Abstract
Personalized therapies are designed to optimize the safety-to-efficacy ratio by selecting patients with higher response rates based on specific biomarkers. Inflammation plays a vital role in the pathogenesis of non-alcoholic steatohepatitis (NASH), a common liver disorder. Eotaxin-1 plays a role in innate and adaptive immune responses. High eotaxin-1 levels are associated with diabetes and fatty liver disease and, therefore, serves as a biomarker for patient selection. The anti-eotaxin-1 monoclonal antibody is tailored for the personalized therapy of patients with inflammatory conditions due to high levels of eotaxin-1. To evaluate the biological activity and immunomodulatory effect of orally administered anti-eotaxin-1. C57B1/6 mice were treated with either oral or intra-peritoneal anti-eotaxin-1 antibody before induction of immune-mediated hepatitis using an injection of concanavalin A (ConA) and checked for liver injury and eotaxin-1 serum levels. Oral administration of anti-eotaxin-1 alleviated the immune-mediated liver injury. Serum alanine aminotransferase levels decreased to 1807 U/L, compared with 19025 U/L in untreated controls and 3657 U/L in mice treated with parenteral anti-eotaxin-1 (P < 0.005). A trend toward reduced serum eotaxin-1 levels was observed in treated mice, ranging from 594 pg/mL in the controls to 554 and 561 pg/mL in mice treated orally and intraperitoneally (P = 0.08, P = 0.06, respectively). Oral administration of anti-eotaxin-1 antibody shows biological activity in the gut and exerts a systemic immunomodulatory effect to alleviate immune-mediated hepatitis. The data suggest that testing for eotaxin-1 serum levels may enable screening patients with high-eotaxin-1 levels-associated NASH.
Introduction
Personalized therapy optimizes the safety-to-efficacy ratio by selecting patients with higher response rates based on specific biomarkers1–3 and is becoming an important modality for improving patient care by enabling the use of optimized therapies for selected groups of patients.4–7 Inflammation plays a key role in the pathogenesis of non-alcoholic steatohepatitis (NASH), a common liver disorder.8–10 Currently, there are no approved therapies for NASH, and several of the drugs tested over the last few years have shown efficacy only in a subset of patients. 11
Eotaxin-1 (CCL-11) is a potent eosinophil chemoattractant that contributes to tissue eosinophilia. 12 It plays a role in innate and adaptive immune responses. The main receptor for eotaxin-1 is chemokine receptor type 3 (CCR3). Recent evidence indicates that eotaxin-1 also binds to other receptors expressed on various cell types, suggesting a more widespread regulatory role for eotaxin-1.
Recent data suggest that eotaxin-1 may serve as a biomarker for obesity, diabetes, and NASH, as well as a potential therapeutic target. Serum levels of eotaxin-1 were found to be elevated in animal models of diabetes.13,14 The autoimmune diabetic syndrome of the diabetes-prone BioBreeding rat serves as a model for type 1 diabetes, 15 and a time-dependent, insulin-independent pancreatic upregulation of CCL-11 was described in this model. Overexpression of pancreatic CCL-11 was associated with enrichment in Th2 CD4+ T cells within the insulitis lesions.15,16 Elevated levels of CCL-11 were described in patients with type 1 diabetes and may be associated with disease-related complications. 17 Circulating eotaxin-1 and eotaxin-1 mRNA levels in visceral adipose tissue were reported to be increased in obese mice and humans. 18 The stromal/vascular component of adipose tissue is the primary source of eotaxin-1. High eotaxin-1 levels were associated with obesity.18–20 Weight loss in humans led to a reduction in plasma eotaxin-1 levels. 18
Eotaxin-1 levels correlated with liver triglycerides in a high-fat mouse model of NAFLD.14,21 Serum eotaxin-1 concentrations in obese patients with non-alcoholic fatty liver disease (NAFLD) were distinctly associated with pro-inflammatory cytokines, tumor necrosis factor-alpha (TNF-α), interleukins (IL-6, IL-1β), vascular endothelial growth factor (VEGF), and platelet-derived growth factor (PDGF) levels.21,22 Eotaxin-1 levels were predictive of insulin resistance, as expressed by homeostatic model assessment (HOMA) score, intima-media thickness, and severity of liver steatosis. 20 In obese patients with normal and ultrasonographic evidence of NAFLD, serum concentrations of IL-17 were related to eotaxin-1 and intimal thickening, suggesting that IL-17, released by the visceral adipose tissue, induces eotaxin-1 secretion via the smooth muscle cells present in the atheromatous vessels. 23
Bertilimumab is a first-in-class humanized anti-eotaxin-1 monoclonal antibody that lowers eotaxin-1 levels.12,22 It is designed for personalized therapy of high eotaxin-1 level-mediated inflammatory conditions. Parenteral administration of bertilimumab is currently under evaluation in clinical trials in patients with high eotaxin-1 levels suffering from inflammatory bowel disease (IBD), bullous pemphigoid, and atopic dermatitis.12,24
The present study aimed to assess the biological activity and the hepatoprotective and anti-inflammatory effects of orally administered anti-eotaxin-1 in a mouse model of immune-mediated hepatitis.
Materials and methods
Animals
Mice were maintained in the animal care facility of the Hebrew University Hadassah Medical School (Jerusalem, Israel). The mice were administered regular diet with free access to water and maintained in a 12-h light-dark cycle. All experiments were conducted according to the guidelines of the Hebrew University-Hadassah Institutional Committee for Care and Use of Laboratory Animals (No. EO/2017).
Induction of concanavalin A (ConA)-induced hepatitis
ConA (MP Biomedicals, USA) dissolved in a solution of 50 mM Tris pH 7, 150 mM NaCl, and 4 mM CaCl2, was injected into the tail vein at a dose of 500 μg/mouse (15 mg/kg). Mice were sacrificed 16 h after ConA injection.
Experimental groups
Approximately 11–12-week-old C57/black male mice were purchased from Harlan Laboratories (Jerusalem, Israel). Mice were separated into three groups (A–C, n = 5) for testing. Mice in the control group A were treated with ConA alone. Mice in group B received 200 ng of anti-eotaxin-1 (BioTechne, Indianapolis, US, MAB420) orally 2 h before ConA administration and mice in group C received intraperitoneal (IP) administration of 200 ng anti-eotaxin-1 2 h before ConA administration. Serum from the three groups of mice was tested for liver enzymes, alanine aminotransferase, and aspartate aminotransferase (ALT and AST), and serum eotaxin-1 levels.
Liver enzyme measurement
Serum AST and ALT levels from all mice in all groups were determined with an automatic analyzer.
Determination of serum eotaxin-1 levels
Eotaxin-1 serum levels were measured by a sandwich ELISA (DuoSet ELISA for mouse CCL11/Eotaxin; R&D systems, Cat. No: DY420). The optical density between the wavelengths of 450 and 570 nm was measured using a microplate reader. Concentrations in test samples were extrapolated from the standard curve.
Statistical analysis
Statistical analysis was performed using the Student’s t-test. A P value <0.05 was considered significant.
Results
Figure 1 shows the effect of both oral and IP administered anti-eotaxin-1 monoclonal antibody on immune-mediated liver injury, as measured by its effect on liver enzymes. Both the oral and IP administration of anti-eotaxin-1 antibody showed a decrease in ALT and AST levels, and a statistically significant association with alleviation of liver injury was observed in groups B and C when compared to the untreated controls in group A (1807 vs 3657 vs 19025 U/L for ALT and 1399 vs 2418 vs 17138 U/L for AST, respectively, P < 0.0001). Moreover, the oral route of administration exerted a more profound effect on the reduction of liver enzymes as compared with the IP route (P < 0.001 and P = 0.34, for ALT and AST, respectively, normal levels up to 50 U/L).

Effect of oral and IP administration of anti-eotaxin-1 antibody on immune mediated hepatitis. There was a significant decrease in liver enzymes (ALT and AST) in treated group B and C compared to group A (P < 0.0001).
Figure 2 shows the effect of administration of anti-eotaxin-1 monoclonal antibody on serum eotaxin-1 levels. Both oral and IP administration showed a reduced serum eotaxin-1 level in comparison with group A, from 594 pg/mL in the controls to 554 and 561 pg/mL, (P = 0.08, P = 0.06) respectively, in mice treated orally and intraperitoneally. However, there was no significant difference in the decrease between the two treated groups B and C (P = 0.38).
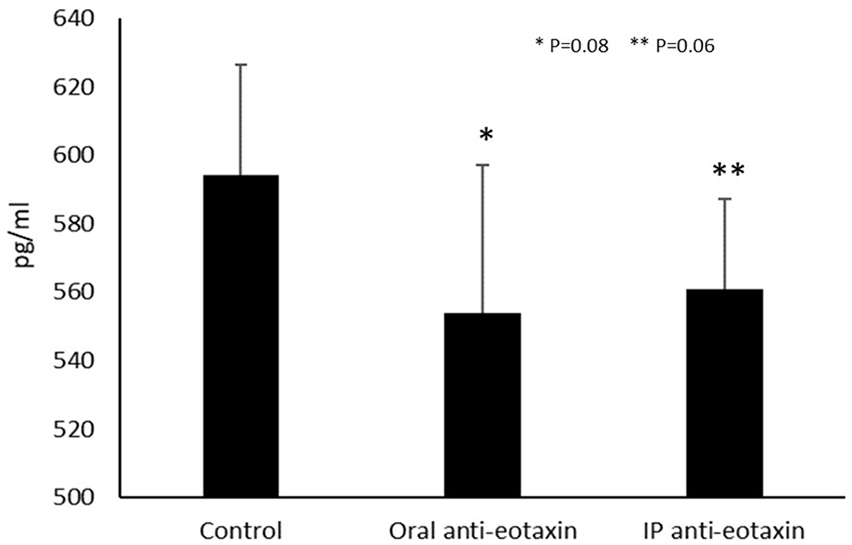
Effect of oral and IP administration of anti-eotaxin-1 antibody on serum eotaxin levels. There was a trend toward reduced serum eotaxin levels in the both orally and IP administered anti-eotaxin-1 antibody compared to control group A (P = 0.08 and 0.06, respectively).
Discussion
The results of the present study show that both orally and parenterally administered anti-eotaxin-1 antibody exert hepatoprotective and anti-inflammatory effects in a mouse model of immune-mediated hepatitis, as assessed by a significant reduction in liver enzymes. The effect was associated with a decrease in serum eotaxin-1 levels. The observed decline in eotaxin-1 levels was not significant, and this result could be due to the interference of the injected anti-eotaxin-1 antibody in the eotaxin-1 ELISA. A similar artifact was reported in pharmacokinetic studies in humans (data not shown).
Eotaxin-1 belongs to the chemokine family and has a role in peripheral immune cell migration, augmentation of the pro-inflammatory cascade, and leukocyte activation. Eotaxin-1 is considered an important mediator in allergic diseases such as asthma and atopic dermatitis.25–29 Furthermore, elevated eotaxin-1 levels are shown in various pathologic conditions, ranging from airway inflammation to Hodgkin’s lymphoma, coronary artery disease, and various autoimmune diseases.25–28,30,31 Eotaxin-1 also plays a role in the pathogenesis of IBD. 12
Eotaxin-1 is strongly associated with insulin resistance, diabetes, and fatty liver disease.14,20,21,23,32–34 It plays a role in the pathogenesis of several chronic liver diseases wherein the immune system contributes to liver damage. Eotaxin-1 is secreted from the liver, and its levels correlate with the degree of lymphocyte recruitment, which augments the inflammatory cascade. 35 ConA injection in mice mimics a model for T cell-mediated liver injury. ConA injection induces expression of eotaxin-1 in the liver and elevates eotaxin-1 serum levels along with those of IL-5. 36 The induction was attenuated in IL-4 (–/–) and STAT6 (–/–) mice. Similarly, eotaxin-1 mRNA levels were elevated in the livers of animals with alcoholic hepatitis. 37 Eotaxin-1 blockade atten-uates ConA-induced liver injury and leukocyte infiltration. 36 The results of the present study, showing alleviated liver damage by parenteral administration of anti-eotaxin-1, lends support to the above data. However, oral administration of anti-eotaxin-1 was not tested previously in this model.
In humans, eotaxin-1 levels were elevated in patients with primary sclerosing cholangitis but were lower in primary biliary cirrhosis and autoimmune hepatitis. 38 Immunohistochemical studies on livers of patients with the drug-induced liver disease showed high expression of eotaxin-1. 39 An elevated eotaxin-1 expression is associated with the pathogenesis of chronic liver diseases, 40 and eotaxin-1 is also up-regulated in senescent human hepatic stellate cells. Eotaxin-1 levels were correlated with hepatic biosynthetic capacity and other inflammatory cytokines, and higher eotaxin-1 levels were associated with hepatic necroinflammation and fibrosis in liver histology. An elevated serum level of eotaxin-1 is present in patients with cirrhosis correlated with the Child-Pugh and model of end-stage liver disease (MELD) score. 40 Serum eotaxin-1 levels were suggested as a biomarker for disease severity in cirrhosis. 40 High levels correlated with liver complications and predicted an unfavorable prognosis.
High levels of eotaxin-1 before anti-viral therapy with pegylated interferon and ribavirin in chronic hepatitis C virus (HCV) patients were predictive of a sustained virologic response. 41 Higher plasma eotaxin-1 levels before HCV anti-viral therapy were also useful in predicting virologic response to HCV treatment with interferon-α + ribavirin in HIV/HCV co-infected patients. 42
The data of the present study show a beneficial effect of oral administration of anti-eotaxin-1. Interestingly, an enhanced statistically significant effect was documented with the oral delivery as compared with the parenteral route. As oral administration is not expected to be associated with clinically significant absorption of the antibody, the mechanism of action of the two routes of administration is different. Parenteral administration of the antibody alleviates inflammation by targeting eotaxin-1 in the blood or tissues. 36 In contrast, the oral mode of administration is more likely to exert a systemic anti-inflammatory effect by exerting a local effect on the gut immune system. Eotaxin-1 is ubiquitous throughout the normal gastrointestinal tract. 43 Eotaxin-1 is a regulator of homing of eosinophils to the gut, 44 and eosinophilic infiltrates in the esophagus, stomach, small bowel, or colon are observed in a variety of disease states. In addition, lower eosinophil numbers are present in the lamina propria of the healthy intestinal tissues. 45 Eotaxin-1-deficient mice had lower numbers of eosinophils in the gut mucosa, and these changes occurred with normal eosinophil numbers in the hematopoietic compartments. Eotaxin-1 is produced by mononuclear cells within the lamina propria of the gut. 46 Oxazolone-challenged L2-IL5 mice developed dose-dependent pan-esophageal eosinophilia, and their esophagi showed increased eotaxin-1. 47 In the dextran sodium sulfate-induced colitis model, increased eotaxin-1 levels localized to inflammatory cells were observed. 48
Oral immunotherapy using non-absorbable antibodies was effective in both inflammatory bowel diseases and NASH.10,49–56 It is based on the assumption that generating signals in the gut may re-direct the systemic immune system to an anti-inflammatory route. One of the major advantages of this method is its ability to induce immunomodulation without immunosuppression. 10 It is hypothesized that oral administration of anti-eotaxin-1 functions similarly by binding to eotaxin-1 in the gut mucosa.
Limitations
Future studies will determine the effect of therapy on liver histology and the cellular and humoral mechanisms which mediate the effect. Power calculation for estimation of sample size selected was not done in this feasibility study.
Conclusions
Oral administration of the non-absorbable anti-eotaxin-1 antibody shows biological activity in the gut and exerts a systemic immunomodulatory effect to alleviate immune-mediated hepatitis. The data suggest that testing for serum eotaxin-1 levels and using oral anti-eotaxin may enable the selection and treatment of patients with high-eotaxin-1 level-associated fatty liver disease.
Footnotes
Animal welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
Mice were maintained in the animal care facility of the Hebrew University Hadassah Medical School (Jerusalem, Israel). The mice were administered a regular diet with free access to water and maintained in a 12-hour light-dark cycle. Or, Or, *.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: YI is a consultant for Immune Pharma
Ethics approval
Ethical approval for this study was obtained from the Hebrew University-Hadassah Institutional Committee for Care and Use of Laboratory Animals (No. EO/2017).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
