ± 0.548 and 0.502 ± 0.340, respectively. The expression of CD19 in the SO2 exposed group was significantly lower than that in the control group (P < 0.05; Figure 2(a) and (b)).

The expression of CD19 protein in the SO2 exposed and control groups. Rats, which were exposed to control air or 600 ppm SO2, were sacrificed and the lungs were removed for western blot (WB) assays. The expression of CD19 was shown as (a) gray values or represented by the (b) blots photograph.
SO2 exposure downregulated CD19+ or CD19+ CD23+ cells in lung tissues
Since CD19 is mainly expressed on B lymphocyte, we examined the percentages of the CD19+ and CD19+ CD23+ cells in lungs in the SO2 exposed and control groups using flow cytometry. The results showed that the percentage of CD19+ cells in control and SO2 exposed groups was 6.490 ± 3.478 and 3.710 ± 0.574, respectively. The percentage of CD19+ cells in the SO2 exposed group was significantly lower than that in the control group (P < 0.05; Figure 3(c)). Moreover, the percentage of CD19+ CD23+ cells in the SO2 exposed and control groups was also measured by flow sytometry and the results showed that the percentages of CD19+ CD23+ cells in the SO2 exposed and control groups were 5.740 ± 3.138 and 3.450 ± 0.544, respectively. The percentage of CD19 + CD23 + cells in the SO2 exposed group was also significantly lower than that in the control(P < 0.05; Figure 3(d)). Taken together, SO2 may reduce B-cells in lung tissues.
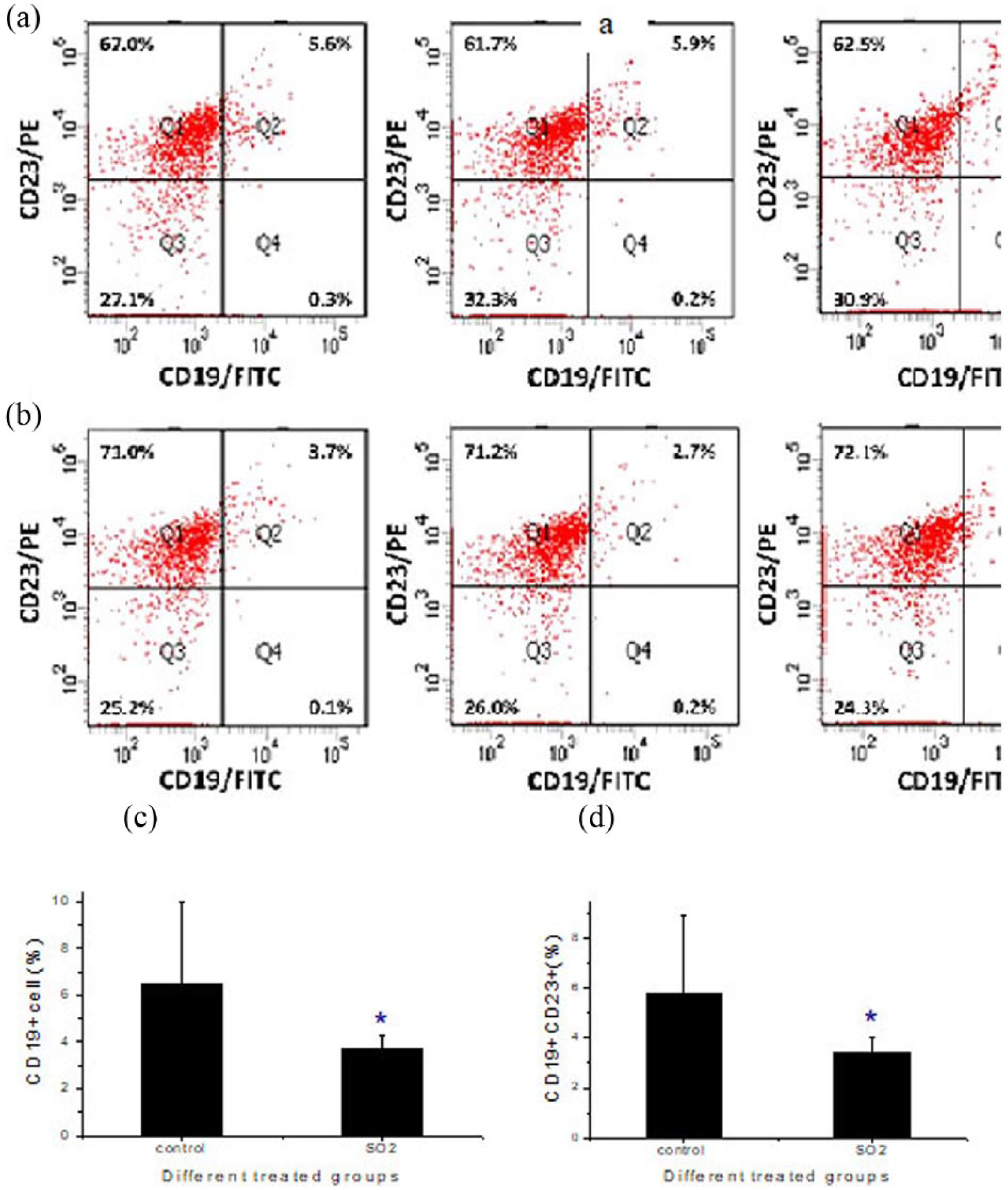
SO2 exposure decreases CD19+ or CD19/CD23+ cells in lung tissues of rats. Bronchoalveolar lavage was performed with 3 mL PBS on rats which were exposed to control air or 600 ppm SO2 and BALF was collected in irrigating solution. The cells from BALF were pooled into the cells from the lung digested with collagenase Ⅳ and analyzed by flow cytometry and recognized by anti-rat CD19-FITC or and CD19-FITC/CD23-PE antibodies. (a, b) The proportion of cells (Q1: CD23+/CD19−; Q2: CD23+/CD19+; Q3: CD23−/CD19−) was shown as representable photographs from the control group (a) or SO2 exposure group (b). Next, the proportion of (c) CD19+or (d) CD19+/CD23+cells was shown.
The IgG, IgA, and IgE both in serum and BALF were not influenced by exposure of SO2
Because CD19 is expressed on B lymphocyte and plays an important role in activation, proliferation, differentiation, and antibody production, we observed whether the antibodies production was influenced by downregulation of CD19 after SO2 exposure. The IgG, IgA, and IgE in serum and BALF of both the SO2 exposed and control groups were detected by ELISA and the results showed that the serum IgG in the control and SO2 exposed groups were 1771.830 ± 83.180 and 1770.381 ± 115.268, respectively (P > 0.05). The IgG in BALF in the control and SO2 exposed groups were 61.010 ± 3.770 and 57.079 ± 8.153, respectively (P > 0.05). The serum IgA in the control and SO2 exposed groups were 63.402 ± 56.272 and 105.468 ± 75.292, respectively (P > 0.05). The IgA of BALF in the control and SO2 exposed groups were 1.585 ± 1.331 and 2.960 ± 2.693, respectively (P > 0.05). The serum IgE in the control and SO2 exposed groups were 40.816 ± 40.652 and 33.839 ± 50.384, respectively (P > 0.05) and the IgE of BALF in the control and SO2 exposed groups were 0.489 ± 0.160 and 0.671 ± 0.332 (P > 0.05). There were no significantly statistical differences in the IgG, IgA, and IgE levels of both in serum and in BALF between the SO2 exposed and control groups (Figure 4(a)–(f)).

The levels of IgG, IgA, and IgE in both serum and BALF were not influenced by downregulation of CD19. The IgG, IgA, and IgE level in serum or BALF was examined by ELISA assays. (a) Serum IgG level. (b) BALF IgG level. (c) Serum IgA level. (d) BALF IgA level. (e) Serum IgE level. (f) BALF IgE level.
SO2 exposure induced lung injury in rats
Histopathological examination with H&E stained histological sections of experimental rat bronchial airways are shown in Figure 5(a)–(f). For the control group, the alveolus and bronchiole structures were clear, with few infiltrated inflammatory cells. For the SO2 group, pathological changes, such as chronic bronchitis, local alveolar hemorrhage, and lymphocytes infiltration, were observed. The results of immunohistochemical analysis showed that CD19+ cells were decreased and CD3+ cells were increased in the SO2 exposed group, compared with the control (Figure 6).
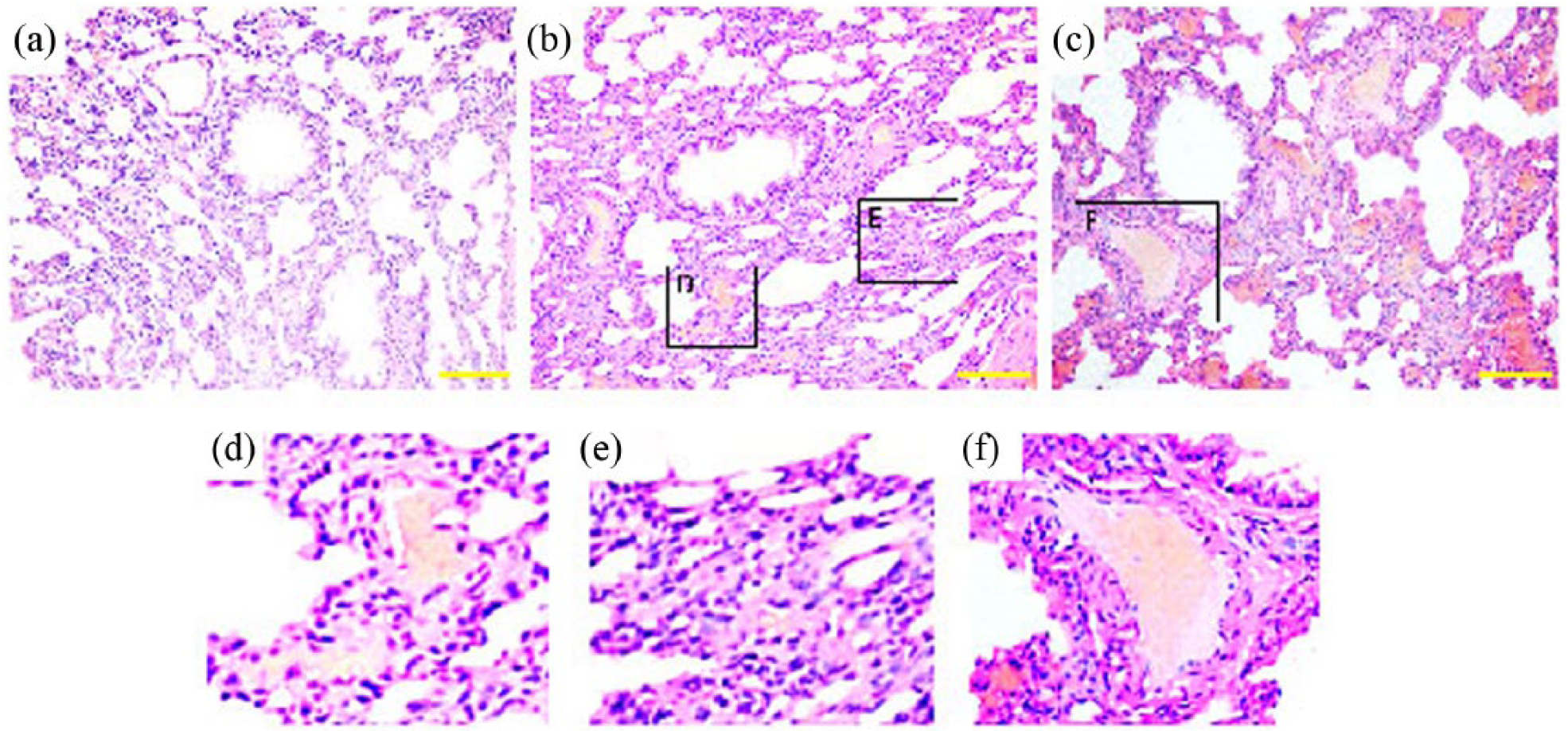
SO2 exposure induced lung injury in rats. Rats, which were treated by control air or 600 ppm SO2, were sacrificed and lung tissues were removed for hematoxylin and eosin (H&E) assays. Results from (a) control group or (b, c) SO2 exposure groups are shown. The (d) local alveolar hemorrhage, (e) lymphocytes infiltration or (f) local alveolar hemorrhage and lymphocytes infiltration are also shown. Original magnification ×200. The scale bars in the figures indicate 100 μm.

Immunohistochemical analysis of lungs in SO2 exposed rats and control. The sections of lung tissues from both SO2 exposed rats and control were stained with anti-CD3 or anti-CD19 antibodies, described as “Materials and Methods.” (a) CD3 for control. (b) CD3 for SO2 exposed rat. (c) CD19 for control. (d) CD19 for SO2 exposed rat. Original magnification ×400. The scale bars in the figures indicate 50 μm.
Discussion
RADS, first put forward and named by Brooks et al., 7 is indicated as a continuous asthma syndrome after an airway is exposed to irritate gas at a high level. Usually, it has a sudden onset, repeated attack, and lasts for a long time. The major clinical manifestations are dyspnea, chest discomfort, cough, nausea, vomit, headache, and so on. The symptoms appear by 24 h after irritation and no incubation for clinical manifestations is observed. The time of symptom appearance varies from several minutes to hours and continuous cough lasts for over 3 months. The major abnormality in lung function is obstructive ventilation function disturbance, and the minor abnormality is mixed ventilation function disturbance. In RADS, the airway provocation test is positive and lung function can be restored to normality by 3–6 months after being away from the irritating environment. RADS can result in long-term sequela and chronic respiratory tract diseases. 8
It is now explicit that bronchial asthma is a heterogeneous disease, usually characterized by chronic airway inflammation. It is defined by the history of respiratory symptoms such as wheeze, shortness of breath, chest tightness, and cough that vary over time and in intensity, together with variable expiratory airflow limitation. 9 There has been little systemic research on the pathogenesis of RADS. The irritation by chemical gases may first cause injury of the airway epithelial cells and fall off, followed by inflammation, edema, and blockage of bronchiole, which are similar to chemical trachitis and bronchitis. 10 However, the difference is that the course of RADS is longer than that of chemical trachitis and bronchitis. The symptoms last for several months after removal of the irritation gases in RADS and the mechanism is unclear. It is reported that the epithelial cells of the lung and the respiratory tract can prompt inflammatory cells to release histamine and the inflammatory medium in the high osmotic pressure environment formed by high ionic strengthen and the released process can occur in several minutes after being stimulated with high osmotic pressure. 11 Further researches indicated that the membrane potential of the cells can be changed in a high cation environment, whereas changes in the membrane potential of the cells can cause increase in the oxygen radical in cell respiratory burst, which is involved in the inflammation process.12,13
Sulfur dioxide (SO2) is one of the main air pollutants, which is formed when sulfur-containing fuel is burned. In some special situations, people may be exposed to a high concentration of SO2, such as an accident of SO2 tank leakage, mine blast, smoke of gunpowder in a war, and smoke of volcanic eruption, which may be harmful to the persons exposed to it. Many studies have indicated that SO2 exposure increases morbidity and mortality.14,15 SO2 is an upper respiratory tract irritant. It has been demonstrated that both acute and chronic SO2 exposure produce a variety of adverse pulmonary effects, including bronchoconstriction, mucus secretion, pulmonary inflammation, bronchial hyperresponsiveness, and morphological changes, such as epithelial thickening.14,16–19 CD19 molecule is an important mark of B lymphocyte and plays a key role in B cell signal transduction, activation, differentiation, and production of antibody.20–22 In this experiment, the effects of inhalation of SO2 on rat CD19+ cells were observed by inhalation of high concentration of SO2 as stimulator to explore its mechanism in RADS formation.
Our results showed that inhalation of high concentration of SO2 can result in airway inflammation in experimental animals. But this inflammation response is different from that of bronchial asthma and manifests as airway inflammation that are mainly pulmonary congestion and lymphocytes infiltration. This inflammation presents the alteration of fine peribronchitis analogous to chronic bronchitis, and eosinophil infiltration was not observed. In humoral immunity, bronchial asthma manifests with increase of immuglobulin (IgE, IgA, IgG) and the increase of CD19 positive cell expression is one of the important factors. However, our results showed that the percentages of CD19+ cells and CD19+ CD23+ cells in lung tissues were significantly decreased after inhalation of a high concentration of SO2. The results of immunohistochemical analysis also showed that CD19+ cells were decreased in the SO2 exposed group. The results showed that the CD19 expressions in lung tissue were also decreased both in mRNA and protein levels after inhalation of a high concentration of SO2, whereas the serum levels of IgG, IgA, and IgE were not influenced by inhalation of a high concentration of SO2. To our knowledge, this is the first time in the world that CD19 expression is reported to be downregulated by inhalation of high concentration of SO2.
In summary, RADS is a non-immunogenicity, chronic airway inflammatory disease caused by irritation of harmful factor and manifests as airway hyperresposiveness. Further studies are necessary to explore the molecule mechanism by which SO2 downregulates the expression of CD19 and why serum and BALF level of IgG, IgA, and IgE were not influenced by SO2.
Footnotes
Acknowledgements
Z.Z. and Z.M contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by (1) The PAL 12th Five years project of medicine of China: BWS12J007(Zhuang Ma, Wenwu Sun).
