Abstract
β-patchoulene (β-PAE) is one of the essential tricyclic sesquiterpenes of patchouli oil while β-patchoulene epoxide (β-PAO) is the oxidative product of β-PAE which can only be found in the oil with long storage period. Our previous researches demonstrated that both β-PAE and β-PAO exert potent anti-inflammatory activity in vivo, but which one is more valuable still remains uncertain. Therefore, this study adopts the model of LPS-stimulated RAW264.7 macrophages to compare β-PAO with β-PAE on the anti-inflammatory activity. According to our results, β-PAO was superior to β-PAE on anti-inflammation as evidence by lowering the protein and mRNA expressions of several pro-inflammatory cytokines including tumor necrosis factor-α (TNF-α), interleukin-12 (IL-12), interleukin-1β (IL-1β), and monocyte chemotactic protein-1 (MCP-1). β-PAO was also better than β-PAE in reducing the productions of nitric oxide (NO) and prostaglandin E2 (PGE2) through inhibiting inducible nitric oxide synthase (iNOS) and cyclooxygenase (COX)-2 signaling pathway. The results above provided experimental basis for the conclusion that β-PAO was more potent than β-PAE in anti-inflammatory activity in vitro.
Introduction
Inflammation is a guarding mechanism against different harmful stimuli, such as allergens, microbial infections, and physical injuries.1,2 It can be produced through activating macrophages, which are able to phagocytose external intruders, being the first defensive line of the innate immune system. In addition, macrophages can release many pro-inflammatory mediators and resulting in the destabilization between pro- and anti-inflammatory cytokines. 3 Such imbalance is generally considered to be the reason for several inflammatory diseases, such as rheumatoid arthritis, atherosclerosis, and allergies. 4 Therefore, effective therapies for inflammation are of significant importance. Non-steroidal anti-inflammatory drugs (NSAIDs) are commonly used to resist inflammation, but its severe side effects including gastrointestinal diseases and liver injury restrict its wide range of clinical application. 5 Therefore, a great deal of study has been conducted in an attempt to seek alternatives against inflammation with less side effects.
According to our previous study, β-patchoulene (β-PAE) is one of the representative hydrocarbon sesquiterpenoids of patchouli oil, while β-patchoulene epoxide (β-PAO) is the tricyclic sesquiterpene that only exists in the long-stored oil. 6 β-PAO was found to share similarity in structure (Figure 1(a)) with β-PAE (Figure 1(b)), and it is reasonable to reckon that β-PAO might be the oxidative product of β-PAE. In addition, both β-PAE and β-PAO have been proven the capability of anti-inflammation in vivo, but which one is better, still remains uncertain. 6 Hence, this study aims to sort out the superior of anti-inflammatory activity using the model of LPS-stimulated RAW264.7 macrophages.
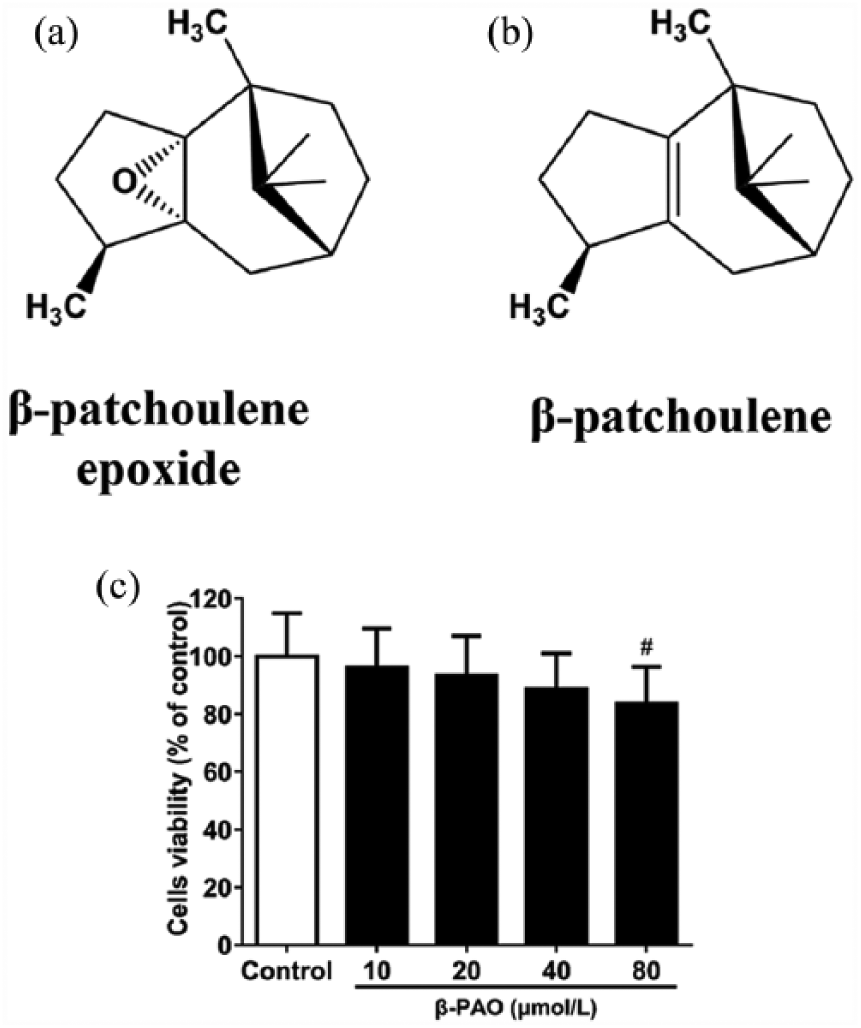
Chemical structure of (a) β-patchoulene epoxide and (b) β-patchoulene. (c) Effect of β-PAO on RAW264.7 cell viability. The viability was evaluated by the MTT assay, and the values were shown as the mean ± SD.
Materials and methods
Plant materials and reagents
β-PAO and β-PAE were isolated from patchouli oil according to published articles.6,7 Lipopolysaccharide (LPS) was obtained from Sigma-Aldrich (St. Louis, MO, USA). RPMI-1640 culture medium, penicillin-streptomycin, and fetal bovine serum (FBS) were provided by Gibco (Grand island, NY, USA). Phosphate-buffered saline (PBS, 0.067 mol/L, pH 7.2) was supplied by Hyclone (Beijing, China). Phosphoric acid, N-(1-naphthyl) ethylenediamine dihydrochloride and sulfanilamide were obtained from Macklin (Shanghai, China). Cell Titer 96® AQueous One Solution Cell Proliferation Assay was provided by Promega (Madison, WI, USA). (Shanghai, China). Enzyme-linked immunosorbent assay (ELISA) kits were obtained from eBioscience (San Diego, CA, USA). Total protein kits were supplied by Nanjing Jiancheng Bioengineering Institute (Nanjing, China). All other chemicals used in this study were of analytical grade available unless stated specifically.
Cell culture and drug treatment
The RAW264.7 cell line was purchased from American Type of Culture Collection (ATCC; Rockville, MD, USA) and cultured at 37°C in RPMI-1640 medium supplemented with 10% FBS and 1% penicillin-streptomycin under a humid circumstance with 5% CO2.
RAW264.7 macrophage cells were seeded in 96-well sterile disposable culture plates at 5 × 104 cells/well for 24 h unless otherwise specified. Afterward, the cells were incubated with β-PAE (20 μmol/L) or different concentrations of β-PAO (10, 20, and 40 μmol/L), respectively, which was dissolved in the solution of 0.1% dimethyl sulfoxide (DMSO) for 2 h. Subsequently, LPS (final concentration is 100 ng/mL, dissolved in phosphate buffer) was added into the medium and incubated for another 24 h. It should be noticed that cells in control group were only treated with serum-free medium of 0.1% DMSO, while LPS group was only treated with LPS but not exposed to other drugs.
3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay
Cell viability was analyzed by Celltiter 96® AQueous One Solution Cell Proliferation assay. After treated with various concentrations of β-PAO for 24 h, the culture medium was discharged and substituted by fresh medium. Next, 20 μL Cell Titer 96® AQueous One solution was added, and the cells were further incubated at 37°C for 4 h. The supernatant was then discarded and the resulting formazan crystals were dissolved in 150 μL DMSO. The optical densities were measured at 490 nm by Thermo Scientific microplate reader. Cell viability was expressed as a percentage of the control group.
Nitric oxide assay
Griess reagent was used to measure the concentration of nitrite, an indicator of nitric oxide (NO) production. Briefly, after LPS treatment, 100 μL supernatant of each well was mixed with the same amount of Griess reagent. Griess reagent comprised equal volumes of 1% (w/v) sulfanilamide in 5% (v/v) phosphoric acid and 0.1% (w/v) N-(1-naphthyl) ethylenediamine-HCl. The mixture was then incubated for 10 min under room temperature. Solution absorbance was measured at 540 nm using microplate reader.
Pro-inflammatory cytokines assay
Measurement of pro-inflammatory cytokines including tumor necrosis factor-α (TNF-α), interleukin-12 (IL-12), interleukin-1β (IL-1β), monocyte chemotactic protein-1 (MCP-1), and prostaglandin E2 (PGE2) were carried out using ELISA kit. Then, the supernatants of every group underwent absorbance detection according to the manufacturer’s instructions with a microplate reader.
Quantitative real-time polymerase chain reaction
To analyze the mRNA expressions of TNF-α, IL-12, IL-1β, MCP-1, inducible nitric oxide synthase (iNOS), and cyclooxygenase (COX-2), total RNA was extracted from LPS-stimulated RAW264.7 cells using TRIzol® reagent, and 2 μg of total RNA was used to synthesize cDNA using RevertAid First Strand cDNA Synthesis Kit (Thermo Scientific, MA, USA). The cDNA was then used as the template for real-time PCR (the primers of PCR are listed in Table 1) with FastStart Universal SYBR Green Master (Rox) (Roche, Basel, Swiss Confederation). Each reaction was conducted in triplicate under the following cycling conditions: 10 min at 95°C, followed by 40 cycles at 95°C for 15 s, and then 60°C for 1 min. The relative expression levels of target genes were normalized by glyceraldehyde-3-phosphate dehydrogenase (GAPDH).
Primer sequences.

COX-2: cyclooxygenase; iNOS: inducible nitric oxide synthase; TNF-α: tumor necrosis factor-α; IL-1β: interleukin-1β; IL-12: interleukin-12; MCP-1: monocyte chemotactic protein-1; GAPDH: glyceraldehyde-3-phosphate dehydrogenase.
Statistical analysis
Statistical analysis was performed using a one-way analysis of variance (ANOVA) followed by an least significant difference (LSD) test for multiple comparisons using Statistical Product and Service Solutions (SPSS) software (version 20.0). All data were expressed as mean ± SD. Values of P < 0.05 were considered statistical significant.
Result
Cytotoxic effect of β-PAO in RAW264.7 cells
In our study, cell viability was measured to assess the cytotoxicity of β-PAO. As shown in Figure 1(c), the result of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay showed that RAW264.7 cell viability was significantly inhibited by β-PAO at 80 μmol/L, indicating that 80 μmol/L β-PAO had cytotoxic effect on RAW264.7 cell. However, other dosages (10, 20, and 40 μmol/L) showed nontoxic effect. Therefore, 10, 20, and 40 μmol/L β-PAO were selected for further experiment.
Effect of β-PAO and β-PAE on LPS-induced NO production and iNOS mRNA expression in RAW264.7 Cells
As shown in Figure 2(a), a significant increase in nitrite concentration was found in LPS group when compared with the control group. Specifically, NO production of LPS group significantly (P < 0.01) increased upon the control group, which could be repressed by β-PAO pretreatment. The superior effect was detected at the dose of 20 and 40 μmol/L, which was comparable to β-PAE (20 μmol/L). Figure 2(b) showed that the mRNA expression of iNOS was dramatically (P < 0.01) elevated after LPS stimulation. However, such trend was markedly attenuated by β-PAO, which was better than β-PAE of identical concentration.
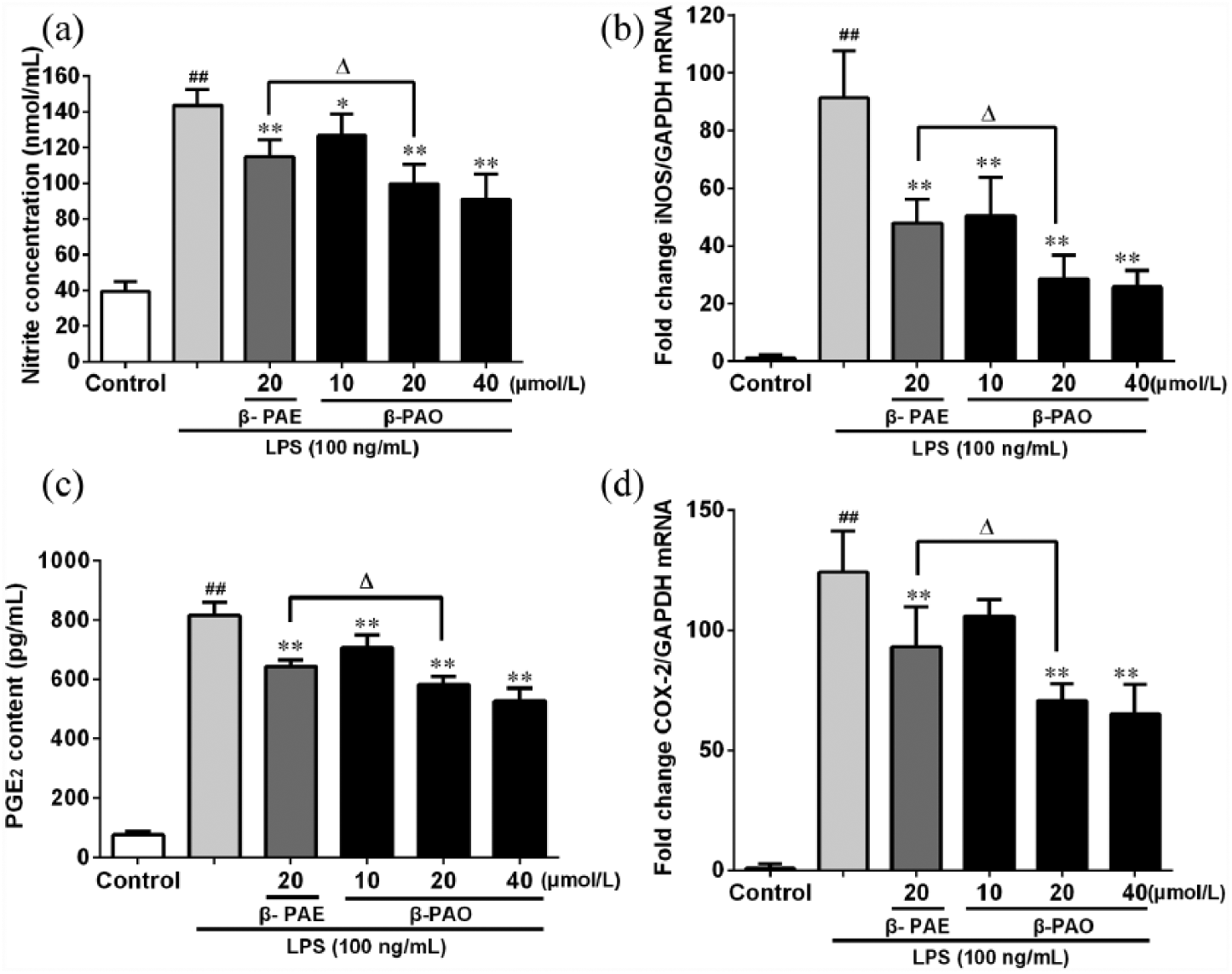
Effect of β-PAO and β-PAE on (a) NO production, (b) iNOS mRNA expression, (c) PGE2 production and (d) COX-2 mRNA expression in LPS-stimulated RAW264.7 macrophages. The amount of NO in the medium was measured using the Griess reagent and PGE2 content was measured with ELISA kit. The mRNA expressions of iNOS and COX-2 were measured with qRT-PCR. Data were expressed as mean ± SD.
Effect of β-PAO and β-PAE on LPS-induced PGE2 production and COX-2 mRNA expression in RAW264.7 Cells
As depicted in Figure 2(c) and (d), PGE2 level and COX-2 mRNA expression of LPS group were both dramatically (P < 0.01) increased when compared with control group. However, they could be reversed by pretreating with β-PAO. It should also be noticed that β-PAO were more effective than β-PAE of identical concentration.
Effect of β-PAO and β-PAE on TNF-α, IL-12, IL-1β, and MCP-1 productions and mRNA expressions in LPS-induced RAW264.7 cells
As shown in Figure 3, TNF-α, IL-12, IL-1β, and MCP-1 levels in LPS group were significantly (P < 0.01) increased, as compared to the control group. However, these could be decreased by β-PAO in a dose-dependent manner, especially when it was up to 20 μmol/L that possessed more noticeable anti-inflammatory effect than β-PAE did. In addition, TNF-α, IL-12, IL-1β, and MCP-1 mRNA expressions were dramatically (P < 0.01) increased after pretreating with LPS when compared to control group. Nevertheless, the trends of TNF-α, IL-12, IL-1β, and MCP-1 mRNA expressions were markedly decreased in medicated groups and closely matched by their ELISA results.

Effect of β-PAO and β-PAE on the protein and transcriptional levels of (a) TNF-α, (b) IL-1β, (c) IL-12, and (d) MCP-1 in LPS-stimulated RAW264.7 macrophages. The TNF-α, IL-1β, IL-12, and MCP-1 contents were measured with ELISA kit and their mRNA expressions were measured with qRT-PCR. Data were expressed as mean ± SD.
Discussion
LPS is a major component of Gram-negative bacteria (e.g. Escherichia coli) cell wall. It stimulates the inflammatory responses through binding to the membrane receptors of macrophages and then triggering the production of many effector molecules including NO and PGE2. NO, catalyzed by iNOS from
Also, cytokines are of significant importance to mediate inflammatory responses. As the most representative pro-inflammatory cytokine, TNF-α can stimulate the macrophage to release MCP-1, IL-12, and IL-1β through binding to the TNF-receptor then exacerbating inflammatory responses. 12 Our results showed that β-PAO could significantly reduce the protein and mRNA expressions of TNF-α, IL-12, IL-1β, and MCP-1 more than β-PAE did. It was indicated that β-PAO was superior to β-PAE when it came to modulate the release of pro-inflammatory cytokines.
In conclusion, we demonstrated that the anti-inflammatory activity of β-PAO was better than β-PAE. Thus, β-PAO is worthwhile to exploit and develop as a new agent for prevention and treatment of inflammatory diseases.
Footnotes
Acknowledgements
The authors are grateful to the researchers who participated in the study. X. W. and J.-L. L. contributed equally to this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by grants from the National Natural Science Foundation of China (No. 81403169); Natural Science Foundation of Guangdong Province, China (No. 2014A030310224); Clutivating Foundation for Major State Project-Department of Education of Guangdong Province, China (No. 2014GKXM035); and Science and Technology Planning Project of Guangdong Province, China (Nos 2014A020221023, 2016A020226049, and 2013A022100001).
