Abstract
Osteoporosis is a chronic multifactorial disease characterized by deterioration of bone mass and is vulnerable to bone fracture. Plasminogen activator inhibitor-1 (PAI-1) is an important molecule for maintenance of optimum bone mass. Several single-nucleotide polymorphisms (SNPs) in PAI-1 have been reported to alter PAI-1 expression and/or the translational level. In this report, we explored the possible role of common PAI-1 gene polymorphisms on predisposition to osteoporosis in a Chinese cohort. A total of 364 post-menopausal Chinese women diagnosed of having osteoporosis and 350 healthy females hailing from similar areas were enrolled in this study. Five common SNPs (−844G > A, −6754G/5G, +43G > A, +9785G > A and +11053T > G) were genotyped by polymerase chain reaction (PCR) followed by restriction fragment length polymorphism (RFLP). Relative expression of PAI-1 mRNA and plasma PAI-1 levels were quantified by reverse transcription polymerase chain reaction (RT-PCR) and enzyme-linked immunosorbent assay (ELISA), respectively. Prevalence of homozygous mutant (5G/5G) and minor allele (5G) of PAI-1 (−675 4G/5G) polymorphism was significantly more frequent in patients than in healthy controls (5G/5G: P < 0.0001, odds ratio (OR) = 3.18; 5G: P < 0.0001, OR = 1.65). Both plasma PAI-1 and relative mRNA expression levels were significantly lower in patients compared to healthy controls. Interestingly, the quantity of plasma PAI-1 and mRNA expression was correlated with PAI-1 (−675 4G/5G) polymorphism: subjects with 4G/4G genotype had elevated PAI-1 in comparison to homozygous mutant, and displayed lower quantity of PAI-1 protein and mRNA values. PAI-1 (−675 4G/5G) mutant is associated with susceptibility to development of osteoporosis in post-menopausal Chinese women. Furthermore, this variant in the promoter region alters plasma protein levels and relative expression of PAI-1.
Introduction
Osteoporosis is a common bone disorder characterized by loss in bone mass, augmented skeletal fragility and increased risk of bone fracture. Osteoporosis is more frequent in post-menopausal women because of decreased production of oestrogen, and this disorder subsequently leads to disability and mortality of the individual. A significant link between osteoporotic fracture and mortality rate in post-menopausal women has been demonstrated worldwide. 1 In China, prevalence of osteoporosis has been reported to be 13% and is more prevalent in older women (>50 years of age). 2 Bone mineral density (BMD) test determines the amount of mineral, mainly calcium, present in the bone and has been correlated with the age, sex and genetic make-up of the subjects. Various studies have demonstrated an association between common gene polymorphisms with BMD scores in different populations.
Plasminogen activator inhibitor-1 (PAI-1) is a vital member of the serine proteases group that inhibits plasminogen activators, including tissue (tPA) and urokinase-type plasminogen activator (uPA), and blocks fibrinolysis pathways leading to hamper conversion of plasmin from plasminogen. 3 The role of PAI-1 in relation to bone loss has been ill defined. Mice deficient in both oestrogen and PAI-1 are protected from bone loss. 4 Another independent study on diabetic mice model showed a significant correlation between reduced bone loss and PAI-1 deficiency. 5 A recent report that demonstrated involvement of plasminogen on bone recovery in mice model 6 further strengthens the link between PAI-1 and bone loss pathogenesis. PAI-1 is a 50-kDa glycoprotein coded by SERPINE-1 located in the long arm of the seventh chromosome (21.3). Several single-nucleotide polymorphisms (SNPs) have been reported in SERPINE-1, but five SNPs (−844G > A, −675 4G/5G, +43G > A, +9785G > A and +11053T > G) have been well investigated in various populations. Among them, −675 4G/5G polymorphism has been shown to regulate expression of the PAI-1 gene.7,8
Studies linking common PAI-1 variants and development of osteoporosis are very limited. A recent report on Korean post-menopausal women showed association of PAI-1 (−675 4G/5G) mutants with susceptibility to osteoporosis. 9 However, another report including Turkish women failed to demonstrate such association. 10 In this study, we enrolled a larger number of post-menopausal cases from a Chinese cohort, genotyped five common polymorphisms (−844G > A, −675 4G/5G, +43G > A, +9785G > A and +11053T > G), quantified expression of PAI-1 in both plasma and RNA levels and investigated the possible association with the development of osteoporosis.
Materials and methods
Subjects
In this hospital-based case–control study, 364 post-menopausal females who were admitted to the Department of Orthopaedics, Jingzhou Hospital of Traditional Chinese Medicine, China, during 2013–2016 and satisfied all inclusion criteria were enrolled. Patients with clinical phenotypes, which affect bone metabolism such as parathyroid, thyroid, liver, and rheumatic diseases, were excluded from this study. Furthermore, patients with a history of surgery-induced menopause were also omitted from the study. Baseline characteristics of each patient were recorded and are shown in Table 1. Dual-energy X-ray absorptiometry was performed in each patient by trained neuro and orthopaedic surgeons as described earlier. 11 Based on World Health Organization (WHO) criteria, patients with mean BMD less than 2.5 SD were diagnosed to have osteoporosis. In all, there were 350 post-menopausal females, essentially healthy and hailing from similar geographical areas without a history of other diseases or under medications known to affect bone mass density. About 5 mL of intravenous blood was collected from each participant; plasma was isolated and used for biochemical assays. The Institutional Human Ethical Committee of Jingzhou Hospital of Traditional Chinese Medicine approved the study protocol. Each participant was informed about the study protocol and the possible outcome signed consent was obtained.
Prevalence of (−675 4G/5G) polymorphism in osteoporosis patients and healthy controls.

HC: healthy controls; CI: confidence interval.
Isolation of DNA and RNA
Whole genomic DNA from all participants was isolated by mini kit as per instructions of the manufacturer (QIAamp DNA Blood Mini Kit, QIAGEN, USA). Furthermore, based on availability of fresh blood samples, total RNA was isolated from 282 subjects (152 patients and 130 healthy controls) using RNeasy kit (QIAGEN). DNA and total RNA samples were stored at −800°C until PAI-1 genotyping and mRNA quantification, respectively.
Genotyping of PAI-1 polymorphisms
Five common PAI-1 gene polymorphisms (−844G > A, −675 4G/5G, +43G > A, +9785G > A and +11053T > G) were genotyped by polymerase chain reaction (PCR) followed by restriction fragment length polymorphism (RFLP) as described earlier. 12 About 20% of the samples were randomly selected, direct sequenced and observed for 100% concordance with the PCR-RFLP results.
Real-time PCR quantification assay
Total RNA was extracted from blood samples (152 patients and 130 healthy controls) by kit and 1 µg was used for synthesis of c DNA. Relative expression of PAI-1 was measured by reverse transcription polymerase chain reaction (RT-PCR) by TaqMan method, as performed earlier. 13 Glyceraldehyde 3-phospate dehydrogenase was taken as reference and relative expression of PAI-1 mRNA was quantified by the delta delta Ct method.
Plasma PAI-1 quantification
Plasma PAI-1 in both healthy controls and osteoporosis patients were measured by enzyme-linked immunosorbent assay (ELISA) as instructed by the manufacturer (R&D Systems).
Statistics
All analysis was conducted by GraphPad Prism Version 6.01. Distribution of PAI-1 polymorphism controls and osteoporosis cases were compared by Fisher exact test. For comparison of genotype and allele, a P value less than 0.01 was considered as significant after Bonferroni correction (0.05/5 = 0.01). Association of plasma protein levels and mRNA expression of PAI gene with PAI-1 (−675 4G/5G) variants was analysed by analysis of variance (ANOVA) followed by Tukey’s post test. Spearman’s rank test was employed to test for a possible link between PAI-1 levels and their expression. Significance was set at <0.05 of probability value. A post hoc power analysis was performed by G power 3.1 software which revealed that the present case–control study had 89% power to detect a genetic association at 0.15 effective size.
Results
Baseline data
In this study, 714 post-menopause females were enrolled, including 350 healthy subjects and 364 osteoporosis patients. Demographic and biochemical characteristics of healthy controls and osteoporosis patients were analysed. The mean age, homocysteine (Hcy), folate levels and body mass index (BMI) were comparable among healthy controls and osteoporosis patients. Fasting blood sugar (FBS) remained elevated in patients compared to controls. In contrast, osteoporosis patients showed a lower mean systolic blood pressure (SBP), diastolic blood pressure (DBP), high-density lipoprotein (HDL) and low-density lipoprotein (LDL) than healthy controls (data not shown).
Prevalence of PAI-1 gene polymorphisms in healthy controls
Five SNPs in PAI-1 (−844G > A, −675 4G/5G, +43G > A, +9785G > A and +11053T > G) were genotyped by PCR-RFLP in 350 healthy Chinese females. Prevalence of genotypes was as follows: −844G > A (GG: 38%, GA: 48% and AA: 14%); −675 4G/5G (4G/4G: 42%, 4G/5G: 48% and 5G/5G: 10%); +43 G > A (GG: 92%, GA: 6% and AA: 2%); +9785 G > A (GG: 90%, GA: 9% and AA: 1%); and +11053T > G (TT: 25%, TG: 52% and GG: 23%). Distribution of genotypes were in Hardy–Weinberg equilibrium (HWE) for −844G > A, −675 4G/5G and +11053T > G polymorphisms; on the other hand, genotypes of +43 G > A (X2 = 47.50, P < 0.0001) and + 9785 G > A (X2 = 8.76, P < 0.003) polymorphisms were deviated from the HWE.
5G/5G genotype is associated with osteoporosis
To access the possible role of PAI-1 variants with predisposition to osteoporosis, prevalence of PAI-1 genotypes was compared among healthy controls and osteoporosis patients. 5G/5G genotype and minor allele (5G) were more frequent in osteoporosis patients compared to female controls (5G/5G: P < 0.0001, odds ratio (OR) = 3.18; 5G: P < 0.0001, OR = 1.65; Table 1), indicating a genetic predisposition for development of osteoporosis. However, distribution of other SNPs (−844G > A, +43G > A, +9785G > A and +11053T > G) was comparable among control and case (data not shown).
Plasma PAI-1 and relative mRNA expression in osteoporosis patients and controls
Plasma PAI-1 and relative expression of mRNA were quantified by ELISA and RT-PCR, respectively, in 152 osteoporosis patients and 130 healthy females. The mean plasma levels of PAI-1 were significantly elevated in healthy controls than in osteoporosis patients (P < 0.0001). Similarly, healthy controls displayed a higher relative expression of PAI-1 mRNA than patients (P = 0.001; Figure 1).

Plasma PAI-1 and relative expression in osteoporosis and healthy controls. (a) Plasma levels of PAI-1 were quantified by ELISA in healthy controls (n = 130) and osteoporosis patients (n = 152) and mean PAI-1 levels were compared by Student’s t-test. Healthy controls displayed higher plasma PAI-1 compared to osteoporosis patients (P < 0.0001). (b) Relative mRNA expression of PAI-1 gene was quantified by RT-PCR in osteoporosis and healthy controls and mean values were compared by ‘t’-test. Similarly, healthy controls had higher mRNA expression than patients (P = 0.001).
Genotype–phenotype relationship
In this study, we observed diminished levels of plasma PAI-1 as well as lowered expression of mRNA in osteoporosis patients compared to healthy controls. In addition, association of −675 4G/5G variants with predisposition to osteoporosis was also noticed. Based on these observations, we hypothesized that there would be a possible association between −675 4G/5G polymorphism and plasma PAI-1 and/or expression of mRNA. To test this, we compared the mean PAI-1 levels and the relative expression of mRNA in different genotypes of PAI-1 (−675 4G/5G) polymorphism and the results are demonstrated in Figure 2. Subjects with 4G/4G genotype displayed a significantly higher plasma PAI-1 and mRNA expression compared to the 5G/5G genotype (Figure 2(a) and (b)), indicating an important functional relevance of −675 4G/5G polymorphism on determination of PAI-1 levels both in terms of protein and mRNA.
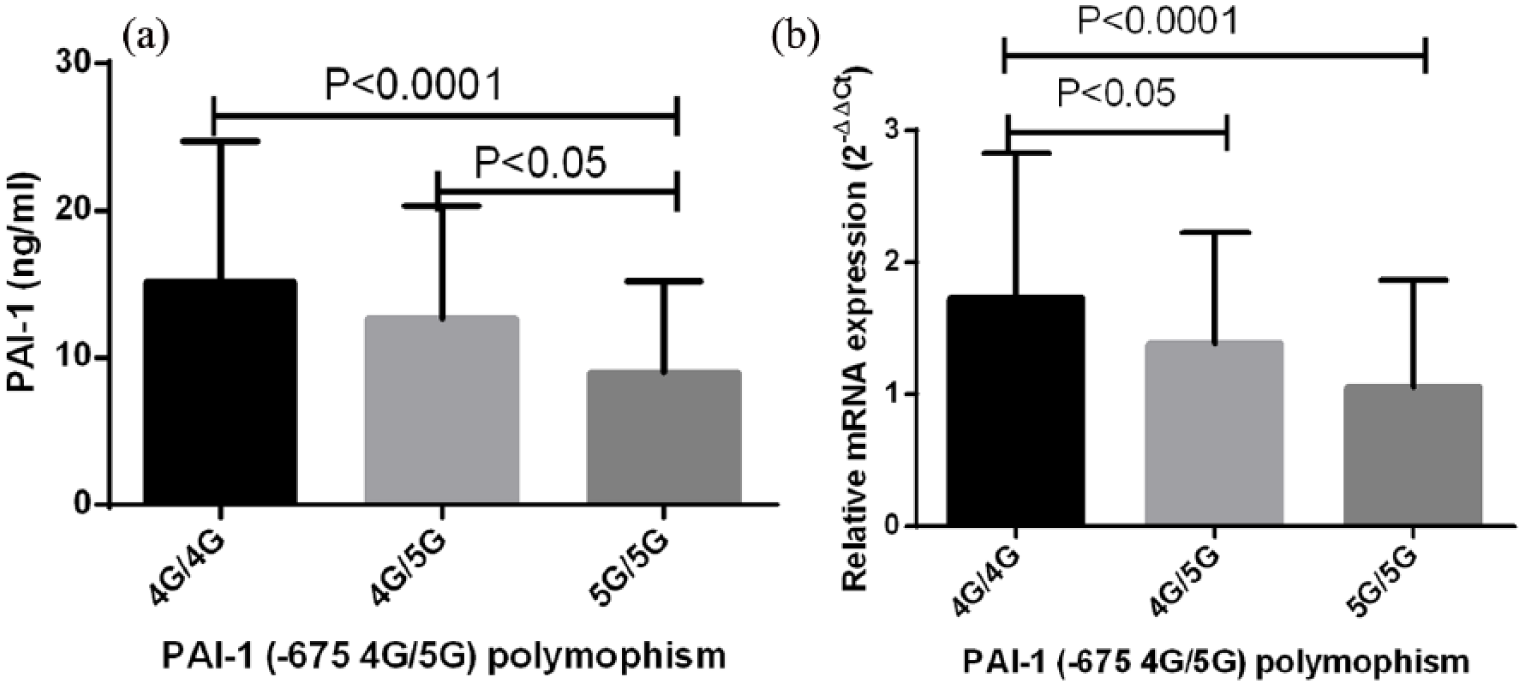
Association of PAI-1 (−675 4G/5G) polymorphisms with relative mRNA expression and plasma PAI-1. Plasma PAI-1 and relative expression of mRNA were compared among different genotypes of PAI-1 polymorphisms (4G/4G: n = 103; 4G/5G: n = 124; 5G/5G: n = 55). Subjects with 4G/4G displayed significantly more (a) plasma PAI-1 and (b) mRNA expression compared with other genotypes. Mean plasma PAI-1 levels and mRNA expression among different genotypes were compared by ANOVA followed by Tukey post test.
Correlation of plasma PAI-1 with mRNA expression
Next, we tested the possible correlation between mRNA expression and plasma PAI-1. As shown in Figure 3, a positive correlation was observed among mRNA expression and plasma levels of PAI-1 (r = 0.85, P < 0.0001).

Correlation of relative PAI-1 expression and plasma PAI-1. Correlation between plasma level of PAI-1 and relative expression or PAI-1 mRNA was analysed by Pearson’s rank coefficient in 282 subjects including osteoporosis patients and healthy females.
Discussion
In comparison to Western countries, the prevalence of osteoporosis is lower in China and more frequent in aged women. In this study, we enrolled 364 osteoporosis patients and investigated the association of common PAI-1 variants with predisposition to osteoporosis development. Furthermore, the genotype–phenotype relationship was also explored. The results showed a significant link of −675 5G/5G genotype with predisposition to osteoporosis development. In addition, correlation of −675 4G/5G polymorphism with plasma PAI-1 and relative mRNA expression was also observed, indicating the importance of 4G/5G variants in susceptibility to osteoporosis and strengthening earlier observations on functional relevance of PAI-1 (−675 4G/5G) polymorphism, respectively.
Among five common polymorphisms in PAI-1 studied in this investigation, −675 4G/5G SNP has been widely investigated in the Chinese population. We observed a higher prevalence of heterozygous mutants (48%) compared to 4G/4G (42%) and 5G/5G (10%). Similar trends in distribution of −675 4G/5G polymorphism in the Chinese population were reported earlier. A higher frequency of heterozygous individuals may be attributed to their possible protective nature against certain infectious diseases. However, reports including healthy subjects from Fujian and Shandong showed a higher prevalence of 5G/5G genotype over wild-type (4G/4G) and heterozygous mutants (4G/5G). 14 It is advised to enrol enough number of samples to decipher the genotypes frequency of a population: we have enrolled a higher number of healthy females (n = 350) compared to earlier studies. 14
PAI-1 down regulates fibrinolysis and maintains the appropriate plasminogen plasmin ratio. The role of PAI-1 in human osteoporosis is ill defined. To our knowledge, this is the first report to quantify the plasma level of PAI-1 and the relative expression of PAI-1 mRNA in osteoporosis patients and healthy female controls. We observed both diminished plasma PAI-1 and mRNA levels in osteoporosis cases than in healthy females. Lowered PAI-1 in osteoporosis cases possibly failed to inhibit both plasminogen activators (tPa and uPa), leading to elevated extracellular matrix erosion by enhancing activation of matrix metalloproteinase (MMP) through raised plasmin levels. 15
A total of five common variants in the PAI-1 gene were genotyped and a possible association with osteoporosis was explored in this study. Similar to an earlier report on Korean post-menopausal women, 9 the −675 4G/5G polymorphism was linked with osteoporosis susceptibility while other SNPs failed to demonstrate such an association. Subjects carrying 5G/5G genotype or minor allele (5G) had 3.18- or 1.65-fold higher chances of developing osteoporosis. However, another independent study on Turkish women failed to demonstrate such association of −675 4G/5G polymorphism with osteoporosis. 10 These discrepancies among studies can be due to different ethnic populations investigated and the number of samples enrolled in the studies. In this report, we have included a greater number of both patients and controls compared to previous reports.9,10
The functional relevance of −675 4G/5G polymorphism has been studied in different populations. Wild-type (4G/4G) is correlated with elevated plasma PAI-1 and mRNA expression. In line with earlier reports, association of 4G/4G with elevated plasma levels of PAI-1 7 and relative mRNA status 13 is seen in both patients and controls. The exact mechanism of how 4G/4G is linked to higher PAI-1 both in protein and mRNA levels is not known. This PAI-1 polymorphism is situated at the promoter region and is believed to alter the binding of certain transcription factors. 8 Furthermore, the stimulation of whole blood with lipopolysaccharide and peptidoglycan showed higher PAI-1 expression in wild-type and heterozygous mutants compared to subjects with a homozygous mutant genetic background for PAI-1 (−675 4G/5G) polymorphism. Altogether, data of this study, along with earlier reports, demonstrate a significant functional role of PAI-1 polymorphism (4G/5G) on determination of plasma PAI-1 and mRNA expression.
In conclusion, PAI-1 polymorphism is significantly correlated with predisposition to osteoporosis and increased a threefold higher chance to develop osteoporosis in subjects harbouring 5G/5G genotype. Furthermore, (−675 4G/5G) variants affect plasma and mRNA expression of PAI-1. Although this study included larger sample numbers from a Chinese cohort, further studies are necessary from different geographical areas to validate our observations.
Footnotes
Acknowledgements
We are grateful to all patients and controls enrolled in this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
