Abstract
Background
COVID-19 patients may have residual pulmonary alterations after the acute disease, with fibrotic-like alterations. Since metalloproteinases (MMP) and their regulators may be involved in inflammation and abnormal repair processing, we aimed to investigate the correlations between MMP-9, a tissue inhibitor of metalloproteinases (TIMP-1) and chest CT abnormalities in acute phase and mid-term follow-up.
Methods
COVID-19 patients with plasma analyses and CT scans performed at acute onset and 3 months after discharge (T post) were evaluated. MMP-9, TIMP-1, and MMP-9/TIMP-1 ratio were analyzed. CT extents of COVID-19 pneumonia and fibrotic-like alterations were visually scored (score range 0–25). Spearman rank correlation analysis (p-value <.05) was computed between acute and mid-term plasma analyses and CT scores.
Results
39 patients were enrolled. At hospital admission, MMP-9, TIMP-1, and MMP-9/TIMP-1 had a median of 240.5 ng/mL, 258.8 ng/mL, and 0.9. The median CT global and fibrotic-like scores were 9 and 6. At T post, MMP-9 and TIMP-1 were not statistically different (p-value <.05). There was a reduction of CT global score (p-value = .00007). A significant correlation was found between MMP-9 and CT global score at hospital admission (ρ = 0.456, p-value = .003) and between MMP-9/TIMP-1 ratio and CT global score at hospital admission (ρ = 0.406, p-value = .009). No other significant correlations were found between plasma enzymes and CT alterations, both in acute and mid-term follow-up.
Conclusion
MMP-9 plasma levels and MMP-9/TIMP-1 ratio correlate with lung involvement during the acute phase. None of the levels of MMP-9, TIMP-1, and MMP-9/TIMP-1 ratio may be adopted as predictors of residual pulmonary alterations in mid-term follow-up.
Introduction
A common manifestation of COVID-19 is lung involvement, often in the form of pneumonia. Of all patients with COVID-19 infection, approximately 15% may develop a severe illness, requiring hospitalization and oxygen therapy, 1 while 5% may progress to a critical condition, developing multiorgan failure, acute lung injury, or acute respiratory distress syndrome, necessitating admission to the intensive care unit. 2 There is evidence supporting vascular damage and immunothrombosis as key factors involved in the pathogenetic mechanisms of severe forms of COVID-19. Postmortem histopathological examinations have revealed diffuse alveolar damage with perivascular infiltration of inflammatory cells, extensive damage to the lining of blood vessels, severe endothelial injury, and thrombosis.3–5 A key factor in these mechanisms is the dysregulation of neutrophil extracellular trap (NET). In fact, there is a robust correlation between neutrophil extracellular trap (NET) and severity of respiratory illness. 6 Activated neutrophils release metalloproteinases (MMPs), a large family of zinc-dependent endopeptidases involved in extracellular matrix (ECM) degradation. These enzymes are inhibited by binding or interactions with tissue inhibitors of metalloproteinases (TIMPs). MMPs, whose expression and activation are increased by cytokines such as IL-1 and TNF-alpha, contribute to the “cytokine storm,” supporting the migration of immune cells to infection sites and promoting platelet and neutrophil activation.7–11 There are 25 members of this family, involved in different neutrophil extracellular trap (NET) changes. For instance, MMP-3 and MMP-9 can promote abnormal repairing process, MMP-7 and MMP-8 may influence the pulmonary levels of profibrotic/antifibrotic mediators, and MMP-8 and MMP-10 are involved in the pulmonary macrophage regulation. 12 Thus, the NET state depends, at least partially, from the different expression of MMPs and TIMPs.
Considering the severe form of COVID-19 as a septic state, MMP-2 and MMP-9 have been proposed as potential biomarkers of disease severity, including for their possible role as therapeutic targets.13–15 In fact, several studies have shown increased MMP-9 levels in critical patients, with higher levels of MMP-9 observed in patients with acute respiratory distress syndrome compared to those with non-severe COVID-19.16,17 Conversely, MMP-2 levels were found to be lower. 18
On the other hand, although chest computed tomography (CT) is considered non-specific for evaluating COVID-19-related pneumonia, it has shown high sensitivity for detecting parenchymal alterations and, therefore, represents the best imaging tool for assessing the degree of lung involvement. 19 Moreover, several CT scoring systems have been proposed to quantify the extent of pneumonia, both using semi-quantitative methods and automated tools.20–22 Furthermore, CT has proven useful in the follow-up of COVID-19-related alterations. For instance, CT abnormalities may resolve after the acute phase or may progress to so-called fibrotic-like alterations, which are parenchymal abnormalities with signs that may be related to fibrosis but can improve over time. 23 However, the underlying factors that may lead to these different outcomes remain unclear. Since MMP activity may be involved in tissue repair and fibrotic processes, we hypothesized that their levels might be related to the different evolution of COVID-19 pneumonia, though few data are available in the literature. 24
Thus, the aim of this study was to investigate whether MMP and TIMP levels could serve as potential biomarkers of fibrotic-like alterations in COVID-19 patients during mid-term follow-up.
Materials and methods
Study design
This single-center retrospective study was conducted at the Department of Radiological Sciences, Oncology and Pathology, Policlinico Umberto I Hospital, “Sapienza” University of Rome. Patients with a confirmed diagnosis of COVID-19 pneumonia, admitted to the hospital between March and June 2020, were considered for enrollment. Inclusion criteria included CT and plasma tests performed at hospital admission and the same examinations repeated 90 days later.
The diagnosis of COVID-19 infection was confirmed using a nasopharyngeal swab and a commercial reverse transcription-polymerase chain reaction (RT-PCR) kit, following the manufacturer’s instructions (RealStar® SARS-CoV-2 Altona Diagnostics, Hamburg, Germany).
The following clinical and laboratory data were collected: age, sex, comorbidities, symptoms and acute respiratory distress syndrome development, and COVID-19 treatment.
This study received approval from the local ethics committee (Lazio, Area 1, Rif. 7226, Prot. 0473/2024). The need for informed consent was waived due to the retrospective nature of the study.
During the acquisition and processing of data, national and international regulations were adhered to.
Blood test for plasma enzymes
Peripheral whole blood samples were evaluated using a Microfluidic Next Generation Enzyme-Linked Immunosorbent Assay (ELISA) to separately determine the levels of MMP-9 and TIMP-1, using the Simple Plex™ Ella Assay (ProteinSimple, San Jose, CA, USA) on the Ella™ Microfluidic System (Bio-Techne, Minneapolis, MN, USA), according to the manufacturer’s instructions and as previously described.25–27 Then, the MM-9/TIMP-1 ratio was computed.
Chest CT analysis
Pulmonary involvement was evaluated by a chest radiologist on axial CT images reconstructed with a sharp kernel and standard lung window, using a semi-quantitative score, as proposed in previous studies.28,29 The score was visually calculated for each of the five lobes, considering the extent of anatomical involvement, as follows: 0, no involvement; 1, <5% involvement; 2, 5–25% involvement; 3, 26–50% involvement; 4, 51–75% involvement; and 5, >75% involvement. The resulting global CT score was the sum of each individual lobar score and ranged from 0 (no involvement) to 25 (maximum involvement).30,31 The prevalent parenchymal alterations were also evaluated, including ground-glass opacity, consolidation, crazy paving, and reticulation. The score was also used to assess fibrotic-like alterations, defined by the association of pulmonary abnormalities with architectural distortions, bronchial dilation, volume loss, or honeycombing,32–35 independently computed.
Statistical analysis
Data were analyzed using statistical software (Prism version 9, GraphPad Software). Continuous variables were expressed as the median and interquartile range (IQR), while categorical variables were expressed as counts and percentages. Patient characteristics were compared using Student’s t test or 2-tailed χ2 test for continuous and categorical variables, respectively. The 2-tailed χ2 test or Fisher’s exact test and the nonparametric Mann–Whitney test were used for comparing proportions and medians. Longitudinal evaluation was performed using the nonparametric Wilcoxon test. The relationship between clinical metalloproteinase plasma levels was assessed using Spearman rank correlation analysis. A p-value <.05 was considered statistically significant.
Results
Patients’ characteristics
Demographic and clinical characteristics of patients.

ARDS: acute respiratory distress syndrome; CT: computed tomography; IQR: interquartile range.
Plasma levels of MMP-9 and TIMP-1
Next generation ELISA, zymography data, and differences between hospital admission and after discharge. Data are shown as the median and interquartile range (IQR).
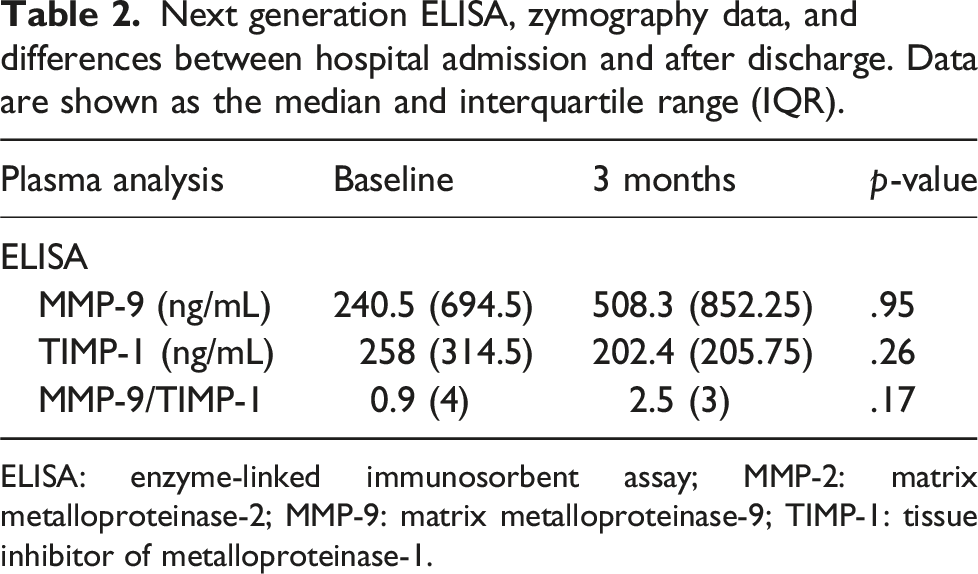
ELISA: enzyme-linked immunosorbent assay; MMP-2: matrix metalloproteinase-2; MMP-9: matrix metalloproteinase-9; TIMP-1: tissue inhibitor of metalloproteinase-1.
CT analysis and correlations with plasma enzymes
In the acute phase of the infection, the median CT global score was 9 (IQR: 5.5), and the most prevalent parenchymal alteration was ground-glass appearance (46.2%), followed by consolidation (33.3%), crazy paving (17.9%), and reticulation (2.6%) (Figure 1). At hospital admission, the median fibrotic-like CT score was 6 (IQR: 4.5). The most common pattern associated with fibrotic-like alterations was consolidation (28.2%), followed by ground-glass appearance (25.6%), crazy paving (17.9%), and reticulation (5.1%). Chest CT findings of COVID-19 pneumonia. Patient 1 (a; b): extensive area of ground-glass opacity in the right upper lobe with a small inner area of consolidation (black arrow) and crazy paving (white arrow) at hospital admission, global CT score of 4 (a); complete resolution after 3 months (b). Patient 2 (c; d): bilateral areas of ground-glass opacities at hospital admission, global CT score of 12 (c); residual abnormalities, mainly sustained by ground-glass opacity, global CT score of 11, and a consolidation with architectural distortions and a bronchial dilation (white arrow), configuring a fibrotic-like appearance (d).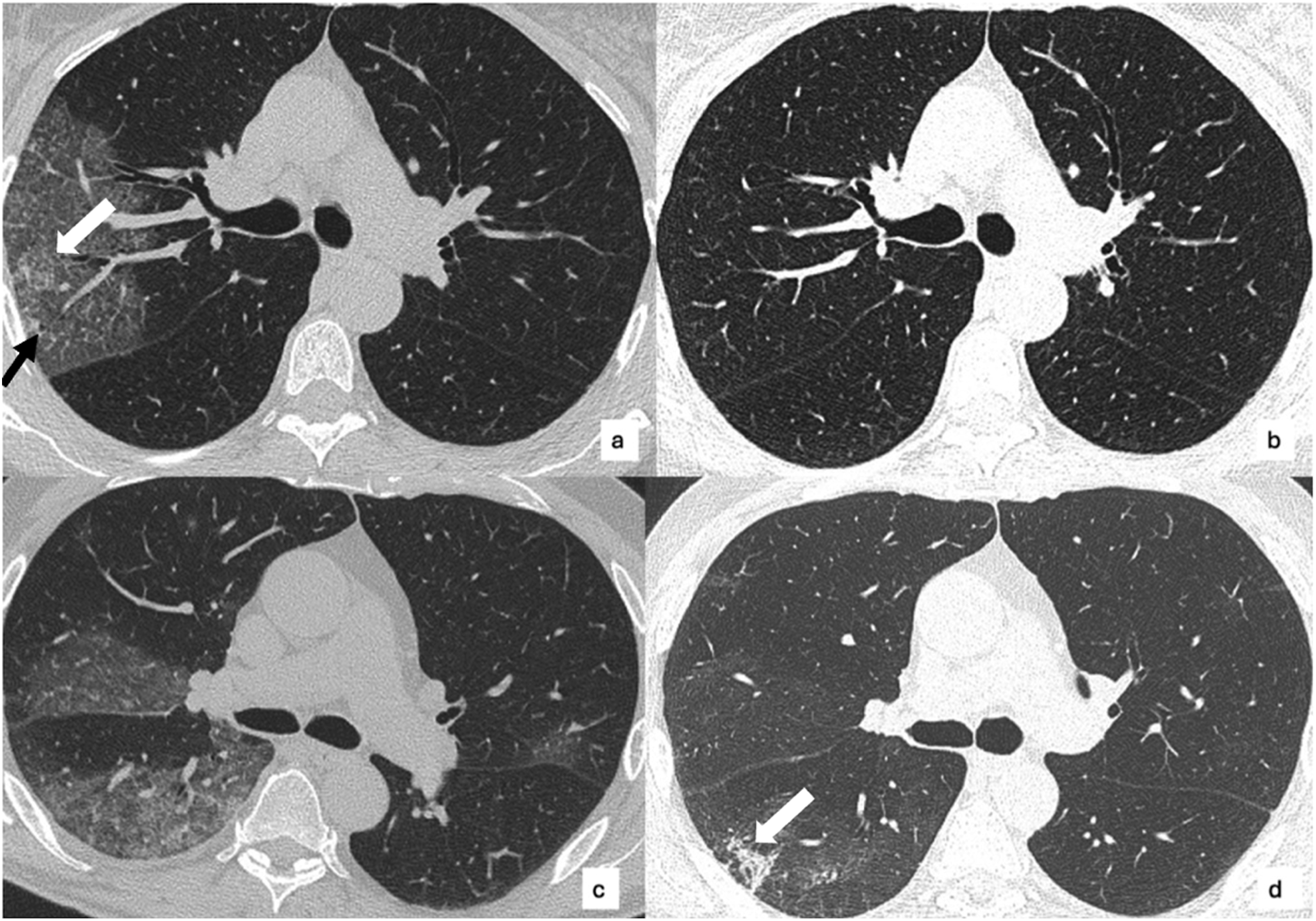
CT score and differences between hospital admission and after discharge.

CT: computed tomography; fib-like: fibrotic-like; GGo: ground-glass opacities; IQR: interquartile range.
At hospital admission, a positive correlation was found between plasma levels of MMP-9 and the global chest CT score (ρ = 0.456, p-value = .003), and between the MMP-9/TIMP-1 ratio and the CT global score (ρ = 0.406, p-value = .009). Scatter plots are reported in Figure 2. No other significant correlations were found, especially between plasma levels of MMP-9 and MMP-9/TIMP-1 at baseline and chest CT scores at T post (Tables 4 and 5). Regarding the differences between baseline and T post, the only significant difference was found for the ΔCT global score, but there were no significant correlations with MMP-9, TIMP-1, and MMP-9/TIMP-1 in the acute phase (ρ = −0.135, p-value = .407 for MMP-9; ρ = 0.207, p-value = .199 for TIMP-1; and ρ = 0.078, p-value = .634) or at T post (ρ = −0.019, p-value = .911 for MMP-9; ρ = −0.031, p-value = .854 for TIMP-1; and ρ = 0.083, p-value = .620). Scatter plots of correlations between MMP-9 and MMP-9/TIMP-1 ratio with CT global score at hospital admission. MMP-9: matrix metalloproteinase-9; TIMP-1: tissue inhibitor of metalloproteinase-1. Correlations between plasma enzymes at baseline and CT analysis. Significant values are in bold. Correlations between plasma enzymes at 3 months and CT analysis.


Discussion
In our study, MMP-9 and MMP-9/TIMP-1 were elevated among COVID-19 patients and associated with COVID-19 severity, in line with previously observed MMPs.36,37
The role of chest CT as a primary imaging tool in the diagnosis and management of COVID-19 has been widely discussed, including the need for standardized radiological reports using various visual scores.38–40 As described, SARS-CoV-2 stimulates the immune system, leading to a cytokine storm, 41 and COVID-19 pneumonia may be associated with massive alveolar damage and loss of lung architecture. This occurs through an alteration of the NET balance influenced by the different expressions and plasma activities of MMPs and TIMPs. More specifically, MMP-9 induces inflammation and degradation of the alveolar-capillary barrier, stimulating the migration of inflammatory cells and destruction of lung tissue, 42 while TIMP-1 is a specific inhibitor of NET degradation enzymes, promoting fibroblast proliferation and exhibiting antiapoptotic and proinflammatory effects.42,43 Thus, the MMP-9/TIMP-1 ratio also plays a crucial role, considering the need for balance in lung tissue damage and repair. Various studies have demonstrated the role of MMP-9 levels and the MMP-9/TIMP-1 ratio in the development of community-acquired pneumonia compared to healthy patients, also showing their relationship to the severity of community-acquired pneumonia. 43 Moreover, Demir et al. revealed higher levels of MMP-9 and TIMP-1 in patients with lung involvement on chest CT scans than in those with no lung involvement, without significant differences related to the extent and severity of lung involvement. 42
However, in our study, plasma levels of MMP-9, TIMP-1, and the MMP-9/TIMP-1 ratio did not correlate with mid-term follow-up CT alterations.
Hence, these results suggest that the plasma enzymes we investigated cannot be adopted as predictors or biomarkers of residual mid-term CT alterations. Since metalloproteinases may be involved in abnormal repair processes, 12 the process underlying the absence of complete restitutio ad integrum after the acute onset seems unrelated to these enzymes. COVID-19 pneumonia typically has a natural course of about 2 to 3 weeks, with described CT alterations such as ground-glass opacities, consolidations, reticulations, and crazy paving. 44 After the acute onset, either complete restitution or residual abnormalities on CT have been observed in survivors. These residual abnormalities may be associated with signs commonly related to fibrosis, such as architectural distortions or bronchiectasis, leading radiologists to describe them as scars or residual fibrotic interstitial lung disease. However, although these alterations were observed several months to even years after the acute onset, they can reduce over time, as observed in previous SARS-MERS infections. For this reason, the term “fibrotic-like changes” was coined for residual parenchymal abnormalities in COVID-19 pneumonia with apparent signs of fibrosis, such as bronchial dilation instead of apparent traction bronchiectasis. 24
In our study, fibrotic-like alterations were mainly sustained by architectural distortions, but bronchial dilation was also observed. The complete course and the underlying mechanisms of fibrotic-like alterations remain to be fully understood. Since MMPs may have a role in the abnormal fibrotic repair processes, we sought to investigate whether MMP-9 and TIMP-1 could play a role. Our results did not confirm this hypothesis, but other MMPs could be further investigated. Moreover, it will be interesting to monitor patients with fibrotic-like alterations over time to verify if long-term residual abnormalities may indeed be considered fibrosis (e.g., progression of fibrotic signs) and to check if there is a relationship with MMP expression in these patients. In fact, since the levels of MMP-9, TIM-1, and MMP-9/TIMP-1 did not change significantly after 3 months, an active inflammation related to COVID-19 could be still present. Hence, a longer follow-up would be desirable, excluding patients with other concomitant causes of inflammation that might influence enzymes levels during that period.
The main limitation of our study is the sample size; in fact, the sample size, computed with a power set at 80% and α = 0.05, based on survival rate of COVID-19 infections admitted to the hospital in 2020 in our area, 45 should be at least 137. Then, longer period of follow-up would be desirable. The retrospective study design limited the availability of CT scans and blood tests at 6 or 12 months. In fact, in many cases, there was no clinical indication to repeat CT or blood tests after 6 or 12 months after hospital admission. Moreover, due to the relatively small size of our study population, we did not verify the possible influence of specific comorbidities, that could influence the results. Lastly, due to logistical limitations, we investigated only some of all possible plasma enzymes that could have a role in mid-term and long-term post COVID-19 lung alterations.
In conclusion, although we confirmed that plasma levels of MMP-9 and the MMP-9/TIMP-1 ratio during the acute phase may be related to the severity of lung involvement on CT, the absence of a significant correlation between MMP-9, TIMP-1, and the MMP-9/TIMP-1 ratio in the acute phase and residual pulmonary alterations on CT after discharge suggests that we cannot recommend their adoption as prognostic biomarkers for predicting the evolution of pulmonary involvement in COVID-19 pneumonia.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Sapienza Università di Roma.
