Abstract
The evidence on whether high-dose new generation proton pump inhibitors (PPIs) including rabeprazole and esomeprazole achieve a higher eradication rate of Helicobacter pylori has not been assessed. The primary comparison was eradication and adverse events (AEs) rate of standard (esomeprazole 20 mg bid, rabeprazole 10 mg bid) versus high-dose (esomeprazole 40 mg bid, rabeprazole 20 mg bid) PPIs. Sub-analyses were performed to evaluate the eradication rate between Asians and Caucasians, clarithromycin-resistance (CAM-R) strains, and clarithromycin-sensitivity (CAM-S) strains of different dose PPIs. We conducted a literature search for randomized controlled trials comparing high-with standard-dose esomeprazole and rabeprazole for H. pylori eradication and AEs. A total of 12 trials with 2237 patients were included. The eradication rate of high-dose PPIs was not significantly superior to standard-dose PPIs regimens: 85.3% versus 84.2%, OR 1.09 (0.86–1.37), P = 0.47. The high dose induced more AEs than those of the standard dose, but didn’t reach statistical significance (OR 1.25, 95% CI: 0.99–1.56, P = 0.06). Subgroup analysis showed that the difference in eradication rate of PPIs between high- and standard-dose groups were not statistically significant both in Asians (OR 0.99, 95% CI 0.75–1.32, P = 0.97) and Caucasians (OR 1.27, 95% CI 0.84–1.92, P = 0.26). Furthermore, there were similar eradication rates in CAM-S (OR 1.2; 95% CI 0.58–2.5; P = 0.63) and CAM-R strains (OR 1.08; 95% CI 0.45–2.56; P = 0.87) between the standard-and high-dose groups. High and standard dosages of new generation of the PPIs showed similar H. pylori eradication rates and AEs as well as between Asian versus Caucasian populations, with or without clarithromycin-resistance. However, further studies are needed to confirm.
Keywords
Introduction
Helicobacter pylori is a major cause of many gastric lesions, such as chronic gastritis, mucosa-associated lymphoid tissue lymphoma, gastroduodenal ulcer, and gastric cancer. 1 Eradication of H. pylori may be an effective measure to prevent these diseases,2,3 especially for gastric cancer, because H. pylori is considered to be one of the important controllable factors affecting the occurrence of gastric carcinoma.2,4 Triple therapies including proton pump inhibitors (PPIs), amoxicillin, and clarithromycin or metronidazole, have been widely used around the world.4–6 However, there has been development of clarithromycin and metronidazole resistance occurred in more than 15% cases in many regions and greatly affected the treatment efficacy. 7 So, searching for better treatments are crucial for the management of gastric diseases.
Adequate acid inhibition is essential for the eradication of H. pylori.8,9 When the pH value in the stomach is 6–8, H. pylori is in an active replication state, which is also very sensitive to antibiotics, thus, in this regard, the antibiotics could reach the most effects.10–12 So, the association between the failure of H. pylori eradication and the PPIs-induced acid inhibition had attracted extensive attention, and several meta-analyses had been published about whether increasing the dose of PPI can improve the eradication efficacy of H. pylori.13,14 A previous meta-analysis 13 showed that high-dose PPIs seems more effective than standard-dose for curing H. pylori infection in 7-day triple therapy. However, there was only one study compared the eradication rate between different dose of the new generation PPIs in the meta-analysis. However, PPIs can be divided into two generations and each displays various acid-suppressing effects and antibacterial activities.15,16 Rabeprazole and esomeprazole, the new generation PPIs, are more potent in acid inhibition by rapid and consistent increase the intragastric pH to 6–8 versus the first generation PPIs (omeprazole, pantoprazole, and lansoprazole), 17 indicating that in such a pH range, H. pylori is more susceptible to antibiotics.18–20 Therefore, the conclusions obtained from this meta-analysis 13 were more applicable to the first generation PPIs. Another meta-analysis 14 compared rabeprazole or esomeprazole with first generation PPIs or with each other, but it emphasized the difference in the eradication rate between esomeprazole and rabeprazole rather than the different doses, while an additional study showed that the low- and high-doses of rabeprazole had a comparable anti-acid secretion effect. 21 Hence, whether high-dose rabeprazole and esomeprazole achieve a higher eradication rate is still uncertain.
More importantly, these studies13,14 did not differentiate the efficacy in different ethnic and Clarithromycin resistance. However, gastric acid secretory capacity might be different in Asian and Caucasian 22 and clarithromycin was acid labile and the efficacy was potentiated by strong acid inhibition.23,24 Thus, we guess that ethnic difference and different resistance to clarithromycin may affect the efficacy of PPIs at different doses.
Therefore, the aim of the present meta-analysis is to compare the eradication rate, safety of high-dose with the standard-dose between the new generation PPIs and also assesses the difference of clinical efficacy between Asians and Caucasians, Clarithromycin resistance (CAM-R) strains, and Clarithromycin sensitivity (CAM-S) strains.
Materials and methods
Search strategy
We searched Pubmed using the keywords of “esomeprazole” or “rabeprazole” or “PPI” or “proton pump inhibitors” and (“helicobacter pylori” or “H. pylori” or “HP”) for trials from 1 January 1990 to 31 December 2020. Search was limited to only human subjects and English studies. We also searched the unpublished trials at relevant web sites (http://www.clinicaltrials.gov) and reference lists from the articles to ensure completeness. In addition, the references lists of studies were also checked for articles missed in the previous searches. Two reviewers (WWG and XZ) independently screened all titles, abstracts, and read full texts for eligible studies. Full-text potentially relevant articles were reviewed independently to determine eligibility.
Inclusion criteria
Inclusion criteria for articles in the meta-analysis were: (i) articles or abstracts had to report the results of comparative, randomized trials, (ii) the eradication regimens had to be new-generation PPIs (rabeprazole and esomeprazole) plus two antibiotics with or without bismuth for at least 7 days, (iii) studies had to include at least two branches of treatment differing only on the PPIs used, (iv)comparing a standard dose of rabeprazole or esomeprazole twice a day with a high dose twice a day between similar triple or quadruple therapies, (v) H. pylori infection had to be determined by biopsy and/or urea breath test (UBT) prior to treatment, (vi) eradication had to be evaluated by biopsy and/or UBT at least 4 weeks after the end of treatment.
The new generation PPIs include rabeprazole and esomeprazole. Regarding the concept of standard and high PPI doses, esomeprazole 20 mg was considered as standard dose.4,6 Different dosages of rabeprazole has been recommended for H. pylori eradication worldwide. Rabeprazole 10 mg twice daily is recommended in Japan, 6 whereas both 10 and 20 mg twice daily are employed in China. 25 Although rabeprazole 10 and 20 mg are both recommended in China, we want to compare the effect of 10 versus 20 mg in order to avoid unnecessary waste. So, we assumed that 10 mg (low dose) as standard-dose for rabeprazole in this article. High-dose PPIs was considered, if at least twice the standard dose of any of the PPIs was used twice a day.
Data extraction
Two authors (WWG and XZ) extracted data independently and discordances were resolved by a third investigator. From the publications we extracted study characteristics, including author identification, year of publication, study design, doses and types of antibiotics, main comparison, sample size for each trail, length of intervention, ethnic. Eradication rates of H. pylori were calculated by intention-to-treat (ITT) when possible, if not, analysis used per protocol data.
Quality assessment
The quality of studies was assessed by two independent reviewers (WWG and XZ) using the components recommended by the Cochrane Handbook for Systematic Reviews of Interventions. 26 Cochrane Risk of Bias Tool assessed the following domains: randomization method, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and other sources of bias.
Statistical analysis
The main comparison was ITT eradication rate using high versus standard-dose of new generation PPIs. For safety measures, analyses were also performed for adverse reaction rates. Sub-analyses were performed to evaluate: (i) the eradication rate of standard and high-dose PPIs in Asian patients versus Caucasian patients, (ii) the eradication rate in CAM-S strains versus CAM-R strains. Amoxicillin and clarithromycin were more acid labile than other antibiotics and sub-analysis was also performed to evaluate the eradication rate in amoxicillin + clarithromycin (A + C) group and non A + C group. The eradication and adverse reaction rates were assessed using odds ratio (OR) with 95% CI. The heterogeneity was assessed with the Q-test and the I2-test. If the Q-test probability was lower than 0.05 (P < 0.05), the studies would be considered heterogeneous. I2 test classified heterogeneity and I2 ⩾50% indicated a substantial level of heterogeneity. If no heterogeneity was observed, a fixed effects model was selected. Meta-analysis was conducted using the Review Manager (Revman Version 5.3, Copenhagen, Denmark).
Results
Studies included
A total of 4827 potentially relevant articles were retrieved. After revising the potentially useful abstracts for the analysis, 16 studies25,27–41were retrieved for detailed evaluation, and 12 studies25,27–37 were eligible for inclusion (Figure 1). A total of 2237 patients were included and the characteristics of the studies were shown in Table 1. Nine studies25,29–35,37compared standard versus high-dose rabeprazole and three studies27,28,36 compared esomeprazole. Ten trials28–37 were treated for 7 days, another two25,27 were treated for 10 and 14 days, respectively. In all studies, PPIs and antibiotics were given twice a day. Four studies25,31,33,35 described the eradication rate in CAM-S and CAM-R. Nine trials25,29–31,33–37 in Asian patients and three27,28,32 in Caucasian patients.
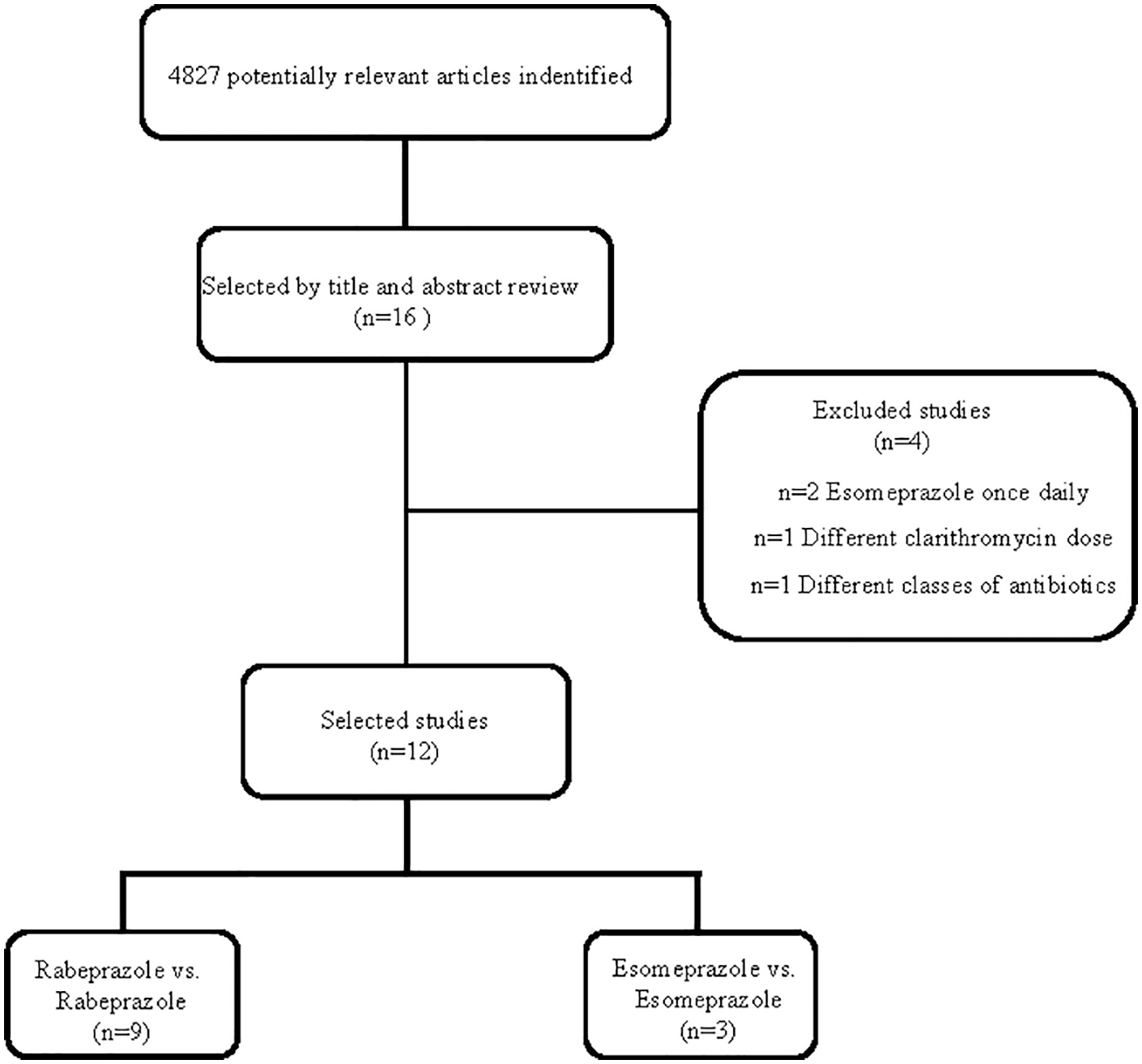
Flow chart demonstrating process of study selection.
Included studies.
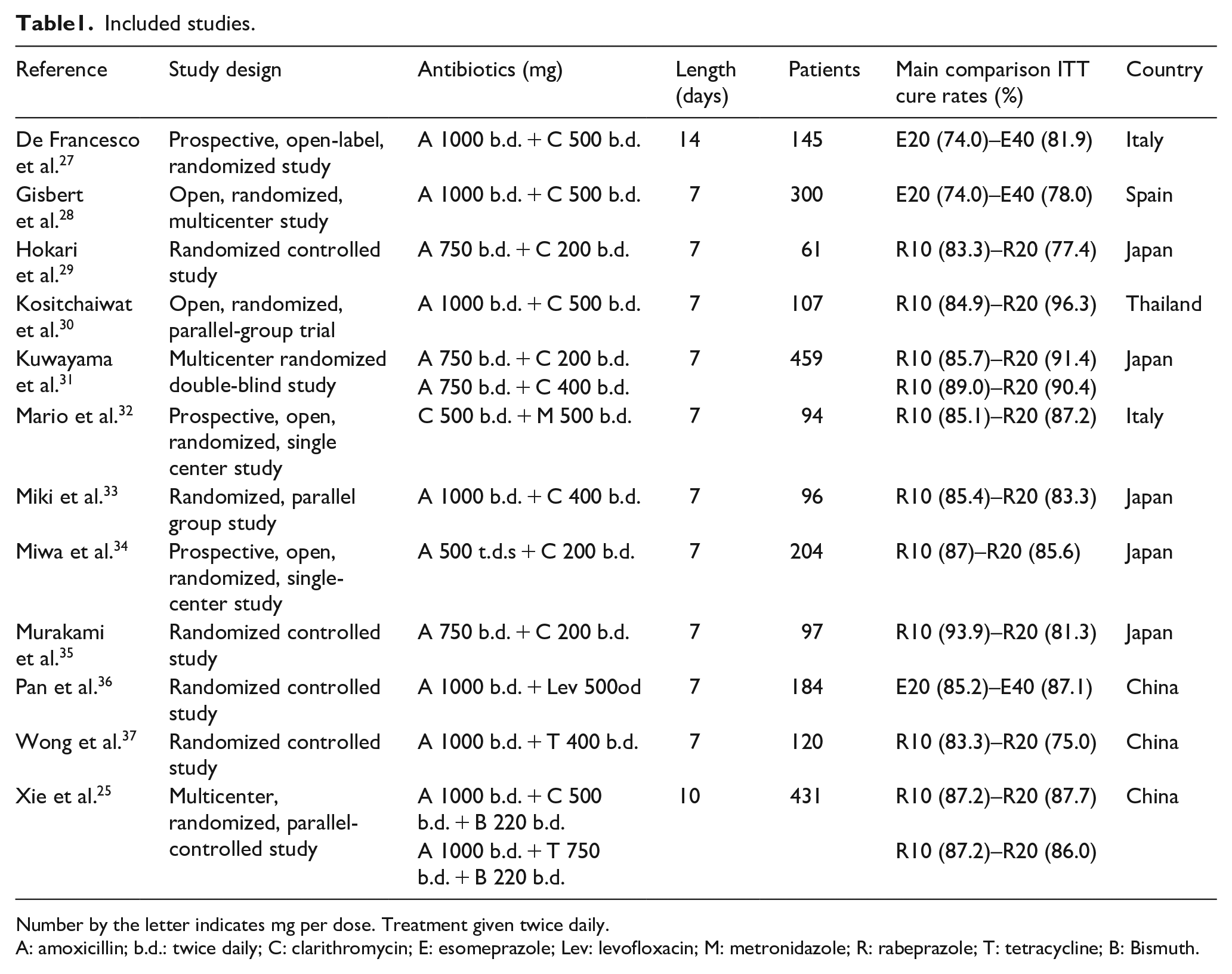
Number by the letter indicates mg per dose. Treatment given twice daily.
A: amoxicillin; b.d.: twice daily; C: clarithromycin; E: esomeprazole; Lev: levofloxacin; M: metronidazole; R: rabeprazole; T: tetracycline; B: Bismuth.
Four studies38–41 were excluded after reading full text. Antos et al. 39 reported the comparison esomeprazole of 40 mg twice daily versus 20 mg twice daily; however, different antibiotics were prescribed in each branch. Anagnostopoulos et al. 40 and Hsu et al., 41 whose study compared esomeprazole 40 mg once a day to esomeprazole 40 mg twice a day. Mansour et al. 38 compared 10 mg rabeprazole taken twice daily with that of 20 mg twice daily, but clarithromycin doses were also different in two groups.
Quality assessment
Risk of bias for the included studies were shown in Table 2. Blinding of participants and personnel was the main source of potential bias. The randomization methods were satisfactory, but only one trial was double blinded.
Risk of bias for the included studies.

Eradication rate of high versus standard-dose PPIs
Patients cured on an ITT basis with a high-dose PPIs were 955/1120 (85.3%) versus 940/1117 (84.2%) with the standard-dose. The OR was 1.09 (95% CI = 0.86–1.37, I2 = 0%) (Figure 2) and no significant difference (P = 0.47) was found in the two branches. Subgroup analyses were performed in trials assessing rabeprazole and esomeprazole, with an OR of 0.99 (95% CI = 0.75–1.32, I2 = 6%, P = 0.96) and 1.32 (95% CI = 0.88–1.97, I2 = 0%, P = 0.18) respectively. The difference in eradication rate of PPIs between high-dose and standard-dose groups in A + C group (85.4% vs 83.2%) and non A + C group (85.1% vs 86.1%) was not statistically significant: (OR 1.18, 95% CI 0.89–1.57, P = 0.25) and (OR 0.93, 95% CI 0.63–1.39, P = 0.72) (Supplemental Figure 1S).

Forest plot of eradication rate of high-dose versus standard-dose regimens.
AEs
A total of nine included studies provided information of AEs. The incidence rate of AEs was higher in high-dose PPIs group in comparison with standard-dose PPIs group (23.0% vs 20.5%). However, no significant difference was found in the two groups (OR 1.25, 95% CI: 0.99–1.56, P = 0.06) (Figure 3).

Forest plot of AEs of high-dose versus standard-dose regimens.
Asians versus Caucasians
Nine studies compared the eradication rate of different dose PPIs in Asian patients and three in Caucasian patients. The difference in eradication rate of PPIs between high-dose and standard-dose groups in Asian (86.7%, 738/851 vs 86.8%, 735/847) and Caucasian (80.3%, 216/269 vs 76.3%, 206/270) was not statistically significant: (OR 0.99, 95% CI 0.75–1.32, I2 = 6%, P = 0.97) and (OR 1.27, 95% CI 0.84–1.92, I2 = 0%, P = 0.26) (Figure 4).

Forest plot of eradication rate of high-dose versus standard-dose regimens in Asians and Caucasians.
CAM-S strains versus CAM-R strains
Four studies (all rabeprazole) compared the eradication rate of different dose PPIs in CAM-S strains and CAM-R strains. The ITT eradication rate for CAM-S strains with high-dose rabeprazole was 318/332 (95.8%) versus 307/323 (95.0%) with the standard-dose. The OR was 1.2 (95% CI = 0.58–2.5, I2 = 0%, P = 0.63) (Figure 5). Curing rates for CAM-R strains with high-dose rabeprazole was 21/44 (47.7%) versus 29/53 (54.7%) with the standard-dose. The OR was 1.08 (95% CI = 0.45–2.56, I2 = 0%, P = 0.87) (Figure 5).

Forest plot of eradication rate of high-dose versus standard-dose regimens in CAM-S strains and CAM-R strains.
Discussion
The present meta-analysis provided evidence that no significant difference in the eradication rate of H. pylori was found in the new generation PPIs including rabeprazole and esomeprazole between high-dose and standard-dose groups. Our results were inconsistent with a previous meta-analysis showing that increasing the dose of PPIs can increase the H. pylori eradication rate. 13 The discrepancy could be because that meta-analysis only compared one study for the eradication rate between different dose of the new generation PPIs. It was known that the differences in acid inhibition lead to different eradication rate and strong acid inhibition increases the efficacy of H. pylori.42–44 Rabeprazole and esomeprazole could provide better control of gastric acid, leading an increase in pH value to the neutral range within a short time than older PPIs when they administered at the standard doses; thus, strengthened the effects of antibiotics.45–47 In addition, 24-h intragastric pH monitoring had shown that 10 and 20 mg of rabeprazole twice daily have a comparable antisecretory effect on the median pH value and percent time of >4.0, >5.0, >6.0, and >7.0 for 24 h. 21 This might explain why the eradication rate in the high-dose group was not better than that in the standard-dose group.
The recommended dose in Japan was rabeprazole 10 mg twice daily, 6 whereas 10 mg twice daily and 20 mg twice daily were employed in China. 25 This variation in recommended doses of rabeprazole might be due to ethnic differences. In the current study, we did perform a sub-analysis of high versus standard PPIs doses between Asians and Caucasians. However, the meta-analysis achieved similar eradication rate in standard and high-dose groups both in Asians and Caucasians. This might be caused by the strong acid-inhibiting effect of the new generation PPIs as we discussed in the introduction section.
CAM-R strains were rising and generally considered to be a major cause of eradication failure.48–50 Many antibiotics, especially clarithromycin, were acid labile and the efficacy was potentiated by strong acid inhibition.23,24 However, our study achieved similar eradication rate between the standard and high-dose groups both in CAM-S and CAM-R strains, which suggests that the rapid and strong inhibitory effect of rabeprazole on acid secretion might not increase the antimicrobial activity of clarithromycin.
The incidence of AEs in our study was similar to that of the previous meta-analysis that comparing high-dose PPIs versus standard-dose in a 1-week triple therapy. 13 Although the safety profile of high and standard-dose PPIs showed no significant difference, there was a small increased trend in high-dose group. One reason might be related to the prolongation of treatment length (one study 10 days, one study 14 days) and another reason might be the addition of bismuth in one study. However, no subgroup analysis was performed due to the small number of relevant studies. Future studies are needed to demonstrate the safety in the bismuth-containing quadruple regimen or in the 10- and 14-day regimen between high- and standard-dose PPIs.
The meta-analysis had a number of limitations. First, the studies had complex diversity of regimens, the type and dose of antibiotics, and the length of treatment. This complexity and the impossibility to sub-analysis data from more similar studies made it hard to assume one unique effect for all studies. Second, the number of studies included in Caucasians was less, so the findings should be interpreted cautiously for Caucasians. Third, only two studies which antibiotics were given for 10 or 14 days, and one that was quadruple therapies. Current guides suggest using triple therapy for at least 10 days or using bismuth quadruple therapies.4,6 However, it is debatable whether the conclusions is still true in these new settings. In addition, only one included study was double blind, and it remained unclear whether allocation concealment was adequate for all of the studies.
Conclusions
Our study revealed a similar H. pylori eradication rate and AEs between high-and standard-dose new generation PPIs (esomeprazole and rabeprazole). Moreover, there were no statistical significance in different PPIs dose between Asians and Caucasians, CAM-S, and CAM-R. More studies are needed for confirmation.
Supplemental Material
sj-docx-1-iji-10.1177_20587384211030397 – Supplemental material for Different dose of new generation proton pump inhibitors for the treatment of Helicobacter pylori infection: A meta-analysis
Supplemental material, sj-docx-1-iji-10.1177_20587384211030397 for Different dose of new generation proton pump inhibitors for the treatment of Helicobacter pylori infection: A meta-analysis by Wenwen Gao, Xiang Zhang, Yanhui Yin, Shuwen Yu and Lu Wang in International Journal of Immunopathology and Pharmacology
Footnotes
Declaration of conflicting interests
No conflict of interest exits in the submission of this manuscript “
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Clinical Pharmacy Research Fund of Shandong Medical (YXHZX019).
Ethics approval
Ethical approval for this study was obtained from *NAME OF ETHICS COMMITTEE OR INSTITUTIONAL REVIEW BOARD (APPROVAL NUMBER/ID)*.
Ethical approval for this study was waived by *NAME OF ETHICS COMMITTEE OR INSTITUTIONAL REVIEW BOARD* because *REASON FOR WAIVER*.
Ethical approval was not sought for the present study because *REASON*.
Not applicable. √
Informed consent
Written informed consent was obtained from all subjects before the study.
Verbal informed consent was obtained from all subjects before the study.
Written informed consent was obtained from legally authorized representatives before the study.
Verbal informed consent was obtained from legally authorized representatives before the study.
Informed consent was not sought for the present study because *REASON*.
*OTHER DETAILS*.
Not applicable. √
Trial registration
*NAME OF TRIAL REGISTRY: TRIAL REGISTRATION NUMBER*.
This randomized clinical trial was not registered because *REASON*.
Not applicable. √
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
