Abstract
Background: Sepsis has been redefined recently as life-threatening organ dysfunction caused by dysregulated host responses to infection and septic shock. Soluble urokinase plasminogen activator receptor (SuPAR) and plasminogen activator inhibitor-1(PAI-1) concentration positively correlate to the activation level of the immune system, and are markers of disease severity and aggressiveness. Objective: The study aimed to identify the blood level of plasminogen activator inhibitor-1 (PAI-1) and soluble urokinase plasminogen activator receptor (SuPAR) in sepsis and its association with mortality. Patient and methods: This is an observational prospective study that enrolled 60 adult patients with sepsis (according to SOFA), admitted to Menoufia and Zagazig university hospitals during the period from December 2019 till October 2020. Plasminogen activator inhibitor-1 (PAI-1) and soluble urokinase plasminogen activator receptor (SuPAR) were checked in all participants. Results: SuPAR and PAI.1 were significant independent predictors of hospital mortality. SuPAR showed sensitivity 100%, specificity 95.9%, and accuracy 94% for prediction of early mortality at a cutoff value of 13.4(pg/ml). While, PAI-1 demonstrated sensitivity 100%, specificity 93.9%, and accuracy of 95% at a cutoff value of 122.5 for predicting mortality. Conclusion: PAI-1 and suPAR were significant predictors of hospital mortality among sepsis patients. The sample size was relatively small, which may have decreased the statistical power of the results of the present study. Hence, additional studies with large sample sizes are required for further validation of the present results.
Introduction
In 1992, sepsis was defined as a systemic inflammatory response syndrome (SIRS) to infection that results from an activation of the innate immune response, regardless of the cause. 1 Sepsis has been redefined again as life-threatening organ dysfunction caused by dysregulated host responses to infection and septic shock as a subset of sepsis in which particularly profound circulatory, cellular, and metabolic abnormalities are associated with a greater risk of mortality than with sepsis alone. 2 Globally, sepsis is common, with an estimated population incidence of 270 cases per-100,000 person yearly and acute mortality of 26.0%. Many reasons suggest even this underestimate the magnitude of sepsis-associated mortality and morbidity. 3
The biomarkers of sepsis can be classified as markers of acute-phase protein (C-reactive protein [CRP], procalcitonin [PCT], and lipopolysaccharide-binding protein), cytokine/chemokine biomarkers (IL-6, IL-8), and markers of other pathophysiologic processes (coagulation factors and soluble cell surface receptors). Also, complement factors (C3a, C5a, and the soluble form of the C5a receptor, sC5aR) have been defined as early markers of sepsis and sepsis severity. CRP and PCT are the most practically used for the detection of bloodstream infections. 4
Plasminogen activator inhibitor-1(PAI-1) is a protein that in humans is encoded by the SERPINE 1 gene, elevated PAI-1 is a risk factor for thrombosis and atherosclerosis. 5 Soluble urokinase plasminogen activator receptor (SuPAR) is a soluble protein form; SuPAR concentration positively correlates to the activation level of the immune system and is present in plasma, urine, blood, serum, and CSF. SuPAR is an indicator of disease severity and aggressiveness. 6 The use of plasma suPAR level enhanced the efficiency of sepsis diagnosis, and the combination of plasma suPAR and APACHE II score improved mortality prediction. 7 Studying long-term outcomes of sepsis is that poor functional status is a risk factor for becoming critically ill as well as a frequent consequence. Many co-morbidities, age, and chronic diseases are risk factors both for sepsis and for impaired quality of life. Therefore, studies work to distinguish between the potentially causal effects of sepsis and that simply describe morbidity and mortality events. 8
Aim of the study
This study aimed to identify the blood level of plasminogen activator inhibitor-1 (PAI-1) and soluble urokinase plasminogen activator receptor (SuPAR) in sepsis and its association with mortality.
Patient and methods
This is an observational prospective study that included 60 adult patients with sepsis admitted to Menoufia and Zagazig university hospitals during the period from December 2019 till October 2020.
All participants were volunteers, and all of them signed written informed consent with explaining the aim of this study before the study initiation.
Approval of the study protocol was obtained by the local Ethical Scientific Committee of Menoufia University’s institutional review board under number (MNF112/2019).
Patients with sepsis were diagnosed based on both of the following criteria
Patients with suspected infection (thorough history taking with clinical status, routine laboratory tests, blood or urine cultures if possible, and radiological images as by (pelvi-abdominal ultrasound, plain chest X-ray, and CT or MRI if possible) who are likely to have a prolonged ICU stay or to die in the hospital identified SOFA score. 2
All subjects were selected according to inclusion and exclusion criteria
Inclusion criteria
Adult patients with sepsis, both sexes, and age more than 30 years.
Exclusion criteria
Pregnant women, cardiac patients, and chronic renal disease.
For all subjects, the following procedures were performed: personal history, past history, present history, and family history. Also, thorough clinical examination: complete general and local examination. In addition, laboratory examination including: Complete blood picture, Kidney functions, and Liver function test.
Specific investigations including: Plasminogen activator inhibitor-1 (PAI-1) and soluble urokinase plasminogen activator receptor (SuPAR).
Methods
3 mL of blood were collected from all patients and a small quantity of plasma (0.2 mL) was isolated within 30 min and stored at −80°C for measurement of SuPAR and PAI-1 levels.
Statistical analysis
Method of calculation and justification for sample size
The sample size calculation
Sample size was calculated using G*power version
N = 60 patients.
Confidence interval = 95%
Alpha = 0.05
Power = 80%
The clinical data were recorded on a report form. These data were tabulated and analyzed using the computer program SPSS (Statistical package for social science) version 21((SPSS Inc, Chicago, IL, USA). The variables were tested using Chi-Squared (χ2) test for qualitative data, Mann Whitney U test for testing quantitative data, correlation coefficient test (Pearson test), multivariate logistic regression analysis, and the ROC (receiver operating characteristic) curves to detect validity of different markers for prediction of early hospital mortality. When p value was less than 0.05, it was considered significant.
Results
Baseline characteristics of studied patients (n=60).
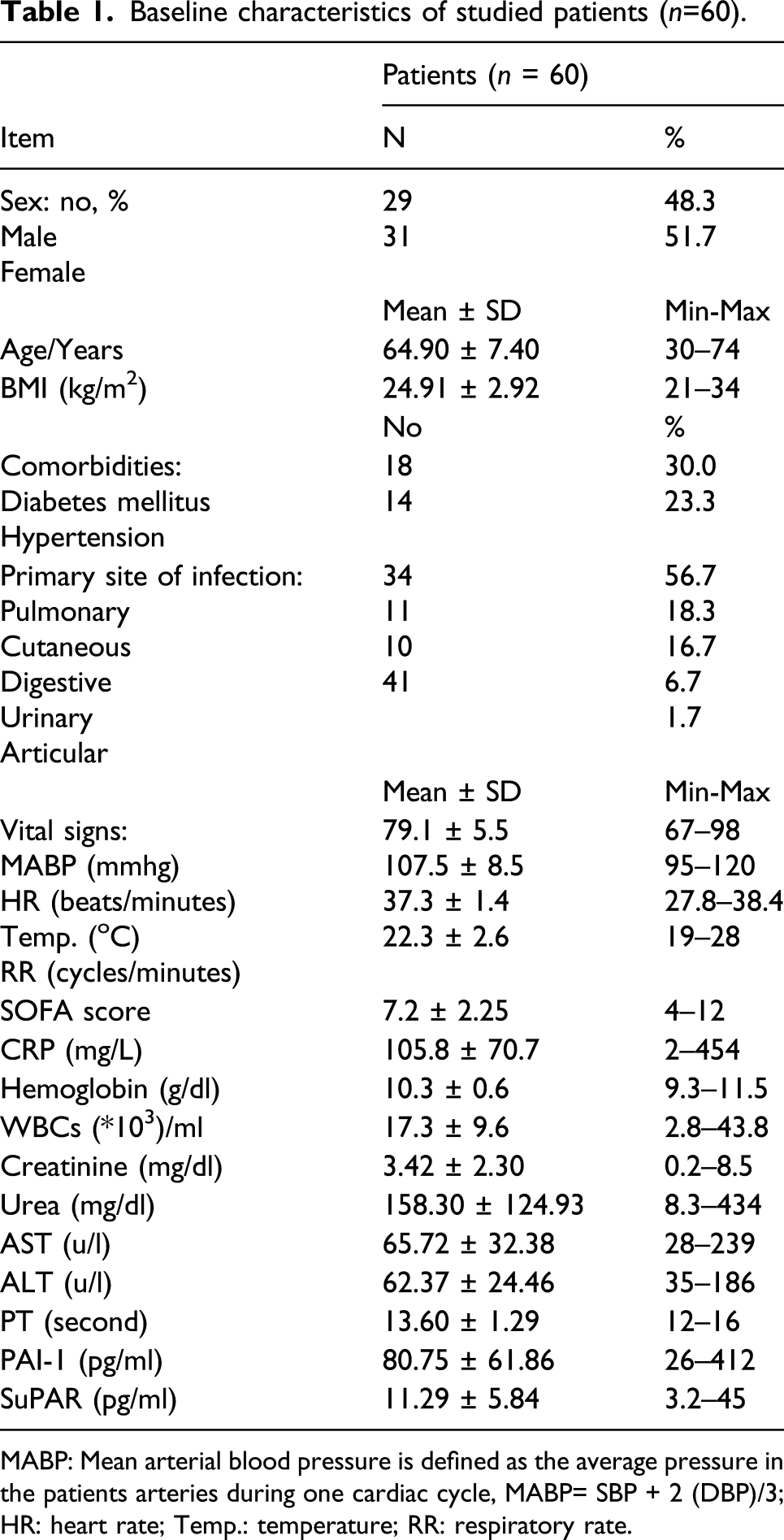
MABP: Mean arterial blood pressure is defined as the average pressure in the patients arteries during one cardiac cycle, MABP= SBP + 2 (DBP)/3; HR: heart rate; Temp.: temperature; RR: respiratory rate.
Comparison between survivors and non-survivors regarding baseline characteristics.

U = Mann Whitney U test, (*) significant.
BMI: body mass index; CRP: C-reactive protein; HB: hemoglobin; WBC: white blood cells; AST: Aspartate transaminase; ALT: Alanine transaminase; PT: prothrombin time; PAI-1: Plasminogen activator inhibitor-1; SuPAR: Soluble urokinase plasminogen activator receptor.
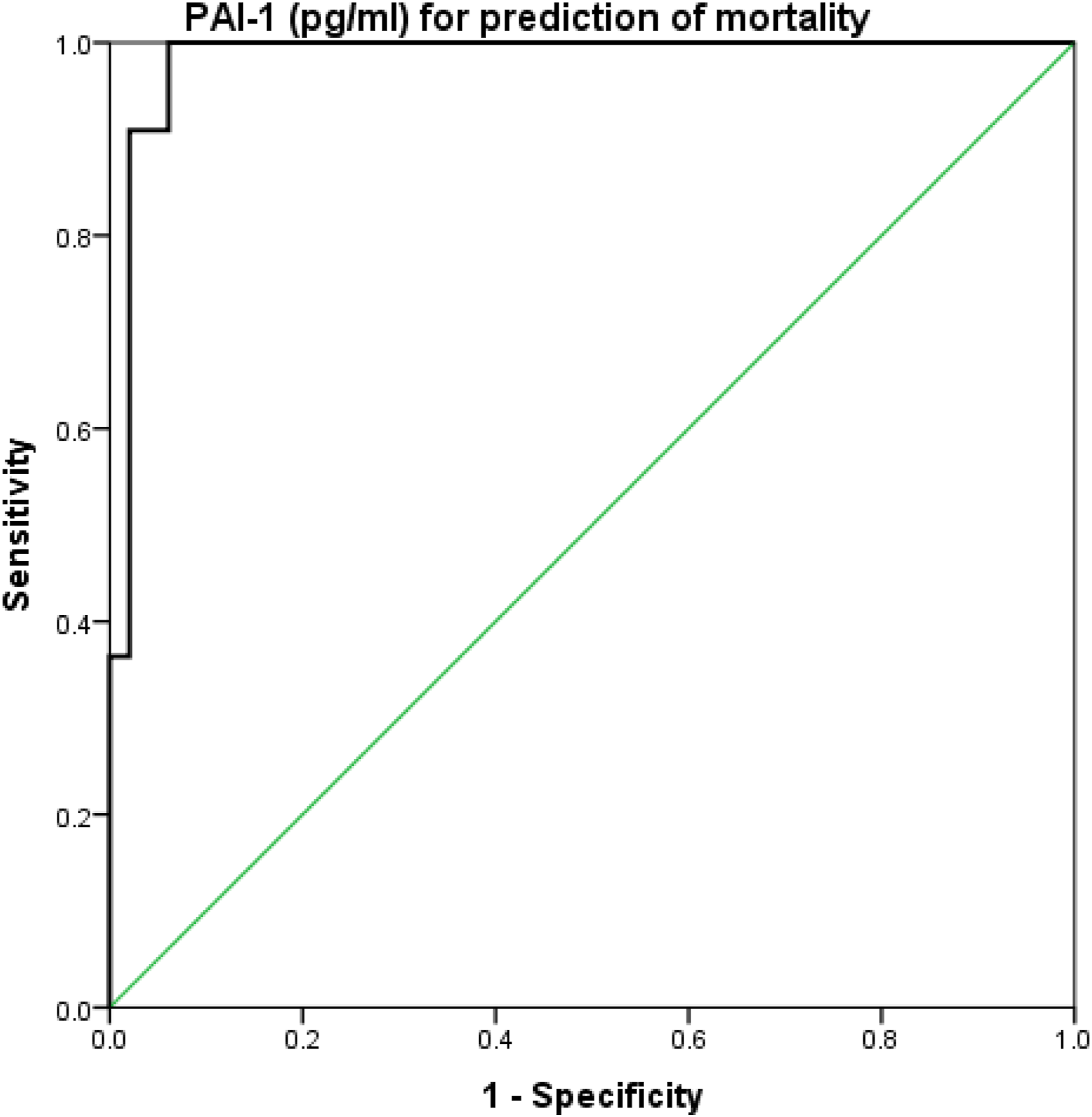
The SOFA score had a sensitivity of 90.9%, specificity of 87.8%, and accuracy of 0.90 at a cutoff value of ≥8.5 for predicting mortality (Figure 1). suPAR had a sensitivity of 100%, specificity of 95.9%, and accuracy of 0.94 at a cutoff value of ≥13.4 for predicting mortality (Figure 2). Finally, PAI-1 had a sensitivity of 100%, specificity of 93.9%, and accuracy of 0.95 at a cutoff value of ≥122.5 for predicting mortality (Figure 3) ROC curve analysis of SOFA score for prediction of mortality. ROC curve analysis of suPAR for prediction of mortality. ROC curve analysis of PAI-1 for prediction of mortality. Cutoff levels of SOFA score, SuPAR, and PAI-1 for predicting early sepsis mortality/7 days. Sens: sensitivity; Spec.: specificity; PPV: positive predictive value; NPV: negative predictive value.


Pearson’s correlation coefficient between PAI-1, SuPAR, and other parameters.

asignificant.
BMI: body mass index CRP: C-reactive protein WBC: white blood cells; AST: Aspartate transaminase ALT: Alanine transaminase PT: prothrombin time PAI-1: Plasminogen activator inhibitor-1 SuPAR: Soluble urokinase plasminogen activator receptor.

Correlation between PAI-1 and PT.

Correlation between PAI-1and SuPAR.
SuPAR level had a significant correlation with each of PT (Figure 6) and SOFA score. Other parameters’ correlations to SuPAR were insignificant, as shown in Table 4. Correlation between SuPAR and PT.
Logistic regression analysis for independent risk of early sepsis mortality/7 days.

asignificant.
PAI-1: Plasminogen activator inhibitor-1; SuPAR: Soluble urokinase plasminogen activator receptor.
Discussion
Plasminogen activator inhibitor type 1 (PAI-1) is a 50-kDa glycoprotein of the serine protease inhibitor family. The primary role of PAI-1 in vivo is the inhibition of both tissue- and urokinase-type plasminogen activators. In addition to this function, PAI-1 acts as an acute-phase protein during acute inflammation. PAI1 is a pivotal player in the pathogenesis of sepsis, a complex clinical syndrome that results from a systemic inflammatory response. 11
The present study showed the mean age of the included patients was 62.1 ± 1 0.7 years and this agreed with the study by
According to the inflammatory labs’ assessment (mean CRP, hemoglobin, and WBCs count), mean creatinine and urea levels, mean AST, ALT, and PT showed that we are similar also to
Our study showed that respiratory rate, SuPAR, PAI-1, and SOFA score were significantly higher among the non-survivor group than the survivor one. While there was no significant difference between the two groups regarding age, sex, heart rate, temperature, SOFA score, CRP, HB, WBCs, creatinine, urea, AST, ALT, and PT. These results agreed with that reported by
While, the current findings disagreed with the study by
The current study revealed that the sensitivity of SOFA score for predicting early sepsis mortality was 90.9%, specificity of 87.8%, and accuracy of 0.90 at a cutoff value of ≥8.5. Also, the sensitivity of SuPAR for predicting mortality was 100%, specificity of 95.9%, and accuracy of 0.94 at a cutoff value of ≥13.4. While the sensitivity of PAI.1 was 100%, specificity of 93.9%, and accuracy of 0.95 at a cutoff value of ≥122.5. In this line, two studies by
The current study revealed that, there was a significant positive correlation between SOFA score and each of suPAR and PAI-1
As mentioned above our study showed that SuPAR, PAI-1, and SOFA score were significant predictors to hospital mortality. This agreed with the study by
On the other hand,
Limitations
One limitation of this study is the limited number of patients as a developing country we have not simply had a registry for all patients and due to environmental and cultural reasons, many critically ill patients from old age not seeking hospital consultation.
Another limitation is the PAI-1 and SuPAR serum levels had been studied a lot in critically ill patients and not a novel hypothesis but to our knowledge, it is the first Egyptian study to elicit its level with early mortality of sepsis and we consider it a trial to use these markers as prognostic predictors not only a diagnostic.
Conclusion
Our study concluded that SuPAR and PAI-1 both can be used for predicting early mortality. Also, SOFA score, PAI-1, and suPAR were significant predictors of hospital morbidity and mortality. The sample size was relatively small, which may have decreased the statistical power of the results of the present study. Hence, additional studies with large sample sizes are required for further validation of the present results.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Approval of the study protocol was obtained by local Ethical Scientific Committee of Menoufia university institutional review board.
Informed consent
All participants were volunteers, and all of them signed a written informed consent with explaining the aim of study before the study initiation
